Abstract
Cardiovascular disease is often associated with chronic kidney disease and vice versa; myocardial vitamin D receptors (VDRs) are among the probable links between the 2 disorders. The vitamin D receptor activator paricalcitol protects against some renal and cardiovascular complications. However, the structural and electrophysiological effects of myocardial vitamin D receptor modification and its impact on the response to ischemia–reperfusion are currently unknown. This work attempted to determine whether obstructive nephropathy induced myocardial changes (in rats) linked to vitamin D receptor deficiency and to ventricular arrhythmias in Langendorff-perfused hearts. Unilateral ureteral-obstructed and Sham-operated rats were treated with either paricalcitol (30 ng/kg/d intraperitoneal) or vehicle for 15 days. In 5 hearts from each group, we found that obstructed rats showed a reduction in VDRs and an increase in angiotensin II type 1 receptor expression (messenger RNA and protein), suffered fibrosis (determined by Masson trichrome stain) and myofibril reduction with an increase in mitochondrial size, and had dilated crests (determined by electron microscopy). These changes were reversed by paricalcitol. In 8 additional hearts per group, we found that obstructed rats showed a higher incidence of ventricular fibrillation during reperfusion (after 10 minutes of regional ischemia) than did those treated with paricalcitol. The action potential duration was prolonged throughout the experiment in paricalcitol-treated rats. We conclude that the reduction in myocardial vitamin D receptor expression in obstructed rats might be related to myocardial remodeling associated with an increase in arrhythmogenesis and that paricalcitol protects against these changes by restoring myocardial vitamin D receptor levels and prolonging action potentials.
Keywords
Introduction
Cardiovascular events occur with increased frequency in patients with chronic kidney disease (CKD). 1,2 Ventricular arrhythmias are particularly prevalent among patients with CKD, even when those patients do not have any kind of electrolyte imbalance. 3 -5 Mortality risk is also increased in patients with CKD who have acute coronary syndrome. 6
The vitamin D receptors (VDRs) are a potential link between these conditions. Vitamin D receptors are expressed in the cardiovascular system, and their activation counteracts myocardial hypertrophy and hypertension by modulating the renin–angiotensin system (RAS), inflammation, and fibrosis. 2 In clinical terms, low levels of serum 25-hydroxyvitamin D are associated with an increase in mortality, in particular with sudden cardiac death and coronary disease. 7 -9 Vitamin D receptor activation by paricalcitol (19-nor-1,25-dihydroxyvitamin D2) increased survival in patients with CKD, independent of their parathyroid hormone and calcium levels, but the mechanism of action for this effect remains unclear. 10
We postulate that CKD, even when kidney function is not significantly impaired, induces myocardial remodeling caused by a reduction in the VDR pathways and that these changes are proarrhythmic, independent of hydroelectrolytic status. To test this hypothesis, we used an animal model of unilateral kidney disease that shows decreases in renal VDR expression and in which paricalcitol treatment restores the VDR pathways. 11 In this study, we evaluated whether reduction in VDR expression occurs also at the myocardial level and whether this modification might be linked to structural changes and/or modifies the electrophysiological response to the ischemia–reperfusion protocol.
Methods and Materials
All experimental procedures were previously approved by the Ethics Committee on Laboratory Animal Research from the Medical Sciences College, National University of Cuyo, Mendoza (IACUL A5780-01), Argentina, in accordance with the guidelines of the Committee of Ethical Animal Experimentation of Argentina. The animals used in the present study corresponded with those previously described by our group; this prior study analyzed changes at the renal cortical level. 11
Surgical Procedure and Pharmacological Treatment
The urinary tracts (left kidney) of female Wistar-Kyoto rats, each animal weighing from 180 to 200 g, were surgically obstructed. Animals were anesthetized with 60 mg/kg of ketamine and 0.1 mg/kg of acepromazine by intraperitoneal injection. Using an aseptic technique, a midline abdominal incision was performed, as previously described. 11 After surgical closure of the abdominal incision, the rats were given free access to water and food. Finally, 15 days after the operation, the hearts and kidneys were harvested from both the obstructed and the Sham-operated animals. Rats were considered to have a successful ureteral obstruction when ureteral dilation was >2 mm (as determined using a millimetric eyepiece). Both groups (Sham and obstructed) were treated either with vehicle (100 μL of propylene glycol/d) or with paricalcitol (30 ng/kg/d in 100 μL of propylene glycol; provided by Abbott Laboratories, Abbott Park, Illinois), both being injected intraperitoneally for 15 days (from surgery until killing). Each of the 4 groups included 5 animals to be used for the molecular and structural studies and 8 animals assigned to the ischemia–reperfusion experiment.
Systolic Blood Pressure Measurement
After weighing the animals, the systolic blood pressure (SBP) was monitored indirectly in conscious, prewarmed, slightly restrained rats. Monitoring was accomplished using the tail-cuff method, and each animal’s SBP was recorded using a CODA device (Kent Scientific Corporation, Torrington, Connecticut) the day before the isolated heart experiments. The rats were trained on the apparatus several times before measurement.
Histological Studies
Myocardial tissue samples from 5 hearts from each group were fixed in 10% phosphate-buffered formalin (pH = 7.1) for 24 to 48 hours before being embedded in paraffin and serially sectioned (5 µm) on a microtome (Leica Microsystems, Deerfield, Illinois). The paraffin sections were subjected to staining with Masson trichrome stain, as previously described. For this experiment, ventricular sections were treated sequentially with Weigert iron hematoxylin solution for 10 minutes, Biebrich scarlet–acid fuchsin for 2 minutes, phosphotungstic acid/phosphomolybdic acid for 10 minutes, and aniline blue for 5 minutes. Tissue was destained in 1% acetic acid for 2 minutes, dehydrated through graded ethanol to xylene, and mounted for examination by light microscopy. For all morphologic evaluations, the observer was blinded to the origin of the histological sections. A standard point-counting method was used to quantitate the fibrosis of the myocardial samples. Ten consecutive fields were randomly selected in the ventricular tissue and were evaluated at ×400 magnification on a 10 × 10 grid-imprinted reticule. Blue-stained interstitial fibrotic areas were assessed by using an image analyzer (ImageJ 1.43; Wayne Rasband, National Institutes of Health, Bethesda, Maryland). All points not counted within cardiomyocytes and vascular space were considered interstitial. The number of grid points containing blue collagen staining in the interstitium was divided by the total number of points in the fields (1000) to obtain the percentage of the fractional area of interstitial collagen deposition. Results were expressed as a percentage of the measured area.
Immunohistochemical Studies
Heart paraffin sections (5-6 μm thick) were dewaxed in xylol, rehydrated, and incubated with 3% H2O2 for 30 minutes to quench endogenous peroxidase activity. After washing in Tris-buffered saline (0.05 mol/L Tris-HCl, 0.15 mol/L NaCl), pH 7.6, and nonspecific blocking with 10% bovine serum albumin for 30 minutes at room temperature, the sections were immunostained to reveal VDR and angiotensin II type 1 receptor (AT1R). Antibodies applied were mouse monoclonal antibody against VDR (D-6) and rabbit polyclonal antibody against AT1R (306; Santa Cruz Biotechnology, Inc, Santa Cruz, California), diluted at 1:500. A commercial immunoperoxidase kit was used (Dako EnVision; Dako Corporation, Carpinteria, California). Positive reactions were evaluated considering the specific location of immunostaining (heart structure and cell compartment: nucleus, cytoplasm, and membrane) and the intensity of the immunoreaction. The negative controls included tissues unexposed to primary antibodies and tissues exposed to control immunoglobulin G. The positive controls were human breast cancer biopsy samples. We used a scoring system slightly modified from one reported previously. 12 Briefly, we used the following intensity scores: 0 = no staining, 1 = weak staining, 2 = moderate staining, and 3 = strong staining.
Electron Microscopy
Immediately upon being separated from their respective organs, tissue samples were fixed by immersion in a fixative solution (1:10). Fixative solution was obtained by diluting 1 phosphate-buffered saline tablet, following the manufacturer’s instructions, in 200 mL of double-distilled water and 2% glutaraldehyde (v/v), 2% of fresh p-formaldehyde (v/v), and 2% of picric acid as saturated solution. After 2 hours at room temperature, the samples were reduced and placed in an osmium tetroxide (OsO4) solution overnight at 4°C. The next day, the samples were dehydrated in alcohol–acetone, grading up to 100%, and embedded in Epon 812 (Sigma, St Louis, Missouri). Ultrathin sections were obtained with an Ultracut microtome (Leitz, Folly Beach, South Carolina) and stained with lead citrate and uranyl, using conventional staining methods. Observations were made and micrographs were created using a Zeiss EM 900 microscope (Zeiss, Oberkochen).
Reverse Transcription-Polymerase Chain Reaction and Semiquantification of Messenger RNA
Total ribonucleic acid from ventricular myocardial tissue was obtained using Trizol reagent (Life Technologies Inc Gibco/Brl Division, Grand Island, New York). Ribonucleic acid of 1 μg was denatured in the presence of 0.5 μg/50 μL oligo (dT) 15 primer and 40 U recombinant ribonuclease inhibitor (Promega, Madison, WI). Reverse transcription was performed in the presence of the mixture, using 200 U of reverse transcriptase in reaction buffer and 0.5 mmol/L of deoxyribonucleotide triphosphate (each), and incubated for 60 minutes at 42°C. The complementary DNA (10 μL) was amplified by polymerase chain reaction under standard conditions. Each sample was analyzed to determine levels of VDR, transforming growth factor β (TGFβ), AT1R, and β-actin (primers in Table 1, Integrated DNA Technologies, Inc, Coralville, Iowa). The VDR, AT1R, and TGFβ signals were standardized against the β-actin signal for each sample, and the results were expressed as a ratio.
Sets of Primers for the RT-PCR Procedure.a

Abbreviations: VDR, vitamin D receptors; AT1R, angiotensin II type 1 receptor; TGFβ, transforming growth factor β; RT-PCR, reverse transcription-polymerase chain reaction.
a Sense and antisense sequences, annealing temperatures, and predicted product size in bp are shown.
Langendorff-Perfused Rat Hearts
After killing, 8 hearts from each group were rapidly excised and kept at 4°C until being connected to a perfusion system (which always took less than 3 minutes). The hearts were perfused at constant pressure of 80 cm H2O with a modified Krebs-Henseleit solution containing (in mmol/L) 121 NaCl, 25 NaHCO3, 1.2 Na2HPO4, 5 KCl, 2.5 CaCl2, 1.2 MgSO4, and glucose. When equilibrated with 5% CO2 in O2 at 36.5°C ± 0.5°C, the pH was 7.4 ± 0.02.
The coronary flow was measured throughout the experiment. It was used as an index of adequate perfusion and as a criterion to assess the efficiency of coronary ligation. A reduction of at least 25% during occlusion was considered satisfactory. In order to validate reproducibility, we reoccluded the artery at minute 10 of reperfusion, perfused Evans blue, weighed the hearts, and after 1 hour of cooling at −20°C, sliced the ventricles transversely from apex to base into 2 mm slices. The zones colored other than blue were considered the areas at risk. The slices were photographed for planimetric analysis (ImageJ 1.43; Wayne Rasband, National Institutes of Health, Bethesda, Maryland) and weighed for adjusted expression of the area at risk. We included only those hearts with an ischemic area greater than 40% of the ventricles in order to guarantee the reproducibility of the reperfusion arrhythmia incidence. 13
Reperfusion Arrhythmias and Action Potentials
After 20 minutes of stabilization, we continuously obtained the surface electrogram equivalent of lead II and epicardial transmembrane potential using a Hewlett-Packard 1500A (Hewlett-Packard Company USA, Palo Alto, California) and a custom-made microelectrode amplifier, respectively. Both signals were digitized with an analog-to-digital converter (NI PCI-6221; National Instruments, Austin, Texas) and recorded using LabView Signal Express 2.5 (National Instruments, Austin, Texas). After a period of 10 minutes, preischemia, the hearts underwent 10 minutes of regional ischemia by ligation of the left anterior descending coronary artery followed by 10 minutes of reperfusion. Ventricular arrhythmias were classified according to the Lambeth convention. 14 We evaluated the incidence and duration of ventricular tachycardia (VT) and ventricular fibrillation (VF) and also the arrhythmia severity each minute using the following scores: 0 = sinus rhythm; 1 = premature ventricular beats or bigeminy; 2 = Salvos; 3 = nonsustained VT (<30 seconds); and 4 = sustained VT (>30 seconds) or VF. 15 We analyzed the following parameters of epicardial transmembrane potentials: action potential amplitude, resting potential, and action potential duration at 90% of repolarization.
Statistical Analysis
Data were expressed as mean ± standard error of the mean, and statistical analysis was performed using analysis of variance (ANOVA) or 2-way repeated-measures ANOVA followed by a Bonferroni posttest and Fisher exact test, as appropriate. Variables not normally distributed were analyzed using the Kruskal-Wallis test followed by Dunn posttest. Statistical analysis was performed using GraphPad Prism 5.0 (GraphPad Software, Inc, La Jolla, California).
Results
Vitamin D Receptor and AT1R Myocardial Expression
The VDR expression in the hearts of the obstructed rats was lower than that found in the hearts of the Sham-operated group (the intensity score for Sham was 2.1; for Sham + Pari, the score was 2.3; for unilateral ureteral obstruction [UUO], the score was 1.0*; and for UUO + Pari, the score was 2.6; P < .05, Sham vs UUO); conversely, AT1R expression was higher (the intensity score for Sham was 1.1; the score for Sham + Pari was 1.2; the score for UUO was 2.5*; the score for UUO + Pari was 1.6; P < .05, Sham vs UUO). Paricalcitol restored VDR and AT1R expression in samples obtained from the UUO + Pari group (Figure 1).

Vitamin D receptor and AT1R expression. A, Representative gels of VDR (227 bp) and AT1R (385 bp) mRNA in myocardial samples obtained from Sham-operated rats treated with vehicle (Sham), from obstructed rats (UUO), and from rats treated with paricalcitol (Sham + Pari and UUO + Pari, respectively). The corresponding housekeeping β-actin (201 bp) is included subsequently. B, Vitamin D receptor–β-actin mRNA ratio shows lower expression in myocardial samples from UUO versus Sham-operated (Sham) rats as well as a higher AT1R–β-actin mRNA ratio (**P < .01 for both). Paricalcitol treatment maintained VDR and AT1R mRNA expression. Results are mean ± standard error of the mean (SEM); n = 5. C, Immunohistochemical staining of VDR and AT1R in myocardial samples. Images were taken at ×400. AT1R indicates angiotensin II type 1 receptor; mRNA, messenger RNA; RDU, relative densitometry units; UUO, unilateral ureteral obstruction; VDR, vitamin D receptor.
Myocardial Structural Remodeling
We did not see any changes in SBP (Sham, 110 ± 5; Sham + Pari, 112 ± 7; UUO, 118 ± 5; UUO + Pari, 116 ± 7; P = not significant [ns]) nor did we see myocardial hypertrophy (relative heart weight in mg/g: Sham, 3.8 ± 0.1; Sham + Pari, 3.7 ± 0.3; UUO, 3.9 ± 0.2; UUO + Pari, 3.7 ± 0.2; P = ns) associated with UUO, but the microscopic structure revealed evidence of increased fibrosis (Sham, 34.7 ± 5.4%; Sham + Pari, 29.5 ± 6.2%; UUO, 68.9 ± 7.4%*; UUO + Pari, 34.1 ± 6.9%; *P < .05, Sham vs UUO) as determined by Masson trichrome staining, the presence of which was consistent with the increase in TGFβ expression at the messenger RNA level (Figure 2). Paricalcitol-treated hearts did not appear to be any different from Sham vehicle-treated ones. Hearts from obstructed rats showed myofibrillar density reduction and mitochondrial edema (Figure 3). However, paricalcitol prevented the loss of myofibrillar content and prevented as well mitochondrial edema.

Myocardial TGFβ expression and Masson trichrome staining. A, Representative gel of TGFβ (294 bp) mRNA. The corresponding housekeeping β-actin is included subsequently. B, Graphical representation of TGFβ–β-actin mRNA ratio shows higher mRNA expression in obstructed rat hearts. **P < .01 Sham versus unilateral ureteral obstruction (UUO). Results are mean ± standard error of the mean (SEM); n = 5. C, Masson trichrome-stained sections of myocardial samples. Magnification: ×400. mRNA indicates messenger RNA; RDU, relative densitometry units; TGFβ, transforming growth factor β.

Electron microscopy: myocardial effect of unilateral obstruction and paricalcitol treatment. The upper row corresponds to electron microscopy photograph (×60 000) from myocardial samples obtained from Sham-operated rats treated with vehicle (Sham) and with paricalcitol (Sham + Pari). The lower row shows data from myocardial samples obtained from rats after 15 days of unilateral obstruction and after having been treated with vehicle (UUO) or paricalcitol (UUO + Pari). UUO indicates unilateral ureteral obstruction.
Coronary Flow and Area at Risk
Coronary flow presented similar values in all of the groups, and the degree of reduction was stable after coronary occlusion. The ischemic area measured at the end of the experiment did not differ between groups (both results are shown in Table 2).
Coronary Flow and Heart Rate During the Experimental Protocol and the Size of Ischemic Area.a
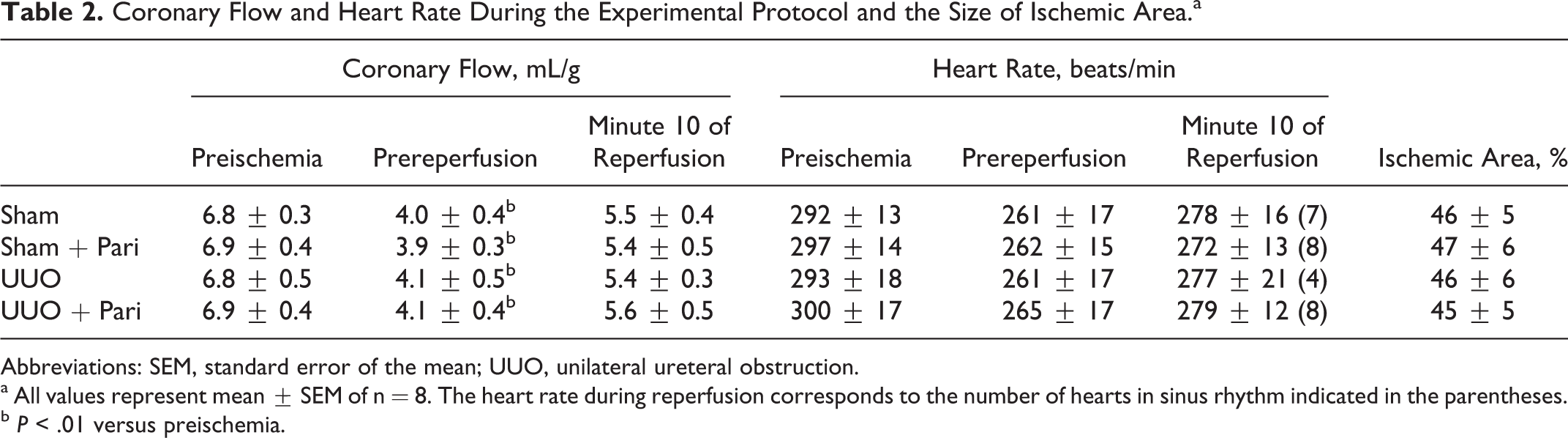
Abbreviations: SEM, standard error of the mean; UUO, unilateral ureteral obstruction.
a All values represent mean ± SEM of n = 8. The heart rate during reperfusion corresponds to the number of hearts in sinus rhythm indicated in the parentheses.
b P < .01 versus preischemia.
Ventricular Arrhythmias
We did not find any differences in basal heart rate between the groups, and all had a reduction of around 20 to 40 beats/min during ischemia (Table 2). Ventricular arrhythmias occurred at the onset of reperfusion in the UUO + vehicle-treated hearts (Figure 4). The administration of paricalcitol reduced the incidence and duration of VF but did not modify VT (Figure 4 and Table 3). Similar durations of VT were followed by a predominant sinus rhythm during reperfusion in all groups other than the UUO + vehicle group, in which VT was followed by sustained VF. These results are supported by a reduction in the severity score (Figure 4), which also indicates that the antiarrhythmic effect was sustained, since vehicle-treated hearts maintained a high level of severity.

Ventricular arrhythmias during reperfusion. A, Incidence of ventricular arrhythmias during reperfusion. White bars indicate the number of hearts that developed ventricular tachycardia (VT) and black bars indicate those who had ventricular fibrillation (VF) for each group. *P = .0406, Sham versus UUO; and + P = .007, UUO versus UUO + Pari, both by Fisher exact test; n = 8 each. B, Arrhythmia severity score throughout the experimental periods. Continuous connecting lines indicate vehicle-treated rats and dashed connecting lines indicate paricalcitol-treated rats. Sham groups are indicated by squares and UUO by circles. *P < .05 Sham versus UUO by 2-way repeated measures analysis of variance (ANOVA). UUO indicates unilateral ureteral obstruction.
Reperfusion Arrhythmias Duration.a

Abbreviation: UUO, unilateral ureteral obstruction.
a All values correspond to the median (first quartile to third quartile) expressed in seconds.
b P < .01 Sham versus UUO analyzed by Kruskal-Wallis test followed by Dunn posttest.
c P < .01 UUO versus UUO + Pari analyzed by Kruskal-Wallis test followed by Dunn posttest.
Transmembrane Potential
Analysis of the transmembrane potential of epicardial cardiomyocytes determined that paricalcitol lengthened the action potential duration at 90% repolarization in both treated groups. This effect was found during preischemia and previous to reperfusion. The UUO hearts maintained short action potential duration during reperfusion. The amplitude and the resting potential were very similar in all groups during the experiment (Figure 5).

Effects of the UUO and paricalcitol treatment on the membrane potential response to ischemia–reperfusion. A, Changes in action potential duration at 90% repolarization (APD90) with ischemia and reperfusion for each group. Data were obtained before ischemia, preischemia (PI), at early ischemia (EI; first 5 minutes) and late ischemia (LI; last 5 minutes), and during reperfusion (REP). The bars represent means ± standard error of the mean (SEM), n = 8. *P < .05; **P < .01; and ***P < .001 for comparisons versus Sham group for the same period. B, The diagonally striped bars represent the resting potential, and the total height of each bar corresponds to the action potential amplitude. The overshot is indicated by the checkered pattern. UUO indicates unilateral ureteral obstruction.
Discussion
The present study relates myocardial VDR reduction in UUO rats with arrhythmogenic remodeling reversed by paricalcitol. This is the first report of a reduction in VDR occurring at the myocardial level, in which the reduction is associated with kidney disease but without any other hemodynamic stimulus, which has been shown to be a VDR expression modifier. Previous reports using the subtotal nephrectomy model of CKD did not find any differences in myocardial VDR expression, but the animals developed myocardial hypertrophy. 16 Others reported that salt-associated hypertrophy induced VDR expression as a counterbalance. 17 Furthermore, a deletion of the receptor induced myocardial hypertrophy. 18 So, the differences here described in respect to previous reports could be attributed to the fact that our kidney disease model partially reduced VDR without any hemodynamic stimulus and/or myocardial hypertrophy. Since we did not find any changes in calcium homeostasis, we speculate that the obstructed kidney might generate inflammatory components that affect VDR expression, but this hypothesis has been confirmed only at the renal level. 19 The specific mechanism involved in myocardial VDR reduction in UUO is not clear and deserves further evaluation.
Our finding helps clarify the role of myocardial VDR expression in the pathophysiological mechanisms linking CKD to cardiovascular disease, which is itself caused by the exacerbation of myocardial angiotensin effects. A long time ago, an original work suggested a possible link between vitamin D and the RAS. 20 Inappropriate stimulation of the RAS resulting from vitamin D deficiency has been associated with hypertension, heart attack, stroke, and hypertrophy of the left ventricle and vascular smooth muscle cells. 21 More recently, experimental and clinical evidence indicates that vitamin D deficiency and tissular angiotensin II (ANG II) upregulation play a pivotal role in the progression of renal disease associated with obstructive nephropathy, 22 and that VDR attenuates obstructive renal injury at least in part by suppressing the RAS. 23 The pleiotropic actions of vitamin D and its analogs are mediated by a specific VDR, a ligand-dependent transcription factor that belongs to the steroid/nuclear receptor gene family. 24,25 In agreement, we find significantly higher AT1R expression and lower VDR expression resulting from UUO. Although our findings are not causative, the remodeling here described is in agreement with the postulated counterbalance between VDR and the RAS at the myocardial level. 18,26,27 Indeed, in a recent report, we showed that AT1R modulated by VDR is involved in the mechanism by which paricalcitol provides renal protection in spontaneously hypertensive rats. 28
Angiotensin II, via the AT1R, is involved in the modulation of ventricular structure and function in CKD. Angiotensin II causes significant proinflammatory activity that induces the expression of cytokines, chemokines, adhesion molecules, growth factors, and reactive oxygen species. 29 Previous reports that associate AT1R and mitochondrial malfunction are in agreement with the ultrastructural changes described here. 30 In addition, the myofibrillar reduction could be attributed to the regulatory effect of vitamin D on the expression of myocardial myosin. 31,32 We attribute these changes to the myocardial VDR pathway because they were prevented in paricalcitol-treated rats. In addition, AT1R blockade interferes with ANG II-mediated TGFβ expression. 33 Vitamin D receptors are negative regulators of TGFβ/Smad signaling. Impaired VDR signaling with reduced expression of VDR and decreased levels of its ligand may thus contribute to hyperactive TGFβ signaling. 34 Important to our understanding, paricalcitol showed a relevant beneficial effect in the reduction of myocardial fibrosis, a key factor in the myocardial dysfunction suffered by patients with CKD. 35
In regard to arrhythmogenesis, it is well known that myocardial remodeling generates electrical instability. 36 The influence of a structural kidney alteration at the myocardial level may not be evident under basal conditions but modifies the response to stressful situations such as ischemia and reperfusion. Unilateral ureteral obstruction rats showed remodeling and an increase in VF during reperfusion. Both the increase in AT1R and TGFβ could underlie the arrhythmogenesis that is observed in UUO rat hearts. The level of AT1R expression plays an important role in ischemia–reperfusion arrhythmogenesis. The AT1R knockout mice do not display ventricular arrhythmias after ischemia–reperfusion. 37 The AT1R is unregulated after ischemia–reperfusion injury. 38 Losartan, a specific AT1R blocker, effectively reduces ischemia–reperfusion arrhythmias in transgenic rats with cardiac AT1R overexpression. 39 The pathway that relates AT1R and TGFβ signaling is involved in a number of human diseases, including heart failure and myocardial arrhythmias. 40 Transforming growth factor β increases sarcoplasmic reticulum Ca2+ content and the frequency of spontaneous intracellular Ca2+ oscillations. 40 Paricalcitol prevented the changes in the pattern of expression, the structural changes, and also the lethal arrhythmias. However, lengthening of the action potential duration of paricalcitol could also explain the antiarrhythmic effect found, as it is known that this variable is a determinant of the myocardial refractory period and the generation of reentry circuits. In fact, it could explain the antifibrillatory effect, due to the interference of microreentrant circuits, without affecting the brief self-limited episodes of VT observed even in Sham-operated hearts, probably because the latter are usually the expression of macroreentrant circuits. 41
We support our electrophysiological findings in a recent publication that describes an action potential lengthening in 1,25-dihydroxyvitamin D-treated, isolated rabbit left atrium that exerts an antiarrhythmic effect. 42 However, we did not identify the mechanisms by which the drug induces action potential prolongation. Since isolated hearts were not exposed to the drug during the experiment, it might be possible to attribute this effect to changes in ionic current expression. We did not find any reports of electrocardiogram changes in patients receiving paricalcitol. The electrophysiological effects of paricalcitol should be further investigated.
We conclude that myocardial VDR pathways are a link between UUO kidney disease and cardiac remodeling and that their presence in a host predisposes that host to lethal arrhythmias during reperfusion. This interesting new model will enable researchers to evaluate the complex interaction of the kidneys and the heart.
Summary
The reduction in myocardial vitamin D receptor expression in unilateral ureteral obstructed rats induced myocardial remodeling associated with an increase in arrhythmogenesis; paricalcitol protects against these changes by restoring myocardial vitamin D receptor levels and prolonging action potentials.
Perspectives
Despite overwhelming data that link sudden cardiac death with CKD, this is a relatively unexplored field in the basic research area, mainly because of the difficulty of finding an adequate experimental model. Furthermore, other cardiovascular effects of VDR expression reduction due to kidney disease could be evaluated using this animal model of UUO. In addition, electrophysiological properties of paricalcitol deserve further investigation in order to clarify whether or not these effects might be responsible for the improved prognosis that has been described in treated patients.
Footnotes
Acknowledgment
Thanks go to Bob Ritchie, Puget Sound Maritime Historical Society editor.
Author Contributions
All authors contributed to conception and design of the study, with substantial contribution to data acquisition, analysis and interpretation of the data, drafting of the article, and critical revision of the article for intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Research and Technology Council of Cuyo University (SECyT), Mendoza, Argentina, and from the National Council of Scientific and Technical Research (CONICET) PIP 2010-2012, both of which were awarded to Walter Manucha. Grant no. PICT 2012-0234 Préstamo BID 2777 OC/AR Walter Manucha.
