Abstract
Sympathetic activation is a hallmark of chronic and end-stage renal disease and adversely affects cardiovascular prognosis. Hypertension is present in the vast majority of these patients and plays a key role in the progressive deterioration of renal function and the high rate of cardiovascular events in this patient cohort. Augmentation of renin release, tubular sodium reabsorption, and renal vascular resistance are direct consequences of efferent renal sympathetic nerve stimulation and the major components of neural regulation of renal function. Renal afferent nerve activity directly influences sympathetic outflow to the kidneys and other highly innervated organs involved in blood pressure control via hypothalamic integration. Renal denervation of the kidney has been shown to reduce blood pressure in many experimental models of hypertension. Targeting the renal nerves directly may therefore be specifically useful in patients with chronic and end-stage renal disease. In this review, we will discuss the potential role of catheter-based renal denervation in patients with impaired kidney function and also reflect on the potential impact on other cardiovascular conditions commonly associated with chronic kidney disease such as heart failure and arrhythmias.
Keywords
The Specific Role of Renal Sympathetic Nerves in Cardiovascular Control
The renal sympathetic nerves consist of efferent sympathetic fibers and afferent sensory fibers. The renal efferent nerves supply all relevant structures including the renal vasculature, the tubules, and the juxtaglomerular apparatus. 1 Accordingly, excitation of renal efferent sympathetic nerves results in (i) urinary sodium and water retention via enhanced tubular sodium reabsorption, (ii) reduction in renal blood flow and glomerular filtration rate (GFR) through neurally mediated vasoconstriction, and (iii) release of renin by stimulation of β1-adrenoceptors on the juxtaglomerular apparatus with concomitant engagement of the renin–angiotensin–aldosterone system. 2 –4
The renal afferent sensory nerves are predominantly located in the renal pelvic wall. 5,6 In contrast to renal efferent nerves, afferent nerves project to the ipsilateral dorsal root ganglia at the level of T6-L2 with the majority of the nerve cell bodies residing at the level of T9-L1. Through integrative processes in the paraventricular nuclei, renal afferent nerve stimulation results in alterations of baroreceptor sensitivity, vagal function and central dopaminergic tone, and an increase in systemic sympathetic nervous activity. 7,8 Increased renal afferent activity is also known to decrease renal efferent activity through the powerful negative feedback control of renorenal reflexes via mechanoreceptor-mediated pathways. 6,9 Furthermore, through a negative feedback loop, increased renal efferent activity increases renal afferent activity. 10
Rationale of Targeting Renal Sympathetic Nerves in Chronic Kidney Disease
Elevated sympathetic nervous system activity has been demonstrated to play a key role in various cardiovascular complications and kidney disease. 11 In chronic kidney disease (CKD), stimulation of renal afferent nerves caused by various mechanisms including ischemia and uremic toxins increases systemic sympathetic outflow via central integrative pathways in the hypothalamus. Sustained sympathetic overactivity per se and the associated rise in blood pressure (BP) are relevant factors contributing to further deterioration of renal function. 12 The kidneys are therefore not only effector organs of sympathetic outflow but also an important modulator of sympathetic nervous system activity. 13
Targeting renal sympathetic nerves in CKD therefore appears as a logical therapeutic option to block the vicious cycle between renal sympathetic nervous hyperactivity and deterioration of kidney function. In fact, attempts were undertaken to modulate renal sympathoexcitation to relieve symptoms of patients with renal failure since the early 1930s. 14 Despite the demonstration of a survival benefit, surgical denervation, most commonly splanchnicectomy, was plagued by a number of complications relating primarily to its nonspecific nature. Meanwhile, studies in many experimental animal models of hypertension have convincingly demonstrated the BP lowering and renoprotective effects of sympathetic inhibition achieved by renal denervation.
Introduction of Catheter-Based Renal Denervation in Human Resistant Hypertension
Surgical sectioning of sympathetic nerves by thoracic and lumbar sympathectomy and splanchnicectomy has been applied successfully to reduce BP and improve the long-term outcome of patients with hypertension. 15,16 Studies in animals have also demonstrated the significant benefit of reduction in sympathetic nervous activity, 17 improvement in natriuresis, 18 and improvement in left ventricular (LV) function. 19 The introduction of catheter-based radiofrequency ablation of renal sympathetic nerves to clinical medicine has been considered as a promising new avenue to device-based therapies for hypertension. Renal sympathetic efferent and afferent nerves located in the adventitia of the renal arteries are the target of this procedure. 5
The Symplicity-HTN trial program was initiated to assess the safety and utility of a catheter-based approach to achieve renal denervation. The initial proof-of-concept study, Symplicity HTN-1, 20 demonstrated the safety and efficacy of catheter-based renal denervation in 45 patients with resistant hypertension. While only minor procedure-related complications were reported, 21 the reduction in office systolic and diastolic BP was substantial (−14/−10, −21/−10, −22/−11, −24/−11, and −27/−17 mm Hg at 1, 3, 6, 9, and 12 months follow–up, respectively, from an average BP of 177/101 mm Hg at baseline. 20 A sustained long-term effect of renal denervation on BP (−32/−14 mm Hg) was recently described in the 36 months follow-up report (Figure 1A). 22 While the office BP fall from baseline did not differ between groups dichotomized by estimated GFR (eGFR > 60 and 45-60 mL/min/1.73 m2), the safety of the procedure could also be confirmed for stage 3b CKD (Figure 1B). The release of norepinephrine (NE) from the renal sympathetic nerves was measured in 10 patients using isotope dilution renal NE spillover methodology. 20 At 30 days after the procedure, the renal NE spillover was decreased by 47%, suggesting a substantial albeit incomplete reduction of renal efferent sympathetic nerve traffic. 20

Thirty-six months of follow-up after renal denervation from Symplicity HTN-1. A, Change in office blood pressure and (B) comparison of change in blood pressure between groups by estimated glomerular filtration rate (eGFR; > 60 and 45-60 mL/min/1.73 m2). Reproduced with permission from Krum et al, Percutaneous renal denervation in patients with treatment-resistant hypertension: final 3-year report of the Symplicity HTN-1 study, Lancet. 2014;383:622-629.
Results from the subsequent Symplicity HTN-2 study, a randomized, controlled clinical trial, were reported in 2010 and 2012. 23,24 Patients with resistant hypertension were randomized to either undergo RDN with continued pharmacological treatment (n = 52) or to continue their established conventional pharmacological treatment alone (n = 54). In line with the results of Symplicity HTN-1, renal denervation reduced office BP by −32 ± 23/12 ± 11 mm Hg at 6-month follow-up (Figure 2), 23 with sustained effects reported at 12-month follow-up (−28 ± 25/10 ± 11 mm Hg). 24 In contrast, no significant change was observed in the control group (1 ± 21/0 ± 10 in office BP and 2 ± 13/7 ± 11 mm Hg in home BP). 23 Mean eGFR was unchanged in both groups at 6 month (0.2 ± 11 mL/min/1.73 m2 in renal denervation group, and 0.9 ± 12 mL/min/1.73 m2 in control group). 23

Blood pressure reduction in Symplicity HTN-2 in the treatment and control groups at 3 and 6 months of follow-up. Reprinted with permission from The Symplicity HTN-2 Investigators, Renal sympathetic denervation in patients with treatment-resistant hypertension (The Symplicity HTN-2 Trial): A randomised controlled trial. Lancet. 1903–1909;376, Copyright (2010).
The results of the latest, largest, and most rigorously designed clinical trial of catheter-based renal denervation, Symplicity HTN-3, have been reported recently. 25 This study was a randomized, blinded, sham-controlled trial. Patients in the control group underwent a renal angiogram and a sham procedure. Stable medication regimens were to be implemented and had to be unchanged for at least 2 weeks prior to enrollment. 24-Hour ambulatory BP monitoring was performed to confirm an average 24-hour systolic BP ≥135mm Hg to exclude white-coat hypertension. Safety and efficacy end points were assessed at 6-month follow-up. During the 6-month follow-up period, the regimen of antihypertensive medication was supposed to be kept stable with medication changes allowed only if deemed clinically necessary. Among 535 uncontrolled hypertensive patients, 364 patients were blindly allocated to treatment group and underwent renal denervation across 88 centers in the United States. At 6 months after the procedure, a significant drop in office systolic BP from baseline had occurred in the treatment group, however, this was not statistically significant when compared to the BP fall observed in the sham procedure group (−14.1 ± 23.9 vs −11.7 ± 25.9 mm Hg, P = .26).
In comparison to Symplicity HTN-1 and HTN-2, the reduction in office BP in the treatment group was less pronounced (−14.1 ± 23.9/−6.6 ± 11.9 in HTN-3 vs −22 ± 10/−11 ± 5 mm Hg in HTN-1 20 and −32 ± 23/−12 ± 11 mm Hg in HTN-2). 23 Furthermore, there was a large BP effect in the sham control group with the drop in office systolic BP being more pronounced than in the non-blinded control group of Symplicity HTN-2 (−11.7 ± 25.3 vs 1 ± 21 mm Hg in HTN-2). 23 Although the pre-treatment BP was similar, the greater range of standard deviation in the treatment group of HTN-3 indicates a wider variation in response. Interestingly, a prespecified subgroup analysis revealed that while no difference in BP changes between RDN and sham control were evident in patients with an African American background, there was a significant difference in non-African Americans, perhaps indicating that racial background may influence the response to the procedure. Of note, patients of African American descent have previously been shown to respond less favorably to treatment with angiotensin-converting enzyme (ACE) inhibitors and beta-blockers. 26
Concerns have also been raised in regard to the operator experience in this U.S. trial. The renal denervation procedures carried out as part of the Symplicity HTN-3 trial were performed by a total of 111 operators throughout the United States. Among them, 31% (34 operators) had only performed 1 procedure, and 85 operators had done less than 5 procedures. 25 Although no significant difference was observed in outcomes between operators who performed <5 procedures and others, this may not eliminate the possible influence of the operators’ learning curve on relatively marginal reduction of BP in the treatment group. From this point of view, ineffective renal denervation might have contributed to the neutral outcome of this study. Furthermore, the absence of tests to assess the degree of renal denervation makes it difficult to investigate this matter further.
The results from Symplicity HTN-3 raised some important issues that need to be resolved in future renal denervation studies. The procedure of catheter-based renal denervation is substantially different from traditional experimental denervation in animals, in which total renal denervation is accomplished by visually stripping and by painting phenol or xylocaine around the adventitia of the renal artery. 18,27,28 In contrast to animal experiments, a reliable test to confirm that renal denervation has been achieved is currently limited to invasive renal NE spillover methodology which is not suitable for wider clinical application. 20,29,30
While evidence for the utility of renal denervation from experimental animal studies is strong, the data from currently available randomized clinical trials are less conclusive and warrant further investigation with a specific focus on improvement in procedural aspects, identification of most suitable patient cohorts, and long-term outcomes.
Renal Denervation and CKD
The results from the aforementioned studies are of major interest for exploration of the therapeutic utility of renal denervation in the context of CKD. In fact, impaired kidney function is a common feature of patients presenting with resistant hypertension. Furthermore, fluid retention is a relevant pathophysiologic component of resistant hypertension and is promoted by increased sympathetic nerve activity, as is increased renin release. Renal denervation therefore appears as a sensible treatment approach in CKD patients with concomitant hypertension. While at this stage no randomized controlled clinical studies are available in this specific cohort, subgroup analyses from larger clinical trials and smaller mechanistic studies have started to explore this further.
While patients in the Symplicity HTN-2 trial had a mean eGFR of 77mL/min/1.73 m2 and patients with CKD and eGFR<45mL/min/1.73 m2 were excluded, 23 this study is important in the current context since it demonstrated that renal function assessed by serum creatinine, eGFR, and cystatin C concentrations was unchanged at 6 months, suggesting that the procedure itself and the associated hemodynamic changes have no adverse effects on the kidneys. Furthermore, longer-term follow-up data of Symplicity HTN-1, in which again no patients with CKD and eGFR < 45 mL/min/1.73 m2 were included, reported that during the first year of follow-up, eGFR remained stable. 21 In patients without newly added spironolactone or other diuretic therapy, eGFR changed by −7.8 mL/min/1.73 m2, for an annualized change of −3.9 mL/min/1.73 m2. In no case did serum creatinine double. Although it was a nonrandomized study, the decline in renal function observed in this 24-month follow-up suggested that there might be an intrinsic beneficial effect of the procedure on the kidney to maintain renal function, which is greater than that achieved via BP reduction alone. In line with this notion is a recent report assessing the influence of renal denervation on renal hemodynamics, renal function, and urinary albumin excretion. 31 In this study, 88 patients with resistant hypertension and normal renal function underwent bilateral renal denervation. Systolic, diastolic, and pulse pressure were reduced by 22.7/26.6 mm Hg, 7.7/9.7 mm Hg, and 15.1/17.5 mm Hg at 3 and 6 months follow-up, respectively. Furthermore, renal resistive index decreased from 0.691 ± 0.01 at baseline to 0.674 ± 0.01 and 0.670 ± 0.01 (P < .05) at 3 and 6 months of follow-up, respectively; the proportion of patients with normal urinary albumin excretion increased by 5% and 12%, whereas proportion of patients with microalbuminuria and macroalbuminuria decreased by 10% and 23%, at 3- and 6-month follow-up without effects on GFR within 6 months.
Whether this approach is safe and effective in patients with an eGFR below 45 mL/min/1.73 m2 remains to be determined in appropriately designed clinical trials. However, preliminary evidence is available from small proof of concept studies (Table 1).
Characteristics of patients with CKD who underwent renal denervation in two published clinical studies.
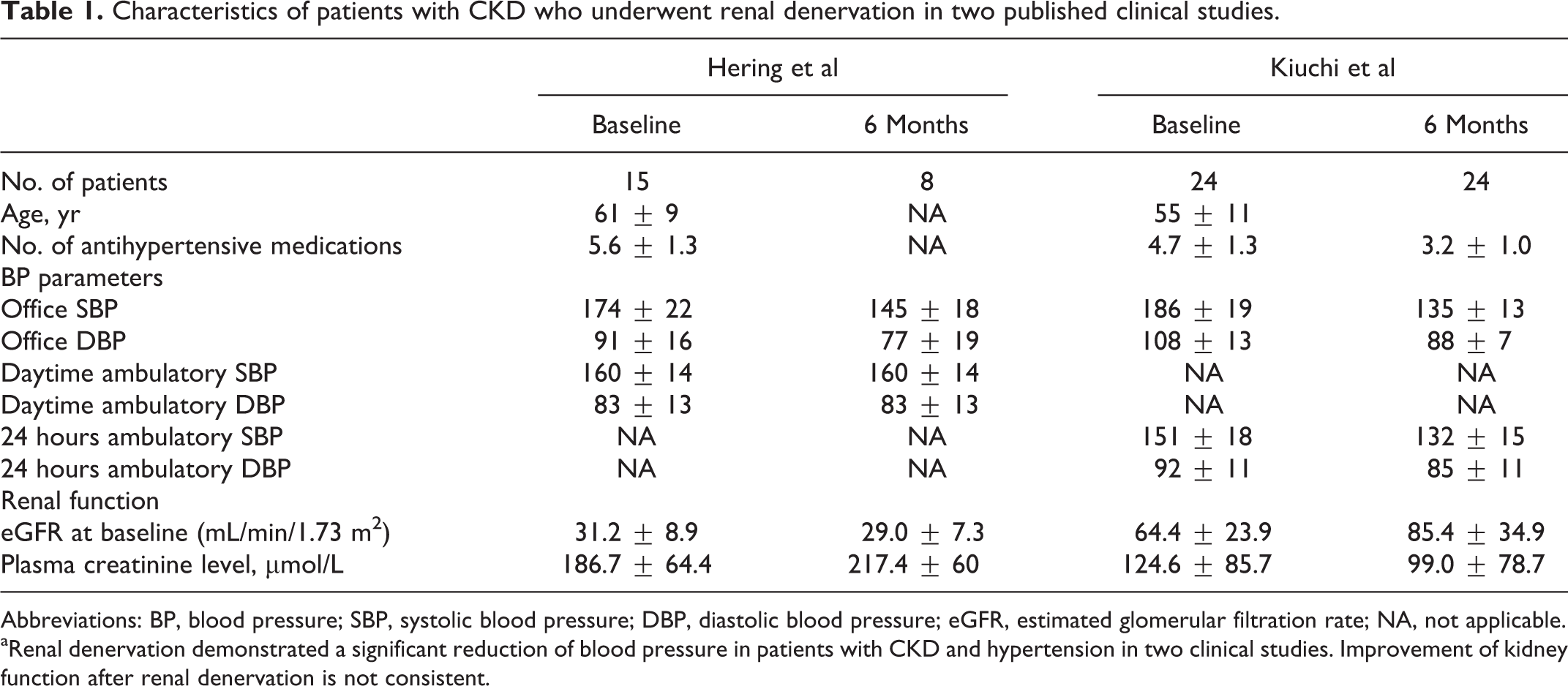
Abbreviations: BP, blood pressure; SBP, systolic blood pressure; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; NA, not applicable.
aRenal denervation demonstrated a significant reduction of blood pressure in patients with CKD and hypertension in two clinical studies. Improvement of kidney function after renal denervation is not consistent.
Hering et al reported the effects of catheter-based renal denervation in patients with resistant hypertension and moderate to severe CKD. 32 (Table 1) An average of 5.0 ± 0.7 ablation treatments per artery were delivered without complications in any of the treated patients. Angiographic evaluation directly after renal denervation did not reveal any compromise of treated arteries. Importantly, eGFR remained stable in this patient cohort with 5 patients being followed up to 12 months. A significant drop in office BP (−34/−14, −25/−11, −32/−15, and −33/−19 mmHg at 1, 3, 6, and 12 months after renal denervation, respectively) was observed without deterioration in renal function and renal blood flow. In contrast to office BP readings, mean 24-hour BP and mean day BP were not significantly reduced after the procedure, possibly related to the limited number of valid ambulatory BP measurements (ABPM) available and substantial intraindividual variability. However, radiofrequency ablation treatment had a considerable impact on nocturnal BP control. In addition, significant reduction in the rate of BP rise, BP power surge, and night-day BP ratios were observed. Renal denervation also diminished mean and maximum night-time BP and restored a physiologic dipping pattern in 9 of 10 patients. The potential clinical relevance of these observations needs to be delineated in future studies.
Kiuchi et al 33 reported similar beneficial effects of catheter-based renal denervation in 24 patients with CKD and refractory hypertension (Table 1). Using an irrigated cardiac ablation catheter, a significant improvement in eGFR (from 64.4 ± 23.9 at baseline to 85.4 ± 34.9 mL/min/1.73 m2 at 6 months of follow-up after denervation) was observed. The reduction in office BP (from 186 ± 19/108 ± 13 at baseline to 135 ± 13/88 ± 7mm Hg at 6 months after denervation) was substantial. A reduction in albuminuria was also reported after catheter-based renal denervation in patients with resistant hypertension. 34
Sympathetic nervous overactivity is pronounced not only in end-stage renal disease (ESRD) but also in early stages of renal disease. 35,36 Urinary albumin excretion, which is a reliable marker of early stage of CKD, correlated positively with elevated plasma NE levels in a cross-sectional study of 495 patients from the general population. 37 Catheter-based renal denervation has been associated with a reduction of albuminuria. 34
Whether these beneficial effects may help to slow the progression of CKD is unknown, however, Ott et al recently provided preliminary evidence from a small uncontrolled study to this effect. A total of 27 patients with CKD stage 3-4 underwent renal denervation for uncontrolled BP, 38 and renal function was evaluated for up to 3 years prior and 1 year after renal denervation. The change in eGFR was calculated by individual regression slopes for each patient before and after renal denervation. Mean baseline BP was 156 ± 12/82 ± 13 mm Hg, despite treatment with 6.2 ± 1.1 antihypertensive drugs. One year after renal denervation, office BP was reduced by 20 ± 20 (P < .001)/8 ± 14 mm Hg (P = .005) and average 24-hour ambulatory BP by 9 ± 14 (P = .009)/4 ± 7 mm Hg (P = .019). Before renal denervation, eGFR declined by −4.8 ± 3.8 ml/min per 1.73 m per year, and after renal denervation eGFR improved by +1.5 ± 10 ml/min per 1.73 m at 12 months (P = .009). These results indicate that in patients with CKD stages 3 and 4, renal denervation does not only decrease BP but appears to slow or even halt the decline of renal function.
A possible mechanism for the potential benefit of renal denervation on renal function may be a a prominent vasodilatation in preglomerular arterioles due to inhibition of renal sympathetic nerve activity. 39 Lohmeier et al could demonstrate that renal denervation increased GFR without a change in fractional sodium reabsorption in dogs with obesity-induced hypertension indicating that renal denervation alters glomerular filtration through the dilation of the renal afferent arteriole. 39
Another possible mechanism might be related to renalase, a protein reported to be secreted by the kidney and relevant for catecholamine metabolism. 40,41 Renalase metabolizes circulating catecholamines, and renalase deficiency may therefore play a role in catecholamone excess. Plasma levels of renalase have been reported to be markedly suppressed in patients with renal failure, and renalase activation induced by catecholamines appears attenuated in renal failure. 42
Interestingly, Jiang et al reported that plasma renalase content and renalase expression in the kidneys were higher in SHR after renal denervation than those in sham operated and other control groups, suggesting that renal denervation induced BP reductions could also be mediated in part by normalized or increased renalase levels. 43
Renal Denervation and End-Stage Renal Disease
While substantial evidence supports the benefits of experimental renal denervation in animal models of ESRD, 12 little is known about the effects of renal denervation in patients with ESRD. The initial proof-of-concept pilot study of catheter-based renal denervation in patients with ESRD on dialysis (average time on dialysis was 3.6 ± 2.6 years) was tested in 12 patients with uncontrolled BP. 44 All patients were on dialysis due to ESRD of various primary renal diseases including nephrosclerosis (n = 4), glomerulopathies (n = 5), immunoglobulin A nephropathy (n = 1), nephrolithiasis (n = 1), and bilateral atrophic kidneys of unknown origin (n = 1). Catheter-based renal denervation was only feasible in 9 patients, whereas the remaining 3 patients were unable to undergo the procedure due to atrophic renal arteries.
The office systolic BP decreased from 166 ± 16.0 to 148 ± 11, 150 ± 14, and 138 ± 17 mm Hg at 3, 6, and 12 months of follow-up, respectively, in patients who had undergone renal denervation (n = 9), whereas in the remaining 3 whose renal artery anatomy was not suitable, BP remained unchanged (176 ± 7 vs 172 ± 6 mm Hg) at 3 months of follow-up. 24-Hour ABPM revealed a significant reduction in systolic BP at 3 months (n = 5). Sympathetic nervous activity was significantly reduced after bilateral renal denervation in 2 patients in whom microneurography and NE spillover measurements were obtained. The average number of antihypertensive medications following renal denervation was reduced from 4.2 ± 1.9 (n = 9) at baseline to 4.0 ± 1.9 (n = 9), 3.7 ± 2.3 (n = 7), and 2.2 ± 1.0 (n = 5) at 3, 6, and, 12 months of follow-up, respectively.
Several case reports have been published confirming possible benefit of catheter-based renal denervation in ESRD. One report describes a patient on hemodialysis treatment who received a kidney transplant around 4 months after his bilateral renal denervation procedure. 44 In the 3 months after RDN, BP was substantially reduced (from 156/95 to 133/81 mm Hg) probably as a results of the demonstrated reduction in renal and total body NE spillover. Consistently, a significant reduction of muscle sympathetic nerve activity (MSNA) was observed and was sustained for 33 months after renal denervation, despite both native kidneys still being in situ but functionally denervated. Another case report of an patient having ESRD with nephrosclerosis due to malignant hypertension demonstrated improved BP control (average office BP from 180 ± 15/105 ± 11 to 155 ± 14/90 ± 10 mm Hg), reduced plasma renin (13.12 to 11.06 ng/mL/h) and ACE activity (22.62 to 14.94 IU/L) at 1 month follow-up. 45 Although these reports have to be interpreted with caution, renal denervation may provide benefits for patients with ESRD. Larger and properly designed clinical trials are now warranted to determine the potential role of this therapeutic approach in ESRD.
Renal Denervation in Heart Failure and Electrophysiological Disorders
Among the various conditions characterized by sympathetic activation, heart failure (HF) plays a crucial role in cardiovascular outcomes and is very common in patients with CKD and ESRD. 46,47 Remarkably, sympathetic hyperactivity is known to occur in the early stage of asymptomatic HF with both preserved and reduced ejection fraction. 48 Given the role of renal denervation in sympathetic hyperactive conditions, renal denervation may possibly improve the outcomes of patients with HF. Surgical renal denervation was shown to improve the LV function of HF induced by myocardial infarction in Wistar rats. 19 Surgical renal denervation also restored natriuresis in response to atrial natriuretic peptide in experimental ischemic HF dogs. 49
Several studies in resistant hypertensive patients have demonstrated that renal denervation reduces LV hypertrophy 50 and LV mass index. 51 The recurrence of atrial fibrillation (AF) has also been suggested to be reduced in hypertensive patients with chronic AF. 52,53
Interestingly, a recent study demonstrated beneficial effects of RDN on atrial electrophysiologic and structural aspects. 54 Renal denervation performed in 14 patients with resistant hypertension not only reduced mean 24-hour BP from 152/84 mm Hg to 141/80 mm Hg at 6-month follow-up (P < .01) but was also associated with increased global conduction velocity (0.98 ± 0.13 m/s to 1.2 ± 0.16 m/s at 6 months, P < .01), shortened conduction time (32 ± 5 ms to 27 ± 6 ms, P < .01), and reduced complex fractionated activity (37% ± 14% to 19% ± 12%, P = .02). The changes in conduction velocity correlated positively with changes in 24-hour mean systolic BP (R(2) = 0.55, P = .01), and there was a significant reduction in LV mass (139 ± 37 to 120 ± 29 g, P < .01) and diffuse ventricular fibrosis (T1 partition coefficient 0.39 ± 0.07 to 0.31 ± 0.09, P = .01) on cardiac magnetic resonance imaging, This study indicates that BP reduction after renal denervation is associated with improvements in regional and global atrial conduction and reductions in ventricular mass and fibrosis. It remains to be determined whether the changes in electrical and structural remodelling are solely due to BP lowering or are due in part to intrinsic effects of renal denervation.
Only a few studies are available that investigated the potential utility of RDN in patients with HF. A first-in-man clinical study of renal denervation in systolic HF, the REACH-Pilot Study, was designed to evaluate the safety of catheter-based renal denervation in patients having HF with reduced ejection fraction. 55 No acute hemodynamic changes occurred that would have interfered with completion of the procedure. In addition, no procedure-related complications were documented. At 6-month follow-up, both symptoms and the 6-minute walk test (by 27.1 ± 9.7 meters) were improved in all patients. Although there was a nonsignificant trend of reduction in both systolic and diastolic BP at 6 months (−7.1 ± 6.9 and −0.6 ± 4.0 mmHg, respectively), no hypotensive episodes were reported.
Ukena et al reported that electrical storm was reduced after bilateral renal denervation in 2 patients with chronic HF. 56 Both patients had treatment-resistant tachyarrhythmias and required a cardioverter defibrillator (implantable cardioverter defibrillator) implantation. The etiology of HF was non-ischemic. Subsequent to renal denervation, both patients had an event-free period up to 5 months. Interestingly, BP was not decreased with renal denervation in either patient.
In line with evidence from animal studies, natriuretic effects of renal denervation might have contributed to the improvement of functional as well as electrophysiological alterations in the failing myocardium in the above clinical studies of HF. However, given the malicious role of sympathetic overactivity, reduced sympathetic nervous activity through renal denervation may well have beneficial effects and deserves further investigation in larger and appropriately designed studies.
Conclusion
As summarized in Figure 3, communication between the kidney and brain involves multiple factors. The etiology of hypertension tends to be multifactorial especially when accompanied by CKD. Several lines of research suggest that renal denervation may exert beneficial effects in the context of CKD. These beneficial effects relate primarily to improved BP control but may also extend to preservation of renal function and reduction of albuminuria. Several comorbidities that commonly exist in patients with CKD such as HF and arrhythmias may also be affected beneficially and strengthen the potential therapeutic utility in CKD. While the experimental evidence to support such notions is convincing, the clinical data available are preliminary and stem mainly from uncontrolled clinical trials and small mechanistic studies and have to be interpreted with the appropriate caution. In light of the results of Symplicity HTN-3 and the questions raised by this study, it will be crucial to perform properly designed randomized controlled studies applying catheter-based renal denervation in an environment with sufficient experience to better understand the potential clinical benefits of renal denervation in CKD.
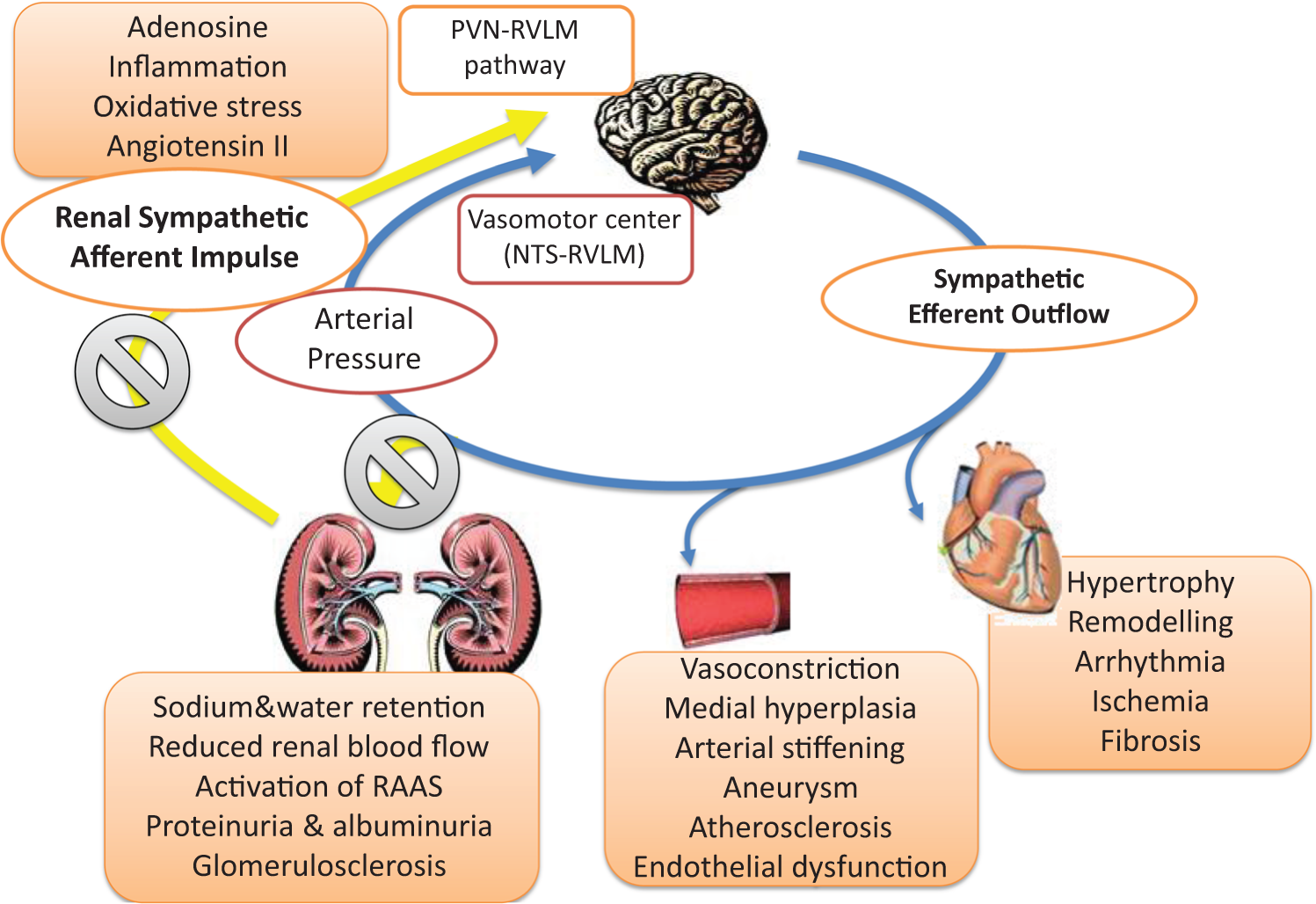
Pathophysiological interactions between the brain and kidney in hypertension resulting in increased total body sympathetic nervous activity as well as blood pressure.
Footnotes
Author Contributions
Markus Schlaich contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Yusuke Sata contributed to design; contributed to acquisition, analysis, and interpretation; and drafted the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Schlaich currently receives research funding from the National Health and Medical Research Council of Australia (NHMRC), Medtronic and Abbott Pharmaceuticals and is supported by an NHMRC Research Fellowships. Professor Schlaich serves on scientific advisory boards for Abbott Pharmaceuticals, Novartis Pharmaceuticals, and Medtronic and has received honoraria and travel support from Abbott, Servier, Novartis, MSD, BI, and Medtronic. Dr Sata has no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
