Abstract
Schizophrenia is a brain disorder that profoundly perturbs cognitive processing. Despite the success in treating many of its symptoms, the field lacks effective methods to measure and address its impact on reasoning, inference, and decision making. Prefrontal cortical abnormalities have been well documented in schizophrenia, but additional dysfunction in the interactions between the prefrontal cortex and thalamus have recently been described. This dysfunction may be interpreted in light of parallel advances in neural circuit research based on nonhuman animals, which show critical thalamic roles in maintaining and switching prefrontal activity patterns in various cognitive tasks. Here, we review this basic literature and connect it to emerging innovations in clinical research. We highlight the value of focusing on associative thalamic structures not only to better understand the very nature of cognitive processing but also to leverage these circuits for diagnostic and therapeutic development in schizophrenia. We suggest that the time is right for building close bridges between basic thalamic research and its clinical translation, particularly in the domain of cognition and schizophrenia.
Keywords
Introduction
Real-world behavior involving attention, sensorimotor integration, inference, and planning requires coordination of neural activity across multiple cortical areas (Cohen and D’Esposito 2016; Cole and others 2013; Sakai and Passingham 2003). Consider an everyday task of crossing a busy street: if one of the traffic lights suddenly goes out, one would need to integrate inputs from passing cars and other pedestrians to dynamically adjust plans of when to cross. Such “on the fly” adjustment requires that a number of specialized cortical areas dynamically interact, resulting in a shift in which sensory streams are prioritized and motor actions are selected. Key to this sensorimotor reorganization is the engagement of associative cortical areas, such as the prefrontal and parietal areas, which add working memory and salience maps to enhance this process and make it efficient (Hwang and others 2019; Murray and others 2017; Zanto and Gazzaley 2013). Thus, a simple action of crossing a street and indeed most mammalian behavior require flexible integration of information computed and stored in a distributed manner across cortical regions. One of the primary goals of neuroscience is to describe the biological basis of distributed cortical processing and inter-areal interactions.
Over the last few decades, research in humans and other animals have revealed a multitude of conditions requiring interaction between cortical areas. For instance, studies have shown that prefrontal areas can direct visual attention by generating “top-down” signals that change how sensory inputs are processed in the visual cortex (Baldauf and Desimone 2014; Bichot and others 2019). Similarly, prefrontal activity is required for challenging cases of visual object recognition known to be processed in inferotemporal cortex (Kar and DiCarlo 2021). Because of extensive direct connections between cortical areas, a major assumption in the field is that these direct corticocortical pathways implement many cognitive operations requiring distributed processing. However, in addition to direct pathways, mounting evidence suggests that the thalamus provides an indirect route for communication between cortical areas, adding flexibility and efficiency (Bourgeois and others 2020; Fiebelkorn and Kastner 2020; Halassa and Kastner 2017; Murray Sherman and Guillery 2011; Pergola and others 2018; Shepherd and Yamawaki 2021; Wolff and Vann 2019). We clarify throughout this perspective how dissecting the circuit mechanisms of these transthalamic corticocortical interactions is likely to yield a more complete mechanistic description of cognition and its dysfunction in psychiatric disorders (Avram and others 2021; Bourgeois and others 2020; Halassa and Kastner 2017; Ouhaz and others 2018; Parnaudeau and others 2018; Roy and others 2022; Steullet 2020; Svoboda and Li 2018).
In addition to its basic scientific value, understanding the role of the thalamus in regulating interactions across cortical areas is of particular relevance to human health. Recent evidence suggests that large-scale interactions across networks in the human brain are severely perturbed in schizophrenia (Dong and others 2018). These brain-based metrics are in line with the symptomatology reported for this disorder involving abnormal reasoning (Leptourgos and others 2017; Weickert and others 2014; Woodward and others 2009) as well as more recent computational models that reveal abnormalities in latent variables underlying inference (Baker and others 2019) and decision making (Nassar and others 2021). By adding studies that show selective vulnerability of associative thalamic structures in schizophrenia (Giraldo-Chica and others 2018; Huang and others 2019) and the known role of these subcortical structures in cortical functional organization, it is reasonable to assume that thalamic dysfunction may underlie some of the cognitive abnormalities in this disorder. Equally importantly, because the thalamus has been utilized as a neurostimulation target in a variety of brain disorders, it may be an untapped pathway for disease correction in schizophrenia, which we also discuss in this perspective.
In the next sections, we first review the anatomy of thalamocortical loops with a focus on associative structures. Then we discuss functional evidence in support of their role in regulating cortical communication based on data collected across a range of animals and tasks. Finally, we look at thalamocortical dysfunction reported in schizophrenia and propose that the unique role of the associative thalamus in gating intercortical interaction makes it an attractive target for circuit specific therapeutic interventions.
Anatomic Organization of Thalamocortical Systems: Focus on Associative Thalamus
The mammalian forebrain is organized into loops connecting the cortex, thalamus, and basal ganglia (Foster and others 2021). Broadly, the cortex is largely composed of recurrent excitatory circuits (Peron and others 2020) arranged in layers (Radnikow and Feldmeyer 2018) that can maintain activity patterns across multiple time scales relevant to driving adaptive behavior, while local cortical interneurons gate and sculpt these activity patterns (Hu and others 2014). In relation to the cortex, the thalamus is a centrally located subcortical region divided into nuclei, which are primarily composed of excitatory neurons (Butler 2008; Hunnicutt and others 2014; Jones 2007). The thalamus receives inhibitory input from a number of sources, including the thalamic reticular nucleus, basal ganglia, and brainstem (Crabtree 2018; Halassa and Acsády 2016; Halassa and others 2014; Lam and Sherman 2011). In primates and carnivores, excitatory thalamic nuclei contain local inhibitory interneurons, but little is known about their function beyond a few well-studied cases (Leist and others 2016; Wang and others 2011). For example, in the lateral geniculate nucleus, interneurons are thought to engage in a push-pull type of operation that ultimately improves the signal-to-noise ratio of thalamocortical visual input transmission (Wang and others 2011). Because much basic physiology is carried out in rodents and the rodent thalamus is devoid of local interneurons outside the lateral geniculate, we know little about the general properties of these circuits. Instead, considerable focus has been placed on the thalamic reticular nucleus, which forms a shell of inhibitory neurons around the thalamus and provides a major source of inhibition across all thalamic nuclei (Crabtree 2018; Pinault 2004).
The excitatory microcircuits within the thalamus diverge majorly from those of the cortex in one key way—an absence of local excitatory recurrent connectivity (Bickford and others 2008; Hunnicutt and others 2014; Zolnik and Connors 2016). Instead, all excitatory inputs to the thalamus originate from extra thalamic sources. This unique property of the thalamus may be central to its role in intercortical communication, as the lack of local excitatory recurrence allows for different task-relevant variables to be effectively separated across neighboring groups of neurons. This may be key for control functions: thalamic ensembles can be conceived as low-dimensional projections of cortical activity patterns that, due to effective separation, may be well positioned to subsequently exert regulatory effects on cortical dynamics within areas (Bolkan and others 2017; Fresno and others 2019; Guo and others 2017; Rikhye and others 2018; Schmitt and others 2017) and across areas (Blot and others 2021; Kastner and others 2020; Jaramillo and others 2019; Mo and Sherman 2019). Testing different aspects of this hypothesis will require an integrative approach to thalamic connectivity, physiology, and task-relevant engagement, which we discuss throughout this piece.
Over the last several decades, there have been many attempts at a general classification for thalamocortical circuits (Halassa and Sherman 2019; Sherman 2012). Although there is currently no comprehensive classification that accounts for all features, we think that three broad categories can be conceived to assist with functional interpretations. These would sit on a continuum between primary sensory and associative thalamic circuits, with higher-order relays in the middle (Fig. 1). Primary sensory thalamic areas, such as the lateral geniculate nucleus (LGN) or ventral subdivision of the medial geniculate body, mostly form topographic and reciprocal loops with primary sensory cortical areas (Hackett and others 2011; Usrey and Alitto 2015). In addition, they receive their main driving input from subcortical sensory areas while their cortical inputs that originate from layer 6 of their respective sensory cortices are largely modulatory (Homma and others 2017; Usrey and Sherman 2019). For example, in primates, corticothalamic projections from the primary visual cortex to the LGN modulates the timing and precision of thalamic transmission without affecting their receptive field properties (Murphy and others 2021), which are inherited from the retina instead (Alonso and others 1996; Hubel and Wiesel 1961). Due to this organization, primary sensory thalamic areas are best described as high-fidelity relays of information from the sensory organs with minimal intrinsic processing, although there is quite a bit of literature on temporal visual processing by the LGN (Saalmann and Kastner 2011; Tang and others 2016).
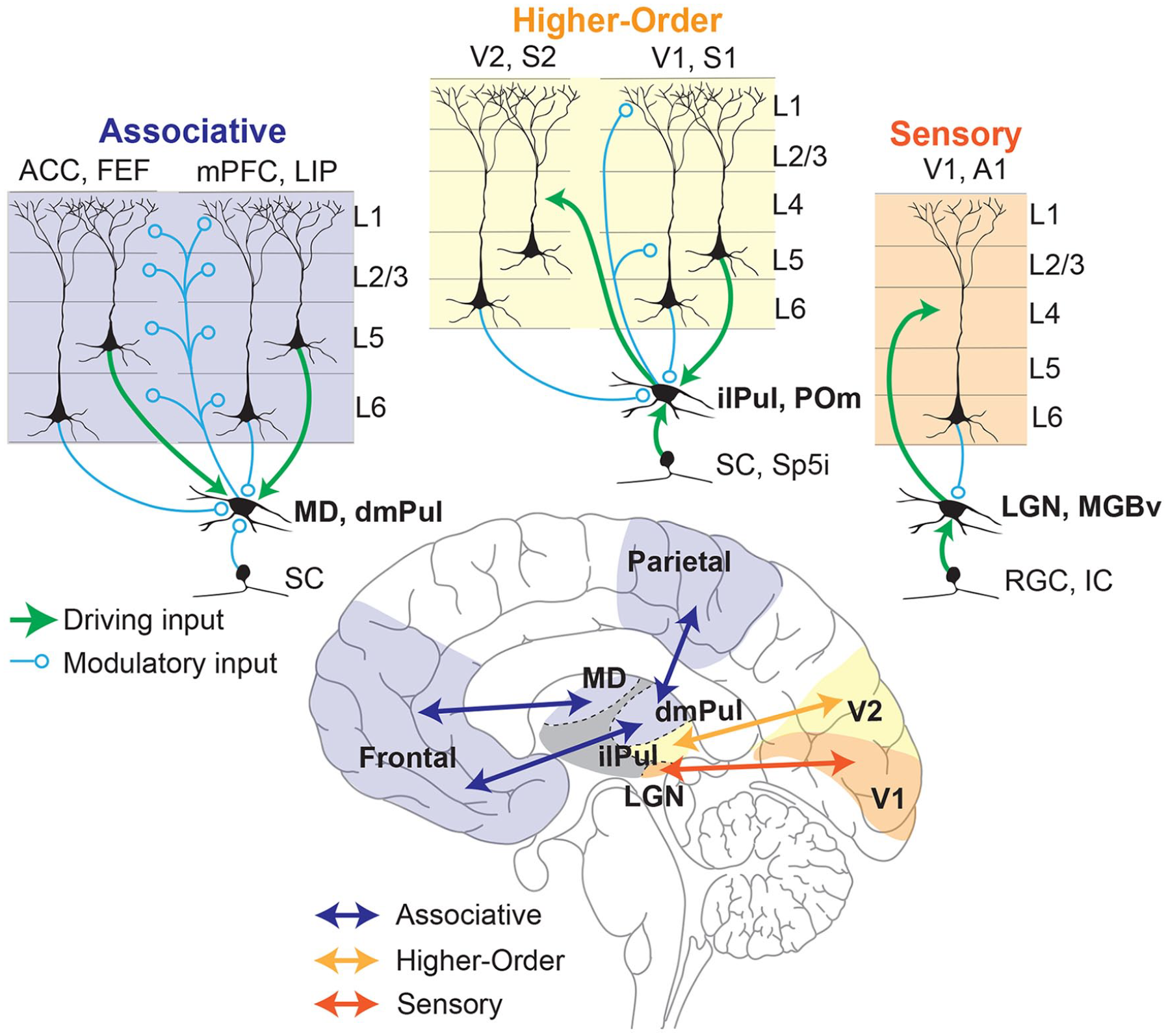
Schematic representation of different thalamocortical circuits within the brain (bottom) with their differences in input/output connectivity highlighted (top). Thalamocortical circuits can be classified in a continuum of circuit motifs based on the origin of their main driving inputs and their cortical outputs. In this scheme (from top right to top left) sensory thalamic nuclei (e.g., LGN) receive their driving input (green) from sensory systems (e.g., retinal ganglion cells [RGCs]) and in turn send driving inputs to their cortical targets (e.g., V1, orange) that terminate mostly in layer 4, thus acting as faithful relays of sensory information to the cortex. Such nuclei receive modulatory inputs (blue) from layer 6 of the cortical areas to which they project (Alonso and others 2001; Murphy and others 2021). Higher-order thalamocortical nuclei (e.g., inferior/lateral pulvinar [ilPul]) can receive their driving inputs from cortical layer 5 neurons (e.g., V1) as well as subcortical areas (e.g., superior colliculus [SC]). These nuclei send modulatory cortical outputs to layers 1 and 5 of the cortical area from which their driving input originates and driving inputs to other cortical areas (e.g., V2, yellow). Such higher-order thalamic nuclei play a functional role in integrating cortical and subcortical inputs to either modulate cortical dynamics or transfer their integrated output to another cortical region (Blot and others 2021; Groh and others 2014; Mease and others 2017). Finally, associative thalamocortical nuclei (e.g., MD) receive their main driving inputs entirely from layer 5 neurons of their cortical partners (e.g., ACC, mPFC, purple) while the subcortical inputs are mostly modulatory (e.g., SC). They in turn modulate cortical dynamics through terminals at all layers of their multiple cortical partners. Associative thalamocortical networks play critical roles in the maintenance and coordination of distributed cortical signals driven by task-dependent inputs and outputs (Bolkan and others 2017; Fiebelkorn and Kastner 2020; Mukherjee and others 2021; Parnaudeau and others 2013; Pergola and others 2018). A1 = primary auditory cortex; ACC = anterior cingulate cortex; dmPul = dorsomedial pulvinar; FEF = frontal eye fields; IC = inferior colliculus; ilPul = inferior/lateral pulvinar; MD = mediodorsal thalamus; LGN = lateral geniculate nucleus; LIP = lateral intraparietal cortex; MGBv = medial geniculate nucleus, ventral subdivision; mPFC = medial prefrontal cortex; POm = posterior medial thalamus; RGC = retinal ganglion cell; S1 = primary somatosensory cortex; S2 = secondary somatosensory cortex; SC = superior colliculus; Sp5i = whisker-sensitive cells of the spinal trigeminal subnuclei, interpolar region; V1 = primary visual cortex; V2 = secondary visual cortex.
It may be surprising to note that most of the thalamus, especially that of primates, is composed of associative areas rather than primary sensory ones. In fact, the two largest thalamic nuclei of the human brain, the pulvinar (dorsomedial division) and mediodorsal thalamus, are largely associative in nature (Halassa and Kastner 2017). Neurons within these areas are largely driven by cortical inputs originating from layer 5 (Usrey and Sherman 2019). Intriguingly, layer 5 cortical neurons that send driving input to associative thalamic nuclei are anatomically distinct from layer 5 neurons that project to other cortical areas, suggesting a distinct function for these pathways from those involved in direct corticocortical communication (Petrof and others 2012).
There are at least two types of functional associative thalamocortical loops that we infer: closed and open loops (Fig. 2). A closed loop indicates functional interactions that are limited to one cortical area and its thalamic partner, whereas an open loop involves multiple cortical areas indirectly connected through a thalamic node. Closed loop interactions have been described for the rodent prelimbic cortex (primate analogue of the dorsolateral prefrontal cortex) and the lateral division of the mediodorsal thalamus (Vertes and others 2015). These interactions involve sustaining cortical representations related to working memory and attentional control signals (Bolkan and others 2017; Guo and others 2017; Schmitt and others 2017), as well as the flexible control/switching of these signals based on changes in input reliability (Mukherjee and others 2021) or temporal context (Rikhye and others 2018). Open loop interactions have been described for the primate pulvinar (Arcaro and others 2015; Fiebelkorn and Kastner 2020; Zhou and others 2016). Higher-order relays are structures that appear to have hybrid properties of sensory and associative thalamic areas. For example, the posteriomedial thalamus of the somatosensory system is driven by layer 5 of the primary somatosensory cortex as well as the brainstem (Groh and others 2014). It has driving and modulatory inputs to higher-order somatosensory and motor cortex (Casas-Torremocha and others 2017; El-Boustani and others 2020). Of note, the primate pulvinar complex is composed of subdivisions that can be thought of as associative (the dorsomedial part) and higher-order relay (inferiorlateral part) (Fig. 1) (Baldwin and others 2017). In nonprimate species, the dorsal equivalent is either underdeveloped or nonexistent (Kaas and Lyon 2007; Zhou and others 2017), rendering the entire pulvinar as a higher-order relay equivalent to the posteriomedial thalamus. The dorsal pulvinar may have evolved in primates alongside the evolution of direct frontoparietal pathways, in line with the “replication principle,” which states that any two cortical pathways that are directly connected are also indirectly connected through the thalamus (Shipp 2003). Electrophysiologic studies in macaque monkeys have focused on a part of the dorsal pulvinar that indirectly connects areas V4 and the inferotemporal cortex, two regions along the ventral visual hierarchy associated with form vision (Saalmann and others 2012; Shipp 2003; Zhou and others 2016). In a combined feature/spatial attention task (Egly and others 1994), this thalamic region shows elevated activity during the delay period of the task when attentional resources are presumably deployed. This elevation appears to also be associated with rhythmicity in the alpha frequency range (10–14 Hz) and accounts for directed functional connectivity between V4 and the inferotemporal cortex (Saalmann and others 2012), indicating a role for the pulvinar in mediating functional corticocortical connectivity (Nakajima and Halassa 2017).
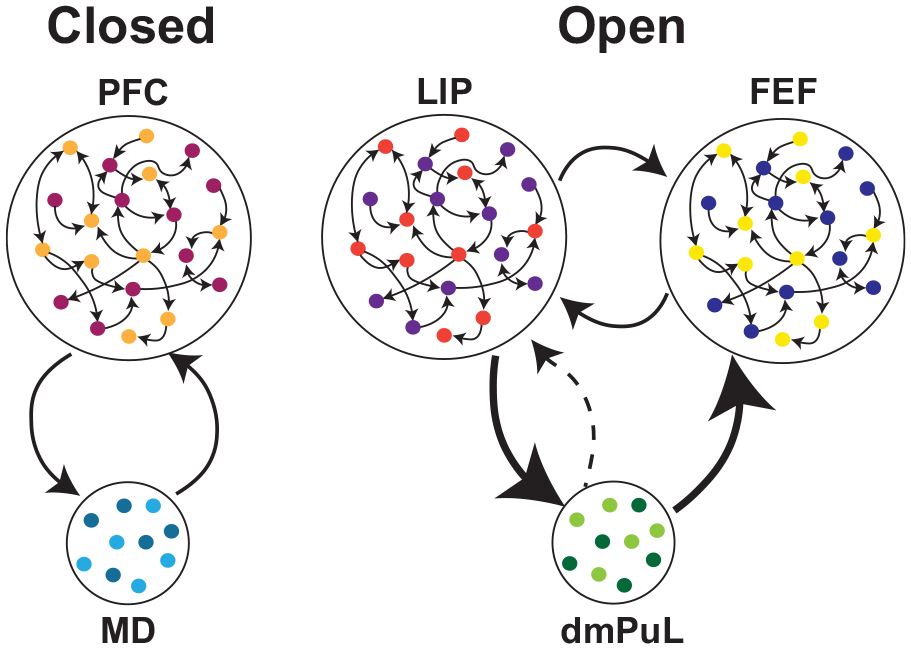
Closed versus open loop associative thalamocortical circuits inferred from function. (Left) A closed loop circuit can be inferred between the mediodorsal thalamus (MD) and the prefrontal cortex (PFC) where the MD uses inputs from the PFC to generate cognitive signals that sustain prefrontal representations as well as mediate flexible control of these signals based on input reliability or temporal context changes (Bolkan and others 2017; Mukherjee and others 2021; Schmitt and others 2017). (Right) A proposed open loop between the lateral interparietal cortex (LIP) and frontal eye fields (FEF) through the dorsomedial pulvinar (dmPuL) for the control of visual attention. Here dmPuL drives increased functional connectivity with FEF and LIP during engaged periods of selective visual attention to emphasize sensory processing, while LIP drives functional connectivity to the dmPuL during periods of disengagement to facilitate motor processing (Fiebelkorn and others 2019; Kastner and others 2020).
Functional Roles of the Associative Thalamus in Cognition
What exactly is the purpose of these thalamocortical loops? While definitive answers are yet to come, we hypothesize that these loops must rely on the distinct architectural features of the cortex and thalamus. To reiterate, the cortex is characterized by local excitatory recurrence and associated endogenous activity, whereas the thalamus is devoid of such features. In addition, one feature that associative thalamic circuits appear to exhibit is input convergence. Specifically, and in contrast to their primary sensory counterparts, associative thalamic circuits appear to integrate signals from distinct cortical sources (Bourgeois and others 2020; Fiebelkorn and Kastner 2019; Hwang and others 2017; Ouhaz and others 2018; Parnaudeau and others 2018; Pergola and others 2018). In some circumstances, the result of this convergence is a dimensionality reduction of the task-relevant variables found in distributed cortical afferents. In other words, they can integrate and transform these signals into novel representations, including temporal context (Rikhye and others 2018), confidence (Komura and others 2013), and uncertainty (Mukherjee and others 2021). Such representations, in turn, may be utilized as cognitive control signals that functionally regulate distributed cortical dynamics rather than relay signals in the classical sense.
To give a concrete example, we expand the discussion on the pulvinar mentioned in the previous section. Specifically, work by Kastner and colleagues has shown that interactions between early and late stations of the dorsal visual pathway in the alpha band range are temporally coordinated by an intermediary connecting pulvinar (Saalmann and others 2012). More recently, the same group showed that this relationship is causal: inactivation of the pulvinar diminishes this corticocortical alpha band coherence in a task-specific manner (Eradath and others 2021). Work by Komura and colleagues (2013), also in macaque monkeys, showed that pulvinar neurons generate “summary statistic”–type responses in a classical visual dot motion task (Fig. 3A). Specifically, when animals are required to make a perceptual judgment on these well-studied stimuli, pulvinar neurons show response patterns that are not reflective of the dominant motion direction, as is the case in cortical area LIP (lateral intraparietal) for example (Roitman and Shadlen 2002; Shadlen and Kiani 2013); rather, their responses reflect the coherence as a scalar quantity regardless of motion direction. Computational modeling of these responses suggests that they may emerge due to convergence of these two motion-sensitive cortical ensembles onto single pulvinar neurons (Jaramillo and others 2019) (Fig. 3B, C). Therefore, responses of pulvinar neurons are not a simple reflection of upstream inputs but under certain conditions can reflect a transformation of a vector input to a scalar signal. Adding Kastner’s work to these findings paints a picture in which the pulvinar may not “relay” such signals but rather generate an output that coordinates activity patterns across distributed cortical ensembles (Bourgeois and others 2020; Kastner and others 2020).
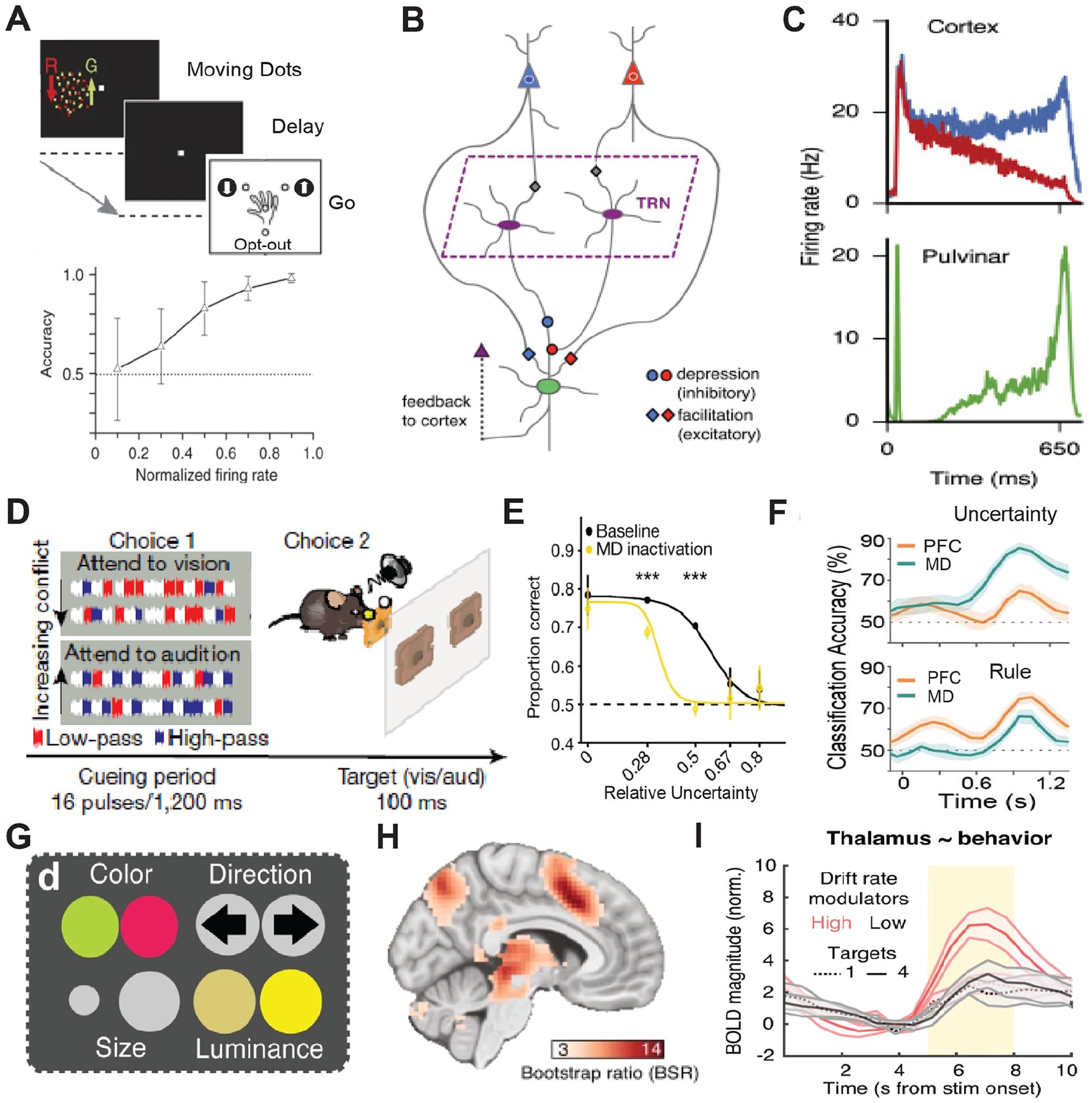
Associative thalamic nuclei integrate convergent cortical inputs into cognitive control signals such as decision confidence or input uncertainty. (A) Top: A random dot motion task with three alternative choices where macaque monkeys had to make a categorical choice based on the relative ratio of dots of one color moving in a particular direction (up or down) to get a reward or opt out for a small but certain reward. Bottom: Activity in the dorsal pulvinar is predictive of an animal’s confidence in choice accuracy. Adapted from Komura and others (2013). (B) Computational model of the dorsomedial pulvinar with two cortical ensembles, differentially sensitive for motion, that converge onto single pulvinar neurons. (C) Simulated pulvinar neurons from the model (bottom) integrate the cortical activity and approximate the absolute value of the difference in rates of the competing cortical neurons (top). Adapted from Jaramillo and others (2019). (D) A rodent attention control task where on each trail an animal has to make a categorical decision on the dominant sound pulse type (high pass vs. low pass) in a mixed string of pulses made of high pass, low pass, and noninformative white noise (choice 1). Subsequently the animal selects the target action (attend to vision vs. attend to audition) that corresponds to the dominant pulse (choice 2). (E) Optogenetic inactivation the mediodorsal thalamus diminishes performance as a function of tracking uncertainty. (F) Mediodorsal thalamic neurons preferentially track input uncertainty while the PFC encodes rules. Adapted from Mukherjee and others (2021). (G) A modification of the random dot motion task where features of the dots vary across four visual dimensions and subjects have to perform a categorical discrimination based on one of the precued features. Subjects were precued to attend to varying number of features (one to four), thus increasing the uncertainty of the feature relevant to the discrimination. (H) Thalamic BOLD magnitude shows correlation to attentional uncertainty and is parametric to it (I). Adapted from Kosciessa and others (2021). BOLD = blood oxygenation level dependent; MD = mediodorsal thalamus; PFC = prefrontal cortex; TRN = thalamic reticular nucleus.
Similar to the pulvinar, the mediodorsal thalamus establishes widespread connections across multiple cortical areas. However, rather than being primarily connected to posterior cortical areas, the mediodorsal thalamus is connected to multiple frontal cortical areas (Li and others 2022; Mitchell 2015). Multiple studies have recently examined mediodorsal function in rodents and showed that neurons within this region have nonrelay properties (Bolkan and others 2017; DeNicola and others 2020; Fresno and others 2019; Hsiao and others 2020; Ouhaz and others 2022; Parnaudeau and others 2013; Rikhye and others 2018; Schmitt and others 2017) that might be similar to the pulvinar studies just described. Specifically, in a cross-modal divided attention task developed in mice, mediodorsal neurons show unique response profiles that are distinct from their connected cortical areas (Fig. 3D, E). In the prefrontal cortex, neurons encode the inputs and outputs of the cross-modal task, whereas thalamic neurons encode changes in input patterns over long and short time scales (Mukherjee and others 2021; Rikhye and others 2018). Encoding of the long-term input changes reflects the statistical regularity of inputs over a multitrial time scale, or the “cueing context.” Encoding of short-term input changes reflects their variance or uncertainty (Mukherjee and others 2021). This may reflect an interesting point of divergence between the mediodorsal encoding of uncertainty (Fig. 3D–F) and the pulvinar encoding the opposite, confidence (Fig. 3A–C). Critically, these short-term summary statistic responses have been found not only in nonhuman animal studies but also in task-based functional magnetic resonance imaging in humans (Kosciessa and others 2021) (Fig. 3G–I). Therefore, although thalamic microcircuitry may be far simpler than that of the cortex, it is nonetheless likely capable of performing important computational transformations that are critical for coordination of distributed cortical signals and in turn cognitive operations. The rodent studies have also shown that the thalamic outputs do not necessarily drive cortical responses in the classical sense but can drive feedforward inhibition (Ferguson and Gao 2018; Mukherjee and others 2020) to slow cortical dynamics in certain contexts or alternatively disinhibit cortical ensembles to implement nonlinear gain control of cortical activity patterns in others (Anastasiades and others 2021; Mukherjee and others 2021).
In summary, the fact that associative thalamic structures provide a mechanism for regulating cortical computations in a precise manner and in turn coordinate activity across large-scale cortical networks led to our proposal where associative thalamic structures could be leveraged to identify and target cognitive control deficits in psychiatric and neurologic disorders. In the next section we focus on schizophrenia as a representative example of this vision.
Mounting Evidence for Associative Thalamocortical Dysfunction in Schizophrenia
Thalamocortical abnormalities are established in schizophrenia and have been shown across multiple stages of the illness, including early psychosis (Avram and others 2021; Woodward and Heckers 2016; Zhang and others 2021). Specifically, structural and functional magnetic resonance imaging have demonstrated abnormalities in patients with schizophrenia (Fig. 4). On the structural end, diffusion tensor imaging has shown that the integrity of thalamocortical connections is diminished, particularly between the areas of the thalamus that are connected to the prefrontal cortex (Hamoda and others 2019) (Figure 4A–C). In fact, reduced frontothalamic connectivity is also detectable in subjects at a high risk of developing schizophrenia (Cho and others 2016; Schleifer and others 2019), suggesting that thalamocortical dysconnectivity may be a potential biomarker as it can precede confirmatory diagnosis. On the functional end, the correlated patterns of activity normally observed between the thalamus and large-scale cortical networks are changed (Dong and others 2018). This phenomenon, which is often referred to as resting state functional connectivity, shows reduced correlated activity (hypoconnectivity) between the thalamus and prefrontal areas (Anticevic and others 2014; Giraldo-Chica and others 2018; Woodward and Heckers 2016; Zhang and others 2021) and increased correlated activity (hyperconnectivity) between the thalamus and sensorimotor areas (Chen and others 2019; Woodward and Heckers 2016) (Fig. 4D, E). Although the exact mapping between these measures and microcircuit function is currently unknown, they clearly suggest that the nature of thalamocortical interactions is changed in schizophrenia and that there is a functional “dysconnectivity” between associative thalamic regions and the prefrontal cortex. Because the hypoconnectivity phenotype has potential circuit parallels based on nonhuman animal studies, we focus on it in this perspective
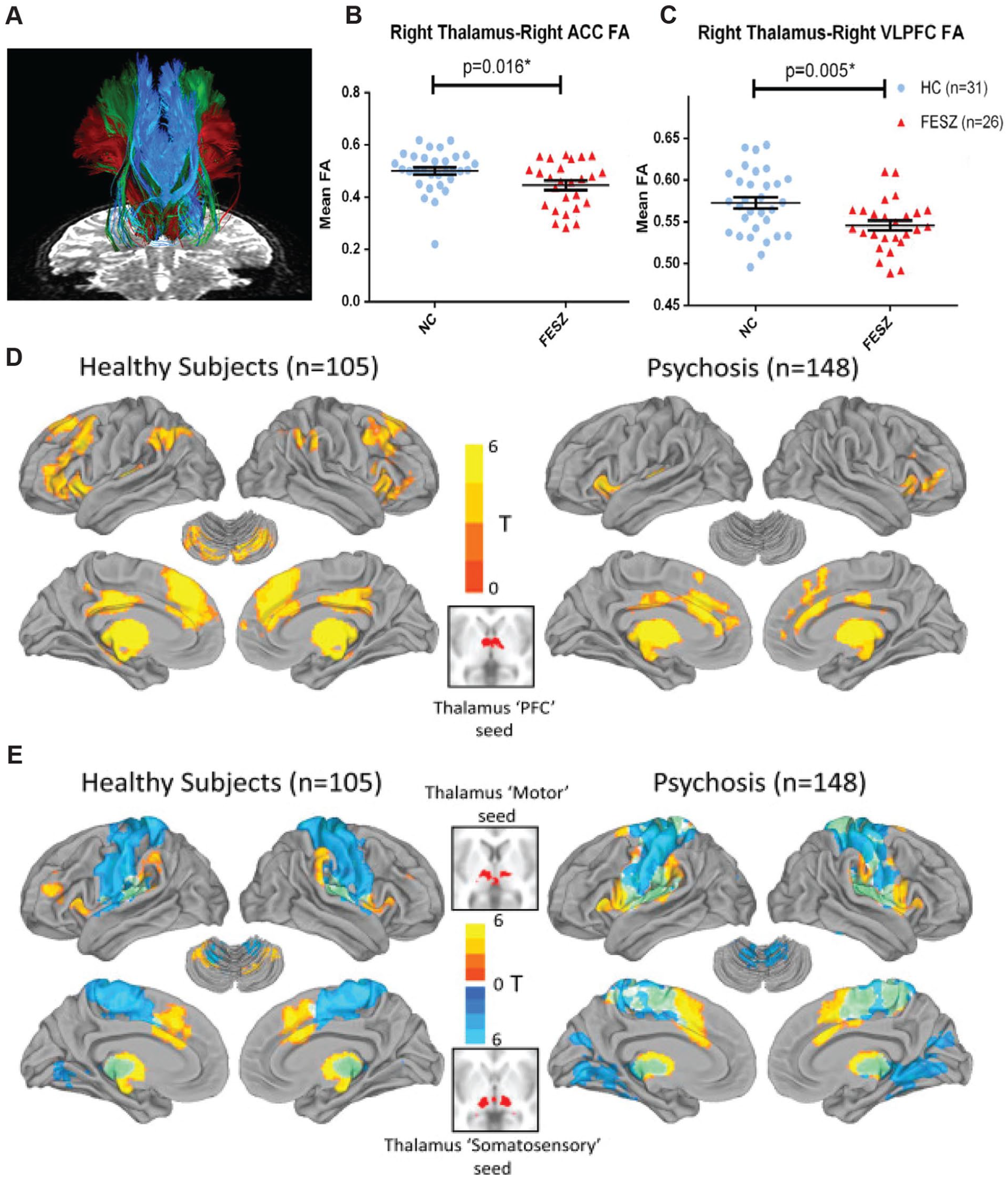
Structural and functional abnormalities in thalamocortical connectivity in schizophrenia. (A) Specific white matter tracts that connect frontal cortical areas—namely, anterior cingulate (ACC), ventrolateral prefrontal cortex (VLPFC), and lateral orbitofrontal cortex—to the thalamus as measured with two-tensor tractography. Thalamus to ACC (B) and VLPFC (C) shows reduced connectivity in patients with schizophrenia. Values are presented as mean ± SEM. Adapted from Hamoda and others (2019). Resting-state functional magnetic resonance imaging comparing functional connectivity between major divisions of the cortex and thalamus of healthy subjects versus patients with schizophrenia shows reduced prefrontal-thalamic connectivity (D) and increased motor (warm colors) and somatosensory (cool colors) thalamic connectivity (E). Adapted from Woodward and Heckers (2016). FA = functional anisotropy ; FESZ = first-episode schizophrenia; HC = healthy controls; NC = normal controls; PFC = prefrontal cortex.
Frontal thalamocortical hypoconnectivity (or reduced coordinated activity) can be interpreted from a perspective that takes into account the advances in circuit neuroscience highlighted earlier. This includes a recent study in mice, which identified two distinct pathways within the mediodorsal thalamus that exert opposing effects on prefrontal circuitry (Mukherjee and others 2021). These two pathways originate from two genetically distinct mediodorsal cell types, one expressing the dopamine D2 receptor and another expressing the kainate receptor Grik4. Leveraging the circuit accessibility of the mouse and using a number of molecular circuit-tracing and identification techniques, this study discovered that the D2+ mediodorsal neurons preferentially innervate prefrontal disinhibitory neurons, positive for vasointestinal peptide (VIP+) (Guet-McCreight and others 2020). In contrast, Grik4+ mediodorsal neurons preferentially innervate prefrontal inhibitory neurons positive for parvalbumin (PV+) (Kim and others 2016). This allows these pathways to preferentially activate or suppress prefrontal activity patterns, a functional consequence that plays out differentially in behavior. In fact, these pathways appear to be required for handling uncertainty in decision making (Bach and Dolan 2012) but are required for distinct types of ambiguous inputs. The Grik4+ pathway appears to be recruited when the incoming evidence is “noisy,” turning on prefrontal inhibition to reduce its impact on the ongoing deliberation process. In contrast, the D2+ pathway is recruited when the evidence is “sparse,” enhancing prefrontal activity to amplify this faint evidence for incorporation into the deliberation process.
It is intriguing that these two pathways straddle two major ideas in schizophrenia: cortical inhibition dependent on parvalbumin interneurons (Lewis and others 2012; Mukherjee and others 2019) and dopamine D2 receptors as the main targets for antipsychotics (Broyd and others 2017). We suggest that these circuit discoveries go one step further: they link these molecular changes to key behavioral and cognitive processes known to be impaired in schizophrenia. Specifically, one of the best documented cognitive changes in schizophrenia is the “jumping to conclusions” phenotype, which is associated with patients making high-confidence judgments on ambiguous inputs (Nassar and others 2021; Ochoa and others 2014; Pytlik and others 2020; Woodward and others 2009). This has been recently linked to applying less scrutiny on internally generated hypotheses and the proneness to delusional thinking (Fouladirad and others 2022). Interestingly, appropriate engagement of the prefrontal cortex in schizophrenia is particularly impacted under conditions of ambiguity (Krug and others 2014) consistent with the possibility that thalamic mechanisms for balancing its excitation and inhibition are impaired. Along these same lines, a number of recent studies have shown that prefrontal-associated networks show abnormally increased responses during initial evidence acquisition but decreased activity during subsequent evidence accumulation, particularly for disconfirmatory evidence (Fig. 5). These brain-behavior changes show correlation to delusional thoughts in clinical ratings (Lavigne and others 2020). Taken together we suggest that the classical notion of a “noisy prefrontal cortex” in schizophrenia (Winterer and others 2004) may thus rely on thalamic mechanisms and in turn be reversed by their targeting. In the final section we discuss how existing techniques for neuromodulation may be implemented to target dysfunctional thalamocortical circuits.
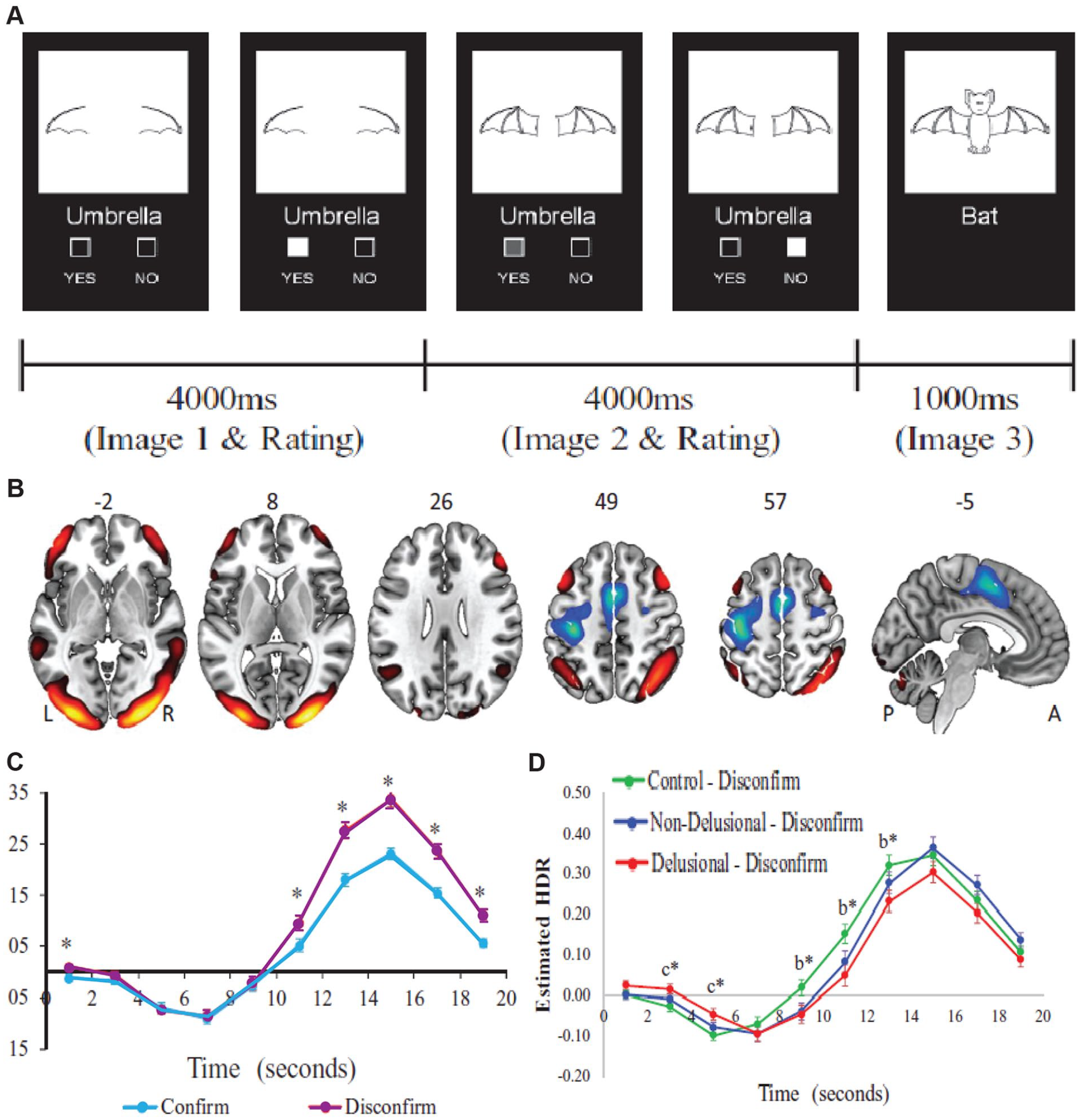
Reduced recruitment of functional brain networks during evidence integration in patients with schizophrenia. (A) In this task, participants were presented with two consecutive partial-line drawings of an object and a word describing it, with the second picture being more complete. Subjects were asked if the displayed word described the full picture upon presentation of the first picture and then to confirm/unconfirm their decision when presented with the second picture. (B) Task-induced activation of the cognitive evaluation network (CEN) which includes the bilateral orbitofrontal and dorsolateral prefrontal cortex, angular and midline temporal gyrus, and occipital lobe. (C) CEN recruitment is higher for disconfirmatory evidence integration (purple) than for confirmatory ones. (D) Patients with schizophrenia and delusion (red) show lower activation of the CEN network as compared with controls (green) as well as nondelusional patients (blue). Values are presented as mean ± SEM. *P < 0.05. bDelusional < controls. cDelusional > controls. Adapted from Lavigne and others (2020).
A Framework for Targeted Thalamocortical Neuromodulation in Schizophrenia
Over the last several years, neuromodulation techniques such as deep brain stimulation (DBS) have evolved into viable therapeutic interventions for treatment of brain disorders (Janson and others 2021; Lozano and others 2019; Schiff 2016). Pioneering work by Schiff and colleagues (2007) has shown that central thalamic DBS in a patient who had been minimally conscious for 6 y after a traumatic brain injury resulted in improved responses to commands, self-feeding, and functional communication. While the exact mode of action of DBS is still under study, the reasons for target selection were grounded in prior research demonstrating that the central thalamus, with its widespread and dense cortical projections, is involved in regulating sleep-wake transitions (Bastos and others 2021; Brown and others 2011), enhancing attentional states (Blumenfeld 2021; Janson and others 2021; Schiff and others 2013), and driving forebrain-wide arousal patterns when stimulated (Shirvalkar and others 2006). Given its mechanistic inspiration and clinical success, could a similar thalamic DBS approach be useful in targeting cognitive deficits in schizophrenia?
As highlighted throughout our perspective, the associative mediodorsal thalamus occupies a central location in prefrontal cortex connectivity and function. Several lines of research in animal models as well as human neuroimaging studies have established the role of the mediodorsal thalamus in regulating interactions within and across prefrontal cortical areas. These studies have further shown that frontothalamic perturbations correlate with a host of dysfunctions characteristic of schizophrenia. Thus, mediodorsal thalamic stimulation with DBS might prove to be a viable therapy, especially in patients resistant to antipsychotic treatment, who account for 30% of the patient population (Chan and others 2021). Indeed, in a recent case study, DBS was targeted to the substantia nigra pars reticulata, a key subcortical input to the mediodorsal thalamus, in a patient resistant to treatment who was experiencing severe hallucinations and thought distortion. A DBS regimen for 24 wk led to a near-complete cessation of hallucinations and thought distortion, and the patient remained stably improved at the one year follow-up (Cascella and others 2021). These results are an excellent proof of principle for using targeted neuromodulation in schizophrenia. It is tantalizing to speculate that this approach may be optimized to preferentially target one of the two mediodorsal thalamic pathways discovered in the mouse work, should they have human analogues.
DBS is an invasive procedure that requires surgical implantation of chronic stimulation electrodes within the brain—a process that can be performed in only a neurosurgical setting and is associated with the risk of infections and hemorrhages (Lozano and others 2019). Thus, in some cases, it may be more viable to look for effective neuromodulation techniques that are minimally invasive.
Transcranial magnetic stimulation (TMS) presents as one such noninvasive technique where an electromagnetically generated electric current is used to depolarize neurons in the brain (Baeken and others 2019). While effective in treating treatment-resistant depression, TMS has provided mixed results in schizophrenia, where it has been targeted to the frontoparietal cortex to treat auditory hallucinations (Marzouk and others 2020), suggesting that further technical refinement is needed before TMS can be effectively used in such clinical settings. Another major drawback of TMS arises from its lack of target specificity and penetrance, restricting its usage to broad cortical regions. However, more recent developments have resulted in improved target localization (Grossman and others 2017). This method shows enhanced spatial selectivity over traditional TMS and therefore could evolve as a potential tool for targeted neuromodulation in patients who are schizophrenic.
A final emerging technology that mitigates the disadvantages of DBS (invasiveness) and TMS (low penetrance beyond the cortex) is focused ultrasonic stimulation (fUS) (Wang and others 2020; Zhang and others 2022). Here, low-intensity ultrasonic sound waves are noninvasively targeted through the skull into the brain with millimeter and millisecond resolution to mediate neuronal activation as well as suppression. While the exact mechanism through which fUS works is yet to be determined, preliminary evidence suggests that it may involve plasticity. fUS triggers voltage-dependent somatic and presynaptic Ca2+ transients within the target area and results in increased expression of neuronal activity–induced transcription factor cFos (Tufail and others 2010). Over the last decade, the number of publications on animal and human studies utilizing focused ultrasound has expanded tremendously (Fishman and Frenkel 2017; Jeong and others 2022; Yang and others 2020). In line with these, Monti and colleagues (2016) recently demonstrated that low-intensity fUS directed to the thalamus over a 10-min period resulted in functional recovery of a patient in a minimally conscious state after traumatic brain injury. While it remains to be seen whether fUS-based neuromodulation would also work in patients who have been in a minimally conscious state long-term, fUS presents us with a potent tool for therapeutic neuromodulation.
We envision a strategy to work similarly for cognitive deficits in schizophrenia, where frontal thalamocortical connectivity is targeted, perhaps even in a closed-loop manner (Fig. 6). Specifically, for each patient, generation of a thalamocortical connectivity model is based on task- and resting-state functional magnetic resonance imaging (Frässle and others 2021; Huys and others 2016). This is followed by fUS of the frontal thalamocortical pathway in the scanner, in which neuromodulation will be titrated via thalamocortical dynamics as neurofeedback signals. A similar approach has been shown to have preclinical efficacy in a rodent model of temporal lobe epilepsy where the hippocampus was modulated with fUS based on the phase of hippocampal theta rhythms (Yang and others 2020). This stimulation paradigm improved the power of the theta rhythms as well as the latency and duration of epileptic seizures in mice. In schizophrenia, there has been a focus on reduced gamma oscillations (Grent-’t-jong and others 2018) and changes in prefrontal excitatory/inhibitory balance (Wengler and others 2020). Therefore, signatures of these processes and their changes in well-controlled tasks can be used as metrics for closed-loop feedback. Along similar lines, selective deficits in closed versus open thalamocortical loops may be discovered in the human brain on the basis of tasks known to preferentially engage the former versus the latter in nonhuman animals. Beyond modulating circuits directly, focused ultrasounds can be used for targeted drug delivery to the brain, either by local opening of the blood-brain barrier or release of drugs from carrier nanoparticles at the site of stimulation (Airan 2017; Carpentier and others 2016; Mainprize and others 2019). Therefore, focal release of D2 antagonists in individual brain regions may better modulate neural circuitry in schizophrenia and allow for increasing dosage with minimal systemic side effects.
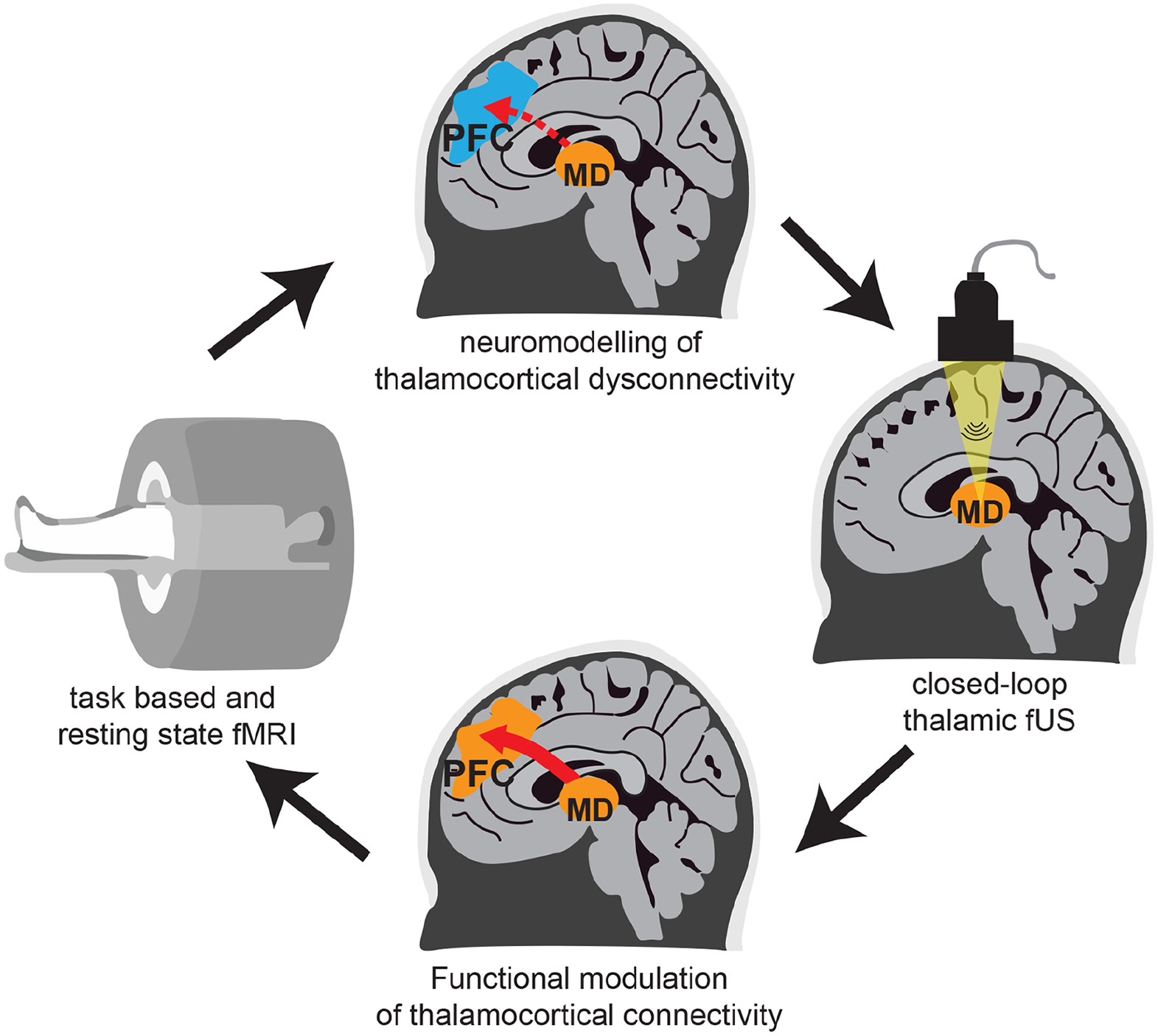
Targeted modulation of thalamocortical dysconnectivity in schizophrenia. Task-based and resting-state functional magnetic resonance imaging (fMRI) are used to stratify patients into different categories of thalamocortical dysconnectivity. Focused ultrasound stimulation of the mediodorsal thalamus is targeted according to this stratification. Functional modulation of dysconnectivity is monitored in real time by using fMRI and behavioral testing to titrate stimulation parameters to individual patients. MD = mediodorsal thalamus; PFC = prefrontal cortex.
Conclusions
The associative thalamus has emerged as a critical node for interactions within and across cortical areas subserving diverse cognitive functions. Ongoing exploration in humans and animal models has provided mechanistic insights into how the associative thalamus implements cognitive operations in the support of flexible behavior. Additionally, we have argued that perturbation of thalamocortical interactions between the mediodorsal thalamus and the prefrontal cortex may form the basis for delusional thinking in schizophrenia. Given the current advances in targeted neuromodulation through DBS, TMS, and focused ultrasound, we are well poised to discover where in the thalamus to target and when in the illness to do so to usher in a new generation of therapy against this disorder.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the authors are supported by grants from the US National Institute of Mental Health (A.M., 1K99MH129613; M.M.H., R01MH120118 and R01MH107680).
