Abstract
Introduction
Metabolic syndrome is a common chronic disorder that is associated with significant cardiovascular disease risk. Clinical studies indicated that cardiac remodeling frequently occurs among patients with metabolic syndrome, with ventricular hypertrophy and heart failure as early manifestations. 1,2 The pathogenesis of left ventricular (LV) hypertrophy or dysfunction in metabolic syndrome is complex. It may involve the interplay of several factors, such as insulin resistance, increase in energy intake, decrease in energy consumption, disturbed fat utilization and storage, aggregation, and ectopic distribution of visceral adipose tissues, and adipocyte hypertrophy. 3,4 Thiazolidinediones are insulin sensitizers that are effective in the improvement of insulin resistance in peripheral tissues, but their effect on cardiac remodeling and function remains controversial. 5
In this study, a lipotoxic rat model was developed by feeding a high-fat (HF) diet. This rat model shares many features with human metabolic syndrome and thus has been used to study metabolic syndromes in several other studies. 6 Through this model, we investigated the effect of pioglitazone, a thiazolidinedione, on HF-diet–induced LV hypertrophy and dysfunction. We also evaluated myocardial expression of renin–angiotensin system following pioglitazone treatment in these animals.
Materials and Methods
Animal Models
A total of 36 male Sprague-Dawley rats (aged 8 weeks, body weight 230-270 g) were housed in groups in an accredited animal house at China–Japan Friendship Hospital. All animals had free access to feed and drinking water. Housing was at a constant temperature of 21°C ± 1°C in a 12-hour light/dark cycle. This study was approved by the institution review board, and all animal care and experimental procedures were in accordance with the guidelines of the Animal Care Committee of the China–Japan Friendship Hospital.
The rats were randomly divided into 3 groups by random drawing of numbers: a control group (n = 12), a high-fat diet group ([HF group] n = 12), and a pioglitazone treatment group (n = 12). Rats in the control group were fed with a normal diet for 30 weeks, with fat accounting for 10% of the total calories. Rats in HF and pioglitazone groups were fed with a HF diet for 30 weeks, with fat accounting for 66% of the total calories. In the pioglitazone group, pioglitazone (10 mg/kg per d) was administered via a gastric tube in the last 6 weeks of the 30-week HF diet treatment. Normal diet was purchased from Keao Xieli Feed Co Ltd (Beijing, China), with a total calorie of 349 kcal/100 g. High-fat feed was prepared by the Keao Xieli Feed Co Ltd, with a total calorie of 445 kcal/100 g and stored in a refrigerator at −20°C.
Hyperinsulinemic–Euglycemic Clamp
At the end of the experiment, hyperinsulinemic–euglycemic clamp technique was performed in all animals as described previously by Kraegen et al. 7 In brief, after fasting for 8 to 10 hours with free access to water, rats were constantly examined with a hyperinsulinemic clamp (12 mU/kg per min of human insulin; Novo Nordisk A/S, Bagsværd, Denmark). Glucose concentrations were clamped at euglycemic levels (5 mmol/L) by intravenous infusion of 20% glucose. Plasma glucose concentration was determined with a Beckman Glucose Analyzer II (Beckman Instruments, Fullerton, California). Glucose infusion rate was used as a measure of insulin sensitivity.
Cardiac Function Evaluation
At the end of the experiment and before euthanasia, echocardiography was performed to measure cardiac structure and function. Under general anesthesia (3% sodium pentobarbital), M-mode echocardiography was performed to measure LV diastolic and systolic dimension, thickness of LV posterior wall in the end-systolic and end-diastolic phase, LV ejection fraction (LVEF), fraction of shortening, cardiac index, and heart rate. Three cardiac cycles were recorded and the mean of the above indices from the 3 cardiac cycles was computed.
Following echocardiographic examination, a small cardiac catheter was inserted into the left ventricle through the carotid artery. The maximum rising and dropping speeds of internal pressure in the left ventricle (+dp/dt max and −dp/dt max) were recorded to represent LV function.
Rats were subsequently euthanized and the heart was harvested. Heart chambers were washed with normal saline and the wet weight of the heart was measured. The left ventricle was isolated and its weight was measured to calculate the ratio of the LV weight and whole heart weight (LV weight index).
Biochemical Measurements
On the same day of echocardiographic examination, venous blood was drawn to measure plasma free fatty acid (FFA) and serum triglycerides. Free fatty acid was determined with enzyme colorimetric analysis using a measurement kit produced by British Randox Corp (Crumlin, UK) and 7170A HITACHI Auto Biochemical Analysis System (Hitachi Co, Ltd, Tokyo, Japan). Biochemical and histopathological studies were also performed on harvested heart tissues. Triglycerides in myocardial tissues were extracted by Folch methods. 7 Myocardial triglycerides were determined with a triglyceride analysis kit (Heguang Pure Medicine Industry Corp, Osaka, Japan) and an Olympus AU400 Auto Biochemical Analysis System (Hamburg, Germany).
Expression of Angiotensin Types 1 and 2 Receptors and Angiotensin II in Myocardial Tissues
Immunohistochemical method was employed to detect angiotensin types 1 and 2 (AT1/AT2) receptors and angiotensin II (Ang II) proteins in myocardial tissues. A 2-step Elivision method was used. Results of coloration were collected through image acquisition using Olympus BX51 microscope (Olympus, Hamburg, Germany). Image analysis system IPP5.1 was used for quantitative analysis. At least 3 tissue slices were required to measure the total area of color-positive sector in order to number the cells expressing these proteins in myocardial tissues and to measure the mean optical density to detect the expression of these proteins in cells per unit area. The expression of the proteins in myocardial tissues was represented with the integral optical density by multiplying the mean optical density with the total area.
Isolation of Total RNA and Real-Time Polymerase Chain Reaction
Total RNA was isolated from the endothelial cells using a Trizol reagent (Gibco BRL, Invitrogen Corp, Carlsbad, California). Complementary DNA (cDNA) synthesis was performed using a ReverTraAce-α Kit (Toyobo Co Ltd, Osaka, Japan), according to the manufacturer’s instructions, in a total volume of 20 mL under conditions of 42°C for 20 minutes, 99°C for 5 minutes, and 4°C for 5 minutes.
Quantitative real-time polymerase chain reaction (PCR) amplification was performed using SYBR Green PCR Master Mix (Toyobo Co Ltd) in a total volume of 50 µL, according to the manufacturer’s instructions. The reaction conditions were 40 cycles of 95°C for 1 minute, 95°C for 15 seconds, 55°C to 60°C for 15 seconds, and 72°C for 45 seconds. The amplification was carried out using an ABI 7300 Sequence Detection System (Applied Biosystems, Foster City, California). The primers were synthesized by Shanghai Sangon Biological Engineering Technology and Services Co Ltd (Shanghai, China). The sequences of the primers are shown as follows. Renin: 5′-GCTACATGGAGAATGGGACTGAA-3′ (forward), 5′-ACCACATCTTGGCTGAGGAAAC-3′ (reverse). Ang II: 5′-AGCACGGACAGCACCCTATT-3′ (forward), 5′-AGAACTCATGGAGCCCAGTCA-3′ (reverse). AT1: 5′-TATCACAGTGTGCGCGTTTCA-3′ (forward), 5′-TGGTAAGGCCCAGCCCTAT-3′ (reverse). AT2: 5′-CCTTCCTGTATTGTTTCGTTGGA-3′ (forward), 5′-CGGCAAGACATAGTCTCTCTCTTG-3′ (reverse).
Statistical Analysis
All values are presented as mean ± standard deviation. The number of animals in each group was determined by a Power analysis prior to the experiment. Statistical significance of differences among groups was evaluated by 1-way analysis of variance (ANOVA). Tukey test was used in conjunction with ANOVA to find which means are significantly different from another. All tests were performed using SPSS program (Chicago, Illinois). Significance was determined by P < .05.
Results
General Findings
All animals completed the experiment. As shown in Table 1, there was no statistically significant difference in the mean body weight and systolic blood pressure between the 3 groups at the beginning of the study (week 0). On week 24, there was an increase in the mean body weight and systolic blood pressure in all groups (P < .01). The mean body weight and systolic blood pressure were similar between the HF and pioglitazone groups (P > .05), but they were higher than that in the control group (P < .01). At week 30, the mean body weight and blood pressure remained higher than in week 0 in all groups (P < .01), and those in the HF and pioglitazone groups were higher than that in the control group (P < .01). There was no statistically significant difference in the mean body weight between pioglitazone and HF groups (P > .05) at week 30, but the mean systolic blood pressure in pioglitazone group was lower than that in HF group (P < .01).
Changes in Body Weight and Systolic Blood Pressure

Abbreviations: BP, systolic blood pressure measured from tail of the animals.
a P < .01 when compared with week 0 in the same group.
b P < .01 when compared with the control group.
c P < .01 when compared with high-fat diet group.
Effect of Pioglitazone on Plasma FFA, Serum, and Myocardial Triglycerides
As shown in Table 2, the levels of plasma FFA, serum, and myocardial triglycerides in the HF group were 50.8%, 56.3%, and 164.5% higher, respectively, than that in the control group (P < .01). The levels of plasma FFA, serum, and myocardial triglycerides in pioglitazone group were 20.5%, 20.1%, and 37.1% lower, respectively, than that in the HF group (P < .01).
Comparison of FFA, Myocardial TG, and Glucose Infusion Rate Among 3 Groups at Week 30

Abbreviations: FFA, free fatty acid; GIR, glucose infusion rate, TG, triglyceride.
a P < .01 when compared with control group.
b P < .01 when compared with high-fat diet group.
Insulin Resistance
At the end of the study, glucose infusion rate in the HF group was lower than in the control group (P < .01) or pioglitazone group (P < .01; Table 2).
Left Ventricular Hypertrophy and Function
At the end of the study, there was no significant difference in the mean heart rate between the 3 groups (P > .05; Table 3). The thickness of LV posterior wall in HF and pioglitazone groups was greater than in the control group (P < .05). Left ventricle weight index in the HF and pioglitazone groups was also higher than in the control group (P < .01). There was no significant difference in wall thickness or LV weight index between HF and pioglitazone groups (P > .05). Left ventricular ejection fraction, fraction of shortening, and cardiac index in HF group were lower than in control or pioglitazone groups. Left ventricular +dp/dt max and −dp/dt max in the HF group were also lower than in the control and pioglitazone groups (P < .01).
Comparison of Ventricular Wall Thickness and Function Among 3 Groups at Week 30
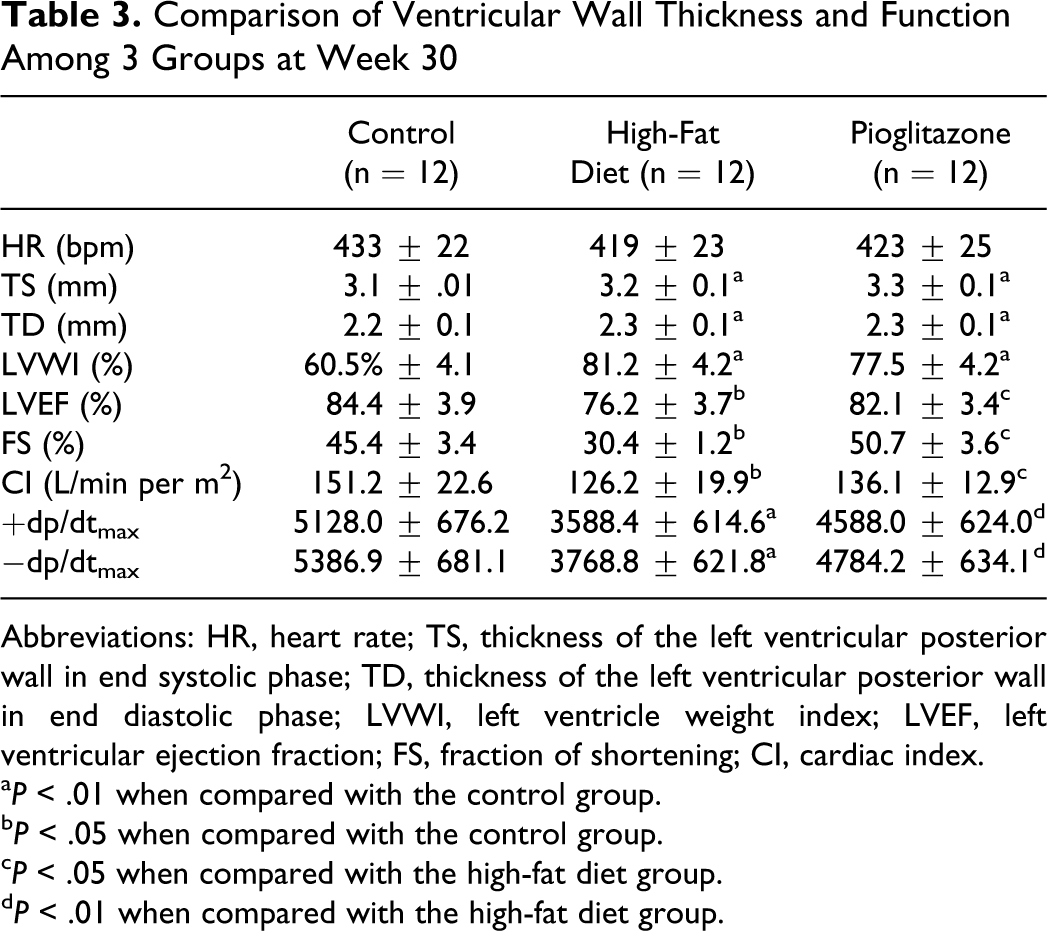
Abbreviations: HR, heart rate; TS, thickness of the left ventricular posterior wall in end systolic phase; TD, thickness of the left ventricular posterior wall in end diastolic phase; LVWI, left ventricle weight index; LVEF, left ventricular ejection fraction; FS, fraction of shortening; CI, cardiac index.
a P < .01 when compared with the control group.
b P < .05 when compared with the control group.
c P < .05 when compared with the high-fat diet group.
d P < .01 when compared with the high-fat diet group.
Expression of Ang II, AT1/AT2 Receptor Proteins, or Messenger RNA in Myocardial Tissues
Immunohistochemistry showed that the expression of Ang II and AT1 in HF group was higher when compared with control and pioglitazone groups (Table 4), with more brownish/yellow colored sites in cytoplasm. The expression of AT2 in the HF group was lower when compared with control or pioglitazone group (Table 4).
Comparison of Optical Density or Expression of mRNA of Renin, Ang II, AT1, and AT2 Among 3 Groups at Week 30

Abbreviations: Ang II, angiogensin II; AT1, angiotensin 1 receptor; AT2, angiotensin 2 receptor.
a P < .01 when compared with the control group.
b P < .05 when compared with the control group.
c P < .01 when compared with the high-fat diet group.
The expression of renin, Ang II, and AT2 messenger RNA (mRNA) in the HF group was higher when compared with the control group (P < .05 or P < .01). The expression of Ang II and AT2 mRNA in the pioglitazone group was lower than that in the HF group (P < .01).
Discussion
In this study, hyperinsulinemic–euglycemic clamp was used to measure glucose infusion rate, which is a gold standard for detecting insulin resistance. In HF group, glucose infusion rate was less than half of that of control group, indicating a high insulin resistance following HF diet. High-fat diet was also associated with an increase in plasma FFAs, serum, and myocardial triglycerides. Pioglitazone treatment was associated with a reduction in plasma FFA, serum, and myocardial triglycerides. The level of glucose infusion rate in the pioglitazone group was 178% higher than in HF group, suggesting that pioglitazone improved insulin resistance in peripheral tissues.
Obesity has been consistently associated with LV hypertrophy and dilatation, and with an increased risk of heart failure. 8,9 Left ventricular hypertrophy in patients with obesity may be caused by several factors, such as the enhancement of sympathetic nerve activity and increase in blood volume or cardiac output. 8,9 In rats, obesity-induced insulin resistance is an important factor for LV hypertrophy and functional damages. 10 In rats treated with HF diet, there was a significant increase in LV end-diastolic pressure and a decrease in LV +dp/dt max. 11 In the present study, echocardiography and cardiac catheterization were performed in these animals to evaluate the impact of HF diet and resultant insulin resistance on ventricular hypertrophy and function. Left ventricular posterior wall thickness and weight index in HF group were higher than in the control group, suggesting HF diet and high insulin resistance are associated with LV remodeling. Left ventricular ejection fraction, fraction of shortening, and cardiac index in the HF group were lower when compared with the control group. Left ventricular +dp/dt max and −dp/dt max in HF group were also lower when compared with the control group. These results indicate that HF diet and increased insulin resistance are associated with a reduction in LV function.
As shown in the present study, insulin sensitizers, such as pioglitazone, are effective in the improvement of insulin resistance in peripheral tissues, 12 but their effect on cardiac function and cardiovascular events remains uncertain. 13 Pioglitazone was found to reduce the composite of all-cause mortality, nonfatal myocardial infarction, and stroke in patients with type 2 diabetes who have a high risk of macrovascular events. 14 In rats fed with normal or HF diet, pioglitazone induced accumulation of FFAs and diacylglycerol in the heart of both the groups, despite concomitant reduction in plasma FFA concentration. 15 The content of triacylglycerol was increased only in the HF group. 15 Pioglitazone was also found to suppress cardiac hypertrophy, inflammation, and interstitial fibrosis in spontaneously hypertensive rats. 16 However, there are few reports on the effect of pioglitazone on HF-diet–induced cardiac dysfunction. In the present study, there was no statistically significant difference between HF and pioglitazone groups in LV posterior wall thickness or LV weight index, suggesting that pioglitazone had little effect on HF-diet–induced LV hypertrophy. However, LV functional indices in the pioglitazone group, such as LVEF, fraction of shortening, cardiac index, LV +dp/dt max, and −dp/dt max, were higher when compared with the HF group. These results indicate that pioglitazone diminished HF-diet–induced LV dysfunction.
A previous study showed that in insulin resistance phase before blood sugar increases to the level of diabetes, cardiac function has already been damaged. 17 The functional damage was related to myocardial ectopic deposits of triglyceride caused by high plasma FFA. 18 When triglyceride was decreased to the normal level following drug therapies, the damaged cardiac function was improved. 19 It is likely that increase in plasma FFA exceeding the storage or oxidation capacities of adipose tissues, and the ectopic deposits of triglyceride of excessive FFA in the form of triglyceride in the heart, is the main cause of heart failure in the presence of insulin resistance and obesity. 20,21 However, the exact molecular mechanisms by which myocardial triglycerides lead to ventricular dysfunction are not clear. There have been studies showing that ectopic deposits of triglyceride in the myocardium may result in ventricular dysfunction through fat cell apoptosis, oxidative stress, and endoplasmic reticulum stress. 22 –24 Renin–angiotensin system is known to play an important role in myocardial remodeling and in functional regulation. In mice overexpressing AT1 receptors, Ang II caused significant cardiac hypertrophy and remodeling with increased expression of ventricular atrial natriuretic factor and interstitial collagen deposition. 25 In rats with chronic congestive heart failure, there was a high level of angiotensin-converting enzyme and Ang II mRNA expression in the myocardial tissues. 26,27 AT2 receptors in the myocardium antagonize the actions of AT1s, inhibiting cell growth and myocardial hypertrophy, promoting cell differentiation and blood vessel dilatation. Chronic AT1 receptor antagonism prevented adverse changes in heart weight, mechanical function, and susceptibility to ischemic/reperfusion injury. 28 In the present study, the expression of renin, Ang II, and AT1 receptors in the HF group was higher, while AT2 receptor expression was lower when compared with the control group. These results suggest that HF diet and myocardial triglycerides may have activated the renin–angiotensin system in the myocardium, leading to ventricular hypertrophy and dysfunction.
In the present study, pioglitazone therapy was associated with a reduced serum and myocardial level of triglycerides. In the pioglitazone group, expression of renin, Ang II, and AT1 receptors in myocardial tissues was lower when compared with the HF group, whereas AT2 mRNA expression was higher. The reduced activation of the renin–angiotensin system following pioglitazone therapy may be a major contributing factor for the improvement in ventricular function in these animals. We did not observe noticeable side effects in the pioglitazone treatment group. However, pioglitazone has been reported to cause edema, heart failure, and weight gain in human participants. 29 At the end of the 6-week treatment, the mean body weight in the pioglitazone and HF groups was similar, but measures of cardiac function, such as LVEF and cardiac index, in the pioglitazone group were better than that in the HF group.
In conclusion, our study found that HF diet leads to insulin resistance in peripheral tissues, ventricular hypertrophy, and cardiac dysfunction in rats. The HF-diet–induced cardiac toxicity seems related to the activation of renin–angiotensin system in the myocardium, probably as a result of myocardial ectopic deposits of triglyceride. Pioglitazone treatment reduced blood pressure, plasma FFAs, serum, and myocardial triglycerides. It also diminished HF-diet–induced ventricular dysfunction in these animals, probably through reducing blood pressure and suppressing the renin–angiotensin system in myocardial tissues.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
