Abstract
Introduction
Cyclosporine A (CsA) has been shown to confer protection against myocardial ischemic–reperfusion injury in small animal models.1–5 Furthermore, results from a small clinical trial support a reduction of infarct size in patients undergoing acute primary percutaneous coronary intervention (PCI) when 2.5 mg/kg CsA was administered before reperfusion. 6 This study also demonstrated sustained benefit on late left ventricular remodeling after 6 months.6,7 The suggested mechanism of protection is inhibition of the mitochondrial transition permeability pore (MPTP), possibly through binding to cyclophilin D. 3 Data regarding infarct reduction are, however, conflicting, and several groups have reported a lack of effect with CsA administration.8–11 Our group has earlier shown that 10 mg/kg CsA administered at reperfusion did not reduce infarct size in a closed-chest porcine model of ischemia and reperfusion, 12 results supported by a recent study by Lie et al in which the same dosing regimen did not confer cardioprotection in an open-chest porcine model. 8 The disparate results surrounding CsA treatment led us to investigate the same dosing regimen used in the clinical study mentioned above in a closed-chest porcine model for ischemia and reperfusion, in order to estimate a possible beneficial effect.
Materials and Methods
Porcine Model of Myocardial Ischemia and Reperfusion
After approval from the local ethics committee, animal experiments were performed in accordance with the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Council of Europe No 123, Strasbourg 1985).
Twenty-four female pigs of a mixed Swedish, Pigham and Yorkshire race (weighing 33-49kg) were used for the closed-chest model of ischemia and reperfusion. The day before the procedure the pigs received 75 mg of acetylsalicylic acid (Trombyl, Pfizer, Sollentuna, Sweden). The animals were sedated with intramuscular tiletamin and zolazepam (Zoletil, Virbac, Carros, France), 6 mg/kg each, suspended in Dexdomitor (Orion Pharma, Esbo, Finland), and 0.5 mg atropine (Mylan AB, Stockholm, Sweden) was administered intramuscularly. General anesthesia was induced by intravenous injection of 3 to 10 mg/kg 2% thiopental (Pentobarbital, Apoteksbolaget, Stockholm, Sweden), and mechanical ventilation with 40% oxygen was performed via oral intubation. Anaesthesia was maintained by a continuous intravenous infusion (4-18 mg/kg per h) of thiopental. Analgesia was assured by continuous intravenous infusion of morphine (MEDA AB, Stockholm, Sweden; 0.5 mg/kg per h). Venous access was established by a 5F introducer sheath through the right jugular internal vein. Monitoring of mean arterial pressure and heart rate was achieved through an arterial cannula in the femoral artery. Respiratory rate and tidal volume were adjusted to keep arterial blood pH, P
At the commencement of catheterization all pigs received 200 units/kg of unfractionated heparin (Leo, Ballerup, Denmark) which was repeated with 100 units/kg every hour. After 60 minutes of stabilization, a 6F JR 3.5 PCI-guiding catheter was inserted via an 8F introducer sheath into the right carotid artery, and under X-ray guidance (OEC 9800 Cardiac General Electric Medical Inc., Utah) placed in the left coronary ostium. An angiogram with Visipaque contrast agent (320 mg/mL; Nycomed-Amersham, Oslo, Norway) was performed and an angioplasty balloon inflated (2.0-3.0 mm in diameter, 10-20 mm long, and 8-14 atmospheres) in the left anterior descending artery distal to the second diagonal branch. Occlusion was verified angiographically and through ST-segment alteration on the electrocardiograph (ECG). After 40 minutes of occlusion, the balloon was deflated and reperfusion verified by contrast injection. The pigs were then kept under anesthesia for 4 hours and thereafter sacrificed.
Intervention Procedure
The pigs were randomized to receive either 2.5 mg/kg CsA (Sandimmune, Novartis, Basel, Switzerland) diluted 1:20 in 5% glucose (Baxter Medical AB) or saline (0.9% NaCl) given as a bolus injection (20 mL) in a central venous catheter 7 minutes before reperfusion. The administration of CsA or saline was blinded to the investigator.
Determination of Area at Risk and Infarct Size
At the end of the experiment, the chest was opened to obtain access to the heart and great vessels. Another coronary angiogram was performed, an angioplasty balloon inflated at the same location as during induction of ischemia and a small suture placed around the balloon. Forty milliliters of 2% Evans Blue was infused via the central venous catheter, followed by an injection of a lethal dose of potassium chloride. The heart was excised, the right ventricle and the atria removed, and the area at risk and the infarct size were determined by staining with Evans Blue and 2,3,5-triphenyltetrazolium-chloride, respectively, as described previously. 10 All measurements were performed by a blinded investigator.
Analysis of CsA Concentration
The concentration of CsA was measured in 3 additional pigs. Samples of whole blood were taken 7, 27, 67, and 127 minutes after the intravenous bolus (ie, 20, 60, and 120 minutes after a presumed reperfusion). The concentration was measured using a chemiluminescent microparticle immunoassay (Architect I2000 SR, Abbott, Abbott Park, Illinois). Levels above 1.2 μmol/L were not determined.
Statistical Analysis
All values are presented as mean ± standard error of the mean (SEM). Statistically significant differences between groups were calculated with Student unpaired t test. Differences in hemodynamics between groups were tested with repeated measures analysis of variance (ANOVA). A value of P < .05 was considered statistically significant.
Results
Baseline Characteristics, Hemodynamics, and Exclusions
Twenty-four pigs were randomized to receive CsA (n = 12; weighing 33-42 kg) or saline (control, n = 12; weighing 33-49 kg). Six pigs (3 in each group) developed ventricular fibrillation between 11 and 20 minutes of ischemia, all of which was successfully treated with direct current cardioversion (150 J). One pig randomized to the control group was excluded due to ventricular fibrillation during reperfusion. Heart rate and mean arterial pressure declined in both groups during the experiments without any significant differences between the 2 groups. Temperature measurement showed no differences over time or between the 2 groups (Table 1 ).
Hemodynamic Data and Body Temperature Before Ischemia and During Reperfusion
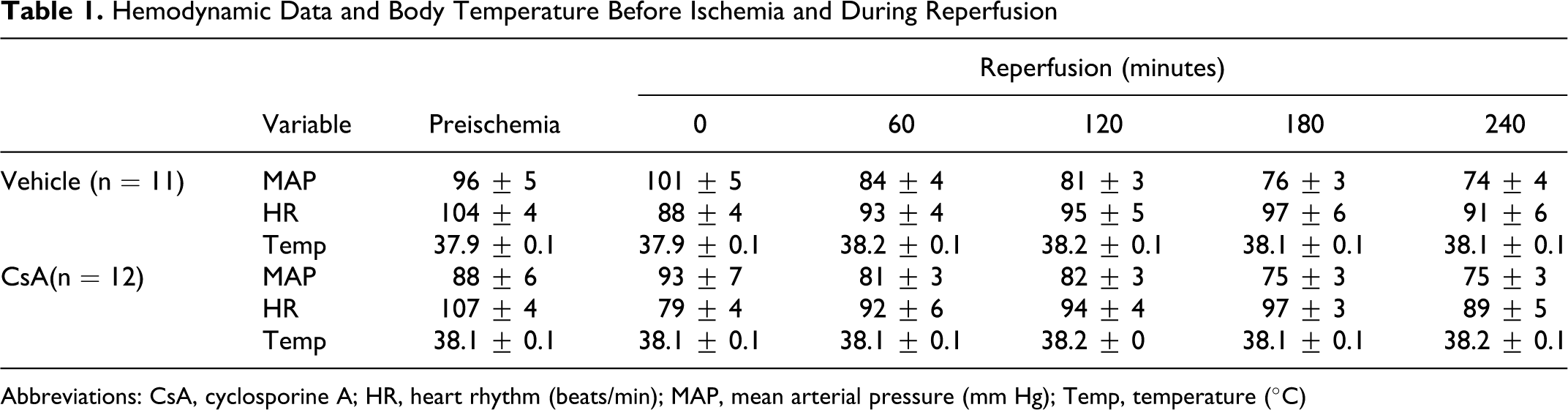
Abbreviations: CsA, cyclosporine A; HR, heart rhythm (beats/min); MAP, mean arterial pressure (mm Hg); Temp, temperature (°C)
Area at Risk and Infarct Size
Area at risk/left ventricle was similar in the 2 groups (22% ± 1% and 20% ± 2% for the control group and the CsA group, respectively; P = .32). Cyclosporine A treatment did not reduce infarct size as a proportion of area at risk compared with saline (51% ± 6% in the CsA group vs 54% ± 6% in the control group; P = .75; Figure 1 ).
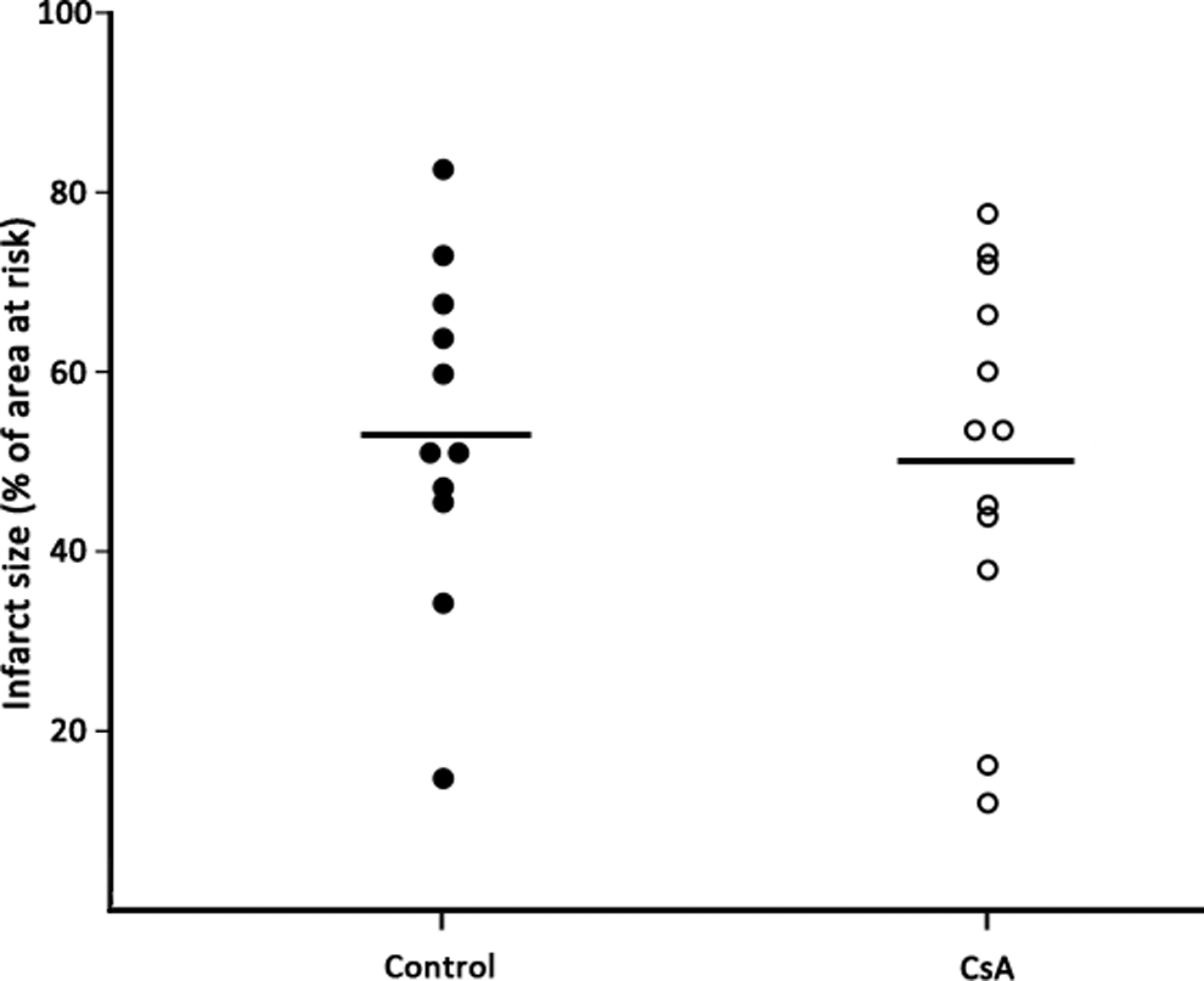
Infarct size. Infarct size as a proportion of area at risk in control (n = 11) and CsA (n = 12) treatment groups. The horizontal bars represent mean infarct size.
Cyclosporine A Blood Concentration
The pharmacokinetics of CsA is presented in Figure 2 .

Cyclosporine A concentration. Whole blood concentration of cyclosporine A (CsA) at different time points after intravenous administration (n = 3). Data are presented as mean ± standard error of the mean (SEM). Points within brackets indicate levels >1.2 mmol/L that were not exactly determined.
Discussion
Cyclosporine A has been appreciated for its ability to reduce ischemic–reperfusion injury in small animal models. Indeed, in this setting, it has served as a model compound for inhibiting MPTP formation and opening, a process suggested to be pivotal for the development of necrosis and apoptosis in reperfused myocardium. 3 The data are, however, conflicting, and negative trials in small animal models have been presented.9–11 Furthermore, consistent evidence of a positive effect in large animal models is sparse. The most promising data were derived in a recent study in minipigs, in which CsA administered intracoronary distal to ligation of the left anterior descending coronary artery reduced infarct size. 13 In this study, however, reperfusion was not performed; hence the results have little bearing on the situation of reperfusion therapy for acute myocardial infarction. Furthermore, the pigs were anesthetized with isoflurane, a compound known to confer cardioprotection with possible interactions with CsA. 12 Finally, the pigs were pretreated with amiodarone, a drug that has been demonstrated to inhibit mitochondrial permeability transition in itself. 14
In contrast, in 2 different porcine models for ischemia and reperfusion carried out by Lie et al and our group, 10 mg/kg CsA administered before reperfusion did not confer protection.8,12 Furthermore, analysis of proapoptotic proteins in myocardial biopsies from our previous study demonstrated that the CsA regimen applied could be potentially harmful. 12 These data evoked suspicion that the dose used in these studies was too high. Indeed, in the only clinical trial on cyclosporine in this setting, a lower dose of 2.5 mg/kg CsA administered before reperfusion reduced infarct size. 6 We therefore applied this reduced dose of 2.5 mg/kg CsA in our closed-chest porcine model of ischemia and reperfusion. We could not, however, demonstrate any positive effect on infarct size.
There are several possible explanations for these disparate results. Cyclosporine A, 10 mg/kg, in our porcine model yielded a whole blood concentration of 4.0 µmol/L 45 minutes after administration. In the clinical study by Piot et al, 2.5 mg CsA/kg administered <10 minutes before reperfusion resulted in a whole blood concentration of approximately 2.1 µmol/L 20 minutes after reperfusion, that is, considerably lower than in our previous porcine study.6,12 Applying the same dosing regimen in our present study, however, yielded an even lower blood concentration of approximately 1.0 µmol/L 30 minutes after administration, indicating differences in the pharmacokinetics of CsA in humans and pigs. Taken together, a dosage in the order of 5 mg/kg CsA might be optimal in pigs, a premise supported by a small study in minipigs. 15 If so, however, this would indicate a narrow therapeutic window. Indeed, as Griffiths et al showed in their rat model, the optimal effect of CsA was achieved at a concentration of 0.2 µmol/L, while this effect was abolished at a concentration of 1 µmol/L. 2 This could be potentially problematic in the clinical setting, with difficulties in prediciting the exact time of reperfusion. In addition to a potential narrow therapeutic window, differences in the distribution to the jeopardized myocardium add further to the uncertainty of clinical usage of CsA. Indeed, one third of patients presenting with acute myocardial infarction have collateral flow as evident on coronary angiograms, as opposed to in pigs, which are without developed collaterals.16,17 This would imply different degrees of ischemia as well as varying distribution of CsA in the myocardium at risk.
In addition to pharmacokinetic considerations, substantial data support the fact that CsA has possible deleterious effects on the ischemic myocardium, including destabilization of hypoxia-inducible factor 1α, modification of cardiac energy metabolism, impaired endothelium-derived vasodilation in coronary arteries, and deterioration of microvascular function.18–22 Furthermore, one cannot exclude the possibility that MPTP is not as crucial in the development of ischemic–reperfusion injury as has been advocated, and that CsA does not in fact confer cardioprotection after ischemia and reperfusion. Finally, in our model we use acetylsalicylic acid to reduce the risk of thrombotic complications during catheterization, and morphine as analgesia. Acetylsalicylic acid has been shown to abolish the cardioprotective effect of statins, whereas morphine in different settings has been shown to reduce myocardial reperfusion injury.23,24 We can therefore not exclude the possibility that this might have interfered with the present results. However, both compounds are, according to guidelines, basic therapy in the clinical setting for the treatment of acute myocardial infarction, making it important to show a possible cardioprotective effect with the concomitant administration of these drugs. 25 Furthermore, we have earlier shown reduced infarct size in the same model with pharmacological intervention on top of the combination of acetylsalicylic acid and morphine. 26
Conclusion
The present study aimed at repeating the positive results of the only clinical study conducted thus far using CsA in order to limit reperfusion injury in the setting of acute myocardial infarction. Regardless of the possible explanations for the diverging results in the clinical setting and the closed-chest porcine model, our study raises important questions concerning CsA as an adjunct therapy at reperfusion, with a possible narrow therapeutic window. Furthermore, the present study, as well as results obtained by other groups, indicates differences with CsA treatment between species. This may be of great importance in translational programs for the development of pharmacological strategies for myocardial protection.
Footnotes
Acknowledgments
We are grateful to Chrichan Månsson (RN) and Karl Forslund (RN) for their excellent technical support.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financed by grants from the Swedish state under the agreement between the Swedish government and the county councils concerning economic support of research and education of doctors (ALF-agreement).
