Abstract
Dronedarone is a new antiarrhythmic drug for patients with nonpermanent atrial fibrillation (AF). A relatively consistent finding in all trials studying dronedarone was a moderate but significant elevation of serum creatinine. Since dronedarone competes for the same organic cation transporter in the distal renal tubule with creatinine, serum creatinine and its derived estimated glomerular filtration rate might not reflect true renal function in patients on dronedarone. We therefore investigated alternative markers for renal function in these patients. We prospectively included 20 patients with nonpermanent AF in whom dronedarone 400 mg twice daily was started. Patients had normal renal function and serum creatinine; serum cystatin C and creatinine clearance were measured before treatment and 10 and 90 days after treatment started. Mean serum creatinine level for all 20 patients at baseline (day 0) was 84.55 ± 12.14 and 87.8 ± 17.59 µmol/L on day 10. This slight increase in all patients was not significant. Patients were now divided into the predefined groups of “increased creatinine” (increase in serum creatinine level > 1 standard deviation) and “not increased creatinine.” Patients with increased creatinine levels (n = 5) showed a significant elevation of serum creatinine levels from day 0 to day 10 (82.4 ± 9.18 to 104.4 ± 12.74 µmol/L; P = .003), whereas change in serum creatinine levels in the not increased creatinine group (n = 15) was not significant. Serum cystatin C levels remained stable in both of these groups (increased creatinine group: 0.76 ± 0.08 to 0.78 ± 0.08 mg/L; P = .65; not increased creatinine group: 0.77 ± 0.108 to 0.77 ± 0.107 mg/L; P = .906). In conclusion, cystatin C represents an easily available and reliable biomarker for estimation of true renal function in patients on dronedarone treatment.
Introduction
Atrial fibrillation (AF) is the most common acquired sustained arrhythmia. 1,2 It is associated with a reduction of quality of life and an elevated risk of stroke and death. 3 Dronedarone is a new noniodinated benzofuran derivative with a chemical similarity to amiodarone 4 used as an alternative antiarrhythmic drug in patients with nonpermanent AF. 5 It was shown to be effective in prolonging the AF-free survival, 6 stabilizing sinus rhythm, 7 reducing heart rate in patients with permanent AF, 8 and reducing number of hospitalizations 9 and the total hospitalization burden 10 due to cardiovascular events in patients with AF. Atrial fibrillation recurrence is less decreased by dronedarone than by amiodarone, but dronedarone showed a better safety profile. 11 Despite these promising results, dronedarone caused an increased mortality in patients with severe heart failure with reduced left ventricular function 12 and in patients with permanent AF. 13
A consistent finding in all trials investigating dronedarone was a significant elevation of serum creatinine levels (2.4%-4.7%) in patients receiving dronedarone compared with placebo. 7 -9,12 In healthy participants, dronedarone was shown to reduce the creatinine clearance by 18% without substantially influencing the glomerular filtration rate (GFR). 14 Creatinine is freely filtrated in the kidney without tubular reabsorption or renal metabolization. However, there is an additive tubular secretion of creatinine into the primary urine (10%-40%), which can lead to an overestimation of creatinine clearance. 14 The increase in serum creatinine levels in patients receiving dronedarone is probably due to inhibition of the organic cation transporter responsible for the tubular secretion. 14 After discontinuation of medication, serum creatinine levels always normalized.
Thus, if a creatinine-based calculation of GFR is applied after administration of dronedarone (eg, according to the Modification of Diet in Renal Disease study or Cockgroft-Gault), an apparently reduced GFR might be deducted. This occurred in the ANDROMEDA trial (European Trial of Dronedarone in Moderate to Severe Congestive Heart Failure9), presenting the estimated GFR based on serum creatinine levels which showed a putative reduction of GFR by 7 mL/1.73 m2 compared to placebo (P = .009). 12
On the basis of these findings, it is recommended to control serum creatinine levels after initiation of dronedarone therapy. Nevertheless, this recommendation does not help to elucidate the true renal function. We therefore tried to investigate alternative markers for renal function in these patients.
Cystatin C represents a well-established parameter in renal function monitoring. 15 Its elimination is completely dependent on glomerular filtration, thus immediately indicating a decline in GFR by an increase in serum levels. 15 Serum cystatin C was shown to be superior to creatinine as a marker of GFR in several studies. 16 We hypothesized that cystatin C serum levels would remain constant in patients showing an increase in serum creatinine levels after initiation of dronedarone therapy. This would indicate a more reliable parameter for monitoring the renal function in these patients.
Methods
Between March 2010 and June 2011, 20 patients received dronedarone for treatment of AF according to guidelines. Patients were excluded when presented with elevated serum creatinine levels (women >84 µmol/L, men >104 µmol/L), previous amiodarone or dronedarone therapy in the last 2 months, hypo- or hyperthyroidism, receiving glucocorticoids, or taking any other cytochrome P450 34A isoenzyme inhibitor. Serum levels of creatinine, cystatin C, and urea were measured before treatment, on day 10, and after 3 months. Creatinine clearance was measured via 12 hour urine samples on the day before treatment and on day 10 of treatment and calculated using a formula (urine volume [mL] × urine creatinine level [mmol/L] ×1000 × 1.73)/(720 min × serum creatinine level [µmol/L] × body surface [m2]) × mL/min). Preliminary pharmacokinetic studies showed that concentrations of dronedarone and its main metabolite SR35021 reach a steady state within 14 days 6 and increase in creatinine levels occurred within 7 to 10 days of treatment. 7,9,12 Blood samples were taken in the morning, fasted, and in horizontal position.
Two groups of patients were defined: patients with an increase in serum creatinine level from day 0 to 10 of more (increased creatinine) or less (not increased creatinine) than 1 standard deviation.
Statistical analyses were generated using GraphPad Prism (Version 5.04, GraphPad Software, Inc, La Jolla, California). Comparison of the 2 defined groups was made by Student t test (level of significance .05).
Results
A total of 20 patients aged 36 to 73 (mean 54) years were included. Patient characteristics and laboratory findings are shown in Table 1. Of all, 17 patients were without any structural heart disease, 1 patient had a coronary heart disease, 1 patient had a mild hypertrophic cardiomyopathy, and 1 patient showed signs of an athlete’s heart. All patients had normal thyroid function.
Basic Characteristics and Laboratory Findings.a
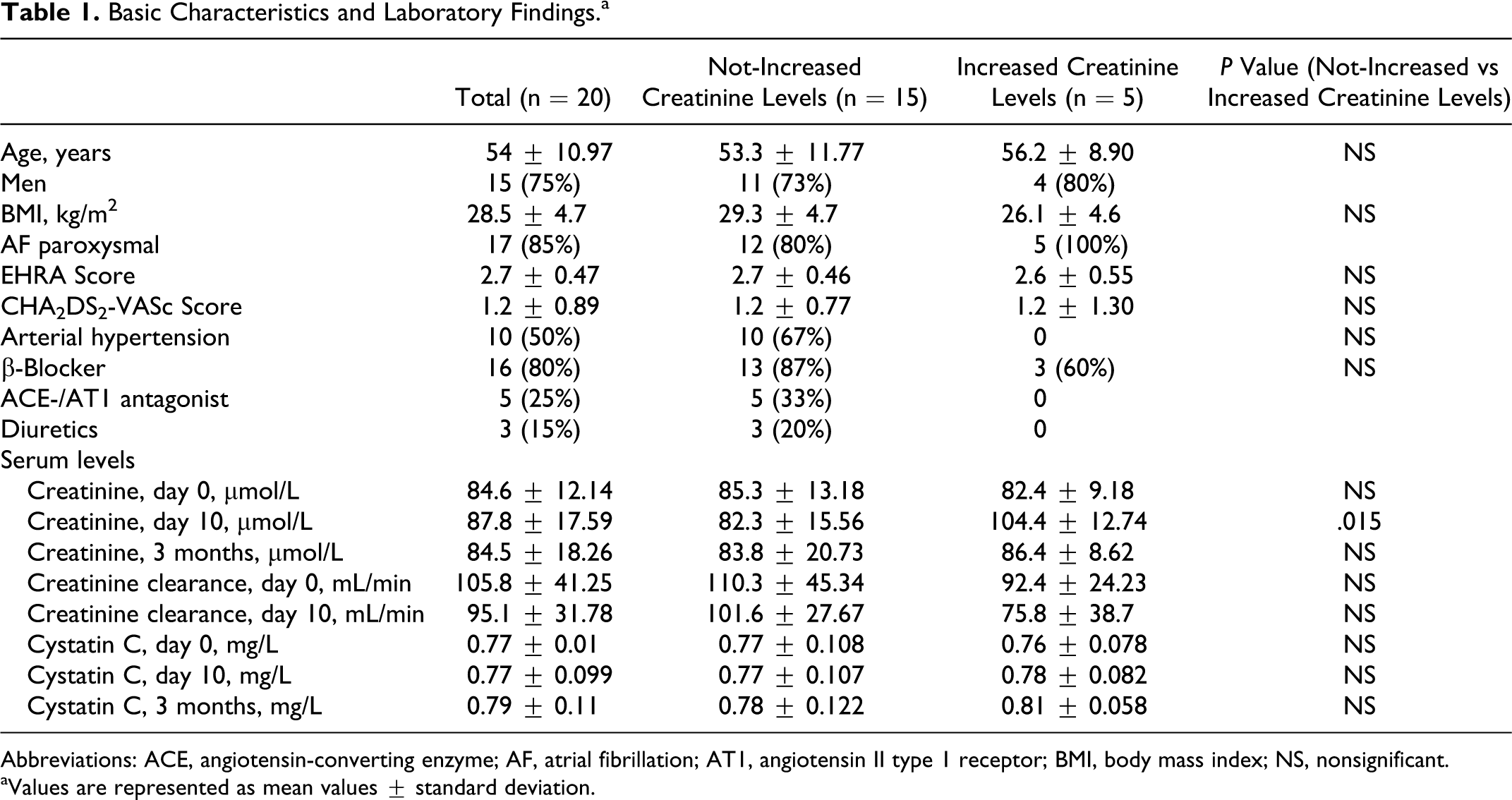
Abbreviations: ACE, angiotensin-converting enzyme; AF, atrial fibrillation; AT1, angiotensin II type 1 receptor; BMI, body mass index; NS, nonsignificant.
aValues are represented as mean values ± standard deviation.
Mean serum creatinine level for all patients at baseline (day 0) was 84.6 ± 12.14 µmol/L and 87.8 ± 17.59 µmol/L on day 10. This slight increase in all 20 patients was not significant. Patients were now divided into the predefined groups of increased creatinine and not increased creatinine. Patients with increased creatinine levels showed a significant elevation of serum creatinine levels from day 0 to 10 (82.4 ± 9.18 to 104.4 ± 12.74 µmol/L; P = .003), whereas change in serum creatinine levels in the not increased creatinine group was not significant. Serum cystatin C levels remained stable in both of these groups (increased creatinine group: 0.76 ± 0.08 to 0.78 ± 0.08 mg/L; P = .65; not increased creatinine group: 0.77 ± 0.108 to 0.77 ± 0.107 mg/L; P = .906; Figure 1). The creatinine and cystatin C levels after 3 months did not show any significant differences, neither compared with baseline values nor in the 2 groups of increased and not increased creatinine. All parameters are presented in detail in Table 1. No serious adverse events were observed during dronedarone treatment. No increase in liver enzymes was observed.

Change in cystatin C serum levels under dronedarone treatment (before and 10 days after beginning of treatment) according to patients with increased serum creatinine (A and C) or not-increased serum creatinine (B and D).
Discussion
Dronedarone is a new antiarrhythmic agent available for the treatment of nonpermanent AF. One detected side effect may be an increase in serum creatinine levels without substantially affecting the GFR. This effect was shown in healthy participants 14 and was a consistent finding in all clinical trials dealing with dronedarone. This effect may have an impact on clinical decision making, since a substantial number of patients with AF may take comedication requiring dose adjustment according to renal function. Common drugs which have to be considered in this respect are angiotensin-converting enzyme (ACE) inhibitors and angiotensin antagonists, requiring dose reduction in case of an increase in creatinine levels. However, dose reduction of ACE inhibitors or angiotensin-antagonists prompted by increased creatinine levels under dronedarone treatment might expose patients to an unwanted and inadequate renin–angiotensin rebound.
In order to facilitate decision making in these patients, we therefore investigated serum levels of an alternative and well-established biomarker for renal function, cystatin C, in patients receiving dronedarone.
Our results showed a significant increase in creatinine serum levels of more than 1 standard deviation (14 µmol/L) in 5 patients compared to 15 patients with a stable serum levels. The creatinine increase is consistent with those increases found in former dronedarone trials. Both ERATO 8 and DIONYSOS 11 reported a mean elevation of serum creatinine in the dronedarone treatment arm of 10.6 and 10 µmol/L, respectively. In the ATHENA trial, the dronedarone group showed an increase in creatinine levels in 4.7% of patients versus 1.3% in the placebo group (P < .001). 9 The EURIDIS/ADONIS trial presented similar results concerning creatinine levels (increase in serum creatinine levels in 2.4% of patients in the dronedarone group vs 0.2% in the placebo group, P = .004). 7 The increase in serum creatinine levels in the dronedarone group in the ANDROMEDA trial (2.8% vs 0% placebo, P = .01) resulted in a significant decrease in estimated GFR of 7 mL/1.73 m2 versus placebo (P = .009). 12 In our study, cystatin C levels remained stable despite serum creatinine elevation. Therefore, cystatin C represents a more reliable, dronedarone-independent marker truly reflecting renal function in patients receiving dronedarone. Nevertheless, despite its efficiency in this constellation, cystatin C remains a considerable cost factor implementing higher laboratory fees.
One limitation of our study surely remains the small number of patients included. This is certainly, among other things, due to the chosen strict exclusion criteria defining the study group. This implies that a somewhat greater number of patients remains unrepresented in this study. Our findings, therefore, do not give any evidence about the course of cystatin C in patients with preexisting impaired renal function. However, the former dronedarone trials also did not include patients with renal impairment.
Conclusion
In conclusion, cystatin C represents an easily available and reliable biomarker for estimation of true renal function in patients on dronedarone treatment. We suggest including cystatin C serum levels for clinical decision making in patients taking dronedarone. Further studies may investigate the relevance of serum cystatin C levels compared to serum creatinine levels in patients with known renal insufficiency under dronedarone treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
