Abstract
Background:
Thrombolysis is an effective treatment strategy for prosthetic valve thrombosis (PVT). Recombinant tissue-type plasminogen activator (rt-PA) is widely used as a thrombolytic agent. Infusion of rt-PA may trigger the production of anti–tissue plasminogen activator (tPA) antibodies (ATAs). We aimed to evaluate the possible relationship between ATA levels and PVT formation, and the role of baseline ATA levels on outcomes of thrombolytic therapy in patients with PVT.
Methods:
This prospective, single-center cohort study included 28 patients with PVT undergoing thrombolysis and 31 controls with normal prostheses. Plasma samples were collected from patients with PVT at baseline and at 15th, 30th, 90th, and 180th days after thrombolysis and from controls at baseline only. The ATA levels were assessed in human plasma by an enzyme-linked immunosorbent assay.
Results:
Baseline ATA-immunoglobulin (Ig) G and IgM were significantly higher in patients with PVT than in controls. The levels of IgM and IgG peaked at 15th and 30th days after rt-PA infusion, respectively. Subtherapeutic international normalized ratio and baseline ATA-IgM were independent predictors of PVT. Thrombolysis failed in 6 patients (21%) in whom baseline IgM levels were significantly higher than successfully lysed patients. Rethrombosis occurred in 9 patients (32%) in whom baseline IgG levels were significantly higher than those without rethrombosis. There was a moderate positive correlation between baseline and 15th-day IgM levels and the dose of rt-PA needed for successful lysis.
Conclusion:
The ATA levels tended to be higher in patients with PVT at the time of initial diagnosis compared to controls without PVT. In addition, such patients with PVT and high ATA levels may be at high risk for failed thrombolysis or rethrombosis.
Keywords
Introduction
Prosthetic valve thrombosis (PVT) is a serious and life-threatening complication of prosthetic valve replacement. 1 Although surgery is recommended as a first-line treatment for management of PVT in guidelines, 2,3 thrombolytic therapy (TT) has been recently performed successfully in this regard. 4 –13 Due to its high fibrin specificity, recombinant tissue-type plasminogen activator (rt-PA) is widely used in the management of PVT. 7 –13 Commercially available rt-PA is produced in Chinese hamster ovary cells using a complementary DNA derived from messenger RNA of human melanoma cells. 14 Like other recombinant molecules (eg, growth factor, growth hormone, and interferons) that induce an immune response when administered to humans, 15 infusion of rt-PA may trigger the production of anti–tissue plasminogen activator (tPA) antibodies (ATAs) probably because even small differences in the glycosylation sites between recombinant and native proteins can augment the antigenicity. 16 In a recent study, Cugno et al reported increased ATA levels in patients who received rt-PA for the treatment of acute myocardial infarction. In the same study, inhibitory effect on functional activity of rt-PA was established with binding studies in patients with high ATA levels. 17 Furthermore, increased ATA levels have been described in patients with autoimmune diseases including systemic lupus erythematosus, 18,19 antiphospholipid syndrome, 20 –22 systemic sclerosis, 23 inflammatory bowel disease, 24 and have been proposed to be responsible for hypofibrinolytic state causing arterial and venous thrombotic complications in these patients.
The presence of ATAs has not been previously evaluated in patients with PVT. On the basis of previous data, 14 –24 we hypothesize that increased ATA levels may be associated with hypofibrinolytic state causing PVT in patients with prosthetic valves. In this study, we aimed to compare the presence and levels of ATAs between patients with PVT and controls with normally functioning prostheses and also to evaluate the possible relationship between ATA levels and outcomes of TT in patients having PVT treated with rt-PA.
Materials and Methods
Study Population
The study was designed involving 2 centers. Samples were collected from patients who admitted to Kosuyolu Kartal Heart Training and Research Hospital in Istanbul, Turkey, over a 3-year period between 2011 and 2014, and laboratory studies were performed by a team in Department of Internal Medicine, University of Milan, in Italy.
Twenty-eight patients with PVT (10 men and 18 women; mean age: 52 [range 25-78]) who were treated with rt-PA and 31 age- and sex-matched healthy controls (11 men and 20 women; mean age: 53 [range 24-78]) with normally functioning prosthetic valves were enrolled in this prospective study. The first 28 patients undergoing TT in our institution and who did not have the exclusion criteria were enrolled in the PVT group and 31 age- and sex-matched patients with normally functioning prostheses were enrolled as controls. Written informed consent was taken from all patients, and the study protocol was approved by the local ethics committee and respected the guidelines of the Declaration of Helsinki.
All patients underwent transthoracic echocardiography (TTE) and subsequently two-dimensional (2D) and real-time three-dimensional (RT3D) transesophageal echocardiographic (TEE) examination. Inclusion criteria were echocardiographic signs of PVT and eligibility for thrombolytic treatment, as described previously. 8 Age- and sex-matched patients with prosthetic heart valves who had undergone TEE and had been proved to be thrombus free and had no previous history of PVT constituted the control group.
During the echocardiographic examination, spontaneous echo contrast was defined as dynamic smokelike echoes within the cardiac cavities with a characteristic swirling motion that could not be eliminated by changes in gain settings. 25 The thrombus area was measured both from atrial and ventricular sides with 2D TEE examination. The 2D TEE study was performed between 0° and 180° angles, where there is less interference with acoustic shadowing and the largest size of thrombus could be measured by tracing the thrombus from atrial to the ventricular side.
Prosthetic valve thrombosis was classified as obstructive or nonobstructive. The presence of obstruction was defined on the basis of Doppler echocardiographic measurements (peak velocity, mean gradient, effective orifice area, velocity ratio, and acceleration time as appropriate). The cutoff values for these Doppler parameters were defined based on the latest recommendations. 26
Patients with known autoimmune, neoplastic, or inflammatory diseases like systemic lupus erythematosus, anti-phospholipid syndrome, systemic sclerosis, inflammatory bowel disease, primary pulmonary hypertension, and type 1 diabetes mellitus were excluded. Pregnant patients were also excluded.
The study included a heterogeneous population with respect to treatment protocols. Various dosing regimens were used in the patients and total doses of rt-PA ranged from 12.5 to 200 mg. Intravenous infusion of rt-PA (Actilyse; Boehringer Ingelheim GmBH, Ingelheim, Germany) without bolus was administered to all patients with PVT. The TT regimens included low dose slow infusion of rt-PA (up to maximum 6 sessions of 25 mg/6 hours rt-PA infusion without bolus dose in 10 patients) 8,9 and low dose ultraslow infusion of rt-PA (up to a maximum of 8 sessions of 25 mg/25 hours rt-PA infusion without bolus dose in 18 patients) 10 protocols. After successful TT, all patients were treated with oral anticoagulant therapy (warfarin) with an international normalized ratio (INR) target between 2.5 and 4. All patients with PVT were followed by TTE every month and by TEE every 6 months for the assessment of potential rethrombosis.
In patients with obstructive PVT, in the absence of fatal or nonfatal major complications as described previously, 8 Doppler documentation of the resolution of increased gradient and decreased valve area, a reduction by ≥75% in major diameter or area of the thrombus, and clinical improvement in symptoms were considered the major criteria for TT success. For patients with nonobstructive PVT, the only criteria was the reduction of thrombus burden by ≥50%. 8 –10 Rethrombosis was defined as readmission of the same patient at a different time with PVT after a successful TT episode.
Sample Collection and Storage
In order to detect ATA levels, plasma samples were collected from all patients with PVT before rt-PA treatment and at 15th, 30th, 90th, and 180th days after the last TT session. Plasma samples were collected from controls only at the beginning of the study.
Blood samples were collected by a clean puncture of an antecubital vein using sodium citrate 3.8% as anticoagulant and then centrifuged at 2500×g for 20 minutes at room temperature. The plasma samples, obtained after centrifugation, were frozen in small aliquots and stored at −80°C until testing.
Anti-tPA Antibody Assay
Plasma levels of anti-t-PA antibodies were measured using an enzyme-linked immunosorbent assay as previously described. 14,19 Briefly, rt-PA was coated overnight onto microtitration plates and, after washing, the residual binding sites were blocked with bovine serum albumin. After further washes, a 1:20 dilution of the plasma samples was added and incubated for 45 minutes at room temperature. After washing, the rt-PA-bound immunoglobulins (Igs) were identified by class-specific mouse monoclonal antibodies, which were detected by means of peroxidase-conjugated antimouse Ig antibodies and revealed by orthophenylenediamine. The results were expressed as units per milliliter (U/mL), referred to an internal standard (plasma collected from a patient with a high anti-t-PA antibody titer) arbitrarily fixed at 100 U/mL. The standard curve was linear until 0.78 U/mL (r = .999), and the same linearity was obtained in the plasma samples from patients and controls. The within- and between-assay coefficients of variation were less than 15%.
Statistical Analysis
Statistical analyses were performed using SPSS for Windows version 19.0 (SPSS Inc, Chicago, Illinois). The variables were investigated with the use of analytical methods (Kolmogorov-Smirnov/Shapiro-Wilk test) to determine whether they were approximately normally distributed. Descriptive statistics were reported as mean, standard deviation, median, minimum and maximum values for continuous variables, and as frequency with percentages for the categorical variables. Group comparisons for continuous variables were tested using the Mann-Whitney U test or Student t test as appropriate. Comparisons of categorical variables were evaluated by the chi-square and Fisher exact test. Friedman test was used to compare the distributions of 3 or more dependent variables. The Bonferroni correction was applied to significance level of pairwise differences for multiple comparisons. The possible predictors identified by univariate analysis were further entered into multiple logistic regression analyses in order to determine independent predictors. Pearson or Spearman correlation analysis was used for assessing correlation between ATA levels and rt-PA dose. The cutoff values were estimated by receiver–operating characteristic (ROC) curve analysis. Significance level was accepted as P < .05 in all statistical analyses.
Results
The study population included 28 patients with PVT (10 men; mean age: 52.1 [range 25-78]) and 31 controls (11 men; mean age: 53.4 [range 24-78]) with normally functioning prosthetic valves. There were no significant differences between the groups in terms of demographic features except anticoagulation status, NewYork Heart Association (NYHA) functional class and presence of spontaneous echo contrast. The demographic features of the study population is presented in Table 1.
Comparison of Study Patients in Terms of Demographic Features.a

Abbreviations: INR, international normalized ratio; LA, left atrium; NYHA, NewYork Heart Association; NOT, nonobstructive thrombus; OT, obstructive thrombus; SEC, spontaneous echo contrast; tPA, tissue-type plasminogen activator; TT, thrombolytic therapy; PVT, prosthetic valve thrombosis; Ig, immunoglobulin.
aBaseline IgG and IgM values are presented as mean and range enclosed in parentheses.
The patients with PVT and controls were compared in terms of baseline IgG and IgM levels. The median baseline ATA-IgG and IgM were significantly higher in patients with PVT than in controls (5.09 U/mL [0.97-94.46] vs 2.65 U/mL [0.10-14.80], respectively, P = .005 for IgG and 28.74 U/mL [1.69-120.50] vs 13.04 U/mL [0.55-70.0], respectively, P = .001 for IgM; Figure 1).

The comparison of baseline ATA-IgG (A) and IgM (B) levels between patients with prosthetic valve thrombosis (PVT) and controls is represented as box plot graphs. Baseline median ATA-IgG (A) and IgM (B) were significantly higher in patients with PVT than in controls. ATA indicates anti–tissue plasminogen activator antibodies; Ig, immunoglobulin.
There were significant differences between patients with PVT and controls without PVT in terms of anticoagulation status, NYHA functional class, presence of spontaneous echo contrast, baseline IgG, and IgM levels. All potential predictors of PVT in the data set were entered into multiple logistic regression analyses. By multivariate analysis, the independent predictors of PVT were subtherapeutic anticoagulation status and high baseline IgM levels (odds ratio [OR]:6.95, 95% confidence interval CI (95%): 1.11-43.92, P = .037; OR:1.08 95% CI: 1.01-1.15, P = .017, respectively; Table 2).
The Results for Multivariate Logistic Regression Analyses of Potential Predictors of Prosthetic Valve Thrombosis.

Abbreviations: AF, atrial fibrillation; CI, confidence interval; INR, international normalized ratio; LA, left atrium; LVEF, left ventricular ejection fraction; NYHA, NewYork Heart Association; SEC, spontaneous echo contrast; Ig, immunoglobulin.
The ATA-IgG and IgM levels obtained from plasma samples of PVT group at baseline and 15th, 30th, 90th, and 180th days were summarized in a cumulative linear graphic. The IgM and IgG levels peaked at 15th and 30th days after rt-PA infusion, respectively (Figure 2). A significant difference was detected between serial IgM levels by Friedman test (P < .001). Post hoc analysis with Bonferroni correction revealed a significant difference between baseline and 15th day IgM levels (P = .001). Similarly, a significant difference was detected between serial IgG levels by Friedman test (P < .015) and post hoc analysis with Bonferroni correction revealed a significant difference between baseline and 30th day IgG levels and 30th and 180th days IgG levels (P = .002; P = .002, respectively).

The ATA-IgG and IgM levels obtained from plasma samples of PVT group at baseline and 15th, 30th, 90th, and 180th days were summarized in a cumulative linear graphic. Each line represents the corresponding immunoglobulin values of each patient in the PVT group. IgM (A) and IgG (B) levels peaked at 15 and 30 days after rt-PA infusion, respectively. p indicates patient. ATA indicates anti–tissue plasminogen activator antibodies; Ig, immunoglobulin; PVT, prosthetic valve thrombosis.
Thrombolytic therapy failed in 6 (21%) patients and rethrombosis occurred in 9 (32%) patients with PVT. Baseline IgM levels were significantly higher in failed TT group compared to successfully lyzed patients (50.46 ± 34.58 U/mL vs 24.28 ± 14.59 U/mL, respectively, P = .023; Table 3). Baseline IgG levels were significantly higher in rethrombosis group compared to remaining patients (12.43 U/mL [2.20-94.46] versus 2.97 U/mL [0.97–46.67] respectively, P = .019; Figure. 3). For rethrombosis, ROC curve analysis provided an area under the curve (AUC) of 0.78, with a sensitivity of 90% and specificity of 60% for baseline IgG levels >3.7 U/mL (P = .02). For TT failure, an AUC of 0.81 was obtained with a sensitivity of 83% and a specificity of 75% for baseline IgM levels >34.2 U/mL (P = .02; Figure. 4).
Comparison of Baseline Characteristics Between Patients With Successful and Failed Thrombolytic Therapy.

Abbreviations: LA, left atrium; NYHA, NewYork Heart Association; NOT, nonobstructive thrombus; OT, obstructive thrombus; SEC, spontaneous echo contrast; TT, thrombolytic therapy; Ig, immunoglobulin.

Box plot graph summarizing the comparison of baseline IgG levels between the patients who developed rethrombosis and the remaining patients. Ig indicates immunoglobulin.
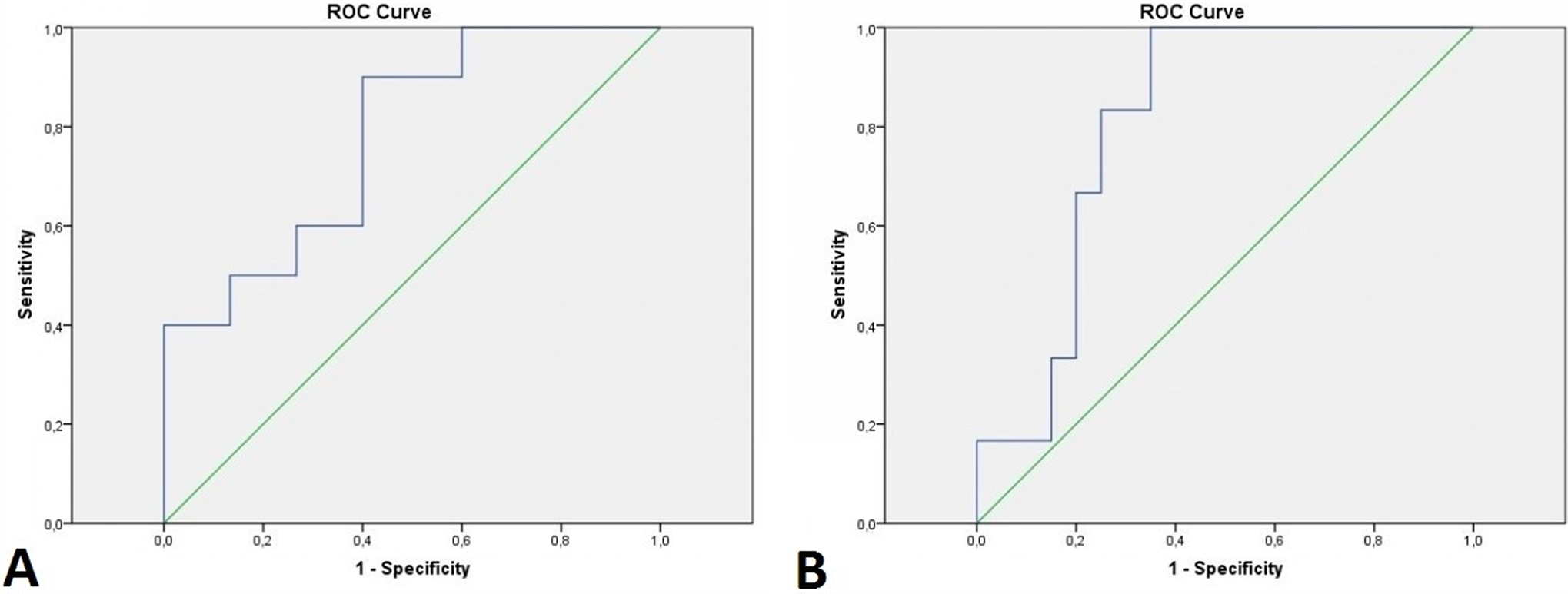
For rethrombosis (A), receiver–operating characteristic (ROC) curve analysis provided an area under the curve (AUC) of 0.78, with a sensitivity of 90% and a specificity of 60% for baseline IgG levels >3.7 U/mL (P = .02), and for thrombolytic therapy (TT) failure (B), an AUC of 0.81 was obtained with a sensitivity of 83% and a specificity of 75% for baseline IgM levels >34.2 U/mL (P = .02).
There were significant differences between successfully lyzed patients and the remaining patients in terms of thrombus area, NYHA functional class, and baseline IgM (P = .041, P = .008, and P = .024, respectively) and a close to significant difference for thrombus type (P = .056; Table 3). Univariate predictors for failed TT were entered into multiple logistic regression analyses. By multivariate analysis, the only independent predictor of failed TT was high NYHA functional class (OR: 11.33, 95% CI: 1.39-92.05, P = .023).
There was no previous rt-PA exposure among patients with PVT except 1 patient in whom baseline IgM and IgG levels were within normal ranges. Nine patients had rethrombosis, and the mean time till the development of rethrombosis was 14.2 ± 7.7 months. Four of them had ceased follow-up examinations after 6 months and were admitted with rethrombosis soon after with a mean INR level of 1.6 ± 0.4. On the other hand, the remaining 5 patients were under strict anticoagulation. Furthermore, factor V Leiden mutation and anticardiolipin antibodies were absent, and homocysteine levels were within normal limits in these patients.
The mean dose of rt-PA used for TT was 90.1 ± 70.9 mg, and mean duration of treatment was 4.5 ± 3.68 days. There was a moderate positive correlation between the baseline and 15th-day IgM levels and the dose of rt-PA (r = .466, P = .038 and r = .547, P = .013, respectively) used in successful TT group (Figure 5). There was no significant association between ATA levels and the type of TT protocol or duration of rt-PA infusion.

Scatter graphs summarizing moderate positive correlations between the baseline (A) and 15th day (B) IgM levels and the dose of rt-PA used in successful thrombolytic therapy group. Ig indicates immunoglobulin; rt-PA, recombinant tissue-type plasminogen activator.
Among patients with PVT, 2 major (peripheric embolism and intraabdominal hemorrhage requiring transfusion) and 1 minor (transient loss of vision) complications were observed during TT. There was no significant difference in terms of ATA levels between patients with complicated and noncomplicated PVT.
Discussion
This prospective, pilot single-center cohort study showed that patients with PVT have increased baseline ATA levels. Furthermore, the level of these antibodies may increase during TT with rt-PA infusion and may interfere with the success of TT, necessitating a higher dose of rt-PA for complete success. Additionally, patients with high baseline ATA levels may have higher risk for rethrombosis.
Hemostasis and fibrinolytic system have opposite effects and work in a natural balance. An increase or decrease in one’s activity may cause a prothrombotic state or serious bleeding. Fibrinolytic system plays an important role in the lysis of formed clot in the circulatory system. Tissue plasminogen activator and urokinase type plasminogen activator are activators of the fibrinolytic system which is inhibited by α-2 plasmin inhibitor and plasminogen activator inhibitor 1 (PAI-1). 27 A decrease in fibrinolytic capacity may result in prothrombotic state. Decreased tPA levels and increased PAI-1 levels were reported in deep venous thrombosis 28,29 and acute myocardial infarction. 30,31 Furthermore, antibodies formed against the tPA molecule may cause a prothrombotic state by decreasing tPA activity. These antibodies may bind the active domains of the tPA molecule preventing the conversion of plasminogen to plasmin as previously demonstrated in 2 patients with the antiphospholipid syndrome and ATA. 20 The presence and the role of such antibodies in patients with PVT have been reported for the first time in this current study.
One of the serious and life-threatening complications of prosthetic valve replacement is PVT. 1 Although surgery is commonly recommended as a first-line treatment strategy for left-sided PVT, 2,3 TT has been recently performed successfully and with low complication and mortality rates. 7 –13 It seems that low dose and slow infusion of tPA may decrease complication and mortality rates without compromising the success rates. 8 –10 On the other hand, longer exposure to antigens may result in increased antibody formation. 32 However, in this study there was no significant relation between ATA levels and the duration of rt-PA infusion.
Endogenous tPA is a serine protease, which is secreted by endothelial cells into the circulation. Autoantibodies to endogenous tPA molecule may be formed without any exposure to exogenous rt-PA and may interfere with endogenous tPA activity. In this study, higher baseline ATA-IgM and IgG levels were detected in the PVT group than in the controls. Among the patients with PVT, only 1 had a history of previous rt-PA exposure and interestingly his ATA levels were within normal range. So his previous exposure did not affect the study results. We hypothesize that autoantibodies to endogenous tPA molecule may be responsible for the hypofibrinolytic state in patients with PVT.
The issue where the ATA come from a baseline in patients with PVT is a matter of concern in which the potential explanations could be speculative. We have previously reported the endothelial dysfunction (ED) in patients with PVT compared with a matched control group and found that patients with PVT had ED which might lead to development of thrombus formation. 33 Although inadequate anticoagulation is the main cause of PVT, heart valve surgery and surgical techniques are inevitable contributing factors due to endocardial fibrosis and the exposure of foreign artificial surfaces such as prosthetic and suture materials to blood stream which promote on the shear rate of the blood flow and may result in unpredictable immune reactions. 34 However, this single observation which might have a range of potential explanations does not allow the conclusion that ED has a unique role in the pathogenesis of PVT that may trigger immune response to foreign elements is deeply speculative and the nature of this study does not address whether ED is cause or effect.
Furthermore, recently in a large-scale cohort, we have reported that neutrophil/lymphocyte ratio, platelet/lymphocyte ratio, and C-reactive protein levels were significantly increased in patients with PVT. 35 Although the underlying mechanism is unclear, this inflammatory process may play a role in the development of ATA in patients with PVT.
As widely known, IgM-type antibodies appear as acute phase antibodies, while IgG type are chronic phase antibodies. In this study, ATA levels were compared at 15th, 30th, 90th, and 180th days after the last rt-PA exposure, and the peak of IgM levels was observed at 15th day, whereas the peak level of IgG was observed at 30th day, consistent with the nature of the antibody types. Cugno et al reported similar results in patients with acute myocardial infarction treated with rt-PA infusion. 17
Rethrombosis was observed after 14.2 ± 7.7 months in 9 of 28 patients with PVT. Baseline IgG levels were significantly higher in these patients. The IgG levels at 180th day were also higher in these patients, but it was not statistically significant. These results suggest that increased baseline IgG levels may be associated not only with PVT but also with rethrombosis. On the other hand, TT failed in 6 patients. In these patients, TT was continued up to 200 mg of rt-PA. Among these patients, 2 were successfully treated with streptokinase, 2 underwent surgery, and the remaining 2 patients were anticoagulated strictly. Baseline IgM levels were significantly higher in these 6 patients compared to the remaining patients. When these patients were excluded, a moderate positive correlation was detected between the baseline and 15th-day IgM levels and the dose of rt-PA used in successful TT group. These results support that IgM antibodies in the acute phase may interfere with rt-PA activity and thus increase the dose needed for successful TT and decrease the success rates.
Due to lack of data about antigenicity of rt-PA, clinicians can hesitate to use antigenic molecules such as streptokinase. However, antibodies against tPA molecule have been recently described in several prothrombotic disorders. On the basis of the study findings, we can conclude that increased ATA levels may be one of the esoteric reasons of PVT and may be related to failed TT and rethrombosis. If the current data are confirmed in further studies, it may be argued that patients with increased levels of ATA could be considered as “resistant” to TT, could require higher doses, or could not be considered as candidate for TT. Presence of high ATA levels in patients with PVT before TT may necessitate treatment with another thrombolytic agent such as streptokinase or redo valve surgery may be considered instead TT with rt-PA infusion.
Study Limitations
The primary limitation was that our study is a nonrandomized, observational study that enrolled relatively small number of patients not assessing very large cohort for risk of PVT. A long-term prospective study where patients are enrolled prior to the PVT event would have been ideal.
The exclusion criteria were based on patient’s history or clinical findings. Antinuclear antibodies, antidouble-strand DNA antibodies, anticardiolipin IgG/IgM, and antiphospholipid antibodies were not available. We did not assess coagulation parameters that could influence the prothrombotic status. While we included only a small number of patients, the power analysis should be interpreted with caution. Further studies are needed to confirm these postulates.
Conclusion
The ATA levels tended to be higher in patients with PVT at the time of initial diagnosis compared to controls without PVT. Patients with abnormally high levels of ATA may have an increased risk for failure of TT with rt-PA and development of rethrombosis. Moreover, the infusion of rt-PA may trigger the production of specific antibodies that bind to rt-PA; further studies are needed to see if these antibodies participate in inducing a hypofibrinolytic state.
Footnotes
Author Contributions
Özkan contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Kalçık contributed to conception and design, contributed to acquisition and analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Gürsoy contributed to conception and design, contributed to interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Öcal contributed to conception, contributed to acquisition, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Griffini contributed to conception, contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Karakoyun contributed to conception, contributed to interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Yesin contributed to conception, contributed to acquisition and analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Gündüz contributed to design, contributed to acquisition and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Astarcıoğlu contributed to conception, contributed to analysis, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Cerşit contributed to conception, contributed to interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Aykan contributed to conception and design, contributed to acquisition, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Cugno contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
