Abstract
Background and Objectives:
Coronary artery calcification (CAC) is associated with future cardiovascular events and/or death of patients on hemodialysis (HD). We investigated whether progression of CAC in patients on HD could be delayed by switching from a calcium (Ca)-based phosphate (Pi) binder to lanthanum carbonate.
Design, Setting, Participants, and Measurements:
The CAC scores were evaluated at study enrollment and after 6 months in 52 patients on HD using calcium carbonate (CC) as a Pi binder. Patients were randomly divided into 2 groups assigned to receive either CC or lanthanum carbonate (LC), and the CAC scores were evaluated after a 6-month treatment period. Progression of CAC was assessed, as were serum levels of Ca, Pi, and intact parathyroid hormone (iPTH).
Results:
Forty-two patients completed the study (23 receiving CC and 19 receiving LC). In the 6 months prior to randomization, all patients were treated with CC. During this 6-month period, the CAC scores increased significantly in all 42 patients. Once randomized, there was significantly less progression in the group treated with LC than with CC. Changes in CAC scores from 6 to 12 months were significantly smaller in the LC group than the CC group (−288.9 ± 1176.4 vs 107.1 ± 559.6,
Conclusions:
This pilot study suggested that LC delayed progression of CAC in patients on HD compared with CC.
Introduction
Coronary artery calcification (CAC) is an important prognostic factor in the general population and patients on hemodialysis (HD). 1 –4 We recently reported that advanced CAC was strongly associated with future cardiovascular events, cardiovascular death, and all-cause mortality in patients on HD. 4 Delaying progression or promoting regression of CAC may therefore improve survival among patients on HD. One key factor leading to CAC progression in patients on HD is calcium (Ca) overload related to the use of Ca-based phosphate (Pi) binders. 5 –8 It remains unclear, however, whether switching from a Ca-based to a non-Ca-based Pi binder can delay the progression of CAC.
In human studies, the non-Ca-based Pi binder sevelamer hydrochloride, a nonabsorbable high-molecular-weight compound with a chemical composition of ([C3H7 N] × [C9H18N2]Y·nHCL)Z, has been found to delay the progression of CAC compared with calcium carbonate (CC) therapy. 3,4,6,9 –12 Clinical studies of the non-Ca-based Pi binder lanthanum carbonate (LC), which contains the rare earth element lanthanum and has a molecular formula of La2(CO3)3⋅4H2O, have indicated that LC is well tolerated and reduces serum phosphorus levels effectively, 13,14 but whether LC delays progression of vascular calcification in patients on HD is not fully known. Thus far, only 2 small studies have reported the effect of LC on vascular calcification. 15,16 One such study investigated the effect of LC on aortic calcification, 15 and consequently there has only been 1 very small study comparing progression of CAC among patients on HD receiving LC or CC. 16
We performed a prospective, randomized trial to determine whether LC delayed progression of CAC in patients on HD, compared with CC. The primary end point of this study was the difference in changes in CAC scores between the CC and the LC groups. We also evaluated whether switching from a Ca-based to a non-Ca-based Pi binder would influence serum levels of Ca, Pi, and intact parathyroid hormone (i-PTH).
Methods
Patients and Study Protocol
This study was performed at a single dialysis center (Shonan Kamakura General Hospital, Kamakura, Japan) upon approval by the ethical committee in our hospital. The study was performed in accordance with the Declaration of Helsinki. Figure 1 presents a flowchart of the study process. Patients were drawn from 68 outpatients on stable HD using CC as a Pi binder in September 2009, from the Dialysis Center of our hospital. Exclusion criteria were pregnancy, malignancy, severe gastrointestinal disease, liver disease, endocrine disease, and arrhythmia. Five patients with arrhythmia were excluded due to the inability to obtain good cardiac images. Among the remaining 63 patients, 52 gave informed consent to 64-row multislice computed tomography (MSCT) for evaluation of CAC and were enrolled in the study.
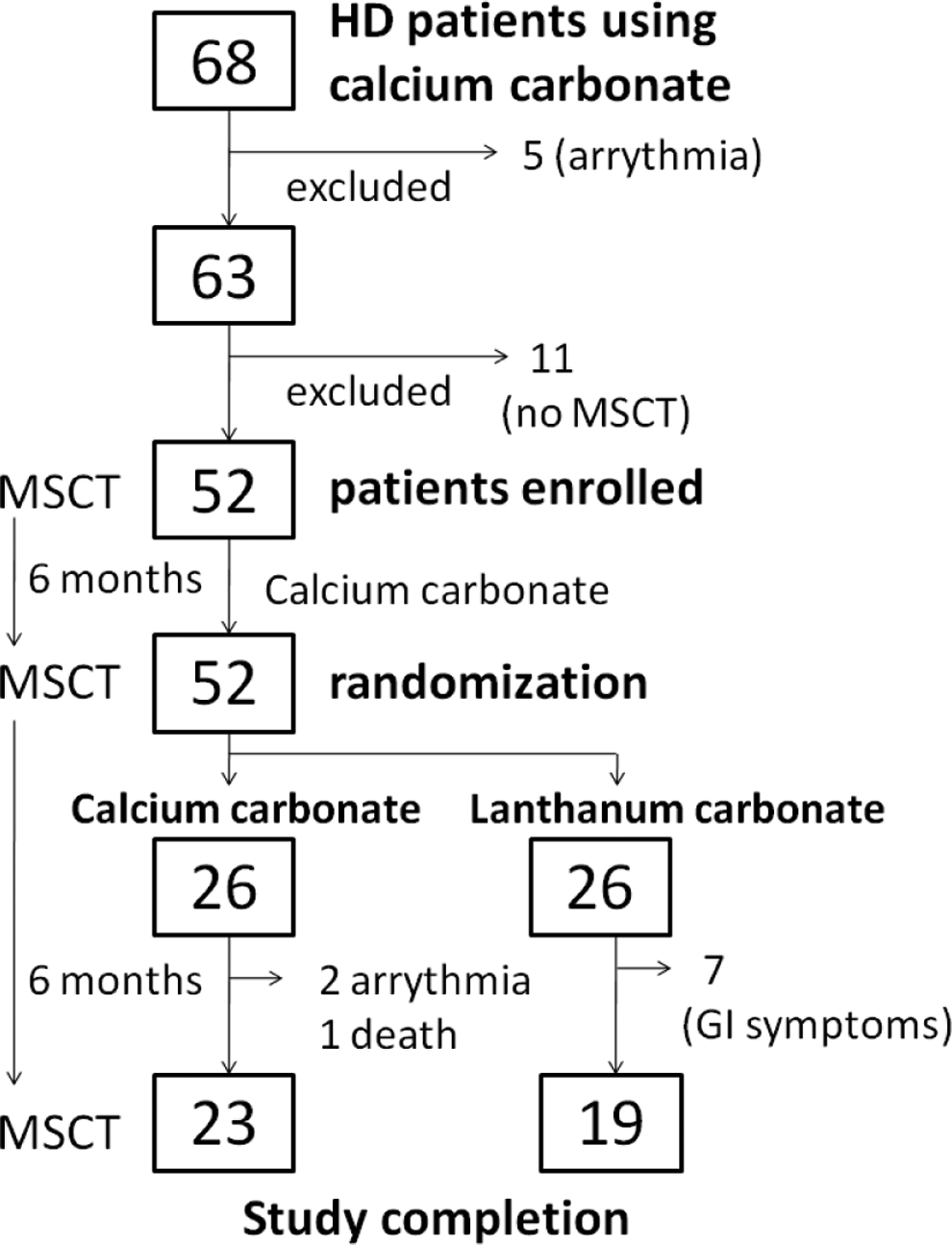
Flowchart of the study process.
All 52 patients underwent MSCT at study enrollment and used CC during the first 6-month lead-in period. Patients underwent MSCT at the end of the lead-in period and were randomly divided into a CC group (N = 26) and an LC group (N = 26). Randomization was conducted using a table of random numbers. Patients assigned to the CC group continued to use CC, and those assigned to the LC group were switched to LC. Phosphate binder assignment was not blinded. The MSCT was performed again after a 6-month interventional period. Serum Ca, Pi, and i-PTH levels were controlled according to the guideline of the Japanese Society of Dialysis Therapy, 17 which recommends Ca of 8.4 to 10.0 mg/dL, Pi of 3.5 to 6.0 mg/dL, and i-PTH of 60 to 180 pg/mL. The Ca, Pi, and i-PTH levels were modulated by medication according to routine clinical practice in our dialysis center. The type of vitamin D used in our study was 1,25-(OH) D3, that is, calcitriol. To address Ca, Pi, and iPTH control, Pi was prioritized and was controlled by a Pi binder (CC or LC). Then, if serum Ca was below 8.4 mg/dL (lower normal limit), vitamin D was newly added or increased to increase Ca absorption from the gastrointestinal (GI) tract. If serum Ca was higher than 10.4 mg/dL (upper normal limit), vitamin D dosage was decreased. Cinacalcet (C22H22F3N·HCl), a Ca-sensing receptor agonist that inhibits secretion of PTH, was added or increased as needed to maintain the levels of i-PTH within their target range.
Blood was drawn from the arterial side of the arteriovenous fistula at the beginning of the first HD session of the week to evaluate the levels of serum Ca, Pi, i-PTH, albumin, C-reactive protein (CRP), total cholesterol (Tchol), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C). Serum Ca and Pi were measured every 2 weeks, and i-PTH was measured every month.
Periodic 240-minute HD was performed in each patient 3 times weekly using polysulfone dialyzers. Blood flow rate (Qb, mL/minute) was set at 4.0 to 4.5 mL/kg body weight, and dialysate flow rate (Qd) was set at 500 mL of dialysis fluid (Kindary 3E, Fuso Pharmaceutical Industries Ltd Osaka, Japan) per minute in all patients. The composition of Kindary 3E (dialysate) was Na+ 140 mEq/L, K+ 2.0 mEq/L, Cl− 114.5 mEq/L, Ca2+ 2.5 mEq/L, Mg2+ 1.0 mEq/L, HCO3 − 25.0 mmol/L, glucose 150 mg/dL, and CH3COO- 8 mEq/L. Low Ca concentration dialysate was used throughout the study period. The diet of patients on HD in our dialysis center was evaluated routinely, and the patients were guided by a specialized dietician, using a diet report to restrict dietary Pi intake to 700 mg/d or less.
After performing Multislice Computed Tomography (MSCT) with a LightSpeed Ultrafast 16 (General Electric Medical System, Tokyo, Japan), we calculated Agatston score 18 according to the method of Horiguchi et al, which we used previously. 19,20 This score is well correlated with that obtained by electron-beam computed tomography. 18 Volumetric data for the entire heart were obtained in helical mode with the following parameters: 1.25-mm collimation width × 16 detectors, gantry rotation speed of 0.5 s per rotation, 120 kV, and 100 mA. Pitch was varied according to the heart rate. Slices that were 2.5 mm in thickness with the center of the temporal window corresponding to 80% of the R-R interval were reconstructed at 2.5-mm intervals. Calcium score, volume, and mass were determined from the MSCT data on a commercially available workstation (Adventure Windows, version 4.4.1; General Electric Medical System, Tokyo, Japan) using CAC scoring software (version 3.5; Smartscore, Tokyo, Japan). To measure the area and peak density of plaques, regions of interest were set by vessel and slice, with the threshold for pixels greater than 130 Hounsfield units, according to the Agatston method. The CAC score was calculated by a radiologist who was completely blinded to patient information, including group allocation.
Statistical Analysis
Continuous variables were expressed as the mean ± standard deviation (SD), unless otherwise specified. Comparison of group mean values was done by
Results
Patient Profile
Among the 26 patients in the CC group, 3 were excluded from the analysis—1 patient died of pneumonia, and 2 had paroxysmal atrial fibrillation that interfered with coronary artery imaging. In the LC group, 7 patients were excluded from the analysis due to the development of GI symptoms that prevented continued LC ingestion. As a result, 23 patients in the CC group and 19 patients in the LC group were included in the final analysis. Table 1 presents the basic characteristics of the 42 patients who completed the study.
Patient Characteristics at Study Enrollment.a

Abbreviations: HD, hemodialysis; IHD, ischemic heart disease; DM, diabetes mellitus; Ca, calcium; Pi, phosphate; iPTH, intact parathyroid hormone; CRP, C-reactive protein; Tchol, total cholesterol; TG, triglyceride; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
aNormal reference ranges are Ca 8.3 to 10.4 mg/dL, Pi 2.7 to 4.5 mg/dL, iPTH 8.7 to 79.5 pg/mL, albumin 3.8 to 5.2 g/dL, CRP < 0.5 mg/dL, Tchol 150 to 220 mg/dL, TG 50 to 149 mg/dL, HDL-C 40 to 100 mg/dL, LDL-C 70 to 139 mg/dL.
Changes in Laboratory Parameters and Medications
Serum levels of Ca, Pi, Ca × Pi product, i-PTH, albumin, CRP, and lipid profile did not show significant intergroup differences during the study period (Table 2). Table 3 presents medications. In the CC group, the initial and final doses of CC were 3000 ± 2500 mg/d and 3000 ± 1700 mg/d, respectively (Tables 1 and 3). In the LC group, the initial CC dose (3000 ± 2500 mg/d) was switched to a final LC dose of 1430.6 ± 652.0 mg/d. At the end of the study, 100% of the patients in the LC group were receiving vitamin D (calcitriol) compared with only 65.2% in the CC group (
Parameters at Randomization and Their Changes in CC and LC Groups.
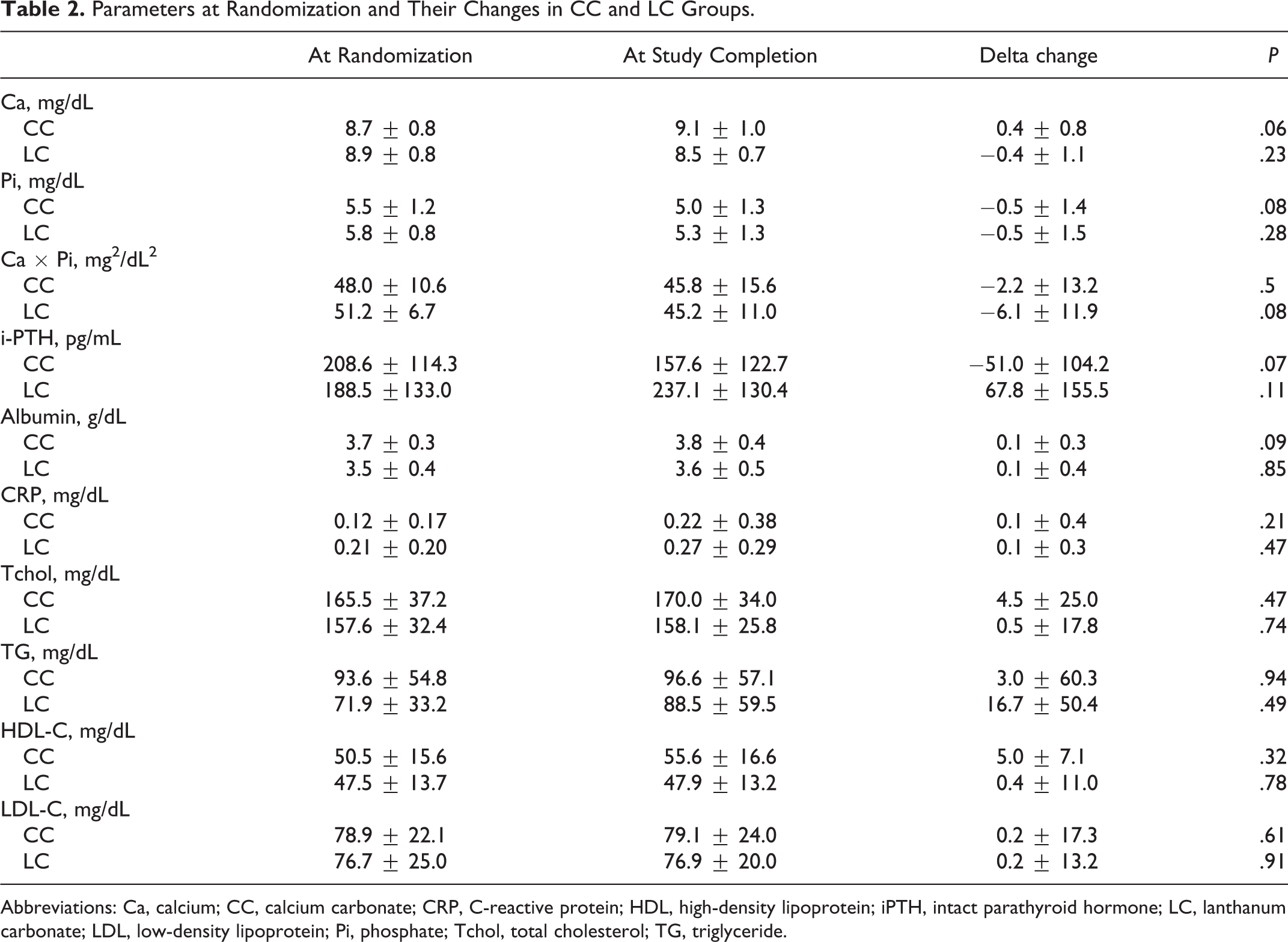
Abbreviations: Ca, calcium; CC, calcium carbonate; CRP, C-reactive protein; HDL, high-density lipoprotein; iPTH, intact parathyroid hormone; LC, lanthanum carbonate; LDL, low-density lipoprotein; Pi, phosphate; Tchol, total cholesterol; TG, triglyceride.
Medications at the End of the Study.

All patients in the LC group required vitamin D to control serum Ca and i-PTH at the end of the study period. Consequently, a difference in CAC scores may have been caused by the institution of vitamin D, rather than discontinuation of CC in the LC group. To consider this possibility, we restricted the analysis to patients who had already ingested vitamin D at randomization (CC group 15 of 23, LC group 13 of 19) and found that the change in log-transformed CAC score between 6 and 12 months was still significantly different (CC group: 0.071 ± 0.165 vs LC group −0.120 ± 0.282,
Change of the CAC Score
Heart rate during MSCT may influence CAC score measurement due to the potential motion artifacts. We did not use a β-blocker to control heart rate at MSCT. However, the heart rates were not significantly different between the CC and the LC groups (CC group: 67.6 ± 7.1/min, LC group: 66.5 ± 8.5/min,
Table 4 presents the CAC scores of the CC and LC groups at study enrollment, at randomization, and at the end of the 6-month intervention period. In the initial 6-month lead-in period, all patients were treated with CC. During this 6-month period, log-transformed CAC scores for all patients increased significantly from 2.625 ± 0.972 to 2.741 ± 0.861 (
CAC scores.

Abbreviations: CAC score, coronary artery calcification score; CC, calcium carbonate; LC, lanthanum carbonate; SD, standard deviation.
a
b
Once randomized, there was significantly less progression in the group treated with LC than with CC. In the CC group, log-transformed CAC scores showed a significant increase at 6 months after randomization (from 2.779 ± 0.736 to 2.880 ± 0.660,
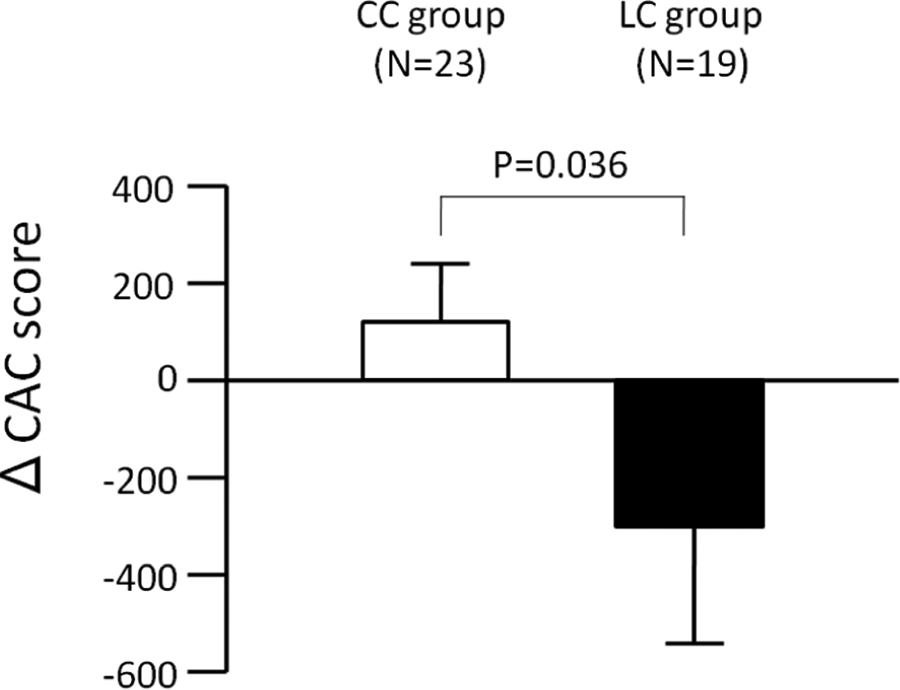
Changes in CAC score (ΔCAC score) in the 6-month intervention period. Open and closed bars represent ΔCAC score in the CC group (N = 23) and the LC group (N = 19), respectively. Data expressed as mean ± SE.. CAC indicates coronary artery calcification; CC, calcium carbonate; SE, standard error.

Percentage changes in CAC scores in the CC and LC groups. Gray bar displays percentage change in CAC scores in the 6-month lead-in period in a total of 42 patients. Open and closed bars display percentage change during the 6-month intervention period in the CC group (N = 23) and LC group (N = 19), respectively. Mean CAC score increased 41.2% in the CC group, while mean CAC score decreased 6.4% in the LC group (
Clinical Outcomes and Adverse Events
During the interventional period, 1 patient in the CC group died of pneumonia and 2 other patients experienced paroxysmal atrial fibrillation. In the LC group, 7 patients developed GI symptoms (nausea, 3 patients; abdominal discomfort, 2 patients; loss of appetite, 2 patients). Table 5 presents all adverse events in the CC and the LC group during the 6-month interventional period.
Adverse Events (AEs) During 6-Month Intervention Period.
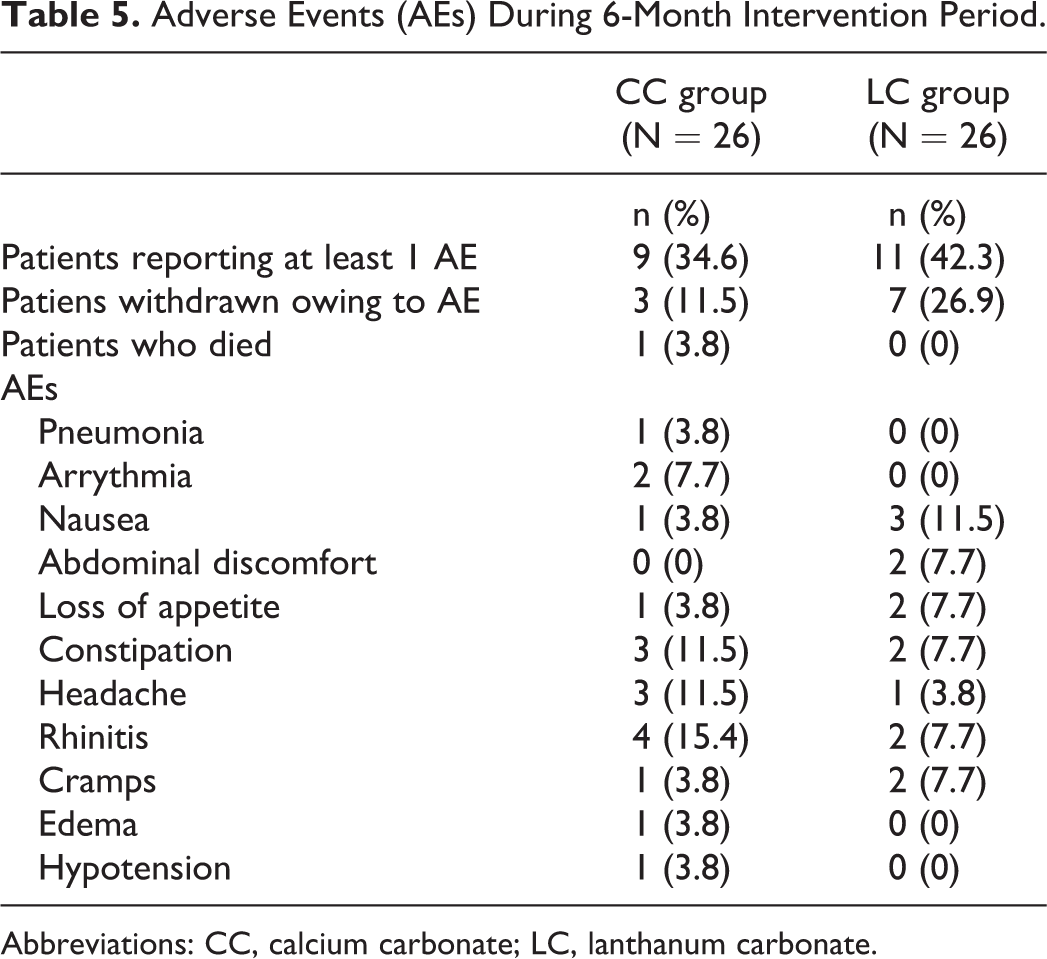
Abbreviations: CC, calcium carbonate; LC, lanthanum carbonate.
Discussion
Our study provided evidence that switching Pi binder from CC to LC delayed the progression of CAC in patients on HD. In the recent years, advanced CAC has been demonstrated to have significant influence on future cardiovascular events, cardiovascular death, and all-cause mortality in patients on HD. 2 –4 Therefore, delaying the progression of CAC may provide considerable benefit in patients on HD.
Switching the Pi binder from CC to LC decreased oral Ca load on patients in the LC group. The dose of CC was decreased from 3000 ± 2500 mg/d at study enrollment to 0 mg/d at the end of study in the LC group. This means that an oral Ca load of 1200 mg/d by daily CC ingestion (on average) was removed in the LC group. As a result of Ca load removal, serum Ca concentration was expected to decrease in the LC group. However, the decrease in serum Ca levels was prevented by the addition of vitamin D (1,25 (OH) D3) in the LC group. All patients in this group eventually required vitamin D by the end of the study. As a result, serum Ca and Pi levels were similar in both the groups throughout the study period. Despite maintenance of serum Pi and Ca levels within the target levels, i-PTH did not remain within the target range in some patients in the LC group. Such patients received cinacalcet to control i-PTH, although the mean i-PTH level was still above the target range in the LC group.
Despite the use of a low Ca dialysate (2.5 mEq/L), the CAC scores increased in patients who were administered CC as a Pi binder. In contrast, LC slowed or prevented the progression of CAC. Long-term use of LC with a low-Ca dialysate may achieve a negative Ca balance and thus affect bone turnover or remodeling. As a result, if LC is used as the sole Pi binder, the optimal dialysate concentration of Ca should be investigated in consideration of Ca balance, bone disease, and changes in CAC. The addition of low-dose CC to LC may have some benefit in this respect.
Serum Pi levels are an important prognostic factor in patients on HD. 20 Furthermore, recent studies have indicated that hyperphosphatemia itself is an important source of inflammation and may contribute to high cardiovascular risk in patients on HD. 21 Unfortunately, the removal of Pi by current dialysis methods is insufficient to maintain Pi levels within the recommended target range, and the majority of patients on HD require oral Pi binders. Aluminum hydroxide is a first-generation Pi binder that displays excellent Pi absorption, 22 but it has been known to cause various problems, including aluminum-associated encephalopathy and osteopathy, in patients undergoing dialysis. After treatment was switched to Ca-based Pi binders, vascular calcification of the aorta, 12,13,23,24 coronary arteries, 1 –12,25 and peripheral arteries 26 became a major problem, despite the use of dialysate with a low Ca concentration. In our study, CAC progressed in the CC group even though the Ca × Pi product was controlled to <50 mg2/dL 2 , and a low Ca dialysate (2.5 mEq/L) was employed. As a result, progression of CAC may be inevitable in patients on HD, and the Ca load created by Ca-containing Pi binders may further accelerate its progression.
Among the third-generation Pi binders (non-Ca-containing binders), sevelamer hydrochloride has been reported to slow the progression of CAC. 3,4,6,9 –12 In our study, LC was also shown to delay the progression of CAC significantly, compared with CC. Although no study has compared sevelamer and LC with respect to progression of vascular calcification, the Pi-binding capacity of LC is stronger than that of sevelamer. 22 A recent study indicated that sevelamer has a pleiotropic effect of reducing CRP in patients on HD. 27 In our study, we found no indication that LC has the same pleiotropic effect as sevelamer in reducing inflammation nor the clinical efficacy to reduce cardiovascular problems. A major limitation of this study was its small sample size and short follow-up period.
Because LC is a metallic compound, concerns might be raised as to whether the therapeutic use may result in deposition and/or toxic side effects, as seen in patients on dialysis with aluminum use in the past. Lanthanum has been reported to accumulate in bones and liver in uremic rats. 28 –30 In humans, however, GI absorption of lanthanum has been reported to be very low (0.00089%) compared with that of aluminum (0.02%-0.14%). 31,32 Plasma concentrations of lanthanum and aluminum in patients on dialysis have also been reported to differ significantly, that is, relatively low levels of lanthanum (approximately 0.6 ng/mL) compared with high aluminum concentrations (100 ng/mL or more). 33 Although 2 years of follow-up study indicated that LC was not associated with bone toxicity similar to that of aluminum, 33 only a prudent dosage is recommended for LC, keeping its metallic nature in mind.
Our pilot study has some limitations. First, the sample size was small, and the observation period was short. Second, CAC scores were widely distributed among the limited number of patients. These factors alone may prevent us from providing a definite conclusion. Third, although our pilot study suggested that LC may delay progression of CAC in patients on HD, compared with CC, long-term clinical outcomes are unavailable. Fourth, dropout in the LC group was high (26%) due to GI intolerance. Indeed, a previous study provided a high percentage of GI side effects in Japanese patients with HD (vomiting 32.4%, nausea 29.0%, stomach discomfort 16.6%, and upper abdominal pain 9.7%). 34 In our study, almost all GI symptoms in the LC group were mild to moderate. However, some patients could not adhere to LC administration. This factor may eliminate any advantage of LC.
In conclusion, our pilot study suggested that LC slows the progression of CAC compared with CC, but a large-scale, prospective, interventional study is needed to confirm this beneficial effect of LC. In the future, the optimal strategy for preventing CAC progression, controlling Ca and Pi balance, and improving bone disease could be clarified by combining all of the available treatments, including LC, low-Ca dialysate, vitamin D, and cinacalcet. Finally, the eventual goal is improvement in outcomes, not the surrogate of CAC progression itself; and outcome studies will be needed in order to determine whether switching Pi binder therapy from CC to LC leads to improved prognosis for patients on HD.
Footnotes
Acknowledgments
The authors thank Mr Satoshi Sekine (Chief Radiology Technician, Department of Radiology, Shonan Kamakura General Hospital) for calculating CAC scores using MSCT data. The authors also thank Mrs Yu Moriya (Secretary, Dialysis Center, Shonan Kamakura General Hospital) for managing the MSCT studies and data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
