Abstract
Lidocaine-sensitive, repetitive atrial tachycardia is an uncommon arrhythmia. The electrophysiologic substrate is still unknown, and the pharmacologic responses have not been fully explored. The aim of this study was to investigate the effects of intravenous adenosine and verapamil in patients with lidocaine-sensitive atrial tachycardia. In 9 patients with repetitive uniform atrial tachycardia, the response to intravenous adenosine (12 mg), lidocaine (1 mg/kg body weight), and verapamil (10 mg) were sequentially investigated. Simultaneous 12-lead electrocardiogram (ECG) was recorded at baseline and continuously monitored thereafter. Tracings were obtained at regularly timed intervals right after the administration of each drug to evaluate changes in the arrhythmia characteristics. Repetitive atrial tachycardia was abolished by intravenous lidocaine in the 9 patients within the first 2 minutes after the end of injection. Adenosine suppressed the arrhythmia in 2 patients and shortened the runs of atrial ectopic activity in 1 patient, while verapamil was effective in 2 patients, 1 of them insensitive to adenosine and the other 1 sensitive to this agent. In 5 patients, the arrhythmia was abolished by radiofrequency ablation at different sites of the right atrium. Lidocaine-sensitive atrial tachycardia may eventually be also suppressed by adenosine and/or verapamil. This suggests that this enigmatic arrhythmia may be caused by different underlying electrophysiologic substrates and that at least in some cases, delayed afterdepolarizations seem to play a determining role.
Introduction
In 2000, we described a previously unreported variety of repetitive uniform atrial tachycardia in 8 patients, which was found highly sensitive to intravenous lidocaine and refractory to most orally tested antiarrhythmic agents and to intravenous verapamil. 1 However, the response to intravenous adenosine, an agent that has been used by others as an approach to the mechanism of uniform atrial tachycardias, 2,3 was not investigated in our patients.
In this study, we assessed the effects of intravenous adenosine and verapamil in a new series of patients with lidocaine-sensitive, repetitive atrial tachycardia. As will be shown, this singular arrhythmia has a variable pharmacologic matrix that contrasts with the stereotyped behavior reported in our former description, a fact that makes its underlying electrophysiologic substrate even more intriguing.
Methods
Of 14 patients referred to our centers during the last 7 years for virtually incessant repetitive bursts of uniform atrial tachycardia, we selected for this report 9 patients (6 men; 3 women; mean age: 43.33 ± 20.24 years old; range: 20-83 years old) in whom the arrhythmia was responsive to intravenous lidocaine. All patients underwent a complete clinical evaluation, with usual laboratory analysis, including thyroid hormone levels, electrocardiogram (ECG) at rest, and bidimensional echocardiogram with color Doppler. Table 1 summarizes the clinical characteristics of patients as well as the coupling intervals of the premature beats initiating the atrial tachycardia salvos and the range of atrial tachycardia cycle lengths observed at rest, as measured in at least 20 salvos. Eight patients complained of palpitations, and 1 had in addition dizziness and a syncopal episode. The arrhythmia was diagnosed 1 to 8 months before referral and 5 patients had been unsuccessfully treated with standard doses of 1 or more antiarrhythmic agents, including class I C sodium channel blockers, β-adrenergic blockers, and diltiazem. The effects of adenosine, lidocaine, and verapamil were acutely tested after discontinuance of any antiarrhythmic agent for at least 5 half-lives.
Clinical and Atrial Tachycardia Features in 9 Patients With Lidocaine-Sensitive Repetitive Atrial Tachycardia

Abbreviations: AT, atrial tachycardia; AHT, arterial hypertension; MVP, mitral valve prolapse; SHD, structural heart disease.
All patients were studied in the fasting state; in 1 of them, the pharmacologic tests were performed during an electrophysiologic study followed by a radiofrequency ablation of the arrhythmia. A venous access was achieved and a 5% dextrose solution was administered at a rate of 1 mL/min. After a baseline 12-lead ECG recording at 25 and 50 mm/s speed with a rhythm strip of at least 1-minute duration, a 12-mg intravenous bolus of adenosine was given, and the ECG was continuously recorded during 2 minutes since the end of the injection. Ten minutes later, intravenous lidocaine was administered in a single dose of 1 mg/kg in 60 seconds. Continuous ECG recordings were obtained right after the end of the lidocaine injection to document changes in the pattern of the arrhythmia and at 1-minute intervals after disappearance of atrial ectopy until baseline conditions resumed. After that, 10 mg of intravenous verapamil was given in 60 seconds. The ECG was continuously monitored and recordings were obtained from the end of verapamil injection to assess any modification in the pattern of the arrhythmia during the first 3 minutes and at 1-minute intervals thereafter during a 30-minute period or until baseline conditions reappeared. We selected adenosine as the first drug to be tested due to its well-known ephemeral action; the second drug we used was lidocaine, whose suppressive effect on the arrhythmia has a well-known duration 1 ; and finally, we administered verapamil since it is not known how long its effect could last in case of arrhythmia suppression.
Criterion for drug efficacy was total suppression of the repetitive burst of atrial ectopic activity, unrelated to changes in sinus node rate. The pattern of the arrhythmia is known to be influenced (suppressed or shortened) by an abbreviation of the sinus rhythm (or atrial pacing) cycle length. Therefore, bradycardia-inducing maneuvers (carotid sinus massage, Valsalva maneuver) were performed at regular intervals whenever disappearance of the arrhythmia was documented after administration of both lidocaine and verapamil to obtain identical or even more prolonged cycle lengths than those preceding the initiation of repetitive atrial salvos at baseline.
The study protocol complied with the Helsinki Declaration and was approved by the Ethical Boards of both Ramos Mejía Hospital and Instituto Sacre Coeur. All patients were informed about the characteristics of the study and signed a written consent.
Results
Intravenous adenosine caused a rapid but ephemeral (lasting 12-25 s) disappearance of the arrhythmia in only 2 cases (see Figure 5), caused a brief but clear-cut shortening of atrial tachycardia salvos (which turned into single and couplets of atrial ectopic activity) in 1 patient, and was ineffective in the other 6 patients. In these patients, a variable degree of atrioventricular (AV) block of the atrial ectopic impulses was documented without discernible changes in the arrhythmia cycle length (Figure 1). In the 3 cases that responded to intravenous adenosine, transient suppression of atrial tachycardia was not dependent on acceleration of heart rate since it occurred before the sinus node cycle length shortening caused by this agent, which was otherwise accompanied by reappearance of the arrhythmia as at baseline.

Case 1. Repetitive atrial tachycardia (mean atrial cycle length: 280 ms) with fast ventricular response at baseline (A) and during the peak effect of IV adenosine (12 mg) (B). Atrial ectopic activity was not modified by adenosine, which instead caused repetitive AV block during the salvos.

From the same patient as in Figure 1. Suppression of repetitive atrial tachycardia by lidocaine. Before stable sinus rhythm (S.R.) resumption, a clear cut prolongation and irregularity of atrial ectopic cycle length was apparent (A). Not a single atrial premature contraction was observed until 10 minutes after lidocaine administration (B).
Repetitive atrial tachycardia was abolished by intravenous lidocaine in the 9 patients within the first 2 minutes after the end of injection (Figures 2, 3, and 6A). This occurred almost systematically with a previous prolongation of the atrial ectopic cycle length and a shortening of the atrial salvos. The suppressive effect of lidocaine on atrial tachycardia persisted in every patient during sinus node slowing obtained by vagal-stimulating maneuvers. The arrhythmia consistently reappeared as isolated premature atrial contractions, couplets, or short runs of atrial tachycardia before reaching the baseline characteristics, which occurred between 9 and 22 minutes after the end of lidocaine injection.
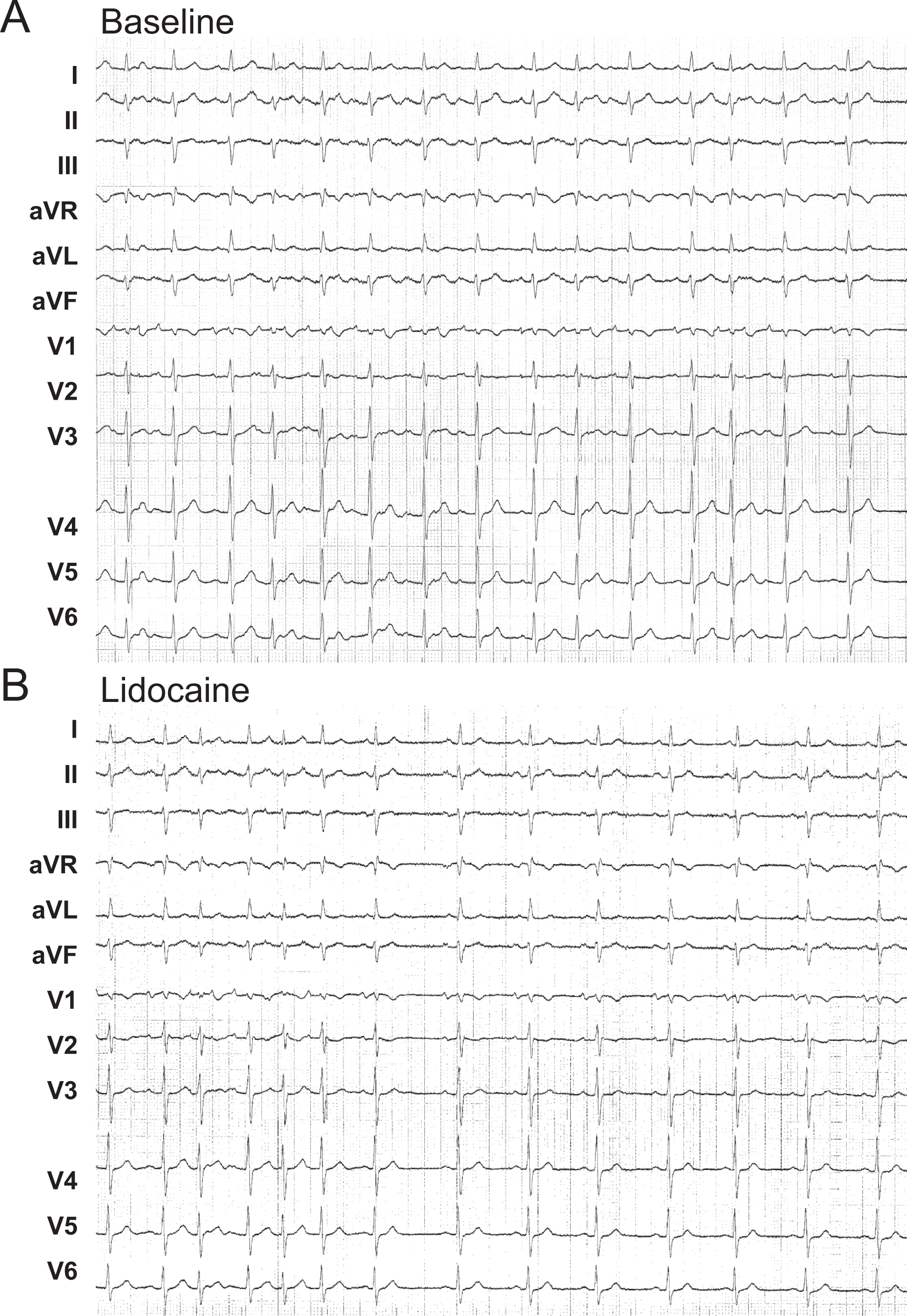
Case 3. Suppression of repetitive atrial tachycardia (A) by intravenous lidocaine (B).
Intravenous verapamil was effective in 2 patients, in whom the arrhythmia was abolished rather gradually, in 1 of them after a brief period of isolated premature atrial ectopy, as shown in Figure 4. In contrast, it only caused an AV conduction impairment during the salvos in 7 patients (Figure 6B). In the former 2 cases, at the time of arrhythmia suppression, sinus node cycle length did not differ from baseline recordings and suppression of the arrhythmia still persisted at slower heart rates, with sinus node pauses lasting up to 1250 and 1620 ms, respectively, induced by vagal-stimulating maneuvers. In these 2 patients, atrial ectopic activity resumed 15 and 25 minutes after verapamil administration.
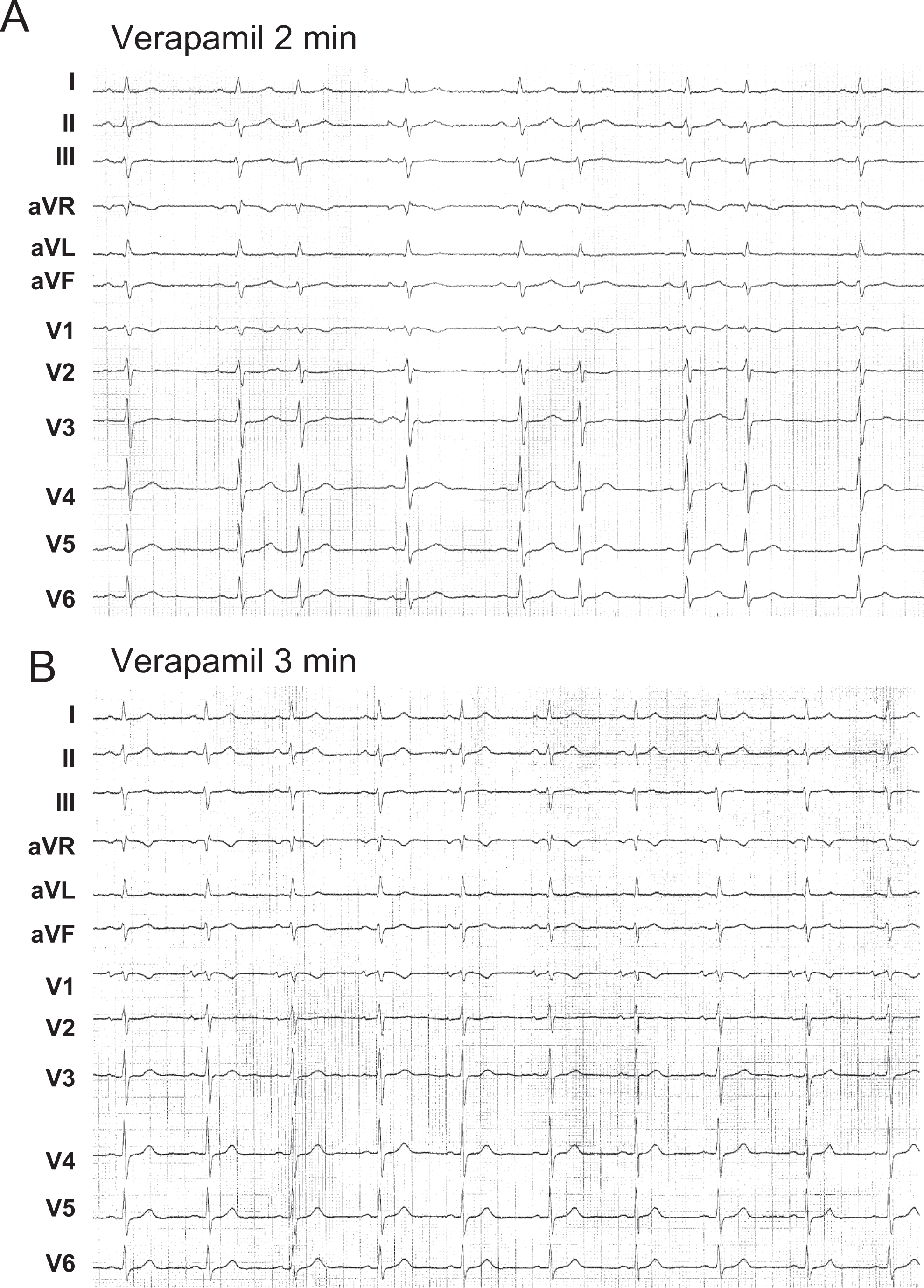
From the same patient as in Figure 3. The effect of intravenous verapamil on repetitive atrial tachycardia. Two minutes after verapamil administration only isolated atrial extrasystoles were present (A), before restoration of stable sinus rhythm (B).

Case 7. Bursts of atrial tachycardia with variable AV conduction (A) were transiently suppressed at the peak effect of intravenous adenosine (B).

From the same case as in Figure 5. Normal sinus rhythm obtained from 1 to 20 minutes after intravenous lidocaine administration (A) and persistence of atrial tachycardia bursts with variable AV conduction after intravenous verapamil administration (B).
Table 2 summarizes the results. In the whole group, there were 5 patients in whom the arrhythmia was only responsive to lidocaine. In 2 patients, atrial tachycardia was also suppressed (one patient) or the atrial ectopic runs were clearly shortened (one patient) by adenosine but was totally insensitive to verapamil. In another patient, the arrhythmia showed the inverse behavior, and in the remaining one, it was sensitive to both adenosine and verapamil.
The Effect of Adenosine, Lidocaine, and Verapamil on Repetitive Atrial Tachycardia

Abbreviations: +, atrial tachycardia suppression; --, atrial tachycardia persistence; ±, shortening of atrial tachycardia bursts.
Treatment and Follow-Up
Of the 2 patients in whom intravenous verapamil suppressed the arrhythmia, only 1 responded to oral verapamil, while the other patient was efficaciously controlled with amiodarone during 18 and 34 months of follow-up, respectively. In both cases, occasional, isolated atrial premature beats were found during follow-up in repeated 24-hour ECG Holter monitoring. In case 1, lidocaine-sensitive atrial tachycardia was treated with mexiletine with excellent clinical results and persistence of only isolated and infrequent atrial premature contractions in several 24-h ECG Holter recordings obtained during a follow-up of 30 months. In 5 patients, the arrhythmia was abolished by radiofrequency ablation, at different sites of the right atrium. In 3 patients, the ablation procedure was guided by electroanatomic mapping of the right atrium with the EnSite NavX system and in the remaining 2 patients by endocardial conventional mapping. The atrial ectopic foci were ablated in the lateral wall at a high/medium level in 3 cases, into the body of atrial appendage in 1 patient and in the fosa ovalis region in the remaining patient. In the successful ablation sites, the local atrial electrogram preceded the onset of the ectopic P waves by 20 to 45 ms. One patient was lost to follow-up after failure of flecainide treatment.
Discussion
Uniform atrial tachycardias are classically classified based on their response to pacing and pharmacologic interventions as due to reentry, abnormal automaticity, or triggered activity. 3 –7 The majority of regular focal atrial tachycardias exhibit electrophysiologic characteristics and pharmacologic responses consistent with triggered activity and, less commonly, with enhanced automaticity. Triggered atrial tachycardias are usually terminated by adenosine 4,8 and also by verapamil. 4 On the contrary, adenosine-insensitive focal atrial tachycardias seem to be caused by micro-reentry. 7
The electrophysiologic mechanism operating in lidocaine-sensitive atrial tachycardia is challenging. In our previous report, 1 its electropharmacologic characteristics were not consistent with any of the mechanisms mentioned above. In fact, in addition to the irregular cycle length, the arrhythmia could be neither consistently induced nor terminated by programmed premature atrial stimulation, which is strong evidence against a reentrant mechanism. Furthermore, it was triggered by slowing down the heart rate and systematically suppressed by atrial pacing at relatively short cycle lengths although longer than those of the tachycardia, a finding that is inconsistent with the presence of delayed afterdepolarizations. However, Markowitz et al 2 reported 4 patients with repetitive uniform atrial tachycardias in whom the arrhythmia was suppressed by adenosine and verapamil, and its mechanism was attributed to delayed afterdepolarizations. Lidocaine was tested in only 1 of these 4 patients and was also effective in supressing the atrial ectopy.
An automatic mechanism due to spontaneous diastolic depolarization was considered unlikely taking into account the absence of “overdrive suppression,” consistently found in our 8 patients previously reported, after interruption of rapid atrial pacing and of the “warming-up” phenomenon during the bursts of atrial tachycardia. An entirely speculative mechanism was then proposed in which early afterdepolarizations or atrial reexcitation due to local differences of membrane potential might play a role. In fact, the first mechanism was described in the midmyocardial (M) ventricular cells 9,10 as depending on long cycle lengths 11 and abolished by mexiletine. 12
The Mechanistic Significance of the Dissimilar Pharmacologic Responses of Lidocaine-Sensitive Atrial Tachycardia
The pharmacologic responses in patients with lidocaine-sensitive, repetitive atrial tachycardia described in this report would imply that they may be caused by different electrophysiologic mechanisms. In fact, transient suppression of the arrhythmia by intravenous adenosine and/or verapamil, as observed in 4 of our patients, strongly suggests that triggered activity due to delayed afterdepolarization was the underlying mechanism. No information is available on the effects of lidocaine on atrial delayed depolarizations. However, Sicouri et al 13 have recently demonstrated that ranolazine, an agent causing sodium channel blockade, as lidocaine also does, abolished triggered activity caused by delayed afterdepolarization in preparations of pulmonary vein sleeves. Thus, in the 4 patients in whom the arrhythmia was suppressed not only by lidocaine but also by adenosine and/or verapamil, triggered activity caused by delayed afterdepolarizations might have been the operating cellular mechanism. Nevertheless, it should be noted that lidocaine has a much lesser degree of atrial selectivity than other sodium channel blockers. 14 From this small sample size, it might be argued that these responses may have been coincidental and not a result of a true suppressive effect of the tested drugs. However, this may be reasonably excluded taking into account that the repetitive bursts of ectopic atrial activity were only separated by 1 to 2 normal sinus beats at baseline in those patients responding to intravenous (IV) administration of these agents (as well as in the remaining 5 patients who did not), as clearly seen in Figures 1A and 3A. Also, the arrhythmia did not recur even at slow sinus rates obtained by vagal-stimulating maneuvers. Besides, the rarity of lidocaine-sensitive atrial tachycardia is a major impediment to performing a randomized placebo-controlled trial for assessment of the pharmacologic substrate of this arrhythmia.
The electrophysiologic mechanism operating in the remaining 5 patients remains obscure. The focal characteristics suggested by the radiofrequency ablation of the arrhythmia in discrete sites of the right atrium might be due to a local re-excitation mechanism, as mentioned in our seminal paper. A distinctive characteristic of lidocaine-sensitive atrial tachycardia is its repetitive, almost incessant, character, which precludes an accurate assessment of the effects of programmed atrial pacing on initiation and interruption of the arrhythmia and therefore its full electrophysiologic exploration.
It is remarkable that not all repetitive atrial tachycardias are lidocaine sensitive. In fact, lidocaine failed to suppress the arrhythmias in 5 of our primary group of 14 patients. In 3 of these patients, the atrial tachycardias were insensitive not only to lidocaine but also to adenosine and verapamil; in 1 patient, both adenosine and verapamil suppressed arrhythmia, while the remaining patient was only sensitive to adenosine.
Limitations
Our study included a small number of patients, which reflects the rarity of lidocaine-sensitive repetitive uniform atrial tachycardia. Furthermore, there are quite similar atrial tachycardias that are insensitive to lidocaine and may respond to adenosine and/or verapamil or even are completely insensitive to all these 3 agents. Further studies in larger populations with repetitive uniform atrial tachycardias are necessary to determine the full pharmacologic responses of these arrhythmias and to obtain a thorough knowledge of the different electrophysiologic mechanisms involved in their origin.
Conclusions
Lidocaine-sensitive, repetitive uniform atrial tachycardia shows a variable response to intravenous adenosine and verapamil. Although many cases are insensitive to these agents, in some instances, adenosine and/or verapamil may also suppress the arrhythmia, suggesting that it can have different underlying electrophysiologic mechanisms.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Consejo de Investigación en Salud, Ministry of Health, Government of Buenos Aires City, the Fundación de Investigaciones Cardiológicas Einthoven and the Pontificia Universidad Católica Argentina “Santa María de los Buenos Aires.”
