Abstract
Background
Calcitriol (CAL), an active form of vitamin D, plays a vital role in controlling cardiac hypertrophy and heart failure. The aim of the present study is to explore the effects of CAL and to elucidate its underlying mechanisms on myocardial injury induced by isoproterenol (ISO).
Methods
Myocardial impairment was induced by the subcutaneous injection of ISO in adult male Sprague-Dawley rats, and the therapeutic effect of CAL was assessed. Biometric and echocardiographic parameters, interstitial fibrosis, oxidant–antioxidant status, and protein expression relevant to the mitochondrial apoptosis pathway were then measured.
Results
Calcitriol treatment improved the cardiac injury resulting from excessive ISO stimulation, as supported by the suppression of the development of myocardial hypertrophy, interstitial fibrosis, and H2O2 level in heart tissue. The decreased superoxide dismutase and catalase activities induced by ISO were also improved by CAL. Finally, the administration of CAL downregulated the protein expression of Bax and caspase-9.
Conclusions
This study provides evidence that CAL ameliorated cardiac hypertrophy, interstitial fibrosis, and oxidative stress in ISO-induced cardiac injury and might play a vital cardioprotective role in such injuries.
Keywords
Introduction
Excessive catecholamines result in one of the main pathophysiological mechanisms related to myocardial injury in cardiovascular diseases including cardiac hypertrophy, ischemia, and heart failure. 1 The pathogenesis of isoproterenol (ISO)-induced myocardial injury is associated with coronary microcirculatory effects, calcium overload, oxidative stress, and apoptosis.1–3 The ISO is a well-known inducer of oxidative stress within the heart. The ISO has been proven to produce free radicals and reduce the activities of antioxidants, which lead to severe impairment in the heart of rats. Cardiac injury induced by the administration of ISO is an easy, standardized method for evaluating the effects of many drugs on the heart.4,5
Vitamin D is known for its essential role in calcium and bone homeostasis. Most of vitamin D3 is synthesized in the skin upon sun exposure. Subsequently, vitamin D3 is converted into 1,25-dihydroxyvitamin D3 [calcitriol (CAL)] by 25-hydroxylase and 1α-hydroxylase. 24-Hydroxylase is responsible for the inactivation of CAL. The vitamin D receptors (VDRs), 1α-hydroxylase and 24-hydroxylase, are present in cardiomyocytes and fibroblasts, and VDR expression is upregulated with the application of a hypertrophic stimulus (endothelin in vitro and ISO in vivo). 6
Studies have reported that the vitamin D-VDR signaling system may play an important role in regulating cardiac hypertrophy and heart failure.7–9 The size of the left ventricular (LV) cardiomyocytes in VDR knockout (VDRKO) mice was markedly increased compared to those in wild-type (WT) mice. 7 Recent studies from Chen and colleagues have shown that VDR-dependent antihypertrophic activity in the myocyte was linked to the suppression of the calcineurin pathway. 8 In spontaneously hypertensive, heart failure-prone rats, low vitamin D status was associated with the progression and final terminal phase of heart failure. The CAL treatment provided protection against the progression of cardiac hypertrophy and heart failure. 9 These data strongly demonstrated that the vitamin D-VDR signaling system exerted an antihypertrophic effect on the heart. However, specific research focusing on the effects of CAL on cardiac ischemic injury due to ISO is still insufficient.
Thus, the present study aims to investigate the effects of CAL on cardiac hypertrophy, oxidant–antioxidant status, interstitial fibrosis, and expression of molecules relevant to the mitochondrial apoptosis pathway in rats with ISO-induced myocardial damage.
Materials and Methods
Animals and Drug Treatment
Adult male Sprague-Dawley rats (weighing 180-220 g) were provided by the laboratory at the Animal Center of the School of Medicine, Xi’an Jiaotong University. Rats were kept under a 12-hour light/dark cycle in a temperature- and humidity-regulated facility. Animals had free access to water and food. All animal care and experimental protocols were conducted in accordance with the Animal Management Rules of the Ministry of Health of the People’s Republic of China and the Guidelines for the Care and Use of Laboratory Animals of the School of Medicine, Xi’an Jiaotong University.
All rats were randomly divided into 3 groups(1) the ISO group (n = 6): ISO (Hefeng Co, Shanghai, China) was injected subcutaneously at 2.5 mg/kg/d for 7 days to induce cardiac hypertrophy, 5 then with normal saline subcutaneously injected at 0.1 mL per injection, 3 times a week for additional 6 weeks. (2) The CAL group (n = 6): ISO administration was the same as in the ISO group, followed with CAL (Roche, Ltd, Basel, Switzerland) subcutaneously injected at 30 ng/100 g, 3 times a week for additional 6 weeks. The dose of CAL was controlled so as not to increase the serum calcium level in accordance with a pilot experiment (data not shown). (3) The control (CON) group (n = 6): rats were subcutaneously injected with normal saline at 0.1 mL per injection for 7 weeks.
Echocardiographic and Biometric Parameters
At the end of 7 weeks, transthoracic echocardiography was performed using a 15L8-S transducer (Acuson Sequoia 512; Siemens, Munich, Germany) with a frequency of 8 to 14 MHz on all rats under pentobarbital sodium anesthesia (40mg/kg, intraperitoneally). Each rat was examined from parasternal left ventricle short-axis view at the level of papillary muscle. The M-mode echocardiography was used to record the motion of interventricular septum (IVS) and left ventricular posterior wall (LVPW). We measured the heart rate (HR), IVS thickness, and LVPW thickness at diastole, and LV end-systolic and end-diastolic dimensions (LVESD and LVEDD). The LV fractional shortening (FS) and ejection fraction (EF) were automatically calculated. All measurements represent the mean of 3 consecutive cardiac cycles.
After the echocardiographic parameters were measured, the body weights were determined. Subsequently, the hearts were dried using filter paper, and total heart weight (HW) was measured. Right ventricular myocardium was isolated by dissection along its septal insertion and discarded. Only LV myocardium was used for further analysis. Cross-section of the middle part of LV free wall was taken for pathological examination. The rest of the myocardial sample was used for the assessment of oxidant–antioxidant status and Western blot. All analyses were performed in all hearts.
Oxidant–Antioxidant Status
The myocardium was prepared into 10% homogenate in ice-cold normal saline. The supernatant was obtained from tissue homogenate by centrifugation (2000 rpm, 4°C, 15 minutes). The activities of superoxide dismutase (SOD) and catalase (CAT), as well as the H2O2 levels, were measured spectrophotometrically according to the Kit Handbook Guidelines (Nanjing Jiancheng Bioengineering Institute, China).
Histopathology
Myocardium tissues in the left ventricle were sampled and fixed in cold 4% paraformaldehyde for 24 hours, embedded in paraffin, and cut into slices 6 μm thick. The cardiac myocyte diameters were measured with the aid of an image analyzer Motic Image Med 6.0 System (Motic, Xiamen, China) in sections stained with hematoxylin and eosin. The diameter of 50 cardiac myocytes with a centrally located nucleus was measured in each heart (n = 6). The mean value was considered as the cardiac myocyte diameter value of the heart. Masson trichrome staining was performed to evaluate myocardial fibrosis. The myocardial fibrosis area was determined using Motic Image Med 6.0 System. 10 The collagen volume fraction (CVF) was calculated as the sum of all connective tissue areas divided by the sum of all connective tissue and muscle areas in the fields of the section (n = 6, 5 fields in each sample were scanned and averaged).
Western Blot
Myocardium samples were prepared by homogenization in NP-40 lysis buffer containing protease inhibitor cocktail (5%, final). The protein concentration was determined using the Bradford assay (Bio-Rad, Hercules, California). Equal amounts of samples were separated on 12.5% polyacrylamide gels and transferred to polyvinylidene difluoride membranes (Bio-Rad) in a transfer buffer containing 20 mmol/L Tris, 150 mmol/L glycine, and 20% methanol. The membranes were blocked with 5% nonfat milk in Tris-buffered saline and Tween 20 (TBST) for 1 hour at room temperature, then incubated with primary antibodies Bcl-2 (1:400), Bax (1:200) and caspase-9 (1:1000; Santa Cruz, California) at 4°C overnight. Membranes were washed with TBST (3 × 10 minutes). A subsequent incubation was performed with horseradish peroxidase-conjugated secondary antibody (1:1000; Santa Cruz) for 1 hour at room temperature. The membranes were washed with TBST (3 × 10 minutes) before exposure with an enhanced chemiluminescence detection system (Amersham, Shanghai, China). The band intensity was quantified by scanning the images of Western blot films using the Chemi-Genius Bio Imaging System (Syngene, Cambridge, UK). The results were normalized through respective levels of β-actin.
Statistics
Data were expressed as mean ± standard deviation. Differences among groups were tested by one-way ANOVA (from GraphPad Prism 4), followed by Tukey's test. A P value <.05 was considered statistically significant.
Results
Calcitriol Decreased the HW, HW/BW Ratio, and Cardiac Myocyte Diameter
The myocardial hypertrophy was induced by ISO administration in both ISO and CAL groups, evidenced by increase in the HW and HW–BW ratios against the control rats. Meanwhile, the HW and HW–BW ratios were significantly lower in the CAL group, compared with the ISO group (Table 1; P < .05). The diameter of LV cardiac myocytes in rats treated with ISO was enlarged compared to the CON group (Table 1; P < .05). This effect was markedly attenuated in CAL group compared with ISO group (P < .05). These data indicate that CAL served as an inhibitor against ISO-induced myocardial hypertrophy. Compared with the CON group, elevated HR was observed after ISO administration. But it seems likely that ISO-induced higher HR is not altered by CAL (Table 1).
Alterations of the Ratios of Cardiac Myocyte Diameter, HW/BW, and Echocardiographic Parameters in CON, ISO, and CAL groups (n = 6).

Abbreviations: BW, body weight; CON, control group; ISO, isoproterenol group; CAL, calcitriol group; HW, heart weight; HR, heart rate; IVS, interventricular septum; LVPW, left ventricular posterior wall thickness at diastole; LVESD, left ventricular end-systolic dimension; LVEDD, left ventricular end-diastolic dimension; EF, ejection fraction; FS, fractional shortening.
aP < .05 versus CON.
bP < .05 versus ISO.
Calcitriol Relieved ISO-Induced Cardiac Hypertrophy Detected by Echocardiograph
In rats treated with ISO, the IVS and LVPW increased by 29% and 35%, respectively, and the LVESD and LVEDD decreased by 31% and 16%, respectively, compared to the CON group (Figure 1, Table 1, all P < .05). These were associated with substantial increment in FS and EF by 21% and 9%, respectively (Table 1, all P < .05). These data indicate that the ISO induced cardiac concentric hypertrophy. Compared with the ISO-treated rats, the rats administered with CAL showed significant improvement in myocardial hypertrophy, with IVS and LVPW lowered by 14% and the LVESD and LVEDD elevated by 22% and 12%, respectively (Table 1, all P < .05). No statistical differences in EF and FS were observed between the ISO and the CAL groups. These data suggest the inhibitory effect of CAL on ISO-induced cardiac hypertrophy.
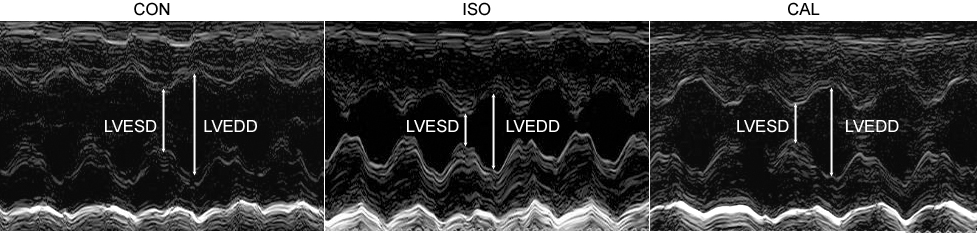
Representative echocardiograph (n = 6). CON indicates control group; ISO, isoproterenol group; CAL, calcitriol group; LVESD, left ventricular end-systolic dimension; LVEDD indicates left ventricular end-diastolic dimension.
Calcitriol Regulated Myocardial Oxidant–Antioxidant Status
Significant reductions in the activities of SOD and CAT by 18% and 22%, respectively, and a significant rise in the H2O2 contents by 59% (Table 2, all P < .05), were found in the myocardium of ISO-treated rats. Compared with the ISO group, the rats administered with CAL reversed the decreased SOD and CAT activities induced by ISO, with activities elevated by 16% and 20%, respectively (all Ps < .05). The CAL treatment also ameliorated the increased H2O2 contents induced by ISO, decreased by 24% (P < .05). Administration of CAL remarkably relieved the reduction of SOD and CAT activities and the increment of H2O2 contents induced by ISO.
Alterations of SOD Activity, CAT Activity, and H2O2 Contents in the Heart Tissue of CON, ISO, and CAL Groups (n = 6).
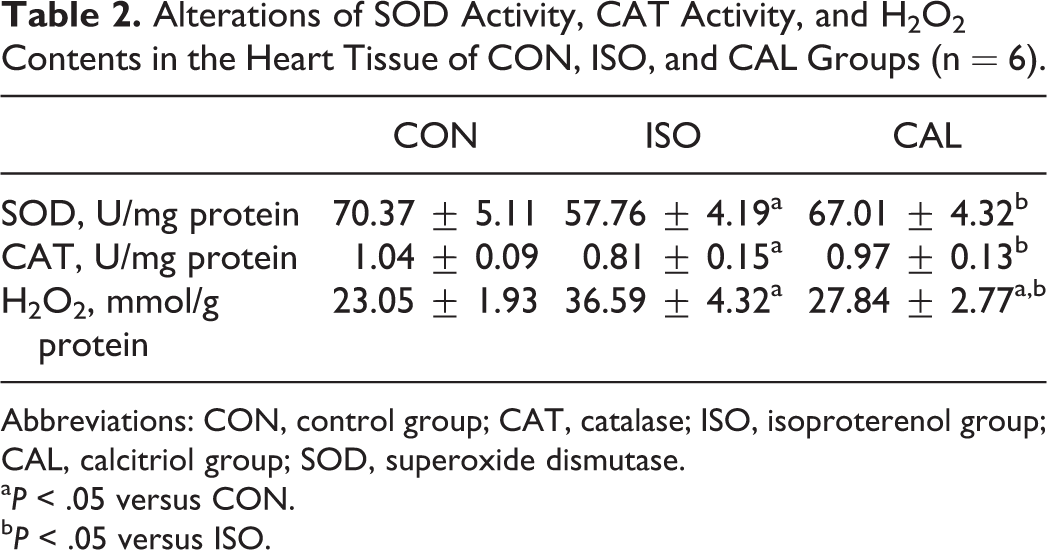
Abbreviations: CON, control group; CAT, catalase; ISO, isoproterenol group; CAL, calcitriol group; SOD, superoxide dismutase.
aP < .05 versus CON.
bP < .05 versus ISO.
Calcitriol Reduced Cardiac Interstitial Fibrosis
Histologically, ISO-treated hearts showed extensive myocardial structural disorder and subendocardial necrosis, which does not extend to the epicardium. As shown in Figure 2, myocardial fibrosis was detected using Masson’s trichrome staining. Blue staining indicates connective tissue. The staining and histogram of CVF revealed that the ISO-treated rats developed marked interstitial fibrosis. The CAL attenuated the ISO-induced collagen deposition.
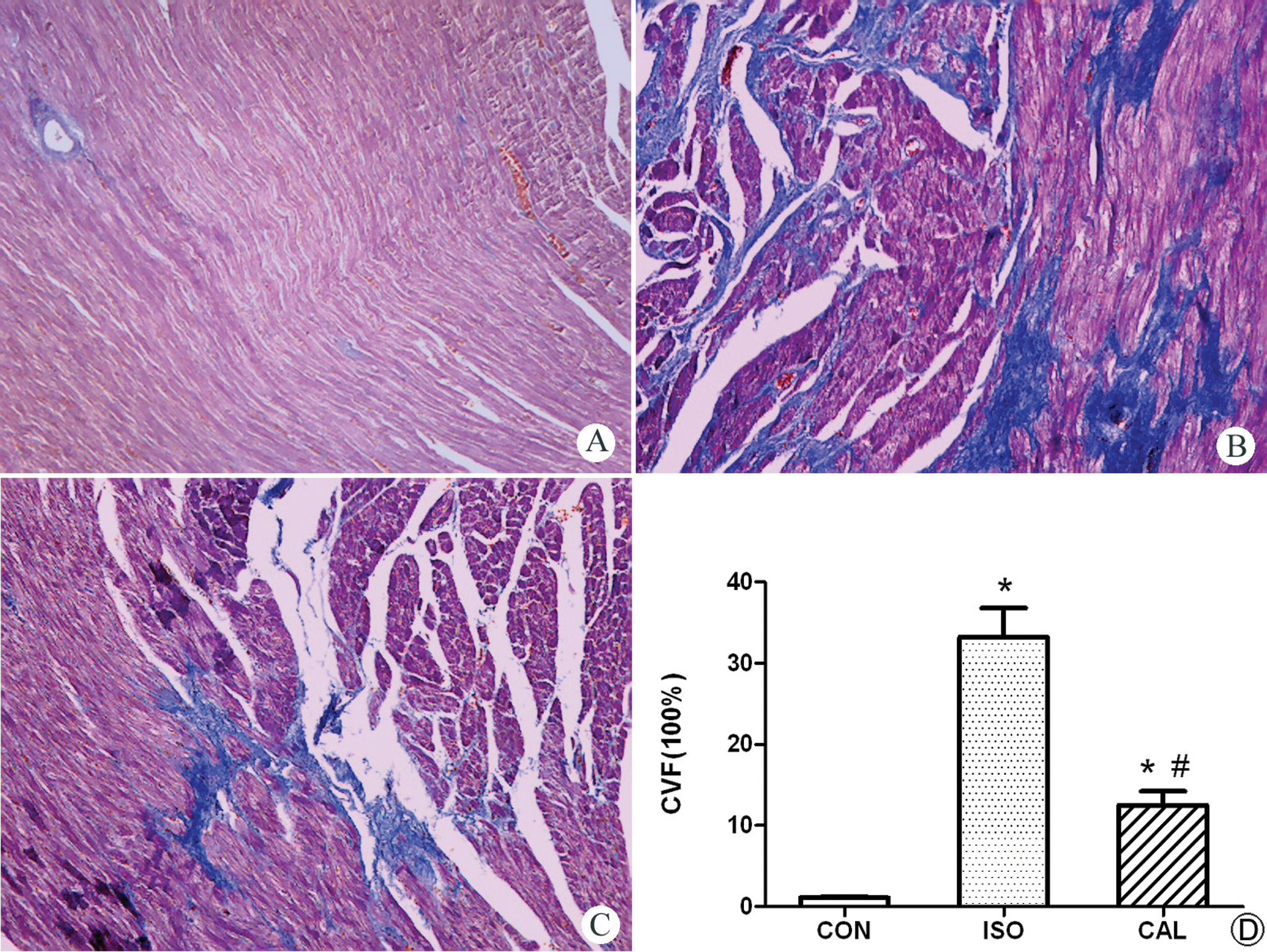
Effect of CAL on cardiac interstitial fibrosis (n = 6). Histopathologic sections stained with Masson trichrome stain (blue: collagen fibers) revealed decreased fibrosis in (C) CAL group compared to that in (B) the ISO group. A, CON group; (D) collagen volume fraction comparison between groups. *P < .05 versus CON; #P < .05 versus ISO (×100). CON indicates control group; ISO, isoproterenol group; CAL, calcitriol group.
Calcitriol Inhibited the Expressions of Bax and Caspase-9 Proteins
Figure 3 shows the expression of Bax and caspase-9 proteins in LV myocardium by Western blot and subsequent quantification analysis. Compared with the CON group, the well-known proapoptotic proteins Bax and caspase-9 were markedly increased in the ISO group (all P < .05). Furthermore, compared with the ISO group, the administration of CAL attenuated the expressions of Bax and caspase-9 in the CON group (all P < .05). No significant differences were observed in Bcl-2 expression among the 3 groups (data not shown).

Effect of calcitriol (CAL) on apoptosis-related protein expression (n = 6). Bax and caspase-9 expressions, as detected by Western blot, indicate a statistical difference in the expression of Bax and caspase-9 proteins between groups (right). *P < 0.05 versus CON; #P < 0.05 versus ISO. CON indicates control group; ISO, isoproterenol group; CAL, calcitriol group.
Discussion
A growing body of evidence affirms that the alterations in vitamin D metabolism are common in patients with heart failure.11,12 We undertook this study to evaluate the influence of CAL on ISO-induced cardiac impairment in rats and to examine the mechanisms involved. The data showed that CAL treatment improved the cardiac effects of excessive stimulation by ISO, as evidenced by extenuated myocardial hypertrophy, interstitial fibrosis, and H2O2 levels in the heart tissue. The CAL improved the impaired SOD and CAT activities caused by ISO. The administration of CAL also downregulated Bax and caspase-9. These results suggest that CAL might play a vital cardioprotective role in ISO-induced myocardial injury.
Chronic stimulation with ISO has been proven to induce cardiac hypertrophy in rats. 5 In the present study, ISO-induced cardiac concentric hypertrophy was indicated by the increase in IVS and LVPW and a decrease in LVESD and LVEDD. The current observations are in accordance with previous research.7–9 The echocardiographic characteristics modified by CAL are confirmed with the depressed cardiac hypertrophy, as confirmed by the HW–BW ratio and cardiac myocyte diameter. The suppression of cardiac hypertrophy, combined with the preservation of cardiac function, represents the beneficial effects of CAL during the progressive transition from compensated cardiac hypertrophy to heart failure under β-adrenoceptor stimulation.
Oxidative stress injury is one of the main underlying mechanisms of ISO-induced myocardial damage.1,13–15 The damage caused by oxidative stress reflects the imbalance between the production of reactive oxygen species (ROS) and the antioxidant defense. Our results are in accordance with previous reports on decreased levels of endogenous antioxidants, such as CAT and SOD, along with increased H2O2 levels following chronic ISO administration.16,17 The CAL treatment reduced H2O2 content and restored SOD and CAT activities in heart tissue, suggesting that the antiperoxidation potency of CAL is involved in heart protection. In addition, the decrease in H2O2 levels caused by CAL may be associated with the inhibition of the angiotensin II pathway. Grimm et al reported that both the circulating and the cardiac renin–angiotensin systems were activated during the initial weeks after the ISO-induced myocardial insult. 18 Angiotensin II initiates oxidative stress in cardiovascular systems by inducing ROS via the activation of nicotinamide adenine dinucleotide phosphate-oxidase.19,20 Vitamin D can act as an inhibitor of the renin–angiotensin system by suppressing renin transcription, both in vitro and in vivo.21,22 We speculate that the inhibition of the angiotensin II pathway by CAL could be another possible mechanism that results in reduced H2O2 production.
In the present study, the overt myocardial fibrosis following ISO treatment was greatly alleviated by CAL. Enhanced oxidative stress in ISO-induced cardiac hypertrophy is associated with elevated extracellular matrix biosynthesis. 15 Therefore, the inhibition of cardiac oxidative stress by CAL is a possible underlying mechanism for the protective effect of CAL against ISO-induced cardiac fibrosis. Furthermore, compared with the WT mice, the VDRKO mice showed increased collagen deposition with a concomitant increase in metalloproteinase (MMP)-2 and -9 activities. This observation in VDRKO mice was correlated with the lowered activity of tissue inhibitors of metalloproteinases (TIMPs). 23 These results indicate that the MMPs/TIMPs system might be another target site for the vitamin D-VDR signaling system in regulating cardiac collagen deposition. However, extrapolating the regulatory mechanism requires further experimental evidence.
The cardiomyocyte apoptosis induced by ISO stimulation plays an important role in myocardial remodeling.24,25 The alteration of apoptosis-related protein signals occurred whether or not the apoptosis was initiated. In our study, the expressions of Bax and caspase-9 were upregulated in ISO-induced rats. The ability of CAL to reduce the elevated expressions of Bax and caspase-9 induced by ISO indicates that CAL might inhibit apoptosis through the modulation of proteins associated with the mitochondrial pathway of apoptosis.
There is extensive evidence to support the role of vitamin D in protecting heart through both the direct and the indirect effects. The direct inhibition of renin expression by CAL has been confirmed both in vitro and in vivo.21,22 The VDRKO is associated with cardiac fibrosis and the MMP/TIMP disturbance. 23 Our data show that CAL can alleviate collagen deposition. Dong et al reported that CAL may improve endothelial function in hypertension by downregulating angiotensin II type 1 receptors and reducing oxidative stress. 26 Vitamin D has indirect actions to maintain cardiovascular function through regulating parathyroid hormone (PTH) and calcium level. The PTH may increase arterial stiffness, 27 and vitamin D-induced transrepression by VDR was confirmed in the human PTH gene promoter. 28 The dose of CAL in the present study was not to increase the serum calcium level in accordance with a pilot experiment.
Taken together, our data substantiate the theory that myocardial injury caused by excessive ISO can be improved by the chronic administration of CAL. This is evidenced by an improvement in the progression of myocardial hypertrophy and interstitial fibrosis, the lowering of H2O2 levels and of the protein expressions of Bax and caspase-9, and the elevation of SOD and CAT activities. The present study suggests the role of CAL as an endogenous cardioprotective steroid.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundamental Research Funds for the Central Universities of China (xjj20100041).
