Abstract
The nitric oxide synthase (NOS)-independent pathway of nitric oxide (NO) production in which nitrite (NO2−) is reduced to NO may have therapeutic applications for those with cardiovascular diseases in which the NOS pathway is downregulated. We tested the hypothesis that NO2− infusion would reduce mean arterial pressure (MAP) and increase skeletal muscle blood flow (BF) and vascular conductance (VC) during exercise in the face of NOS blockade via L-NAME. Following infusion of L-NAME (10 mg kg−1, L-NAME), male Sprague-Dawley rats (3-6 months, n = 8) exercised without NG-nitro-L arginine methyl ester (L-NAME) and after infusion of sodium NO2− (7 mg kg−1; L-NAME + NO2−). MAP and hindlimb skeletal muscle BF (radiolabeled microsphere infusions) were measured during submaximal treadmill running (20 m min−1, 5% grade). Across group comparisons were made with a published control data set (n = 11). Relative to L-NAME, NO2− infusion significantly reduced MAP (P < 0.03). The lower MAP in L-NAME+NO2− was not different from healthy control animals (control: 137 ± 3 L-NAME: 157 ± 7, L-NAME + NO2−: 136 ± 5 mm Hg). Also, NO2− infusion significantly increased VC when compared to L-NAME (P < 0.03), ultimately negating any significant differences from control animals (control: 0.78 ± 0.05, L-NAME: 0.57 ± 0.03, L-NAME + NO2−; 0.69 ± 0.04 mL min−1 100 g−1 mm Hg−1) with no apparent fiber-type preferential effect. Overall, hindlimb BF was decreased significantly by L-NAME; however, in L-NAME + NO2−, BF improved to a level not significantly different from healthy controls (control: 108 ± 8, L-NAME: 88 ± 3, L-NAME + NO2−: 94 ± 6 mL min−1 100 g−1, P = 0.38 L-NAME vs L-NAME + NO2−). Individuals with diseases that impair NOS activity, and thus vascular function, may benefit from a NO2−-based therapy in which NO bioavailability is elevated in an NOS-independent manner.
Introduction
The cardiovascular response to exercise is characterized by a multitude of neural, humoral, and mechanical components serving to elevate cardiac output and redistribute blood flow (BF), and thus O2 delivery (
NO is synthesized endogenously in a reaction catalyzed by the NO synthase (NOS) family of enzymes or the 1-step reduction of nitrite (NO2−) to NO, the latter being a NOS-independent pathway (reviewed by Lundberg and Weitzberg 3 ). Recent evidence from murine models suggests that the availability of NO2− may be upregulated via ingestion of nitrate (NO3−)-rich food stuffs (ie, beetroot juice), thus elevating NO bioavailability (following the reduction of NO3− to NO2− and finally NO) resulting in improved skeletal muscle vascular, metabolic, 4 –6 and contractile 7 function. These results extend to humans as several laboratories have demonstrated ergogenic effects of dietary NO3− supplementation in healthy 8 –13 and diseased 14 –17 populations. Interestingly, while these studies employ a dietary means of increasing endogenous [NO2−], vasoactivity of the directly infused anion is evident in humans 18 –21 and animals, 22 –25 suggesting that bolus delivery may afford an expedited method of augmenting vascular and metabolic control in vivo.
Bearing in mind the beneficial impacts of dietary NO3− supplementation on exercise performance, and the vascular effects of NO2− infusion highlighted earlier, it is logical to consider that direct infusion with NO2− may also impact skeletal muscle vascular control during exercise. Furthermore, when considering that NO2− reduction to NO is potentiated in low PO2 and/or pH environments, 18 bioactivity of NO2− may be further facilitated (or relied upon) when NOS function is reduced or completely abolished and O2 transport is impaired (as is the case in many pathological conditions). If direct NO2− infusion augments exercising skeletal muscle vascular function independent of NOS, NO2− therapy could emerge as an attractive means of restoring NO bioavailability in various cardiovascular diseases in which NOS function is compromised.
Despite these prospects, there are no investigations into the effects of NO2− infusion on exercising skeletal muscle vascular control under conditions of NOS blockade. Therefore, the purpose of this investigation was to determine the impacts of NO2− infusion on skeletal muscle vascular control during exercise in rats with NOS blockade elicited via L-NAME. We tested the hypothesis that, relative to the L-NAME condition, treatment with NO2− would restore exercising mean arterial pressure (MAP) and total exercising hindlimb skeletal muscle BF and vascular conductance (VC) to values observed in healthy young adult rats (with intact NOS function).
Methods
Ethical Approval
All procedures employed in this investigation were approved by the Institutional Animal Care and Use Committee of Kansas State University and were conducted under the guidelines established by The Journal of Physiology. 26 A total of 16 young adult male Sprague-Dawley rats (~3 months of age, Charles River Laboratories, Wilmington, Massachusetts) were maintained at accredited animal facilities at Kansas State University on a 12:12-hour light–dark cycle with food and water provided ad libitum. All rats were familiarized with running on a custom-built motor-driven treadmill for 5 min day−1 at a speed of 20 m min−1 up a 5% grade for ~5 days. In an effort to minimize the unnecessary use of additional animals, control BF, VC, blood gas, [lactate], and plasma [NO2−]/[NO3−] values reported herein represent animals from recently published work (n = 11, 27 ) and followed the same experimental procedures as detailed subsequently.
Surgical Instrumentation
On the day of the experiment, rats were anesthetized initially with a 5% isoflurane-O2 mixture and maintained subsequently on 3% isoflurane/O2 mixture. A catheter (PE-10 connected to PE-50, Intra-Medic polyethylene tubing, Clay Adams Brand, Becton, Dickinson and Company, Sparks, Maryland) was placed in the ascending aorta via the right carotid artery. A second catheter was surgically placed in the caudal (tail) artery as described previously. 28 Both catheters were tunneled subcutaneously through the dorsal aspect of the cervical region and exteriorized via a puncture wound in the skin. The incisions were closed, anesthesia was terminated, and the rats were given a minimum of 60 minutes to recover. 29
L-NAME Infusion
Rats were then placed on the treadmill and, following a ~5-minute resting period, NG-nitro-L arginine methyl ester (10 mg kg−1, L-NAME; n = 8, Sigma Chemical, St Louis, Missouri) was administered to each rat via the caudal artery catheter to inhibit NOS. This dose has been used extensively in our laboratory and has demonstrated inhibition of NOS via attenuation of acetylcholine-induced reductions in MAP. 30,31
Exercise Protocol and Measurement of Hindlimb Skeletal Muscle BF
Following L-NAME infusion, the caudal artery catheter was connected to a 1-mL syringe chambered in a Harvard infusion/withdrawal pump (model 907, Cambridge, Massachusetts) and the carotid artery catheter was connected to a pressure transducer (Gould Statham P23ID, Valley View, OH, USA) maintained at the same height as the animal. Approximately, 3 minutes post-L-NAME infusion, exercise was initiated and treadmill speed was increased progressively over a ~30-second period to a speed of 20 m min−1 (5% grade, ~60%
NO2− Infusion
Following a 30-minute recovery period, a bolus infusion of sodium NO2− (7 mg kg− 1 body mass, L-NAME + NO2−; n = 8, Sigma Chemical, St Louis, Missouri) was administered to each rat via the caudal artery catheter. The exercise and microsphere infusion protocols (radio-labeled differently from the first) were then repeated (condition L-NAME + NO2−).
Blood Sampling and Measurement of Plasma [NO3−] and [NO2−]
Immediately following microsphere infusion but prior to the termination of exercise, a ~0.3-mL blood sample was drawn from the carotid artery catheter for determination of blood pH, PO2, and % O2 saturation (Nova Stat Profile M, Nova Biomedical,). For plasma [NO3−] and [NO2−], following the termination of exercise ~0.8 mL of blood was drawn into heparinized tubes and rapidly centrifuged at 5000g at 4°C for 6 minutes. Plasma was then extracted and frozen immediately at −80°C for later analysis via chemiluminescence as described previously. 4,5,27,33
Determination of BF and VC
Rats were euthanized via pentobarbital sodium overdose (≥50 mg kg−1). The thorax of each rat was opened and accurate placement of the carotid artery catheter was confirmed before the internal organs and 28 individual muscles and muscle parts of the hindlimb were excised.
Radioactivity of each tissue was determined with a gamma scintillation counter (Packard Auto Gamma Spectrometer, model 5230, Downers Grove, Illinois). Tissue BF was then calculated using the reference sample method 28 and expressed as mL min−1 100g−1. VC was then calculated by normalizing BF to MAP and expressed as mL· min−1 100g−1 mm Hg−1.
Statistical Analysis
Results were compared among (control vs L-NAME and control vs L-NAME + NO2−) and within (L-NAME vs L-NAME + NO2−) groups using a priori unpaired and paired 1-tail Student t tests, respectively, corrected for multiple comparisons. Values are expressed as mean ± standard error of the mean.
Results
Mean Arterial Pressure, HR, Plasma [NO3−] and [NO2−], and Blood Gases
Relative to control, post NO2− infusion plasma [NO2−] (control: 0.17 ± 0.2, L-NAME + NO2−: 306.8 ± 38.7 µmol/L, P < 0.01) and [NO3−] (control: 17.8 ± 1, L-NAME + NO2−: 152.5 ± 35 µmol/L, P < 0.01) were significantly elevated. Relative to control, MAP was significantly higher in the L-NAME condition (Figure 1, P < 0.03). Following NO2− infusion, MAP was reduced significantly when compared to the L-NAME condition (P < 0.03). Exercising MAP was not different between control and L-NAME + NO2− groups (P = 0.36). Relative to the control and L-NAME + NO2− conditions, exercising HR was significantly lower in the L-NAME condition (control: 528 ± 12, L-NAME: 493 ± 37, L-NAME + NO2−: 520 ± 33 beats min−1, P < 0.01).

Exercising mean arterial pressure (MAP), systolic blood pressure (SBP), diastolic blood pressure (DBP), and pulse pressure (PP) values for control, L-NAME, and L-NAME + NO2− conditions. *P < 0.03 vs control, #P < 0.03 vs L-NAME. Note: control values represented are from previously published data.
There were no differences in arterial PO2, PCO2, or % O2 saturation during exercise. Arterial blood [lactate] during exercise was greater following NO2− infusion (3.8 ± 0.5 mmol/L) compared to control (2.7 ± 0.4 mmol/L) and L-NAME only (2.1 ± 0.3 mmol/L) conditions, (P < 0.016).
BF and VC
L-NAME significantly reduced exercising total hindlimb skeletal muscle BF and VC (Figure 2, P < 0.03). Following NO2− infusion, total hindlimb skeletal muscle VC was restored to levels observed in control rats (Figure 2, P < 0.03 L-NAME vs L-NAME+NO2−, P > 0.10 control vs L-NAME + NO2). There were no differences in total hindlimb skeletal muscle BF during exercise in L-NAME vs L-NAME + NO2− or control versus L-NAME + NO2− conditions (Figure 2 bottom panel, P > 0.03).
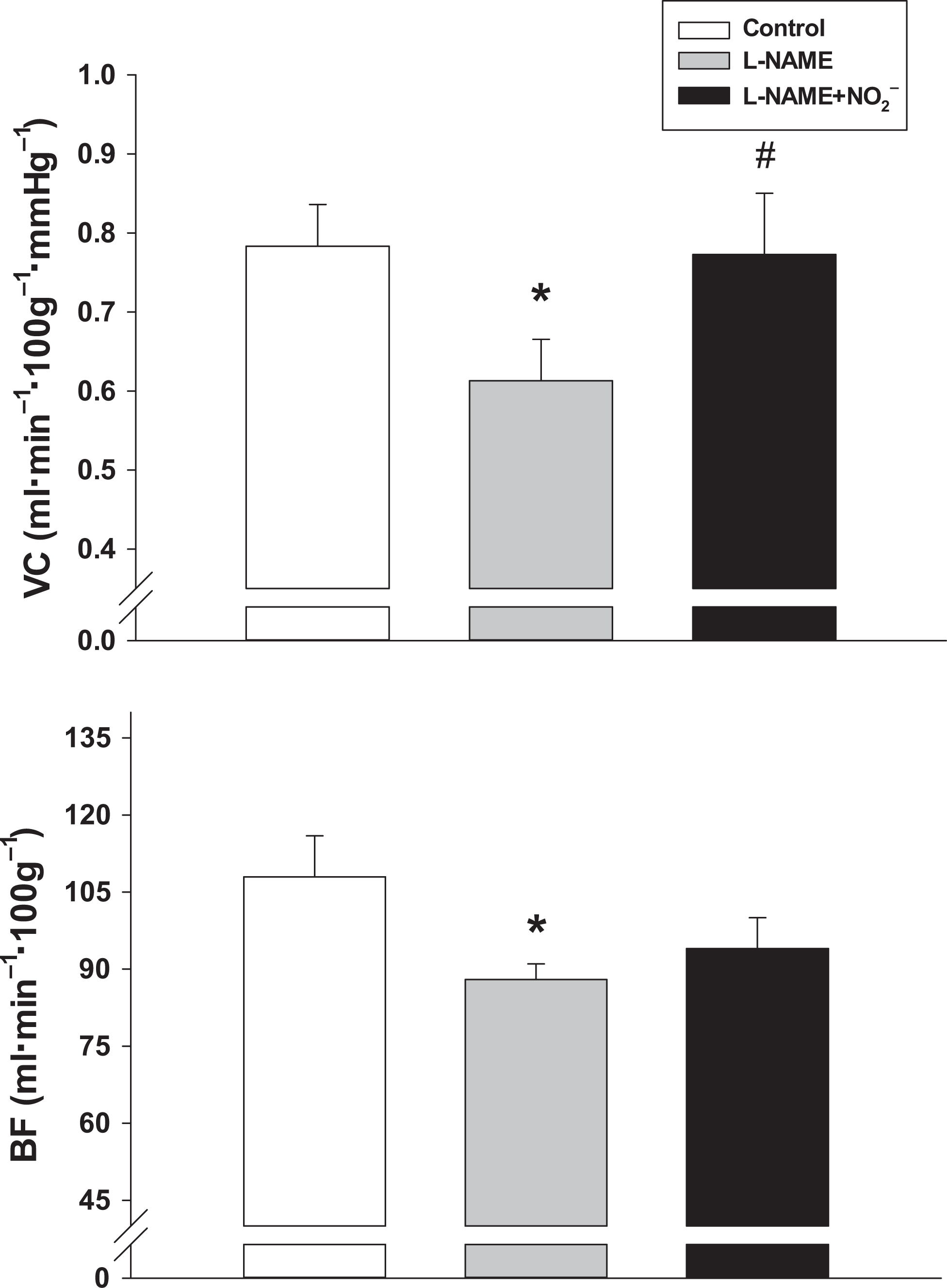
Total hindlimb skeletal muscle blood flow (BF) and vascular conductance (VC) for control, L-NAME, and L-NAME + NO2− conditions in rats during submaximal locomotory exercise. *P < 0.03 vs control, #P < 0.03 vs L-NAME. Note: control values represented are from previously published data.
Relative to control, L-NAME-treated rats had lower BF in 5 and VC in 15 of the 28 individual hindlimb muscles and muscle parts, whereas this was the case for only 3 muscles (BF and VC) in the L-NAME+NO2− condition (Table 1, P < 0.03 for all). Moreover, following NO2− infusion, VC in 19 of the 28 individual hindlimb muscles and muscle parts was increased significantly when compared to the L-NAME condition (P < 0.03, Table 1).
Effects NO2− Infusion (7 mg kg−1) on Exercising Hindlimb Skeletal Muscle BF (mL min−1 100g−1) and VC (mL min−1 100g−1 mm Hg−1) in Rats With NOS Blockade (L-NAME).a
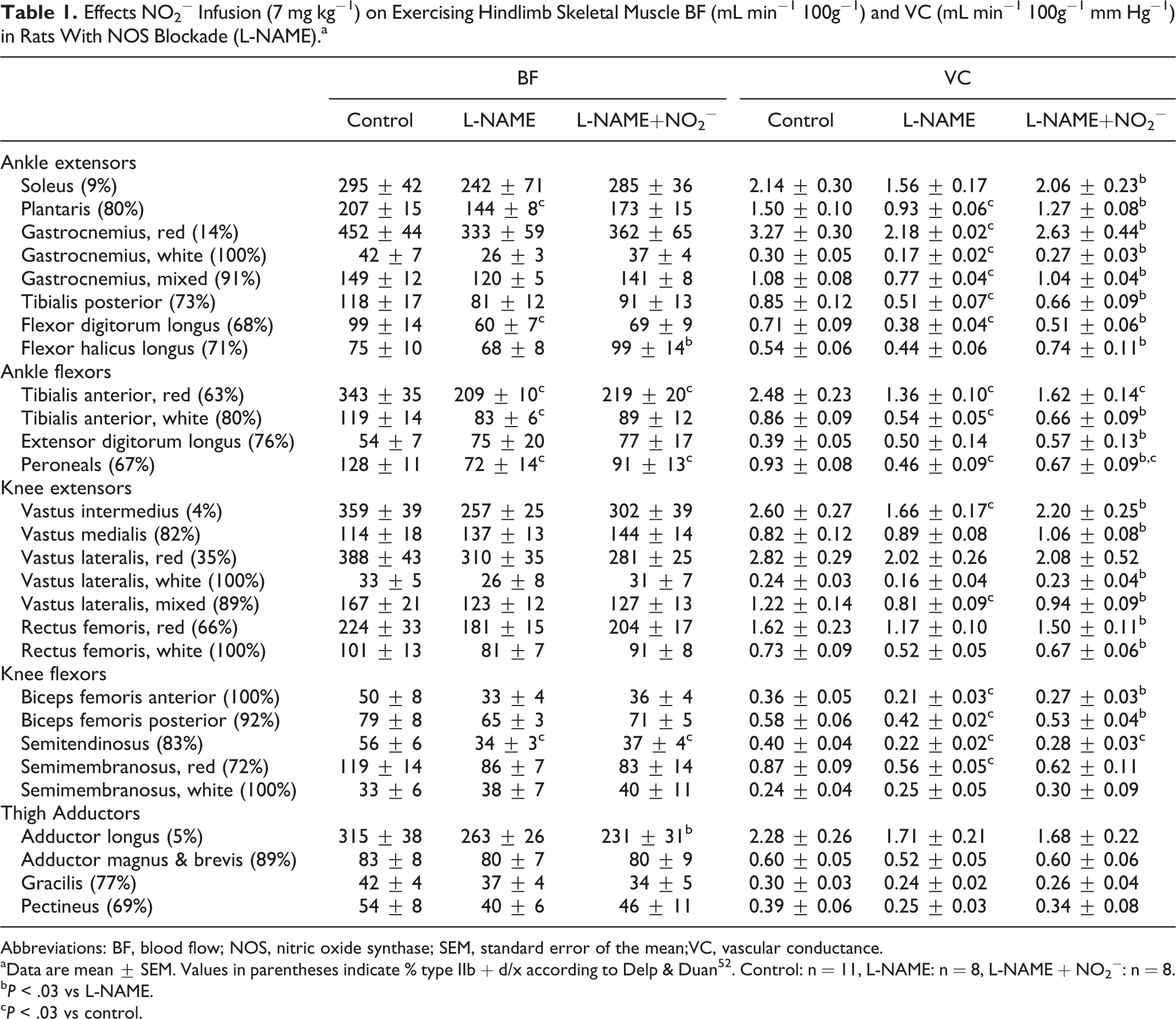
Abbreviations: BF, blood flow; NOS, nitric oxide synthase; SEM, standard error of the mean;VC, vascular conductance.
aData are mean ± SEM. Values in parentheses indicate % type IIb + d/x according to Delp & Duan 52 . Control: n = 11, L-NAME: n = 8, L-NAME + NO2−: n = 8.
bP < .03 vs L-NAME.
cP < .03 vs control.
Relative to control, BF and VC were lower in the adrenals and pancreas while VC was lower in the kidneys, stomach, and small intestine in rats treated with L-NAME (P < .03, Table 2). Following NO2− infusion, renal and adrenal BF and VC were lower when compared to control animals, while renal and adrenal BF was reduced when compared to L-NAME (P < .03, Table 2).
Effects of NO2− Infusion (7 mg kg−1) on Exercising BF (mL min−1 100g−1) and VC (mL ·min−1 100g− 1 mm Hg− 1) in the Kidneys and Organs of the Splanchnic Region.a

Abbreviations: BF, blood flow; SEM, standard error of the mean; VC, vascular conductance.
aData are mean ± SEM. Control: n = 11, L-NAME: n = 8, L-NAME + NO2−: n = 8.
bP < .03 vs control.
cP < .03 vs L-NAME.
dindicates arterial, not portal, BF and VC.
Discussion
The principal original finding of this investigation is that, in the face of NOS blockade, NO2− infusion restored exercising MAP and hindlimb skeletal muscle VC to levels observed in young adult healthy rats with intact NOS function. While NO2− infusion did not increase BF when compared to the L-NAME condition, it did abolish the lower BF induced by L-NAME. Elevations in VC and reductions in MAP could serve to reduce afterload and thus reduce the work of the heart during exercise. These results demonstrate that NO2− may serve as a powerful modulator of vascular control in vivo, independent of NOS function and thus may hold promising therapeutic potential, particularly in diseases with impaired NOS function and chronically elevated MAP.
Effects of Inorganic NO2− Infusion on Skeletal Muscle BF and VC and MAP
An abundance of research has focused on defining the vasoactive/cardioprotective roles of NO2− with many studies suggesting that the reduction of NO2− to NO compliments the well-understood NOS pathway of NO production, particularly when NOS function becomes uncoupled or otherwise impaired (reviewed by
34,35
). The vascular responses to NO2− infusion presented herein support this notion. Similar to what has been reported previously in our laboratory,
36,37
infusion with the comprehensive NOS blocker L-NAME increased MAP ~15% and decreased skeletal muscle VC ~26% during exercise. Consistent with our hypothesis, infusion with NO2− (7 mg kg−1) restored MAP and VC to levels similar to those observed in healthy control animals. One potential explanation for these effects of NO2− could be the lower PO2/pH environment present within the skeletal muscle following NOS inhibition.
33
Such environments facilitate (or uninhibit) NO2− reduction to NO in vivo,
18,38
which may allow local NO2− to support the blood–myocyte Po2 gradient (via ↑
One striking aspect of this investigation, in which acute NO2− infusion was employed, was that the augmented skeletal muscle VC was observed in muscles and muscle parts that span the full spectrum of fast- and slow-twitch fiber types (Table 1). This is in contrast to investigations using short-term dietary NO3− supplementation as a means of increasing circulating [NO2−]. Specifically, there is a fiber-type preferential effect of dietary NO3− supplementation as rats given NO3−-rich beetroot juice for 5 days exhibited elevated skeletal muscle BF and VC exclusively in muscles and muscle portions comprised of ≥ 66% type IIb + d/x muscle fibres. 27 Accordingly, beetroot juice elevates PO2mv during muscle contractions in the gastrocnemius (fast-twitch) but not soleus (slow-twitch) muscles. 33 The substantial array of muscles and muscle portions exhibiting a vasoactive response to NO2− infusion herein suggests that the fiber-type preferential effects observed following dietary NO3− supplementation may be conferred via changes in protein expression that require a longer period of elevated NO2− exposure to manifest. This idea is supported by evidence from Hernandez et al 7 in which the improvements in fast-twitch skeletal muscle force production evoked by NO3− supplementation were attributed to elevations in calcium handling proteins (ie, calsequestrin 1 and the dihydropyridine receptor), which were present following multiple days of dietary NO3− supplementation.
Additionally, the discrepancies in the vascular responses to NO3− versus NO2− treatment could be related to the relative impacts of NOS inhibition in fast- versus slow-twitch muscles. Skeletal muscles comprised predominantly of slow-twitch fibers demonstrate the greatest deficits in BF and
Clinical and Therapeutic Implications
In healthy individuals, eNOS is the primary endogenous source for NO2− and NO.
43
Endothelial dysfunction becomes evident early on in many diseases, including CHF (reviewed by Poole et al
2
) and peripheral artery disease (reviewed by Brevetti et al
44
) and thus likely limits vascular and metabolic function via attenuated NO production from both NOS-dependent and -independent pathways.
43,45
As evidenced by Hirai et al,
46,47
reduced NO from NOS dramatically impairs the matching of skeletal muscle
Experimental Considerations and Potential Limitations
A surprising result of the present investigation was the rise in exercising blood [lactate] following NO2− infusion (~41% and 81% greater vs control and L-NAME, respectively). Lower levels of NO may act as a useful brake on mitochondrial activity via competitive binding to complex IV of the respiratory chain.
48
In contrast, high concentrations of NO have been associated with adverse effects on cell respiration via nitrosylation of mitochondrial electron chain complexes, specifically complex I.
49
In addition, NO works to inhibit complex IV (cytochrome oxidase), thereby reducing cellular O2 consumption. Both of these effects may prove beneficial in certain environments or situations when O2 delivery becomes reduced as reductions in tissue
Furthermore, considering that NOS was acutely inhibited in the present investigation, the impacts of NO2− infusion may differ when administered to specific models of vascular diseases that have been developed chronically, as this would more closely mimic specific etiologies. Additionally, due to the relatively long half-life and bioactivity of L-NAME metabolites (~20 hours in rats 51 ) the experimental design was limited to a fixed sequence and therefore, an ordering effect cannot be ruled out. Future investigations in which NO2− is employed in healthy control animals would also provide further insight into the bioactivity of NO2− in animals with intact NOS function and could shed light on how a NO2−-based intervention may impact healthy cardiovascular function.
Conclusion
These data highlight the potential for NO2− to act independently of NOS and improve skeletal muscle vascular control during exercise. Considering the multiple cardiovascular diseases that impair NOS function, therapies that increase [NO2−] may result in improved skeletal muscle vascular control during exercise. However, the NO2− induced changes in blood [lactate] seen during exercise herein suggests that the reduction of NO3− to NO2−, accomplished via facultative anaerobes in the mouth following dietary NO3− consumption, may provide the controlled release of NO2− needed to elicit the most beneficial vascular and metabolic changes during exercise. It is anticipated that future investigations into the vascular impacts of both NO2−- and NO3−-based therapies will provide crucial insight into the potential benefits, and limitations, of both interventions.
Footnotes
Author Contributions
SKF, CTH, AMJ, TIM, and DCP contributed to conception and design of the experiments.
SKF, AAG, CTH, JLW, AJF, TDC, TS, JDA, AMJ, TIM, and DCP contributed to collection, analysis, and interpretation of data. SKF, CTH, TDC, JDA, AMJ, TIM, and DCP contributed to drafting the article and revising it critically for important intellectual content. All the authors have approved the final version of the article.
Acknowledgments
The authors would like to thank Ms K. Sue Hageman for her excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These experiments were funded by a Kansas State University SMILE award to TIM, and American Heart Association Midwest Affiliate (10GRNT4350011) and NIH (HL-108328) awards to DCP.
