Abstract
Neural activity in the beta band is increasingly recognized to occur not as sustained oscillations but as transient burst-like events. These beta bursts are diverse in shape, timing, and spatial distribution, but their precise functional significance remains unclear. Here, we review emerging evidence on beta burst properties, functional roles, and developmental trajectories and propose a new framework in which beta bursts are not homogeneous events but reflect distinct patterns of synaptic input from different brain regions targeting different cortical layers. We argue that burst waveform shape carries mechanistic and computational significance, offering a window into the dynamic integration of specific combinations of cortical and subcortical signals. This perspective repositions beta bursts as transient computational primitives, rather than generic inhibitory signals or averaged rhythms. We conclude by outlining key open questions and research priorities, including the need for improved detection methods, investigation into developmental and clinical biomarkers, and translational applications in neuromodulation and brain–computer interfaces.
Keywords
Introduction
Neuronal activity generates electric currents that can be measured by various methods to study brain function. These methods (Box 1) include invasive intracortical electrode recordings, as well as noninvasive techniques such as electroencephalography and magnetoencephalography (MEG). Electroencephalography and MEG use sensors placed on or near the scalp to measure neural field activity, which reflects the aggregate postsynaptic electrical activity from large populations of neurons. This neural field activity comprises a combination of aperiodic (nonoscillatory) components and periodic (rhythmic) activity across multiple frequency bands, as illustrated in Figure 1a. Particular frequency bands have been associated with many cognitive, sensory, and motor processes, with examples including successful memory retrieval with theta band activity (4 to 8 Hz; Herweg et al 2020), attentional mechanisms with alpha band activity (8 to 13 Hz; Foxe and Snyder 2011), sensorimotor functions with beta band activity (13 to 30 Hz; Kilavik et al 2013), and perceptual binding with gamma band activity (>30 Hz; Csibra et al 2000).
Electrophysiologic methods for measuring neural activity.

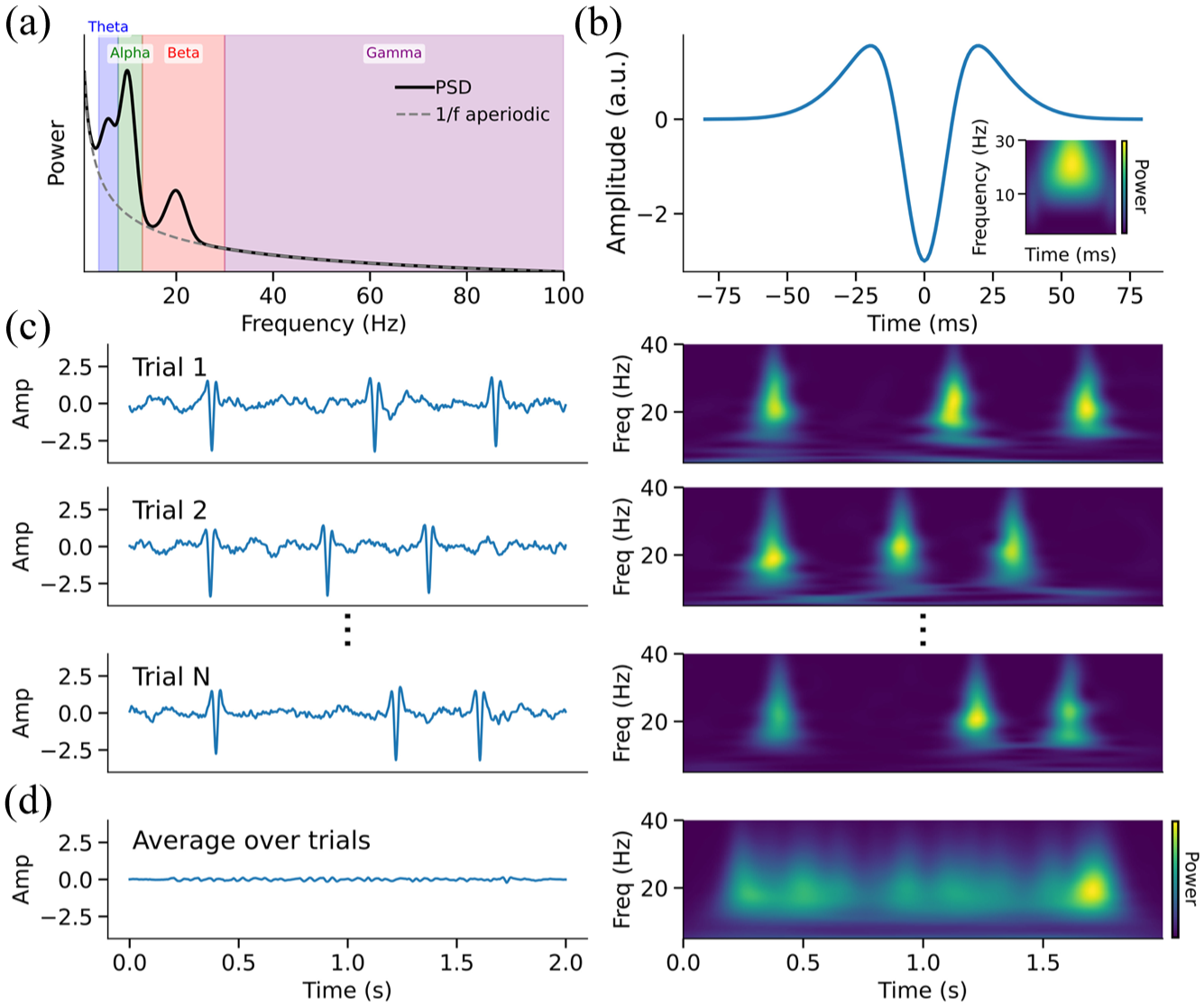
From oscillations to bursts: aperiodic vs periodic activity, burst waveforms, and trial-level dynamics. (a) Canonical power spectral density (PSD) shows periodic components (theta, alpha, beta, gamma) and aperiodic 1/f activity. Beta power appears as a broad peak superimposed on the aperiodic slope. (b) Simulated beta burst waveform generated by summing narrow and broad Gaussian components, approximating temporally aligned synaptic inputs to different cortical layers. Inset shows the corresponding time–frequency (TF) representation, revealing a brief, spectrally constrained beta burst. (c) Simulated single-trial time series (left) and corresponding TF decompositions (right), each containing transient beta bursts embedded in pink noise. The precise timing of bursts vary across trials. (d) Averaging across trials in the time domain (left) abolishes the transient burst structure, while averaging in the TF domain (right) retains elevated beta power. This illustrates how trial-averaged TF metrics can misrepresent beta bursts as sustained induced oscillatory activity.
Traditionally, the analysis of periodic neural activity has relied heavily on signal processing techniques such as the Fourier and Hilbert transforms. These methods assume that neural activity is sinusoidal and stationary (Donoghue et al 2022) and therefore characterize it as consistently oscillatory or rhythmic. However, recent empirical evidence challenges this conventional view, demonstrating that activity in various frequency bands often manifests as discrete transient events, or “bursts,” rather than sustained oscillations (Figure 1b; Jones 2016; Quinn et al 2019; Tal et al 2020). Consequently, standard trial-averaged analyses, which measure mean power within frequency bands, may obscure the dynamic burst-like activity in individual trials (Figure 1c and 1d).
Notably, neural activity in the beta band appears to occur predominantly as transient bursts rather than sustained oscillations (Feingold et al 2015; Sherman et al 2016; Little et al 2019). This insight has prompted a shift in the field, with burst-based analysis increasingly becoming the standard framework for analyzing beta band activity from multiple cortical networks, especially the sensorimotor system. Yet until recently, beta bursts have been considered homogeneous events and analyzed primarily through time–frequency methods that focus on spectral power, duration, and frequency span. However, because beta bursts are fundamentally transient and nonoscillatory events, we argue that they are best viewed in the time domain through their waveforms. Time domain analyses reveal significant diversity in burst shapes and rate dynamics, suggesting that individual bursts may represent distinct computational events reflecting various underlying neural processes.
In this article, we review the current literature concerning sensorimotor beta burst properties, dynamics, hypothesized functions, and development. We focus on the sensorimotor system because it is the most extensively studied in the context of beta bursts, but our goal is to develop a broader framework for understanding cortical beta bursts more generally. We propose that beta bursts are not homogeneous phenomena but instead are distinct “fingerprints” reflecting specific combinations of synaptic inputs from different regions targeting different layers of the cortex. This perspective emphasizes the computational significance of burst waveform shapes, which we argue index the spatial and temporal patterning of convergent inputs and provide a window onto the circuit-level operations occurring at a given moment. While we draw primarily on findings from the sensorimotor system, we suggest that this framework can be extended to beta bursts observed in other cortical regions. Finally, we discuss how this refined perspective can advance our understanding of the functional significance of beta bursts, and we outline important avenues for future research.
Bursts of Activity in the Beta Band
Discovery
The beta frequency band has long been associated with sensorimotor processes, and systematic changes in beta amplitude, such as event-related desynchronization and synchronization, are typically observed before, during, and after movement in trial-averaged analyses (Pfurtscheller 1981; Pfurtscheller and Lopes da Silva 1999; Cassim et al 2000; Jurkiewicz et al 2006). However, the notion that cortical beta activity manifests as transient bursts rather than sustained oscillations has a long but largely overlooked history. To our knowledge, the earliest known description of beta bursts comes from Kamp et al (1972), who reported transient beta events in intracranial recordings from the human frontal cortex and even presented examples of burst waveforms. Despite this early observation, their work received little attention and was rarely cited in subsequent literature. More prominent physiological investigations of transient beta activity emerged in the 1990s, notably through the work of Murthy and Fetz (1992, 1996), who described brief coherent beta activity in the sensorimotor cortex of behaving monkeys. Yet, it was only in the past decade that the concept of beta bursts gained widespread traction, largely due to influential contributions by Stephanie Jones and colleagues, who combined human MEG recordings, computational modeling, and animal data to formalize a mechanistic model of burst generation (Jones 2016; Sherman et al 2016), offering a cross-scale framework that links cellular-level synaptic dynamics with macroscale patterns observed in noninvasive recordings.
Recent animal and human research has reinforced this burst-centric perspective of beta band activity, demonstrating that the slow, sustained patterns of beta amplitude typically observed in trial-averaged data do not accurately reflect the underlying trial-by-trial dynamics of cortical activity (Sherman et al 2016; Little et al 2019). Instead, beta activity in sensorimotor regions manifests as discrete focal bursts, characterized by stereotyped, wavelet-like shapes in the time domain (Figure 1b; Sherman et al 2016; Little et al 2019; Bonaiuto et al 2021). The recognition of beta bursts as transient and focal events challenges previous assumptions of beta activity as fundamentally oscillatory, highlighting the limitations of aggregating data across trials without consideration for the nature of the underlying neural activity (Figure 1c and 1d; Jones 2016).
From Homogeneity to Diversity in Sensorimotor Beta Bursts
Early research on sensorimotor beta bursts largely treated them as homogenous phenomena, with analyses focused on overall burst rates, durations, and their timing relative to behavioral events (Shin et al 2017; Little et al 2019; Wessel 2020). These studies have produced variable findings. For example, Wessel (2020) found a larger frontocentral beta burst rate for successful vs failed action stopping, whereas Jana et al (2020) did not find a significant difference. These discrepancies could be partly due to methodological differences in burst detection methods (Box 2). This variability also underscores the limitations inherent in treating beta bursts uniformly without considering their diversity.
Methods to detect beta bursts.

Subsequent studies started to highlight significant heterogeneity in beta bursts, emphasizing diversity in their time–frequency domain features, such as power, peak frequency, duration, and frequency span (Duchet et al 2020; Enz et al 2021; Szul et al 2023; West et al 2023; Zich et al 2023). However, the use of these time- and frequency-based characteristics likely persists from the traditional assumption that beta activity fundamentally reflects oscillatory processes. Consequently, this approach has limitations in fully capturing the transient nonoscillatory nature of beta bursts and in linking empirical observations to specific neural mechanisms.
More recently, investigations have shifted toward examining the waveform shapes of individual beta bursts in the temporal domain (Figure 2; Karvat et al 2020; Kosciessa et al 2020; Rayson et al 2023; Szul et al 2023). This approach better aligns with biophysical models that explicitly describe burst generation as resulting from the confluence of temporally aligned synaptic inputs across different cortical layers (Sherman et al 2016; Bonaiuto et al 2021). Importantly, individual burst waveforms exhibit substantial diversity along a continuum, rather than forming discrete clusters. Analytical methods such as principal component analysis (PCA; Rayson et al 2023; Szul et al 2023) and convolutional dictionary learning (Power et al 2023) have been employed to characterize these bursts in terms of waveform patterns or motifs (Figure 2a and 2b). Bursts with varying waveforms within these motifs are differentially rate modulated before, during, and after real and imagined movement (Figure 2g and 2h), suggesting distinct computational roles in sensorimotor processes (Rayson et al 2023; Szul et al 2023; Papadopoulos et al 2024a, 2024b). However, the precise functional significance of these waveform shapes and their underlying computational processes remain critical open questions.
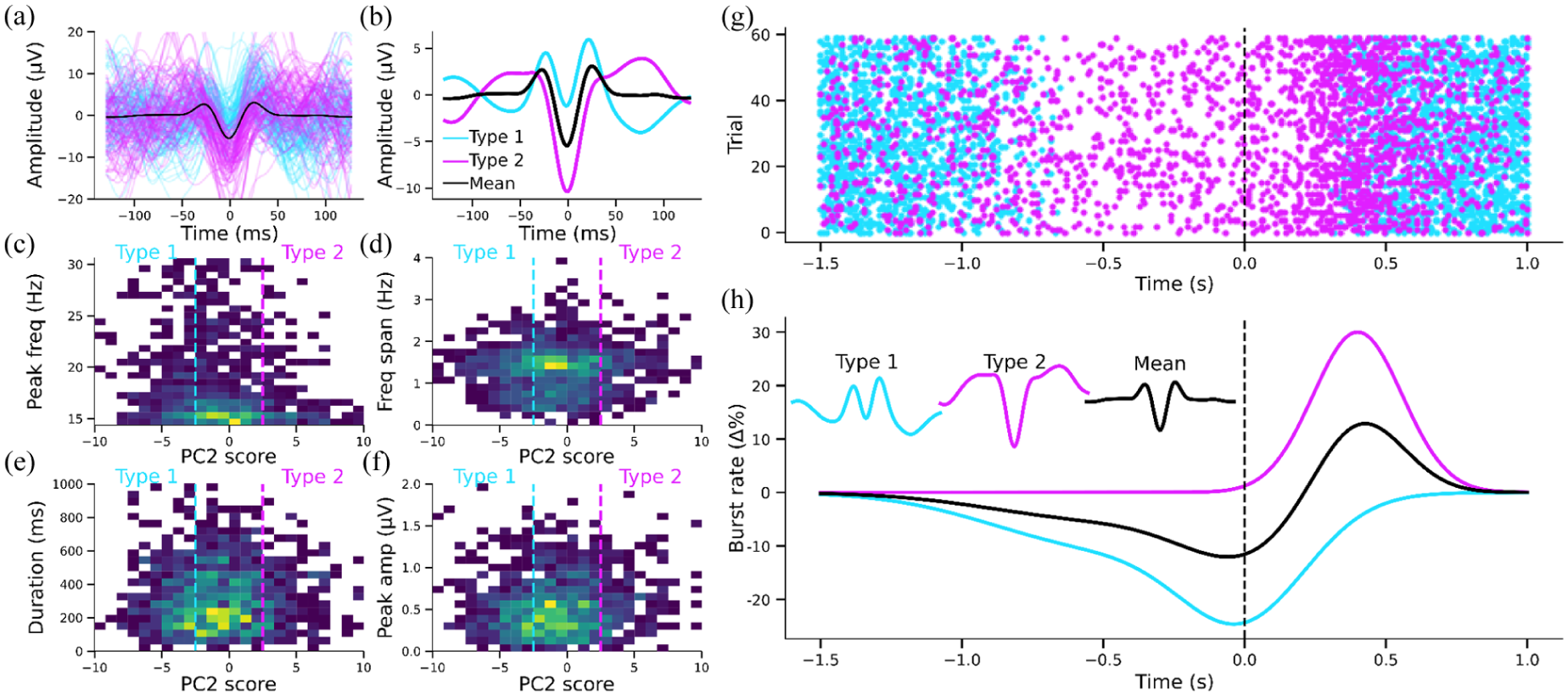
Beta bursts exhibit diverse waveform shapes that are not captured by standard time–frequency features and show distinct temporal dynamics. (a) Sample empirical beta burst waveforms from the tails of a principal component obtained from principal component analysis (PCA) on electroencephalography burst waveforms illustrate continuous shape variability. (b) Average empirical waveforms from low and high bins along a single principal component (PC2, explaining 21.67% of waveform variance) demonstrate consistent shape differences. (c–f) Two-dimensional histograms (the color indicates burst density) show that variation along this PCA axis is not well explained by conventional time–frequency features (peak frequency, frequency span, duration, or amplitude). (g) Simulated data from a toy model illustrates how bursts with different waveform shapes (colors) can vary in their rate relative to movement offset (time 0). (h) Corresponding baseline-corrected rate profiles show that different waveform types can be differentially modulated by task events. These differences would be obscured if bursts were treated as homogeneous events without accounting for shape.
Potential Computational Roles for Cortical Beta Bursts: Insights from the Sensorimotor System
Sensorimotor beta activity has long been proposed to serve multiple critical functions in motor control and sensorimotor integration. Early theories, based on trial-averaged oscillatory amplitude, linked beta to the maintenance of the sensorimotor state (Engel and Fries 2010), the inhibition of motor output (Salmelin et al 1995; Zhang et al 2008), the gating of sensory input (Buchholz et al 2014), top-down control (Buschman and Miller 2007; Siegel et al 2012), and sensory feedback processing (Lalo et al 2007). More recent studies, focusing on transient beta bursts, have shown that bursts tend to track changes in beta power: increasing in rate prior to voluntary movement, decreasing during motor execution, and rebounding postmovement, but their precise timing is more predictive of response time and error than either beta power or burst rate (Little et al 2019). Beta bursts have been associated with increased cortico- and intermuscular coherence during static posture holding, suggesting a role in coordinating functional muscle groups during motor tasks (Simpson et al 2024). Additionally, premovement beta bursts have been shown to predict trial-by-trial motor performance, pointing to their involvement in the anticipatory modulation of movement based on previous outcomes (Bracco et al 2025). In pathologic contexts such as Parkinson disease, prolonged beta bursts are associated with bradykinesia and rigidity, implicating altered burst dynamics in disrupted motor control (Tinkhauser et al 2017; Cagnan et al 2019; Lofredi et al 2019). These observations suggest that beta bursts are closely tied to behaviorally relevant computations, but exactly which computations remains an open question.
While many of these proposed functions assume a unitary role for beta bursts, accumulating evidence of their diversity challenges this view. An emerging theoretical perspective suggests that beta bursts represent transient computational building blocks involved in different sensorimotor and cognitive operations (Spitzer and Haegens 2017; Lundqvist et al 2024). Spitzer and Haegens (2017) suggested that beta activity and therefore potentially beta bursts reflect the reactivation of specific neuronal ensembles that support top-down executive control by selectively reengaging relevant representations. In contrast, Lundqvist et al (2024) proposed that beta bursts implement transient, spatially patterned functional inhibition, supporting cognition through the coordination of task-relevant excitability across neural populations. These spatiotemporal burst patterns are thought to underlie flexible cognitive operations, including working memory, attention, and executive control.
We extend these perspectives by proposing that beta bursts reflect transient patterns of synaptic input arising from coordinated interactions between cortical and subcortical sources. This proposal builds on the biophysical modeling framework developed by Jones and colleagues (Sherman et al 2016) and supported by empirical MEG data (Bonaiuto et al 2021). These studies suggest that beta bursts in the sensorimotor cortex arise from temporally aligned synaptic inputs originating from distinct thalamic sources: broad proximal inputs targeting basal dendrites of pyramidal cells in superficial and deep layers thought to arise from lemniscal thalamic nuclei, combined with stronger and more temporally narrow distal inputs targeting apical dendrites in superficial layers. Within this model, the characteristic beta burst waveform arises when a broad (~100 ms) proximal synaptic input drives current flow up the dendrites and is interrupted by a shorter (~50 ms) stronger distal input targeting apical dendrites of layer 2/3 and layer 5 pyramidal neurons, pushing current flow downward to create the stereotypical trough of the beta burst waveform. Although subthreshold, these inputs can recruit layer 2 interneurons, which provide prolonged GABAb-mediated inhibition onto pyramidal cells (Law et al 2022), thereby modulating cortical excitability.
Given the motor cortex’s extensive and diverse afferent connectivity, including inputs from cerebellar- and basal ganglia–recipient thalamic nuclei, as well as dense corticocortical projections (Shipp 2005, 2007), we hypothesize that the observed diversity of beta burst waveform shapes in the motor cortex may reflect the integration of multiple, functionally distinct input configurations. From this perspective, individual beta bursts are not merely generic inhibitory signals but rather could serve as distinct fingerprints of specific circuit-level computations, if future work confirms consistent mapping between waveform shape and input configuration. For example, some bursts may reflect reactivation of internal models, while others implement transient inhibitory gating or error processing (Moreau et al 2025) or initiate sensorimotor transformations. Thus, different beta burst waveforms may index distinct computational primitives, arising from unique patterns of laminar input that reflect the connectivity and functional demands of the sensorimotor cortex.
Alternative Mechanisms of Burst Generation
Alternative models propose that beta bursts can be generated purely through intracortical interactions between excitatory and inhibitory neurons modulated by intrinsic membrane currents, such as the M current (Jensen et al 2005), or via thalamic inputs that drive spiking and oscillatory dynamics within local excitatory–inhibitory networks (Mirzaei et al 2017). These alternative mechanisms remain valuable considerations in addressing burst diversity in the beta and other frequency bands (Schmidt et al 2023).
Beta Bursts and Development
Understanding how beta bursts develop offers a unique opportunity to disentangle their underlying neural mechanisms and computational roles. The first year of life is a period of heightened brain plasticity, marked by dramatic changes in sensorimotor skill (Adolph and Franchak 2017; von Hofsten and Rosander 2018). Therefore, early infancy represents a particularly ideal point in development to examine how beta bursts change alongside functional and anatomic maturation in different neural circuits and how this relates to behavior.
Despite the theoretical promise of this approach, research on sensorimotor beta band activity in early development remains sparse (Cuevas et al 2014; Perone and Gartstein 2019). Existing infant studies have focused on spectral power changes during action observation and/or execution (van Elk et al 2008; Meyer et al 2011) and commonly use an adult-defined frequency band for analysis (eg, Samson-Dollfus et al 1983; Niemarkt et al 2011). The latter is problematic given well-documented developmental shifts in beta peak frequency in later childhood (Johnson et al 2019; Trevarrow et al 2019), recently demonstrated beta shifts in infants and young children (Figure 3a; Rayson et al 2022, 2023; Wilkinson et al 2024), and the frequency shifts long known to occur in the alpha band across early development (Marshall et al 2002).
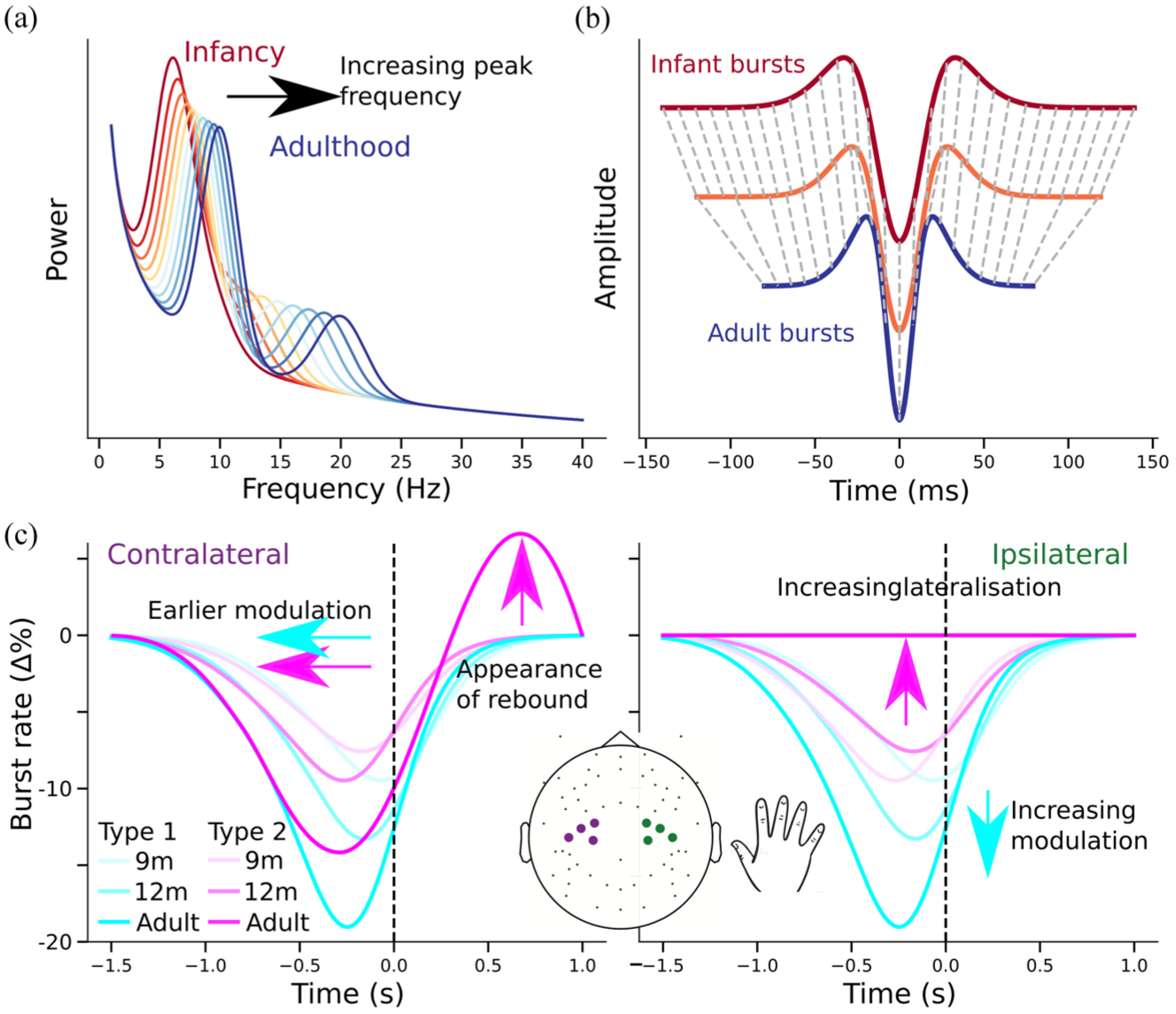
Developmental changes in beta burst shape and modulation. (a) Alpha and beta peak frequencies increase from infancy to adulthood. (b) Infant beta bursts have the same mean waveform shape as adult bursts but stretched in time. Dashed lines indicate correspondences revealed by dynamic time warping. (c) Toy examples of burst rate modulation in contralateral and ipsilateral sensorimotor regions show how 2 burst types (colors) change from broadly overlapping and weakly modulated in infancy to more distinct, lateralized, and selectively modulated in adulthood. These developmental shifts support the hypothesis that beta burst waveform shapes reflect functionally specific input patterns that mature over time. Notably, the postmovement beta rebound—robust in adults—is attenuated or absent in infancy and only gradually emerges across childhood and adolescence, potentially reflecting the late maturation of thalamocortical and cortico-subcortical circuits (Gaetz et al 2010; Johnson et al 2019; Trevarrow et al 2019).
However, recent research on infant beta bursts has now begun to fill this gap. Using electroencephalography, Rayson et al (2022) showed that, as in adults, sensorimotor beta activity in 12-mo-old infants occurs in discrete bursts rather than sustained oscillations. As compared with adults, infant bursts had lower peak frequencies and higher amplitudes. While they lasted longer in absolute time, they were shorter in terms of cycles, indicating potential differences in the temporal structure of generating mechanisms. Importantly, overall beta burst rate decreased during grasp execution in adults but not in infants, suggesting that task-related modulation of burst rate emerges with development.
Building on this work, Rayson et al (2023) extended the analysis to 9-mo-old infants and introduced a detailed waveform shape analysis using PCA, adapting methods developed for adult data (Szul et al 2023). Bursts from all age groups had a similar median waveform shape, but infant bursts were longer in duration and had a lower peak frequency. Burst waveforms from each age group were aligned to a common time course by dynamic time warping (Figure 3b) and characterized along multiple principal components reflecting continuous variability in shape. Only bursts with specific shapes that markedly differed from the median were changed in rate during movement, and this effect became earlier, more pronounced, and more lateralized with age (Figure 3c).
These beta burst findings in infants indicate that specific features of their development, such as waveform shape, lateralization, and movement-related rate changes, may follow the known maturational trajectories of underlying sensorimotor brain networks (Erberich et al 2006; Xiao et al 2018). They also support the idea that developmental studies can now serve as a powerful tool to test competing hypotheses about beta burst function. For example, such studies will be valuable for addressing questions about 1) whether distinct waveform shapes reflect distinct computational roles via examination of how changes in different types of beta burst predict motor skill improvement or 2) if burst properties are shaped by thalamocortical vs corticocortical inputs with diffusion-weighted magnetic resonance imaging to track how changes in brain connectivity predict changes in bursts features.
A New Framework for Understanding Beta Burst Dynamics
Clearly, previous research demonstrates the importance of acknowledging the bursty vs oscillatory nature of sensorimotor beta activity and the potential for research on beta bursts to reveal important insights into sensorimotor processes. However, what is also clear is that so far we know very little about the function of different types of beta bursts or the type of low- vs high-level processes that they reflect. Therefore, we propose an expanded theoretical framework (Figure 4) that reconceptualizes sensorimotor beta bursts as dynamic computational events arising from the coordinated interplay of temporally aligned synaptic inputs across cortical microcircuits. We propose that beta burst waveforms provide a fingerprint of the spatiotemporal pattern of inputs and thus index particular underlying computational processes. This framework builds on the foundational model developed by Jones and colleagues (Sherman et al 2016) while incorporating recent evidence on burst diversity, laminar organization of cortical afferents, and developmental trajectories.
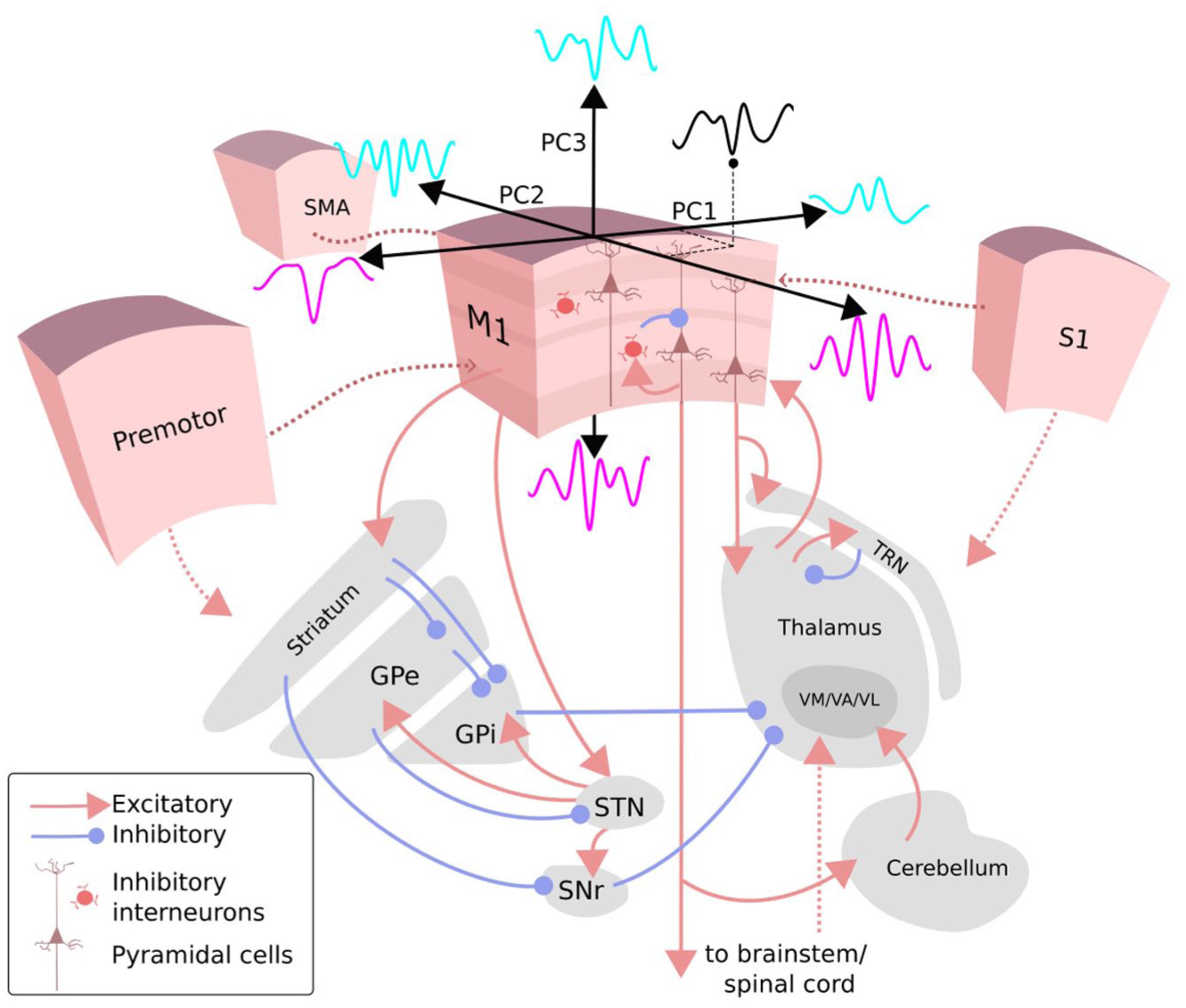
Proposed neural circuits involved in generating diverse beta burst waveforms in sensorimotor cortex. Different beta burst waveforms (illustrated schematically in cyan and magenta) may reflect distinct spatiotemporal patterns of synaptic input from cortical and subcortical regions targeting specific cortical laminae. These inputs arise from multiple sources, including corticocortical projections from the somatosensory (S1) and premotor cortex as well as the supplementary motor area (SMA); thalamocortical projections from the ventromedial (VM), ventral anterior (VA), and ventrolateral (VL) thalamic nuclei; and subcortical circuits involving the basal ganglia (striatum, external globus pallidus (GPe), internal globus pallidus (GPi), subthalamic nucleus (STN), substantia nigra pars reticulata (SNr)) and the cerebellum. The thalamic reticular nucleus (TRN) modulates thalamocortical transmission. Through convergent and recurrent interactions across these pathways, the motor cortex integrates distinct afferent signals across laminae, potentially giving rise to the diverse beta burst waveform shapes observed in electrophysiologic recordings.
Circuit Mechanisms of Beta Burst Waveform Diversity
In the somatosensory cortex, biophysical modeling and empirical MEG data suggest that beta bursts arise from temporally aligned thalamocortical input patterns targeting different cortical layers (Sherman et al 2016; Bonaiuto et al 2021; Law et al 2022). We propose that beta bursts can also result from coordinated reciprocal interactions between cortical and subcortical regions, particularly in motor and associative areas where cortico-subcortical loops may influence their timing, amplitude, and waveform characteristics.
The motor cortex, due to its central role in motor control and learning, receives diverse inputs from frontal, premotor, somatosensory, basal ganglia, and cerebellar regions. These inputs differ in their laminar and dendritic targets (Hooks 2017; Geng et al 2022; Koster and Sherman 2024). Corticocortical projections from somatosensory cortex (originating in layer 5) primarily target upper layers (layer 2/3 and layer 5A), while inputs from frontal areas predominantly project to deeper layers (Hooks et al 2013; Hooks 2017). Additionally, motor cortex layer 5 pyramidal cells project extensively to subcortical targets, including the thalamus, brainstem, and spinal cord (Weiler et al 2008).
These diverse afferent connectivity patterns suggest that beta burst waveforms in the motor cortex may reflect the integration of multiple, functionally specific input configurations (Geng et al 2022). Several interacting loops may contribute to this diversity. The motor cortex forms reciprocal connections with the thalamus through cortico-thalamo-cortical loops, characterized by lamina-specific input–output organization (Shepherd and Yamawaki 2021). It also sends outputs to the subthalamic nucleus (STN) via the hyperdirect pathway and indirectly via basal ganglia circuits. The reciprocal loop between the globus pallidus externus and STN has been proposed to propagate beta bursts within basal ganglia networks (Mirzaei et al 2017). Shared thalamic inputs to cortical and basal ganglia regions may facilitate coordinated communication between these structures (Kuramoto et al 2009). While STN bursts have been observed to precede thalamic bursts during action suppression, the directionality between cortical and STN bursts remains unclear, with evidence for cortical- and STN-leading dynamics (Diesburg et al 2021). Cerebellar inputs further converge with basal ganglia outputs onto overlapping thalamic nuclei, providing additional integrative pathways (Koster and Sherman 2024). Peripheral waveform features in beta bursts may thus reflect the engagement of these modulatory cortical and subcortical loops.
We propose that different beta burst waveform shapes represent distinct underlying circuit-level computations, reflecting diverse combinations of cortical and subcortical inputs. Examining burst waveform shapes, particularly their temporal domain features such as sharpness and nonsinusoidality, may help differentiate bursts with different functional origins and roles. Increased waveform sharpness, for example, has been linked to pathologic states such as Parkinson disease (Cole et al 2017) and used to characterize the subthalamic burst termination (Yeh et al 2020).
While this discussion has focused primarily on the motor cortex due to its rich cortico-subcortical connectivity and functional relevance, we suggest that this framework may generalize to other cortical regions exhibiting burst-like beta activity. In prefrontal and frontal cortices, beta bursts have been linked to cognitive control and working memory (Rodriguez-Larios and Haegens 2023; Lundqvist et al 2024), including the suppression of task-irrelevant information (Lundqvist et al 2016; Lundqvist et al 2018), the active clearance of working memory contents (Lundqvist et al 2018), and the detection of salient or unexpected stimuli (Tatz et al 2023). Frontal beta bursts also support response inhibition, with frontal beta bursts predicting successful stopping in stop signal tasks (Wessel 2020; Muralidharan et al 2022). Importantly, recent work has shown that such bursts are embedded in larger cortico-striato-thalamo-cortical loops, suggesting that the underlying mechanistic architecture may be conserved across domains (Liljefors et al 2024). While the applicability of this framework to temporal, parietal, and visual cortices remains less clear, given differences in local circuit motifs and subcortical projections (Sherman and Guillery 2011), task-modulated beta bursts are observed in parietal and visual cortex as well, particularly in the context of perceptual processing for working memory (Liljefors et al 2024). Whether these bursts reflect similar principles of input convergence or emerge via distinct circuit motifs remains an open and testable question.
The New Framework: Beta Burst Waveforms as Fingerprints of Cortical Computations
As described earlier, the model developed by Jones and colleagues (Sherman et al 2016) characterizes beta bursts as events that emerge when strong, transient excitatory inputs to superficial cortical layers are temporally aligned with broader, weaker synaptic drives to deeper layers. Our expanded framework suggests that the observed diversity in beta burst waveforms, now well documented across multiple studies (Karvat et al 2020; Kosciessa et al 2020; Power et al 2023; Rayson et al 2023; Szul et al 2023; Papadopoulos et al 2024a, 2024b), reflects diverse patterns of synaptic input that indicate distinct computational processes within cortical-subcortical networks.
This perspective represents a significant paradigm shift from traditional interpretations that viewed beta activity either as primarily inhibitory signals or as sustained oscillatory rhythms. We propose instead that beta bursts are temporally precise integration events that reflect the convergence of heterogeneous neural inputs with distinct functional roles. Different waveform motifs serve as functional signatures, indicating the timing and composition of synaptic inputs from diverse cortical and subcortical sources. Biophysical modeling and high-precision MEG studies show that these inputs target specific cortical layers in distinct temporal patterns during burst generation (Sherman et al 2016; Bonaiuto et al 2021), with superficial layers predominantly active around the burst peak and deeper layers showing prominent activity at burst onset and offset. Importantly, different sources of synaptic input target distinct combinations of cortical layers, generating different spatial configurations of synaptic currents. These differences drive current flow in different directions within the cortical column, producing burst waveforms with distinct shapes that potentially reflect the laminar profile and temporal alignment of those inputs, although empirical validation is still needed. Finally, the framework extends beyond feedforward models of thalamocortical input to incorporate closed-loop interactions between cortex and subcortical structures. In particular, basal ganglia–thalamocortical circuits form recurrent loops with motor cortex, where afferent and efferent projections influence burst dynamics. Computational evidence suggests that the precise timing and laminar targeting of cortical inputs can shape the resulting waveform (Szul et al 2023), supporting the idea that beta bursts reflect transient patterns of integration across distributed motor circuits.
Developmental and Clinical Implications
Our framework provides a mechanistic explanation for the developmental changes observed in beta burst characteristics from infancy to adulthood (Rayson et al 2022, 2023). The systematic modulation of specific waveform motifs during sensorimotor tasks across development suggests that beta bursts reflect the progressive maturation of cortical networks involved in sensorimotor control. The transition from broad, bilateral, and weakly modulated bursts in infancy to more focal, lateralized, and selectively modulated bursts in adulthood likely mirrors the maturation of thalamocortical and corticocortical connectivity, tracking improvements in motor precision and interhemispheric specialization. These developmental changes in burst diversity and rate dynamics may also serve as sensitive biomarkers for neurodevelopmental and movement disorders. Moreover, burst-resolved decoding methods, particularly those that leverage waveform features, show promise for enhancing brain–computer interface performance by enabling more accurate and responsive decoding of motor imagery (Papadopoulos et al 2024a, 2024b).
Testable Predictions
This expanded framework generates several testable predictions.
Manipulation of specific inputs to different cortical layers should predictably alter beta burst waveform characteristics and associated motor behavior.
Disorders characterized by abnormal basal ganglia–thalamocortical signaling (eg, Parkinson disease) should show distinctive alterations in beta burst waveform diversity rather than simply changes in overall burst rate, duration, peak frequency, or amplitude.
Targeted neuromodulation approaches that restore typical burst waveform diversity should more effectively improve motor function than those targeting only burst rate, duration, peak frequency, or amplitude.
Developmental interventions focusing on normalizing specific beta burst types may enhance motor learning in children with movement disorders.
This reconceptualization of beta bursts as dynamic computational events resulting from the integration of diverse inputs provides a richer theoretical foundation for understanding their role in sensorimotor processing. By moving beyond simple oscillatory models and models that capture only the “average burst,” this framework offers deeper insights into how beta bursts may contribute to the coordination of complex motor behaviors and how their dysfunction may relate to neurologic and developmental disorders.
Challenges and Limitations
Inferring mechanistic drivers from beta burst waveforms remains an underdetermined inverse problem. Different combinations of laminar and subcortical input can, in principle, give rise to similar cumulative dipole moment dynamics, introducing degeneracy into the mapping between input pattern and waveform shape. While biophysical models provide a principled scaffold (Sherman et al 2016; Bonaiuto et al 2021; Law et al 2022), they do not yet resolve this ambiguity, particularly for regions with complex patterns of cortical and subcortical afferent input such as the motor cortex. However, recent advances in simulation-based inference offer a promising route toward resolving this degeneracy by enabling the estimation of posterior distributions over synaptic input parameters that give rise to observed waveforms, even in the absence of an explicit likelihood function (Tolley et al 2024).
Comprehensive theories of sensorimotor beta bursts must contend with the bilateral expression of sensorimotor beta activity. While task-related modulation is typically stronger contralaterally (Jurkiewicz et al 2006), ipsilateral beta bursts are consistently observed and exhibit meaningful changes across development and task context (Rayson et al 2023). These events may reflect distinct functional processes, such as interhemispheric coordination or motor inhibition, but they may also share underlying motif structure with contralateral events. It remains an open question whether specific beta burst waveform shapes are expressed independently across hemispheres or instead reflect coordinated dynamics across a shared latent circuit. Resolving this ambiguity will require future studies to track beta burst motifs bilaterally.
While biophysical modeling suggests that beta bursts may cluster into discrete categories defined by specific patterns of synaptic input (Sherman et al 2016; Bonaiuto et al 2021; Law et al 2022), empirical analyses based on PCA often reveal continuous variation in waveform shapes (Rayson et al 2023; Szul et al 2023; Papadopoulos et al 2024a). These continuous axes, however, are not without structure: thresholding along principal component dimensions frequently reveals burst types with distinct rate dynamics and behavioral correlates. This pattern suggests a hybrid view in which burst motifs occupy identifiable regions of a continuous low-dimensional space, with within-motif variability reflecting graded differences in input timing, amplitude, or convergence. PCA has proven effective for uncovering this structure, but its orthogonality constraints and emphasis on maximizing explained variance can bias the decomposition toward components ordered by dominant frequency content. As a result, components encoding mechanistically relevant features may not rank among those explaining the most variance. Alternative dimensionality reduction methods, such as variational autoencoders or generalized contrastive PCA (de Oliveira et al 2025), may better capture latent structures shaped by circuit architecture. In addition, task-informed techniques, such as principal component regression (Bair et al 2006) or demixed PCA (Kobak et al 2016), may improve interpretability by aligning components with functionally meaningful axes.
Conclusion and Future Directions
Beta activity is now widely recognized to occur in transient bursts rather than sustained oscillations. The next major challenge is to understand the functional diversity within these bursts. While most studies continue to treat beta bursts as homogeneous events defined by rate or power, growing evidence highlights rich variability in waveform shape, timing, and spatial distribution. We suggest that these features reflect distinct circuit-level processes and are not merely noise.
Our central proposal is that beta burst waveform shapes are mechanistically meaningful. They act as fingerprints of temporally precise and spatially distributed patterns of synaptic input. This framework expands on earlier dual-input laminar models by suggesting that bursts with different shapes reflect distinct combinations of convergent cortical and subcortical signals. Beta bursts should therefore be viewed not as uniform events but as transient, functionally distinct episodes of integration across neural circuits.
Developmental evidence supports this perspective. The shift from broad, weakly modulated bursts in infancy to more focal, lateralized, and selectively rate-modulated bursts in adulthood parallels the maturation of thalamocortical and corticocortical pathways. These changes suggest that features of beta burst waveforms may follow neural circuit development and could serve as sensitive indicators of emerging motor specialization or atypical developmental trajectories. One potential mechanism underlying the increase in beta peak frequency over infancy and childhood (Johnson et al 2019; Trevarrow et al 2019; Rayson et al 2022, 2023) is the increased myelination of thalamocortical projections during development (Barkovich et al 1988). In the Jones model, beta bursts result from the summation of 2 distinct thalamic synaptic drives targeting different cortical layers, and the shape of the burst waveform reflects the width of the temporal distribution of these inputs (Sherman et al 2016). As myelination increases, conduction delays become more uniform (Salami et al 2003), reducing temporal dispersion and potentially producing sharper burst waveforms with higher peak frequencies.
Beta bursts have significant clinical and translational potential. In Parkinson disease, prolonged burst duration has been associated with motor impairment (Tinkhauser et al 2017), and it remains an open question whether burst waveform shape is also altered. If so, this could reflect changes in the underlying input patterns or circuit architecture and guide more fine-grained closed-loop stimulation or neurofeedback interventions. Moreover, recent studies have shown that decoding beta bursts based on waveform features improves performance in brain–computer interface systems as compared with traditional power-based methods (Papadopoulos et al 2024a, 2024b). This suggests that targeting specific burst motifs, rather than simply suppressing or enhancing beta activity, may enable more precise and effective neuromodulation strategies. Pathologic burst shapes could first be identified offline by comparing medication on vs off states (Agouram et al 2025) and subsequently detected online by convolving the incoming signal with the target waveform (Papadopoulos et al 2024a), enabling rapid, shape-specific detection for use in closed-loop deep brain stimulation or neurofeedback paradigms.
We outline several priorities for future research that will be essential for validating, refining, and extending the proposed framework:
Methodological standardization: The field needs consensus on burst detection algorithms and standardized benchmarks for evaluating them. In parallel, improved methods are needed for characterizing burst waveform shape in ways that map onto interpretable parameters of mechanistic models of burst generation. Methods should also enable linking waveform-specific burst dynamics to behavior, allowing for more direct testing of computational hypotheses.
Mechanistic integration across scales: Computational models, invasive recordings, and high-resolution imaging techniques such as laminar MEG (Bonaiuto et al 2021) should be used to link specific burst motifs to their circuit-level origins. Invasive recordings can relate burst dynamics to neural spiking activity, providing the ground truth at a microcircuit level. Laminar MEG can resolve the spatial distribution and propagation of bursts across cortical layers and regions. Computational models are essential for bridging these methods, enabling inference about the synaptic and circuit mechanisms underlying burst waveform diversity.
Developmental and clinical translation: Longitudinal studies are needed to track how burst diversity evolves with motor development and underlying brain structure. Such studies can test whether changes in cortical and subcortical connectivity predict the emergence of specific burst waveform dynamics, which in turn predict improvements in motor skill. This approach offers a more causal test of how distinct projections contribute to burst generation and the functional roles of different burst types. In clinical populations, altered connectivity or circuit dysfunction may be reflected in abnormal burst waveform dynamics, providing mechanistic insight and targets for intervention.
Cross-region generalization: Future work should test whether burst waveform diversity supports specialized functions across different cortical regions, including frontal, prefrontal, temporal, parietal, and visual areas (Lundqvist et al 2016; Lundqvist et al 2018; Wessel 2020; Muralidharan et al 2022; Rodriguez-Larios and Haegens 2023; Tatz et al 2023; Liljefors et al 2024). A related direction involves examining, in the context of the proposed framework, the relationship between cortical and peripheral beta bursts detected in motor units (Bräcklein et al 2022; Abbagnano et al 2025). This could offer insights into how central and peripheral systems coordinate during motor control and how burst dynamics propagate across levels of the motor hierarchy.
In summary, sensorimotor beta bursts provide a tractable window onto dynamic neural computation and may serve as a prototype for understanding burst dynamics across other brain regions. We propose that their diversity in waveform shape, timing, and spatial distribution reflects an underlying diversity of neural inputs and functional roles. By moving beyond averaged measures and toward mechanistic models grounded in burst diversity, we can gain the tools to probe neural circuits with greater specificity, across development, disease, and intervention. Future research focusing on burst motifs, their propagation patterns, and cross-frequency interactions will deepen our understanding of how beta activity supports behavior. Combining developmental, computational, and clinical approaches will enable not only theoretical advances but also the development of burst waveform-based biomarkers and interventions to improve health and function across the life span.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Research Council under the European Union’s Horizon 2020 research and innovation program (ERC consolidator grant 864550).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
