Abstract
Epilepsy is a common neurological disorder associated with alterations in cortical and subcortical brain networks. Despite a historical focus on gray matter regions involved in seizure generation and propagation, the role of white matter (WM) network disruption in epilepsy and its comorbidities has sparked recent attention. In this review, we describe patterns of WM alterations observed in focal and generalized epilepsy syndromes and highlight studies linking WM disruption to cognitive and psychiatric comorbidities, drug resistance, and poor surgical outcomes. Both tract-based and connectome-based approaches implicate the importance of extratemporal and temporo-limbic WM disconnection across a range of comorbidities, and an evolving literature reveals the utility of WM patterns for predicting outcomes following epilepsy surgery. We encourage new research employing advanced analytic techniques (e.g., machine learning) that will further shape our understanding of epilepsy as a network disorder and guide individualized treatment decisions. We also address the need for research that examines how neuromodulation and other treatments (e.g., laser ablation) affect WM networks, as well as research that leverages larger and more diverse samples, longitudinal designs, and improved magnetic resonance imaging acquisitions. These steps will be critical to ensuring generalizability of current research and determining the extent to which neuroplasticity within WM networks can influence patient outcomes.
Epilepsy is defined by the presence of recurrent and unprovoked seizures and affects approximately 50 million people worldwide (Bell and others 2014). Once considered predominantly a gray matter disease, epilepsy is now understood to affect white matter (WM) networks throughout the brain, typically characterized by loss of WM microstructure and disrupted network connectivity. These widespread alterations are observed in patients whose seizures originate in localized regions of the brain (i.e., focal epilepsy) as well as those whose seizures originate broadly and often bilaterally (i.e., generalized epilepsy). Although the origins of WM injury in epilepsy are still debated, its consequences are now better appreciated, with converging studies demonstrating a contribution of WM disconnection to neurobehavioral comorbidities, measures of disease severity, and postsurgical outcomes.
In this review, we summarize new literature describing patterns of WM network alterations in adults with common focal and generalized epilepsy syndromes, including temporal lobe epilepsy (TLE), extratemporal focal epilepsy (ExE), and genetic generalized epilepsy (GGE). We focus our review on results obtained from diffusion-weighted magnetic resonance imaging (dMRI) since dMRI has become the most widely used noninvasive method for interrogating WM microstructure and architecture in human neuroscience. We then provide evidence from dMRI research that WM alterations may underlie common cognitive and psychiatric comorbidities in epilepsy, as well as aid in the prediction of postoperative cognitive, seizure, and visual field outcomes. Finally, we address new data using advanced dMRI sequences and analytic procedures (e.g., machine learning), which may accelerate our understanding of the neurobiology of epilepsy and lead to enhanced predictions of patient-specific outcomes. Box 1 presents definitions of key terms used throughout this review.
Definitions and Key Terms
Diffusion-weighted MRI (dMRI): A form of magnetic resonance imaging that generates images that utilize the diffusion patterns of water molecules, which allows for the detection of microstructural details of normal or abnormal anatomy of a given region in vivo and noninvasively. An extension of dMRI called diffusion tensor imaging (DTI) tractography is used widely for reconstructing white matter tracts in the brain.
Fractional anisotropy (FA): The most common summary measure of microstructural white matter integrity used in DTI studies that assesses the degree of anisotropy of water molecules, from which alterations in axonal diameter, fiber density, and myelination of white matter can be inferred. It ranges from 0 (i.e., isotropic movement of water molecules equally restricted in all directions) to 1 (i.e., anisotropic movement of water molecules, e.g., in fiber bundles in which a diffusion occurs only along one axis and is fully restricted in all other directions). Although reduced FA is typically conceptualized as reflecting reduced myelin content, some findings suggest that increases in WM connectivity can reflect pathological wiring, although the origin of this is not well understood.
Mean diffusivity (MD): Another measure of microstructural integrity of white matter defined as an inverse measure of the membrane density that is more sensitive to cellularity, edema, and necrosis. Describes the rotationally invariant magnitude of water diffusion within tissue and can be affected by any disease process that affects the restriction of the barriers to the motion of water.
Radial diffusivity (RD): Reflects diffusivity perpendicular to axonal fibers and is influenced by changes in the axonal diameters or density. RD is thought to be more strongly related to myelin abnormalities (i.e., demyelination).
Structural connectome (SC): A comprehensive and individualized white matter analysis approach that examines a map of brain network connectivity. This requires measuring the strength of region-to-region connections within an individual. Connectome-based models have the potential to provide more fine-grained information about patterns of abnormal cortico-cortical connectivity underlying cognitive impairments than long-range tractography methods.
Graph theory: A mathematical framework that allows for the quantitative modeling and analysis of the topological properties of complex interconnected systems, which has made a considerable impact on understanding brain connectivity. Such an approach has been pivotal in a shift toward understanding temporal lobe epilepsy as a network disorder.
Hippocampal sclerosis (HS): A lesion characterized by cell loss and gliosis in the hippocampal formation usually seen as atrophy, increased signal, and loss of internal architecture of the hippocampus on MRI.
Parahippocampal cingulum (PHC): The inferior segment of the cingulum (a white matter tract projecting from the cingulate gyrus to the entorhinal cortex), which has been implicated in episodic memory. The PHC runs along the ventral aspect of the hippocampus.
Fornix: A C-shaped white matter bundle that serves as the main output tract of the hippocampus and plays a role in transmitting information from the hippocampus to the mammillary bodies and to the anterior nuclei of the thalamus. The fornix is believed to play an important role in cognition and episodic memory.
Uncinate fasciculus (UF): A curved relatively short fiber that connects the prefrontal and anterior temporal regions. Although its exact function is not understood, it has been associated with episodic and working memory, as well as with language (mainly semantic processing) and socio-emotional processing.
Inferior longitudinal fasciculus (ILF): A long-range associative white matter tract that connects the occipital and temporo-occipital areas to the anterior temporal lobe. The ILF serves as the ventral visual stream important for visual recognition (e.g., objects, faces, places) as well as an “indirect” route for language. The ILF is thought to support multiple cognitive functions including object and face recognition, lexical and semantic processing, and emotion processing.
Inferior fronto-occipital fasciculus (IFOF): The IFOF is a “direct” language route as part of the ventral stream, proposed to connect the occipital cortex to the anterior temporal and inferior frontal cortices. The IFOF is thought to play a role in semantic processing via direct connections of basal-temporal areas with frontal and temporal-parietal cortex, with stimulation leading to disrupted semantic processing (i.e., semantic paraphasias or errors in speech that are related to an object’s meaning).
Arcuate fasciculus (AF): An association tract that connects the temporal and inferior parietal cortices to the frontal cortex, and specifically connects the inferior frontal gyrus (i.e., Broca’s area) and the superior temporal gyrus (i.e., Wernicke’s area). The AF is considered as part of the dorsal pathway for language and is implicated in several language functions (e.g., syntax, repetition, phonological processing, and prosody).
Superficial white matter (SWM): A thin layer of white matter just underneath the cortex, composed of short U-shaped association fibers that provide cortico-cortical connections between adjacent gyri, and represent most of the brain’s white matter connections. SWM is thought to play a role in brain maturation and neuroplasticity. Despite its importance in white matter connectivity, the application of SWM to neurological disease in humans (e.g., epilepsy, autism, Alzheimer’s disease) has been only recently applied.
Perforant path: The major input to the hippocampus that provides connections from the entorhinal cortex to hippocampal subfields including the dentate gyrus, CA1 and CA3, and the subiculum. This path has a major role in memory retention and retrieval.
Anterior temporal lobectomy (ATL): The most common resective surgery for medication-resistant temporal lobe epilepsy introduced in the 1950s. ATL achieves seizure freedom in 60% to 80% of patients and requires removal of the anterior portion of the inferior and middle temporal gyri, the uncus, a portion of the amygdala, and the anterior 2 to 3 cm of the hippocampus and adjacent parahippocampal gyrus.
Perisylvian: Regions of the brain responsible for language found around the lateral sulcus (i.e., Sylvan fissure) of the left hemisphere that include deep white matter tracts that connect fronto-temporo-parietal regions.
Executive function: A broad category of higher-level cognitive abilities including working memory, set-shifting, and inhibition.
Interictal psychosis: Psychosis that occurs in approximately 6% of individuals with epilepsy, with the onset not during or immediately following a seizure. Symptoms of interictal psychosis in epilepsy overlap with symptoms in schizophrenia, such as paranoid delusions and hallucinations.
Drug-resistant epilepsy: When a person has failed to become seizure-free with adequate trials of two antiseizure medications.
Quadrantanopia: A loss of vision in one quarter of the visual field. A homonymous superior quadrantanopia, which presents as a loss of vision in the same upper quadrant in both eyes, is common in patients who undergo ATL due to damage to the inferior optic radiations of the temporal lobe (i.e., Meyer’s loop). Individuals can compensate for the vision loss by tilting their head to bring the affected visual field into view.
Wallerian degeneration: An active process of injury-induced degeneration of the distal end of an axon after neuronal loss or death. Seizure-induced damage from abnormal neural firing and hyperexcitability may cause secondary white matter degeneration along the seizure propagation pathway. Using DTI, early axonal breakdown has been attributed to reduced parallel diffusivity, whereas later myelin degradation is attributed to elevated perpendicular diffusivity.
WM Network Abnormalities Within and Across Epilepsy Syndromes
The presence of WM abnormalities in epilepsy has long been observed, with earlier studies identifying WM hyperintensities, as well as global or regional WM volume loss in patients with different epilepsy syndromes. However, the extent of these abnormalities and their intrinsic patterns were not fully appreciated until the advent and application of dMRI tractography in the early 1990s. In particular, dMRI has emerged as the method of choice for interrogating WM structure in epilepsy due to its ability to derive quantitative measures of individual fiber tract integrity and characterize the adverse effects of epilepsy on cortico-cortical disconnection, even in the absence of direct injury to the cortex.
However, epilepsy is not a single disorder. Instead, the epilepsies are a group of disorders that are unified by a common symptom (i.e., seizures) that can originate from almost anywhere in the brain. For this reason, WM regions, tracts, and networks affected by epilepsy do not follow one uniform pattern, but rather have some syndrome-specific features with abnormalities that are often most pronounced proximal to the seizure focus. Two recent, large-scale studies have well-characterized these patterns for the most common epilepsy syndromes (Hatton and others 2020; Slinger and others 2016), and therefore, each pattern is only briefly summarized below.
TLE
TLE is the most common focal epilepsy syndrome in adults and, therefore, has received the most attention. In TLE, seizures most commonly arise from the hippocampus and other medial temporal lobe structures. For this reason, attention has focused on hippocampal efferent and afferent tracts, including the parahippocampal cingulum and fornix, as well as the uncinate fasciculus (Fig. 1A). These tracts are among the most affected in patients who have gliosis and cell loss in the hippocampus (i.e., hippocampal sclerosis [HS]), and in those with an early age of seizure onset and longer disease duration, with effects larger on the side ipsilateral to the seizure focus (Hatton and others 2020). In addition, temporo-limbic tract alterations in TLE appear to follow a centrifugal pattern such that microstructural abnormalities increase along each tract as they approach the seizure focus (Concha and others 2012). This pattern implies that WM alterations are likely intrinsic to the TLE syndrome rather than general to epilepsy or secondary to treatment-related effects (e.g., antiseizure medications [ASMs]). However, other WM association tracts that course through the temporal lobe (e.g., inferior longitudinal fasciculus) and those distal to the seizure focus (e.g., corpus callosum, external capsule) also show marked WM changes bilaterally in TLE, providing evidence for broad network pathology in patients with focal epilepsy that could represent developmental (i.e., poor myelination of WM tracts) or iatrogenic (e.g., ASM) factors.
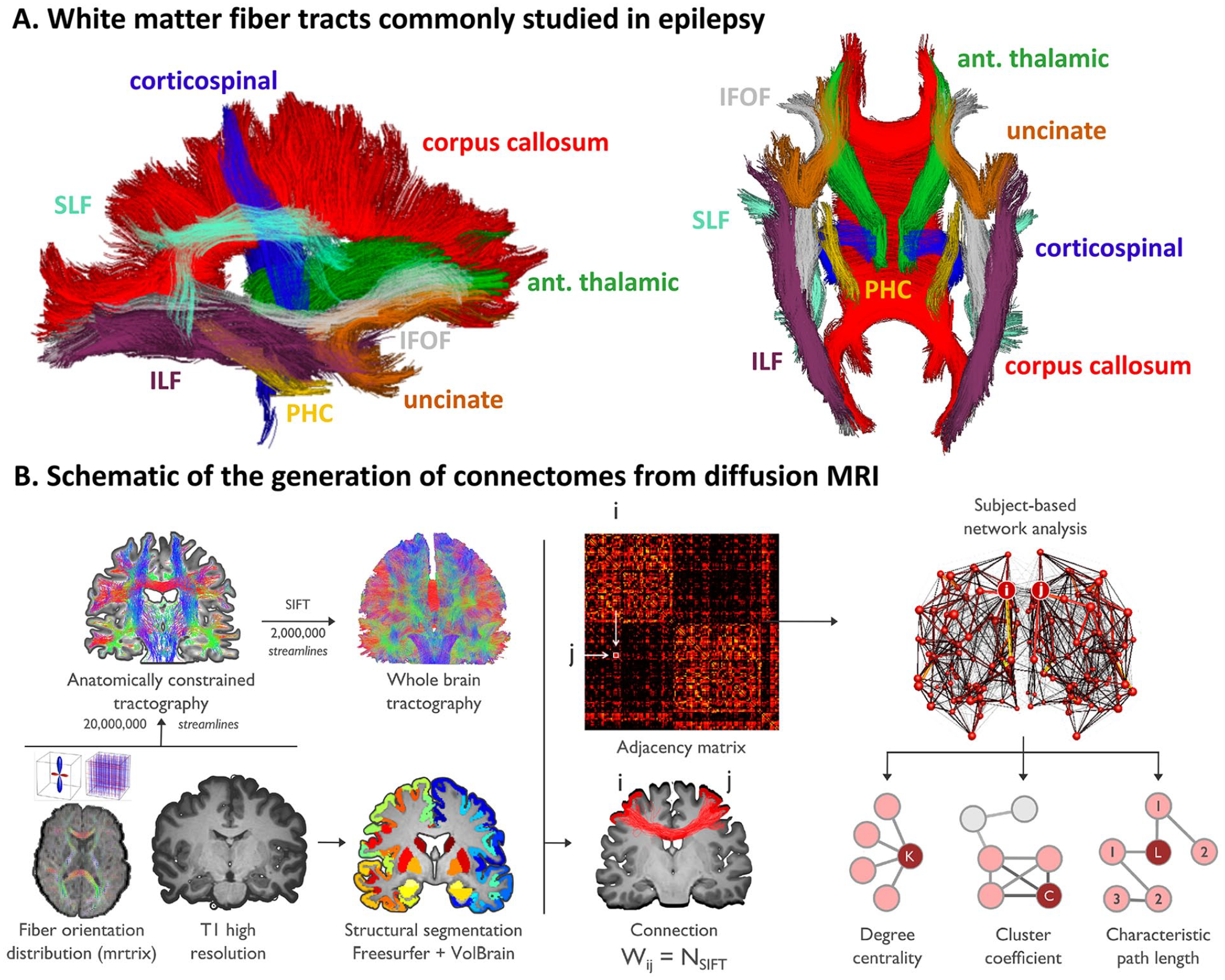
White matter tracts of interest and depiction of structural connectome. (A) DTI-derived fiber tracts that are commonly studied in relation to clinical and cognitive outcomes in epilepsy. Ant. thalamic = anterior thalamic radiations; corticospinal = corticospinal tract; ILF = inferior longitudinal fasciculus; IFOF = inferior fronto-occipital fasciculus; PHC = parahippocampal cingulum; uncinate = uncinate fasciculus; SLF = superior longitudinal fasciculus. Note, the SLF can be further segmented into the arcuate fasciculus (which originates from the posterior temporal cortex), and the first, second and third branches of the SLF that originate from the inferior parietal cortex. (B) Schematic showing the construction of a diffusion MRI connectome. Preprocessed dMRI data are analyzed in an automatically parcellated anatomical space. Adjacency (i.e., connectivity) matrices are then generated by systematically assessing pairwise associations between pairs of all regions (with regions i and j given as an example). Connectivity matrices are equivalent to brain graphs, where brain regions correspond to nodes and structural connections correspond to edges. Connection weight (Wij) is defined as the number of fiber tract connections between two nodes (i and j). The final step (top right) includes graph theory analysis based on the adjacency matrix to extract brain network topological organization (i.e., degree centrality, cluster coefficient, characteristic path length). Adapted from Rodríguez-Cruces and others, 2020, with permission.
ExE
Similarly, patients with ExE harbor a focal epilepsy syndrome with seizures originating from one or more extratemporal areas of the brain, typically in the frontal lobes. Although studies in ExE, such as frontal lobe epilepsy (FLE), are more scarce, decreases in fractional anisotropy (FA) and increases in mean diffusivity (MD) have been shown throughout the frontal lobe WM and frontostriatal fibers, with marked alterations along midline bundles and tracts, including the genu and body of the corpus callosum (Widjaja and others 2014), anterior corona radiata, dorsal cingulum, and external capsule (Hatton and others 2020). Associations between clinical variables and WM disruptions in ExE have been less consistent, with some studies demonstrating that an early age of onset and/or longer disease duration is associated with poorer WM network integrity (Lin and others 2020; Wang and others 2011) and others not finding associations (Hatton and others 2020). The heterogeneity within ExE makes this syndrome challenging to study as a single group, and clinico-diffusion correlations more difficult to capture.
GGE
GGE includes several related syndromes with generalized seizure onset, including juvenile myoclonic epilepsy (JME), where a predominant genetic contribution is suspected. Although patients with GGE do not have visible structural abnormalities on MRI, thalamocortical dysfunction is often present and accompanied by morphological alterations (Bernhardt and others 2009; Whelan and others 2018). Studies of WM disruption in GGE have suggested greatest alterations in fronto-midline fibers, including the genu and body of the corpus callosum, anterior corona radiata, external capsule (Hatton and others 2020), and in thalamocortical pathways (Keller and others 2011; Lee and others 2014). In addition, alterations in pre-supplementary motor area to prefrontal connectivity patterns have been observed and appear unique to GGE syndromes (Vollmar and others 2012). However, there is some evidence that WM alterations in GGE are less severe and widespread than those observed in focal epilepsy (Slinger and others 2016).
Despite these syndrome-specific features, new results from the Enhancing NeuroImaging and Genetics through Meta-Analysis (ENIGMA)-Epilepsy working group have revealed striking similarities in WM compromise across these common epilepsy syndromes. In 1249 patients with TLE, FLE, and GGE compared to 1069 healthy controls, WM alterations were observed within 36 of 38 association, commissural, and projection fibers (Hatton and others 2020;– Fig. 2). Across patient groups, reductions in FA and increases in MD were greatest in fronto-central WM, including the genu and body of the corpus callosum, dorsal cingulum, and external capsule. Although the severity of these alterations varied across syndromes and was most pronounced in TLE with HS, bilateral alterations in many anterior midline fibers were uniform across groups. Although the underlying mechanism(s) that leads to this shared midline pathology is unknown, one possibility is that midline WM is more vulnerable to the direct impact of seizures (locally in GGE and FLE or from seizure propagation via the thalamus in TLE). Another possibility is that midline WM is more vulnerable to neurological or neuropsychiatric injury in general. In support of the latter, Hatton and others (2020) observed very similar patterns of WM disruption between epilepsy and several neuropsychiatric disorders (e.g., bipolar, schizophrenia, depression), with the body and genu of the corpus callosum affected across all disorders. Indeed, several studies have demonstrated cross-disorder connectomic vulnerability, revealing that hub regions that are highly connected and potentially important for communication tend to be disproportionally affected by disease (van den Heuvel and Sporns 2013). Broad patterns of microstructural alterations shared across epilepsy syndromes were not previously appreciated due to a tendency of the field to segregate studies according to single epilepsy syndromes. Although some syndrome-specific findings were evident, shared patterns of WM injury could explain why cognitive and psychiatric comorbidities can be quite similar in two patients with different syndromes but heterogeneous within a syndrome. It is these clinico-diffusion associations that are the focus of this review.
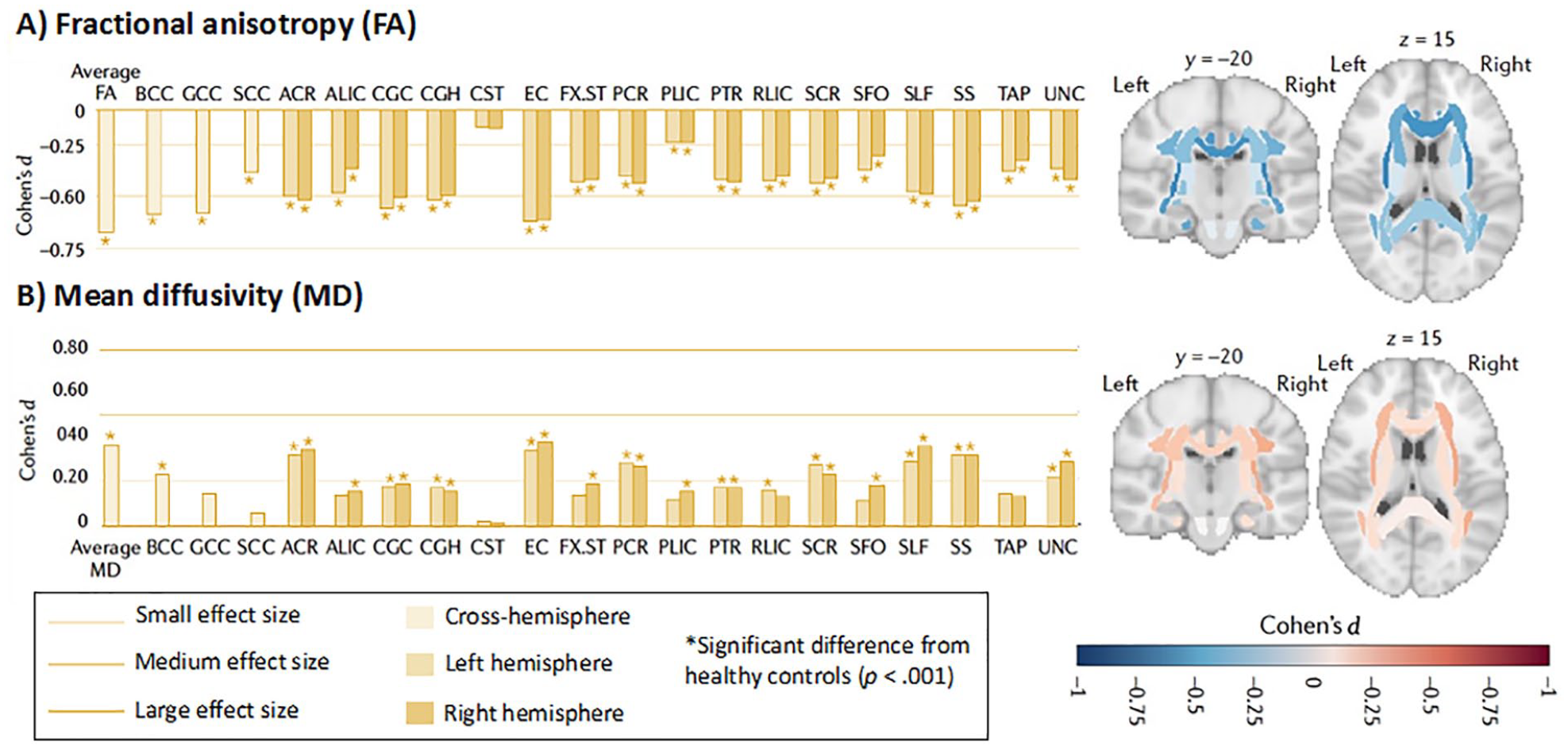
White matter microstructural differences between all epilepsy syndromes compared with healthy controls. All values represent Cohen’s d effect size estimates for differences in (A) fractional anisotropy (FA) and (B) mean diffusivity (MD) between the epilepsy group and healthy controls. Positive effect sizes reflect diffusion values greater than controls; negative effect sizes represent values lower than controls; y and z values represent the slice number for the coronal and axial planes, respectively. Across all epilepsies, the greatest effects on FA were observed in the body of the corpus callosum (BCC) and genu of the corpus callosum (GCC), external capsule (EC), cingulum, and corona radiata. Greatest effects on MD were observed in the EC, anterior corona radiata (ACR), and superior longitudinal fasciculus (SLF). ALIC = anterior limb, internal capsule; CGC = dorsal cingulum; CGH = parahippocampal cingulum; CST = corticospinal tract; FX- ST = fornix; PCR = posterior corona radiata; RLIC = rostral limb, internal capsule; SCC = splenium corpus callosum; SCR = superior corona radiata; SFO = superior frontal occipital fasciculus; SS = sagittal stratum; TAP = tapetum; UNC = uncinate fasciculus. These data are from the ENIGMA-Epilepsy working group (over 2,000 participants). Adapted from Hatton and others, 2020, with permission.
From WM Tracts to WM Networks
Extrapolating from the study of specific WM tracts, a mounting literature has aggregated connectivity information across multiple regions to study macroscale brain network reorganization in epilepsy. These studies have utilized approaches from complex systems analyses such as graph theory, as a formalism to examine changes in WM network topology (Larivière and others 2021; Tavakol and others 2019). Such macroscopic analyses initially generate systematic representations of connectivity, so-called “connectomes,” based on WM tract properties between all pairs of cortical and subcortical regions (Fig. 1B). The topology of the resulting connectomes can then be analyzed at a global scale (by studying network clustering that relates to local communication efficiency, or by studying path length that reflects global efficiency), by examining submodules within the networks through network decomposition techniques, or by studying network embedding of individual regions, with hub mapping being a prominent example of the latter. Complementing graph-theoretical network descriptions, complementary approaches from network neuroscience have emerged, including the use of network communication models that assess how a structurally wired connectome can generate brain dynamics (Girardi-Schappo and others 2021), or the study of spatial trends in network organization, also referred to as connectivity gradients (Huntenburg and others 2018).
Similar to the study of WM tracts, the most robust connectomics literature has focused on TLE. These studies have described shifts in cortical as well as subcortical network topology in TLE at global, modular, and nodal scales. One of the earliest graph theoretical studies in TLE reported reductions in both global and local efficiency in a group of left TLE patients relative to controls, in addition to alterations in hub topography in TLE (Liu and others 2014). These studies have been extended to assess the utility of connectome measures to predict postoperative seizure outcome (see “Postsurgical Seizure Outcomes” section and Table 4). Other studies provided connectome-level evidence for a broad association between degree of mesiotemporal pathology and WM alterations in TLE. In one study, the authors observed overall more marked network reorganization in patients with more severe HS (based on histopathology) compared with TLE patients with only subtle hippocampal pathology (Bernhardt and others 2019; Fig. 3). Other recent investigations used connectome-informed dynamic communication models, underscoring that the alterations in the brain’s WM architecture may relate to delayed dynamic signal flow, and ultimately cognitive impairments across multiple domains in TLE (Girardi-Schappo and others 2021).
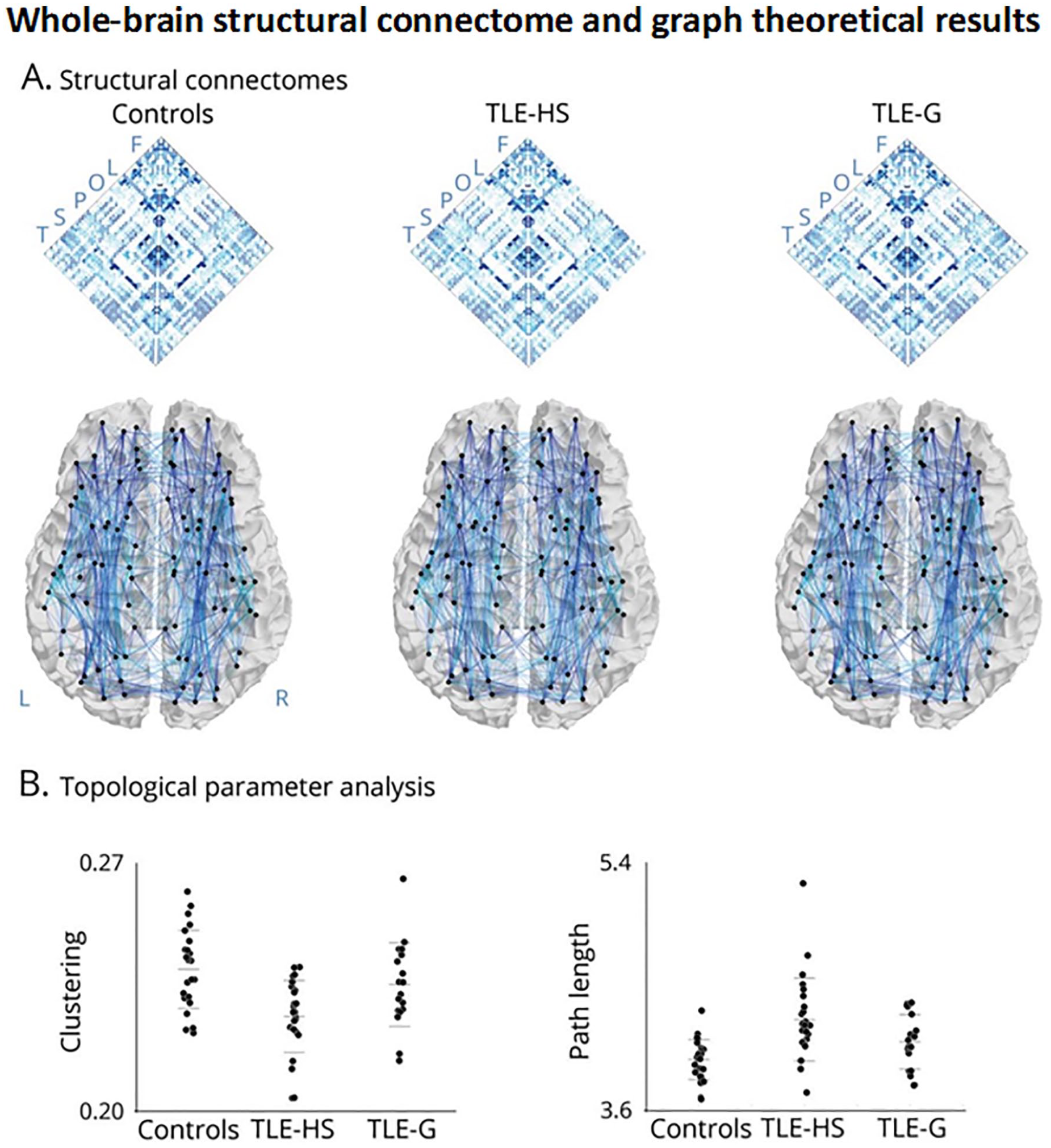
Group differences in network topology. Panel A shows whole-brain structural connectomes in healthy controls, temporal lobe epilepsy (TLE) patients with hippocampal sclerosis (TLE-HS), and TLE patients with isolated gliosis (TLE-G). Maps were generated using diffusion tractography between all regions. Letters refer to regional groupings of the nodes (F = frontal; L = limbic; O = occipital; P = parietal; S = subcortical; T= temporal). Panel B depicts whole-brain graph theoretical results showing a markedly increased path length and decreased clustering coefficient in TLE-HS compared with controls and TLE-G, whereas those with TLE-G are only moderately affected compared with controls. Reproduced from Bernhardt and others, 2019, with permission.
Connectome analyses of WM organization in other epilepsy syndromes are less frequently reported. In GGE, a recent study showed bi-hemispheric alterations in several connectivity parameters compared with controls, and demonstrated an association between network architecture and drug response in patients (McKavanagh and others 2021). These findings are complemented by a connectome-informed machine learning study in JME, which found that structural connectome and conventional dMRI measures can discriminate between patients and controls with more than 80% accuracy (Lee and others 2021). Finally, a recent study applied computational modeling to structural and functional connectome data in both GGE and TLE patients, and identified increases in subcortical drive contributing to cortical dynamics in GGE, while TLE patients presented with reduced subcortical drive and imbalanced excitation-inhibition of cortical microcircuits (Weng and others 2020). This study suggests a potentially important differentiation between focal and generalized epilepsy syndromes at macro- and microscales.
WM Associations with Cognitive and Psychiatric Comorbidities
WM Associations with Cognition in Epilepsy
WM integrity is critical for the integration of cortico-cortical networks that support cognition. However, only recently has compromise to specific WM tracts and networks been linked to domain-specific cognitive impairments in epilepsy (for reviews, see Allone and others 2017; Leyden and others 2015). The majority of work has focused on cognitive impairment in TLE, but new data addressing how WM injury disrupts cognition in FLE and JME are now emerging. A review of TLE studies between 2005 and 2014 is provided in Leyden and others (2015). We provide an update on the state of the field, focusing on studies from 2015 to 2021 for TLE, and studies of other epilepsy syndromes not previously reviewed (Tables 1 and 2).
Studies Examining White Matter Associations with Cognition.
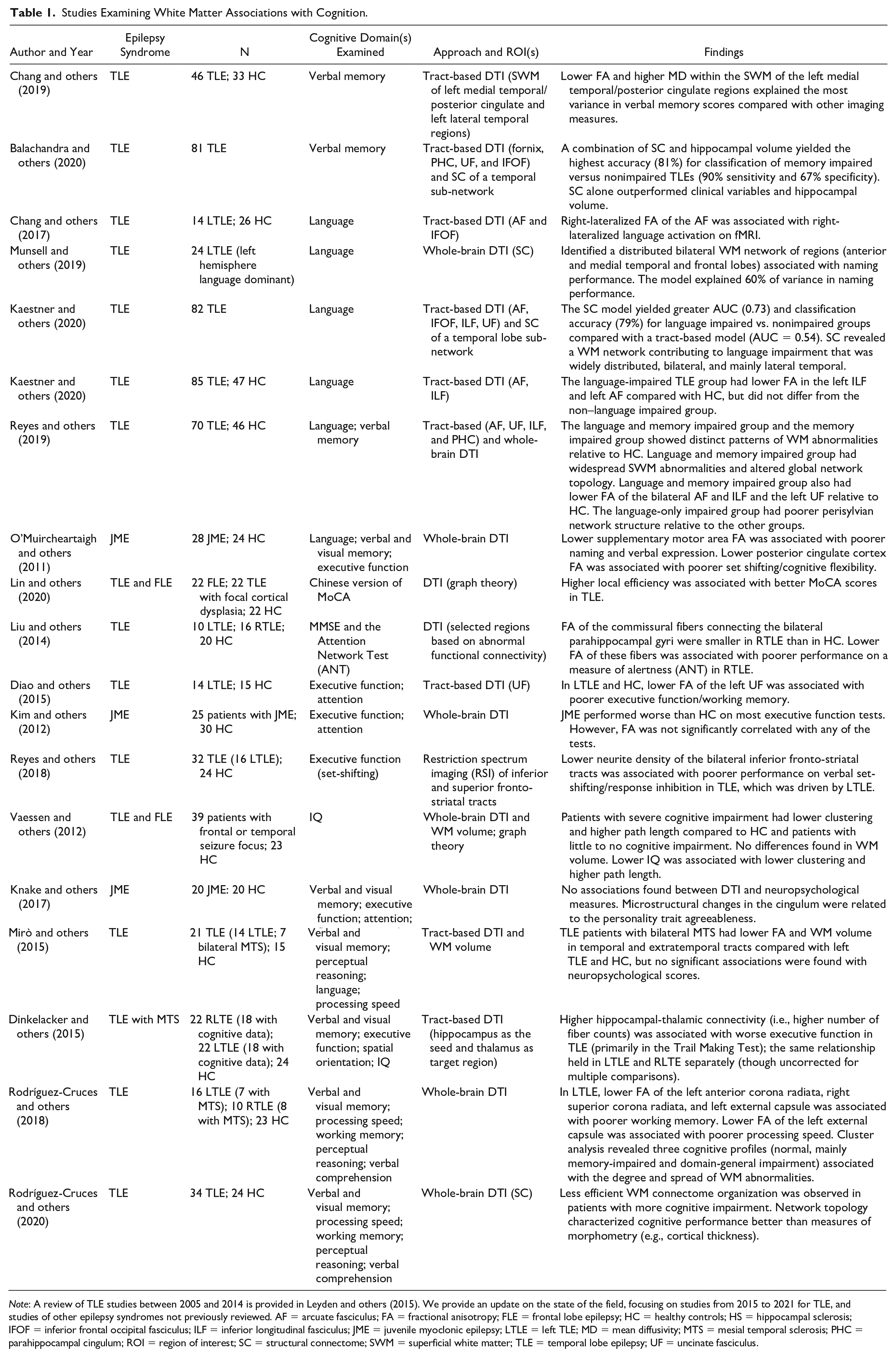
Note: A review of TLE studies between 2005 and 2014 is provided in Leyden and others (2015). We provide an update on the state of the field, focusing on studies from 2015 to 2021 for TLE, and studies of other epilepsy syndromes not previously reviewed. AF = arcuate fasciculus; FA = fractional anisotropy; FLE = frontal lobe epilepsy; HC = healthy controls; HS = hippocampal sclerosis; IFOF = inferior frontal occipital fasciculus; ILF = inferior longitudinal fasciculus; JME = juvenile myoclonic epilepsy; LTLE = left TLE; MD = mean diffusivity; MTS = mesial temporal sclerosis; PHC = parahippocampal cingulum; ROI = region of interest; SC = structural connectome; SWM = superficial white matter; TLE = temporal lobe epilepsy; UF = uncinate fasciculus.
Studies Examining White Matter Associations with Postoperative Change in Cognition.
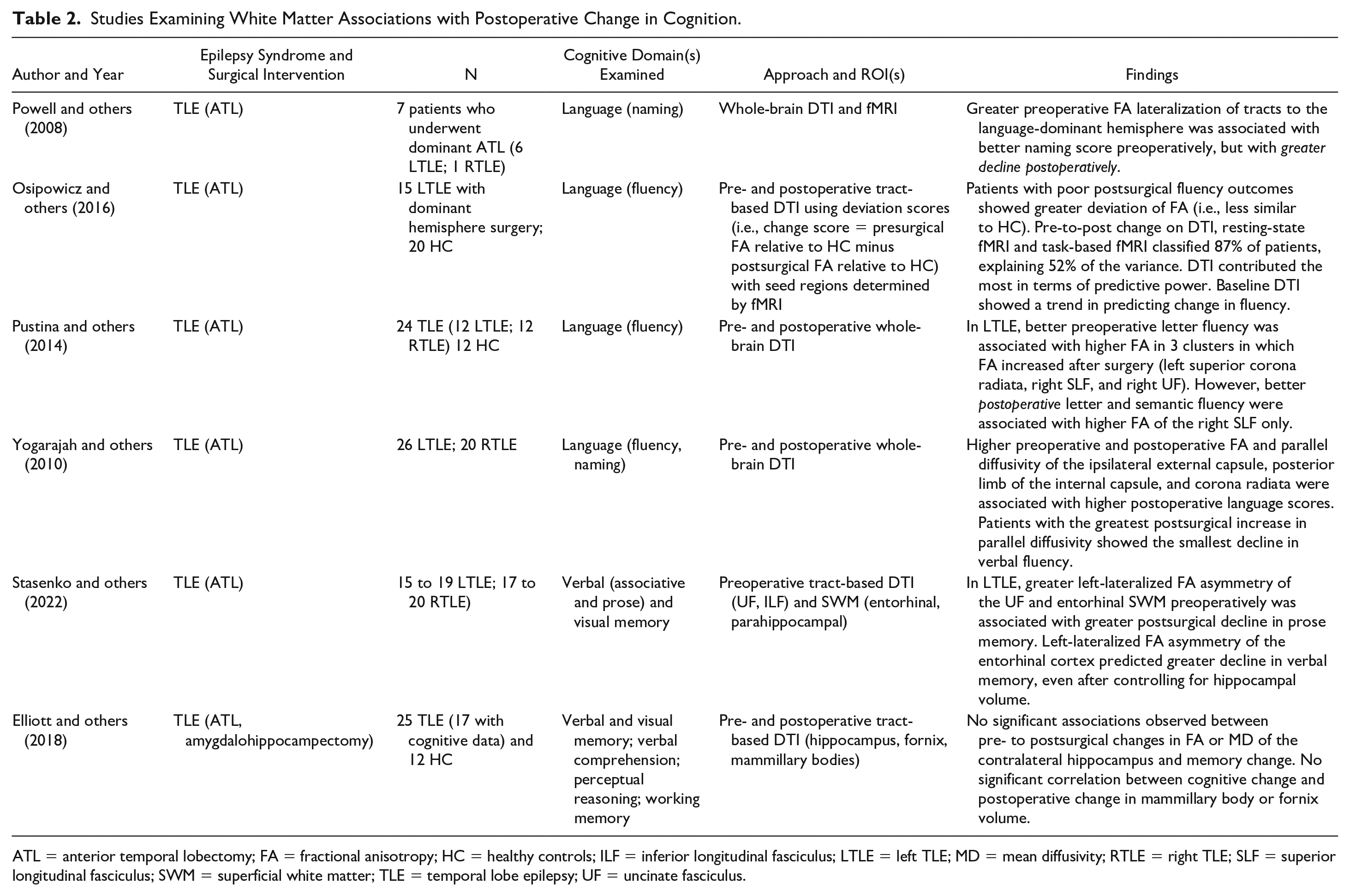
ATL = anterior temporal lobectomy; FA = fractional anisotropy; HC = healthy controls; ILF = inferior longitudinal fasciculus; LTLE = left TLE; MD = mean diffusivity; RTLE = right TLE; SLF = superior longitudinal fasciculus; SWM = superficial white matter; TLE = temporal lobe epilepsy; UF = uncinate fasciculus.
Memory
It is well established that the hippocampus and its projections are critical to learning and memory. However, an emerging literature has characterized how broader WM network disruption contributes to memory impairments in epilepsy (Table 1). Damage to temporo-limbic association tracts, including the uncinate fasciculus, inferior longitudinal fasciculus, parahippocampal cingulum, and inferior fronto-occipital fasciculus, is most commonly associated with impairments in verbal learning and memory in TLE (for review, see Leyden and others 2015). A few studies have also examined the superficial WM (SWM) or U-shaped WM fibers directly beneath the cortex that are important for maintaining short-range cortico-cortical connectivity. These studies have revealed that microstructural loss within the left entorhinal, broader medial temporal, and posterior cingulate SWM also contributes to verbal memory impairments in TLE and may explain more of the variance in memory performances than functional oscillations or cortical thinning in adjacent cortex (Chang and others 2019). In particular, the entorhinal WM contains major afferent connections from the entorhinal cortex to CA3 and the dentate gyrus of the hippocampus via the perforant path and angular bundle. These WM tracts are known to be important for episodic memory encoding (e.g., pattern separation), and damage to which likely disrupts a critical memory circuit in TLE.
Leveraging network models of WM connectivity, Balachandra, Kaestner and others (2020) found that a structural connectome of a temporal subnetwork (i.e., temporal to extratemporal connections) was able to classify TLE patients as verbal memory-impaired versus non-memory-impaired with 81% accuracy. The connect-ome’s strong performance may reflect its ability to identify temporo-limbic and association tracts commonly implicated in memory, in conjunction with short-range connections connecting adjacent temporal lobe cortex (Fig. 4).
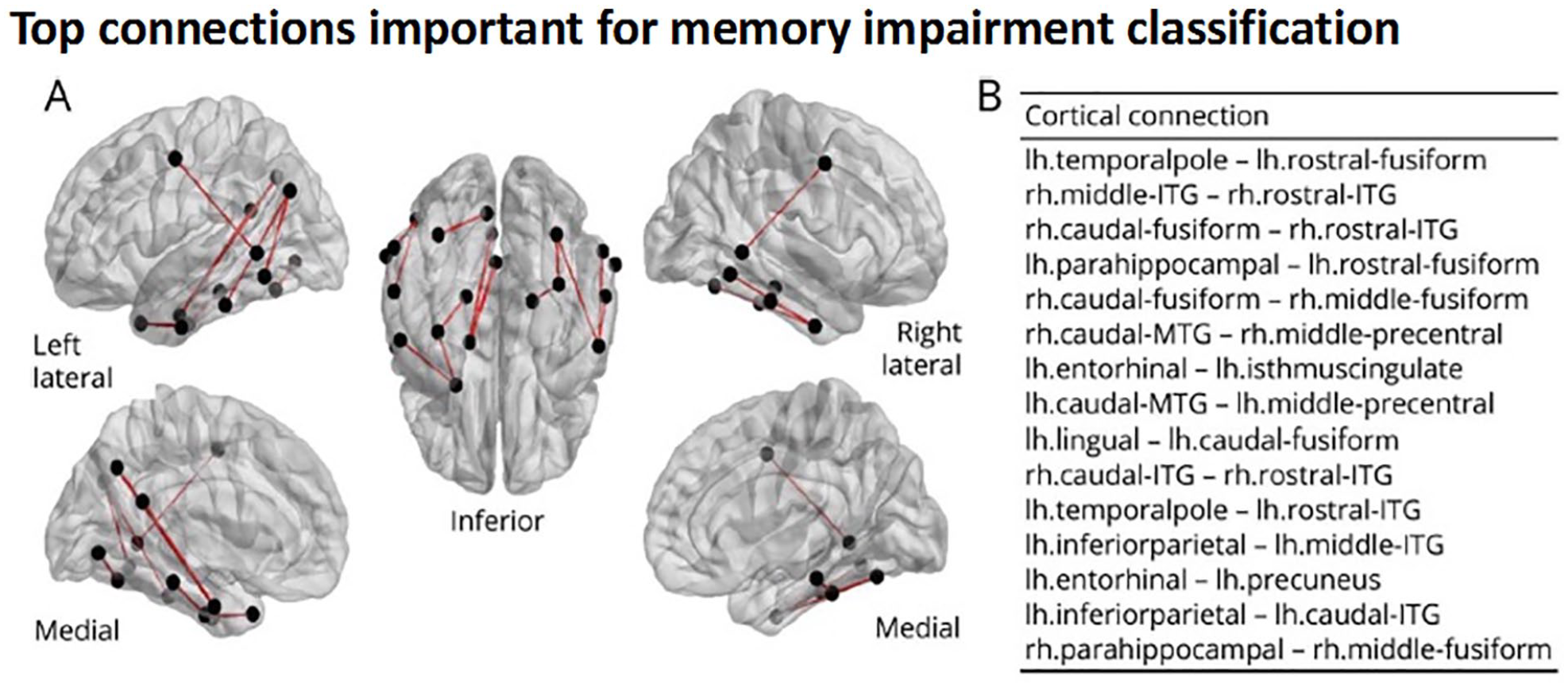
Structural connectome predicts verbal memory in temporal lobe epilepsy. Comprehensive white matter neuronal network mapping (i.e., the structural connectome) was able to predict verbal memory impairment in TLE and highlighted the importance of short-range temporal-temporal connections to memory. Panel A shows a glass brain visualization of the top 15 connections important for classification of patients as memory-impaired versus unimpaired. Panel B shows names of top 15 most important connections ordered by most important (top) to least important (bottom). ITG = inferior temporal gyrus; lh = left hemisphere; MTG = middle temporal gyrus; rh = right hemisphere. Reproduced from Balachandra, Kaestner and others, 2020, with permission.
Associations between WM and visual memory are scarce, with only two studies reporting that damage to the right uncinate fasciculus (Diehl and others 2008) and right parahippocampal gyrus WM (Yogarajah and others 2008) is associated with visual memory impairment in TLE.
Pre-to-postoperative associations with memory
Anterior temporal lobectomy (ATL) is the most common surgical procedure performed for treatment of drug-resistant TLE. However, ATL involves the removal of the anterior hippocampus, amygdala, lateral temporal cortex, and sub-adjacent WM, leading to a high risk for postsurgical memory decline in many patients (Sherman and others 2011). Only two studies have examined WM associations with postoperative memory decline (Table 2). One study highlighted the importance of a frontotemporal tract transected during surgery (i.e., uncinate fasciculus) and the integrity of WM beneath the entorhinal cortex to memory decline following ATL. A second study did not find associations between WM integrity in the ipsilateral temporal lobe (i.e., fornix) and memory decline (Elliott and others 2018). However, the surgical sample in the second study was small and the surgeries were heterogeneous, limiting interpretability of the results. Thus, while some data support the importance of the medial temporal lobe WM to postoperative memory outcomes, there are not enough data to draw reliable conclusions.
Language
Language impairments in TLE have been frequently associated with disruption to both perisylvian (e.g., arcuate fasciculus) and extra-sylvian (e.g., inferior longitudinal fasciculus) WM fibers. Left hemisphere fibers along the dorsal stream are important for mapping auditory sounds to articulatory (motor) representations (e.g., arcuate fasciculus), whereas fibers in the ventral stream are typically implicated in mapping auditory speech sounds to meaning—that is, lexical semantic processing (e.g., inferior longitudinal fasciculus and inferior fronto-occipital fasciculus). Although these left hemisphere frontotemporal tracts are implicated in language performance both in healthy individuals and TLE, right hemisphere fibers also correlate with language performance in TLE, including the right arcuate fasciculus, inferior fronto-occipital fasciculus, superior longitudinal fasciculus, and uncinate fasciculus (McDonald and others 2008; Pustina and others 2014). This suggests (1) right hemisphere contributions to language and/or (2) potential reorganization of language to the right hemisphere in some patients with a left-sided seizure focus. In support of the importance of right hemisphere networks to language, Kaestner and others (2020) demonstrated that using a structural connectome and machine learning (XGBoost), a broad, bilateral pattern of WM abnormalities contributed to naming and fluency impairments in TLE. Although lateral temporal connections between superior temporal gyrus and pars opercularis were the most important features (i.e., fibers from the arcuate fasciculus), other widely distributed and interhemispheric connections also emerged. Similarly, Munsell and others (2019) identified a distributed, bilateral WM network of regions that predicted naming performance in left TLE patients who were all left-hemisphere dominant for language, suggesting that right hemisphere WM contributions to language were not solely secondary to language reorganization.
Neuroplasticity of language networks
A remarkable characteristic of the human brain is its ability to reorganize in response to injury. With language, this is most frequently observed as an interhemispheric shift, making asymmetry of WM tracts a popular method for probing language reorganization in epilepsy (Ellmore and others 2010). For most healthy individuals, the left hemisphere is dominant for language. However, patients with left TLE in particular show reduced left-lateralization of language networks (i.e., a more symmetrical or right-lateralized representation) on fMRI and in WM integrity measured with dMRI. However, reduced asymmetry in perisylvian WM integrity sometimes but not always corresponds to reduced asymmetry in language activations on fMRI (e.g., Chang and others 2017; Powell and others 2007; but see Rodrigo and others 2008). The mixed findings highlight the complexity of language reorganization in left TLE, and our need to better understand reorganization of WM language networks and how it relates to functional reorganization and language performance.
Pre-to-postoperative associations with language
Only a few studies examined the association between WM integrity and postoperative language outcomes. Powell and others (2008) found that greater preoperative asymmetry of frontotemporal WM to the language-dominant hemisphere was associated with greater naming decline postsurgery, suggesting that direct surgical disruption to (presumably healthy) temporal lobe WM leads to decline in naming. Another study demonstrated that patients with pre-to-postsurgical decline in fluency had FA microstructure that looked less like that of controls (i.e., more abnormal), with abnormal WM profiles explaining more variance in language outcomes than language activation on fMRI (Osipowicz and others 2016).
Other research has demonstrated associations between postoperative verbal fluency and higher FA of the right superior longitudinal fasciculus (Pustina and others 2014)—a finding that may reflect a compensatory interhemispheric shift in language networks to the contralateral hemisphere. However, there is also evidence that greater pre-to-postsurgical increases in parallel diffusivity in the ipsilateral ventromedial temporal lobe are associated with better postoperative language scores in left TLE (Yogarajah and others 2010). Taken together, the extant literature suggests that better language outcomes following ATL depend on both inter- and intra-hemispheric shifts in WM integrity in key dorsal and ventral language tracts. Although surgery incurs a risk of language decline, there appears to be potential for microstructural and functional reorganization in both ipsilateral and contralateral hemispheres that may help to mitigate language decline.
Executive Function
Executive dysfunction is observed in a third to half of patients with TLE and has a higher prevalence in JME and FLE. However, unlike for language and memory, there is less consistent evidence linking specific WM tracts/regions to executive dysfunction in epilepsy. In adults, working memory impairments have been associated with damage to the superior longitudinal fasciculus, cingulum, and temporal lobe WM (Winston and others 2013) as well as the uncinate fasciculus (Diao and others 2015). In addition, poorer performance on set-shifting and response inhibition—two components of executive function—has been associated with lower neurite density of the bilateral inferior frontostriatal tracts (Reyes and others 2018). However, in another TLE study, poorer set-shifting performance was associated with heightened hippocampal-thalamic connectivity, interpreted to reflect a pathological increase of WM connectivity leading to less efficient executive function (Dinkelacker and others 2015). These mixed results are unsurprising given that executive function is not a unitary construct, with different studies measuring different aspects of executive function. Interestingly, no study has examined the relationship between pre-to-postsurgical changes in executive function and WM connectivity. This would be a fruitful area for exploration as there is some evidence for postsurgical improvement of executive function (Sherman and others 2011) and, separately, normalization of frontotemporal FA (e.g., Pustina and others 2014).
Cognitive Phenotypes
The majority of studies have focused on cognitive domains in isolation as well as specific tracts, guided by a priori hypotheses regarding structure-behavior relationships. However, recent studies have moved toward an examination of cognitive phenotypes, or patterns of cognitive impairment, and examined how these phenotypes map onto whole-brain microstructural pathology (Reyes and others 2019; Rodríguez-Cruces and others 2018; Rodríguez-Cruces and others 2020;– Fig. 5). These studies have identified three to four distinct cognitive phenotypes in TLE that have unique patterns of deep and superficial WM network abnormalities, some of which correspond to previously reported a priori tracts. Most interesting is the observation that patients with a cognitively intact profile do not differ from healthy controls in WM network pathology, lending further validation to the biological relevance of these phenotypes and the importance of WM integrity to cognition.
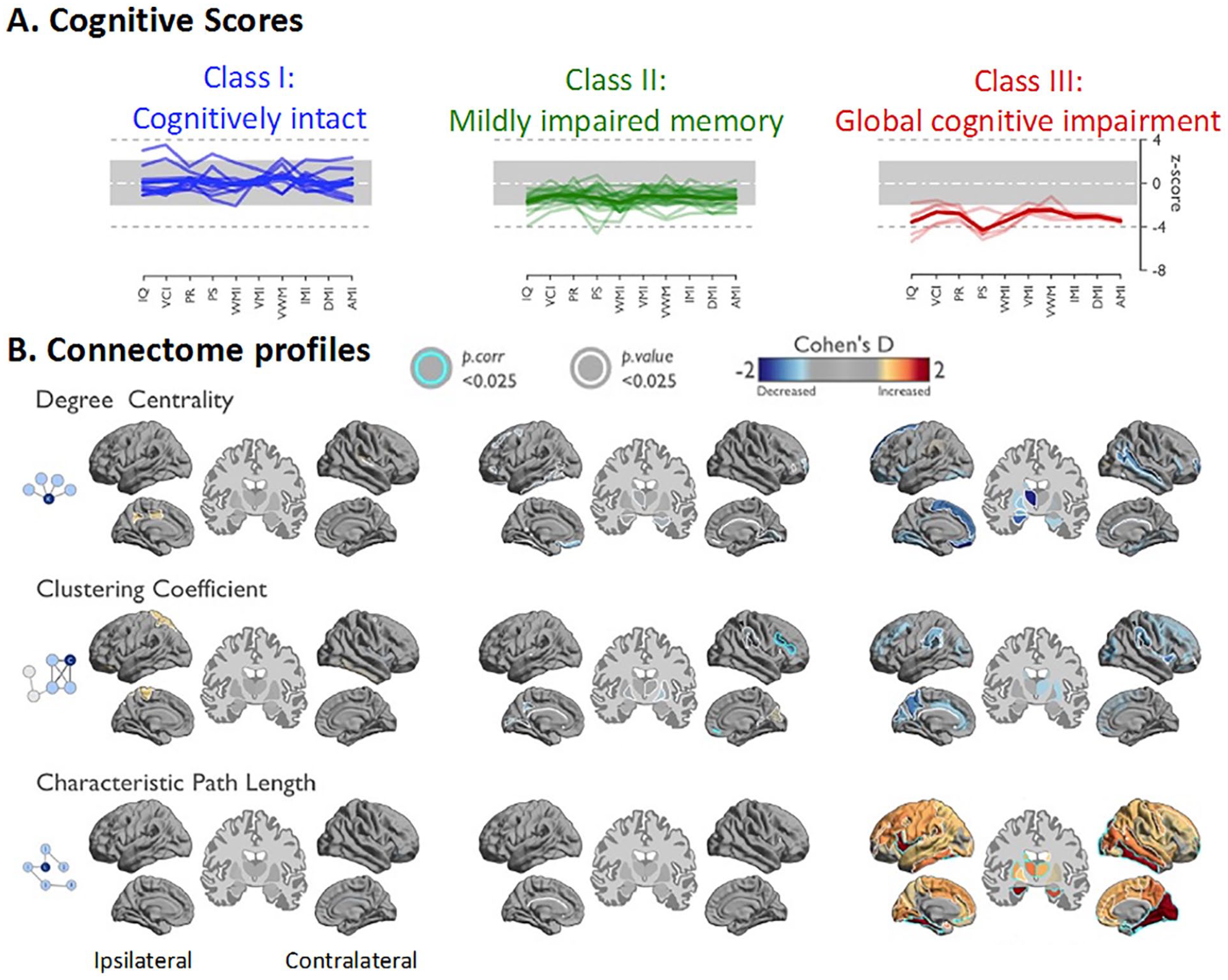
Multidomain cognitive phenotyping and whole-brain white matter connectome. Patients with less efficient WM network organization showed more pronounced cognitive difficulties. WM connectome metrics were more closely associated with cognitive function than cortical thickness. (A) Hierarchical clustering of cognitive profiles converged on three cognitive classes in the temporal lobe epilepsy cohort: Patients in Class 1 had cognitive scores within normal range, those in Class 2 showed mild impairment in memory-specific domains, and Class 3 displayed pronounced impairment across all domains, with prominent reduction of processing speed. (B) Gradual network organization abnormalities were observed across classes with most marked changes in Class 3, intermediate differences in Class 2, and only subtle changes in Class 1. Class 2 showed decreased clustering in the contralateral suborbital sulcus and inferior frontal sulcus. At a connectome-wide level, Class 3 showed the most marked increases of characteristic path length, while Classes 1 and 2 were rather normal. In Class 3, path length increases were most marked in the lateral and medial temporal lobes in both hemispheres, the ipsilateral frontal, and the contralateral occipital lobe. Modified from Rodríguez-Cruces and others, 2020, with permission.
Associations with Psychiatric Comorbidities
Depression and Anxiety
Depression affects approximately one out of four patients with epilepsy. Although once thought to reflect a reaction to psychosocial stressors associated with epilepsy, research now supports a bidirectional relationship between TLE and depression (Kanner and others 2012), with WM abnormalities as one potential contributor. In a recent systematic review of the neuroimaging correlates of depression in epilepsy (Elkommos and Mula 2021), three studies examined WM microstructure. Two studies reported that WM abnormalities in fronto-temporo-limbic regions were associated with increased depressive symptoms in TLE (Kavanaugh and others 2017; Kemmotsu and others 2014). However, a third study did not find a significant difference between TLE with depression and anxiety compared with TLE alone in a post hoc analysis (Stretton and others 2015; Table 3). In sum, there is some evidence that fronto-limbic network dysfunction may underlie a bidirectional link between TLE and depression, and this may influence which patients present with depression. However, prospective, longitudinal studies are needed that directly compare TLE with depression to TLE without depression and track whether the evolution of WM changes corresponds to the evolution or severity of depressive symptoms. Future investigations of these associations in other epilepsy syndromes is also important.
Studies Examining White Matter Associations with Psychiatric Outcomes.
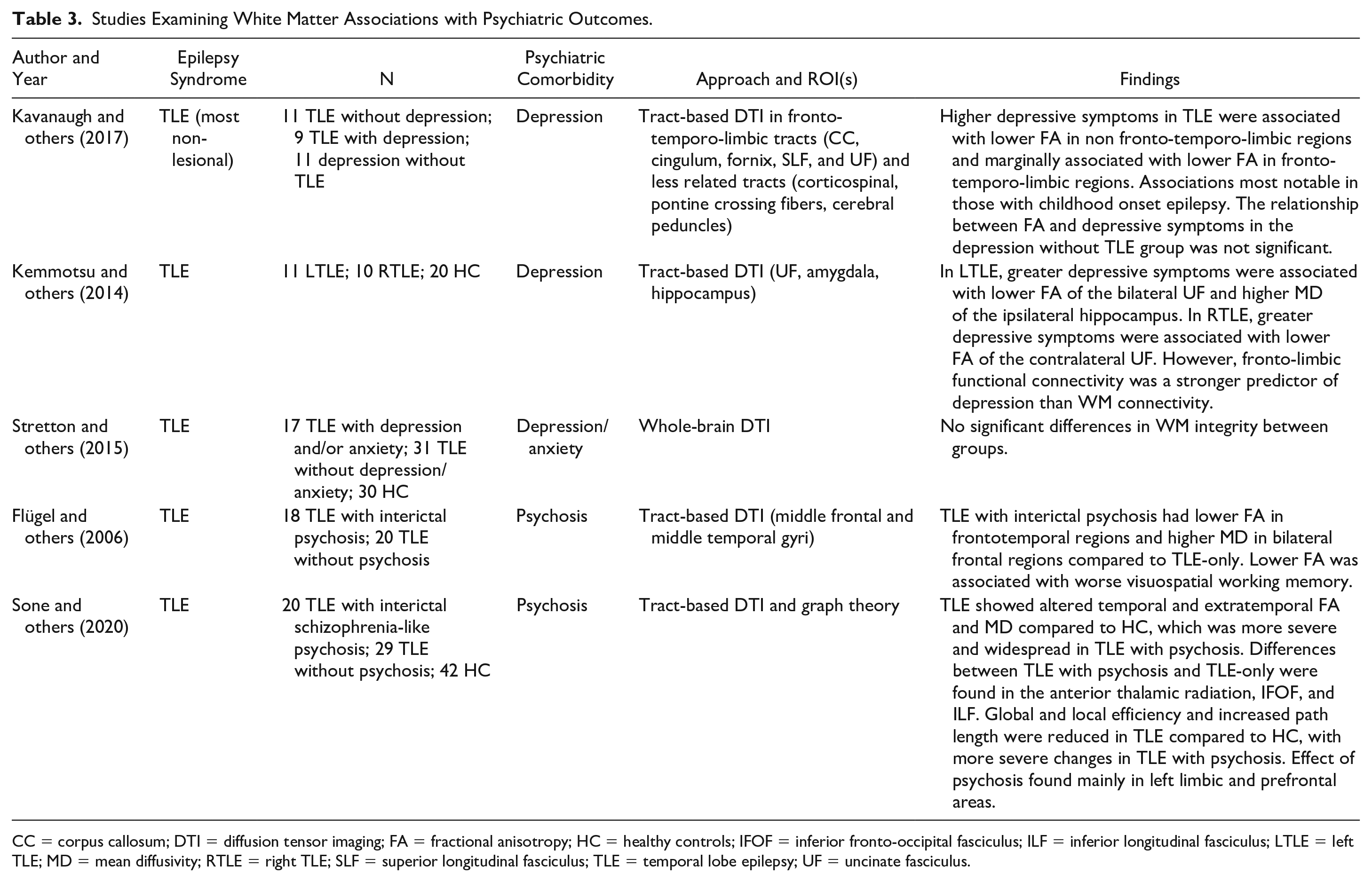
CC = corpus callosum; DTI = diffusion tensor imaging; FA = fractional anisotropy; HC = healthy controls; IFOF = inferior fronto-occipital fasciculus; ILF = inferior longitudinal fasciculus; LTLE = left TLE; MD = mean diffusivity; RTLE = right TLE; SLF = superior longitudinal fasciculus; TLE = temporal lobe epilepsy; UF = uncinate fasciculus.
Interictal Psychosis
There is a prevailing view that a strong link exists between TLE and psychosis, and that damage to gray and WM may give rise to psychosis in epilepsy. A systematic review reported an almost eightfold increased risk of psychosis in epilepsy relative to the general population, with an even higher risk in TLE (Clancy and others 2014). Psychosis in epilepsy is classified as ictal or postictal if it is closely linked to seizure occurrence. Conversely, interictal psychosis is not temporally related to seizures and may not necessarily resolve in-between seizure episodes. A recent study found differences between TLE with versus without interictal psychosis in several temporo-limbic tracts (inferior fronto-occipital fasciculus, inferior longitudinal fasciculus) and the anterior thalamic radiations (Sone and others 2020). In the same study, a graph theory analysis found that TLE with psychosis had a greater reduction in global and local efficiency compared with controls, with the effect of psychosis primarily in left limbic and prefrontal areas. A previous tract-based study reported lower FA in bilateral frontotemporal regions and higher MD in bilateral temporal regions in TLE with psychosis compared with TLE alone (Flügel and others 2006). Thus, psychosis in epilepsy may be associated with a distributed pattern of temporo-limbic pathology, not restricted to the mesial temporal lobe.
WM Associations with Seizure Laterality, Drug Resistance, and Postsurgical Outcomes
Seizure Onset Laterality
Identifying the side of seizure onset is a crucial step in the presurgical evaluation of a patient with epilepsy. This presents a challenge for many patients with TLE whose seizures may not clearly lateralize on scalp-EEG, or for whom subtle epileptogenic lesions are not visible on conventional MRI. For this reason, dMRI has been proposed as a clinical decision support tool that could be used to map the underlying seizure networks and increase confidence in seizure laterality.
A number of studies have examined the utility of using dMRI to identify the side of seizure onset (e.g., Ahmadi and others 2009; An and others 2014; Concha and others 2012; Nazem-Zadeh and others 2014; Nazem-Zadeh and others 2016; Table 4). These studies have obtained accuracies from 71% to 91% for discriminating patients with right from left TLE using frontotemporal WM tracts alone and reflect the tendency for patients with unilateral TLE to have greater WM tract damage on the side ipsilateral to the seizure focus and proximal to the seizure onset zone.
Studies Examining White Matter Associations with Seizure Lateralization, Drug Resistance, Postsurgical Seizure Outcomes, and Postsurgical Visual Field Deficits a .
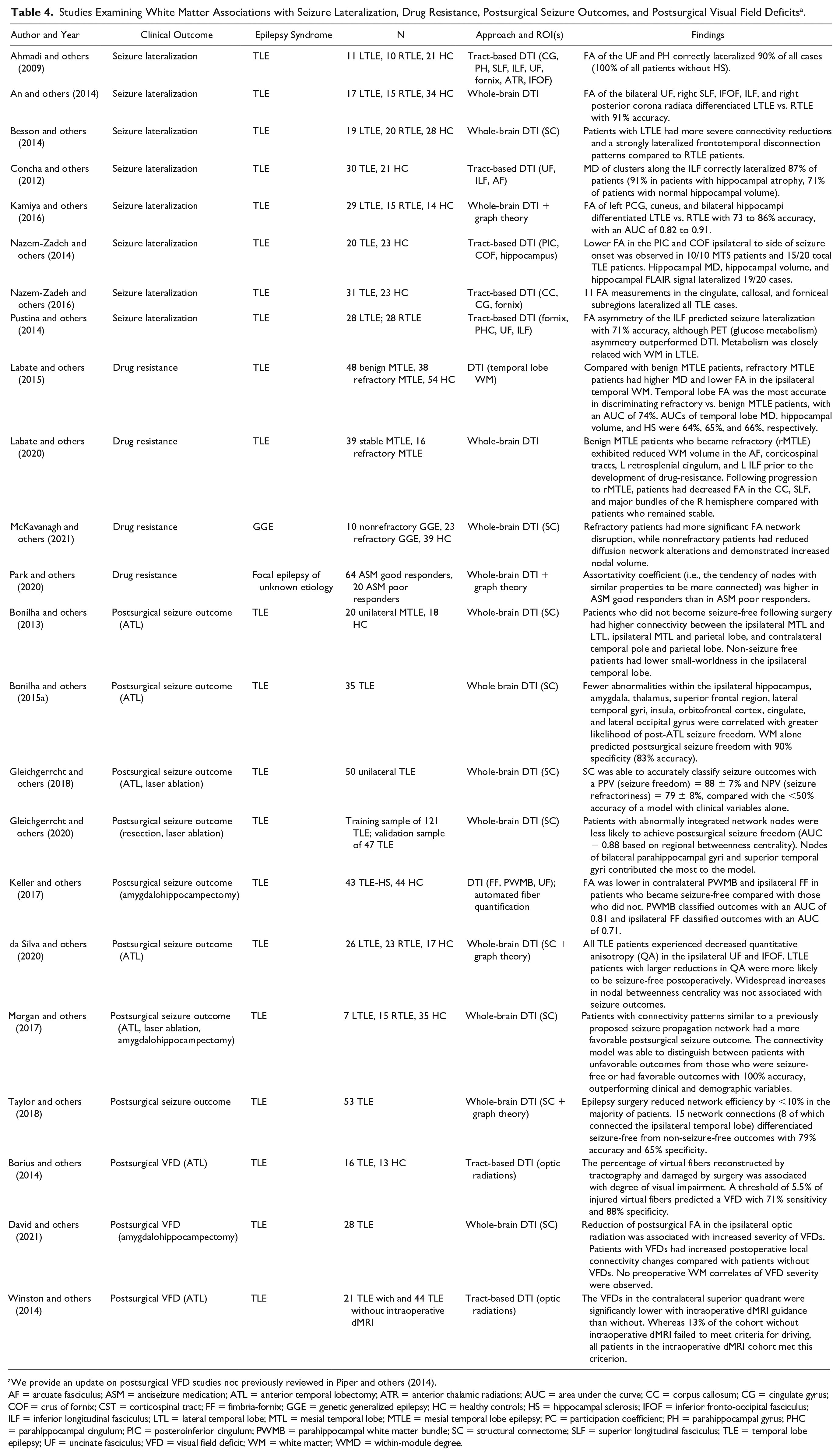
We provide an update on postsurgical VFD studies not previously reviewed in Piper and others (2014).
AF = arcuate fasciculus; ASM = antiseizure medication; ATL = anterior temporal lobectomy; ATR = anterior thalamic radiations; AUC = area under the curve; CC = corpus callosum; CG = cingulate gyrus; COF = crus of fornix; CST = corticospinal tract; FF = fimbria-fornix; GGE = genetic generalized epilepsy; HC = healthy controls; HS = hippocampal sclerosis; IFOF = inferior fronto-occipital fasciculus; ILF = inferior longitudinal fasciculus; LTL = lateral temporal lobe; MTL = mesial temporal lobe; MTLE = mesial temporal lobe epilepsy; PC = participation coefficient; PH = parahippocampal gyrus; PHC = parahippocampal cingulum; PIC = posteroinferior cingulum; PWMB = parahippocampal white matter bundle; SC = structural connectome; SLF = superior longitudinal fasciculus; TLE = temporal lobe epilepsy; UF = uncinate fasciculus; VFD = visual field deficit; WM = white matter; WMD = within-module degree.
Beyond tract-based studies, Besson and others (2014) used a structural connectome approach to demonstrate differences between left and right TLE, with more severe alterations in left TLE patients, who showed a strongly lateralized frontotemporal disconnection pattern. Using graph theory, Kamiya and others (2016) found decreased local efficiency in the left posterior cingulate gyrus, left cuneus, and bilateral hippocampus in left TLE. In contrast, only the right hippocampus showed altered network properties in right TLE. In this study, a support vector machine correctly classified between 73% and 86% of patients as having left versus right seizure onset. Taken together, preliminary evidence suggests that both tract-based and structural connectome measures of network pathology aid in lateralization of the seizure focus in TLE, with moderate to high classification accuracy across studies. With further refinements in machine learning algorithms and larger samples, dMRI may serve as clinically useful for augmenting pre-surgical seizure lateralization.
Drug Resistance
Only 60% of patients with epilepsy respond to the first two ASMs and less than 4% respond to further ASM trials. The remaining 30% to 40% are defined as “drug resistant” and present a considerable treatment challenge. Labate and others (2015) found that patients with drug-resistant mesial TLE had more severely reduced temporal lobe FA compared with patients with benign mesial TLE, irrespective of the presence of HS. In fact, temporal lobe FA was able to differentiate between refractory versus benign TLE with an AUC of 0.74. In a follow-up study, patients whose mild mesial TLE eventually evolved into refractory mesial TLE had distinct microstructural alterations in the corticospinal tracts, superior longitudinal fasciculus, left cingulum, and left inferior longitudinal fasciculus prior to the development of drug resistance (Labate and others 2020). These data suggest that greater WM pathology both within and beyond the temporal lobe may predispose patients to develop drug-resistant seizures. Identifying these patterns at the onset of epilepsy could help guide treatment decisions early, including identifying patients who are not likely to gain seizure control from ASMs and who should be considered for surgery or other nonpharmacologic treatments.
Postsurgical Seizure Outcomes
TLE and other focal epilepsies represent a spectrum of disorders with a wide range of postsurgical seizure outcomes (i.e., seizure-free vs. not), even in patients with similar preoperative clinical features (Coan and Cendes 2013). Keller and others (2017) found that patients with TLE who had greater preoperative pathology in the ipsilateral dorsal fornix and contralateral parahippocampal WM were more likely to have poor seizure outcomes relative to those with less pathology. Furthermore, pathological changes in the ipsilateral fornix and uncinate were beyond the margins of the resection in patients with poor seizure outcomes, suggesting that insufficient disconnection of the temporal lobe epileptogenic network may lead to persisting seizures. Gleichgerrcht and others (2020) quantified whether brain regions were situated on efficient communication pathways in the whole-brain network (i.e., regions with high betweenness centrality) to map patient-specific reorganization in structural hubs. Combining these measures with supervised machine learning, that study found that nodes most strongly associated with seizure freedom included the bilateral parahippocampal and superior temporal gyri (Fig. 6 Panel A). Bonilha and others (2015a) used a structural connectome model to predict post-ATL seizure outcomes in TLE with 77% sensitivity and 89% specificity (Fig. 6 Panel B). In a similar study, network connections that contributed the highest accuracy were located not only in the ipsilateral temporal and extratemporal regions but also in the contralateral hemisphere (Bonilha and others 2013). These data suggest that broad WM network abnormalities both ipsilateral and contralateral to the seizure focus may increase risk for poor seizure outcomes, implying incomplete resection of the epileptogenic network as detected by dMRI.
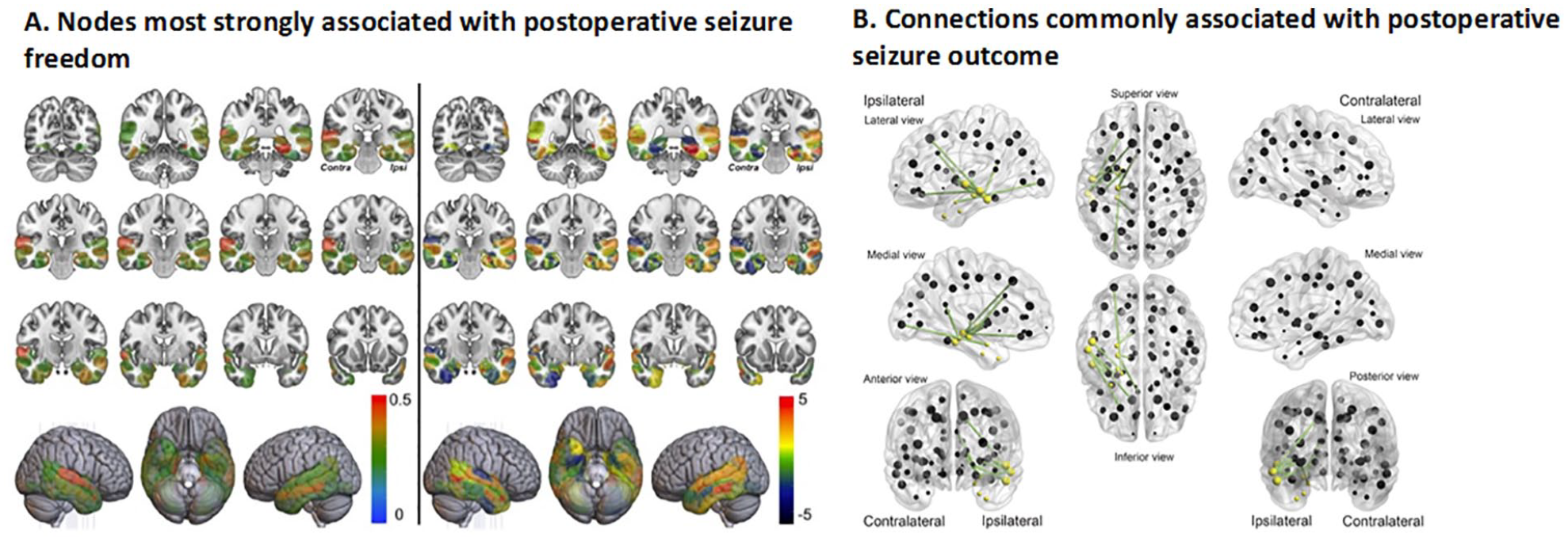
Structural connectome and network topography as biomarkers for estimating postsurgical outcomes in patients with TLE. (A) Network integration in the medial and lateral temporal regions was related to postsurgical seizure outcomes, such that patients with abnormally integrated network nodes were less likely to achieve seizure freedom. The left panel of A illustrates feature importance for classification for a model of betweenness centrality (BC)—the degree to which other regions rely on a particular node for efficient (i.e., shortest amount of steps needed) flow of information. A higher BC indicates a more highly integrated region within the network. The ipsilateral parahippocampus, contralateral superior temporal gyrus, and bilateral entorhinal regions showed the highest importance. The right panel demonstrates group differences in BC between seizure-free and non-seizure-free patients. Positive t-values indicate higher values in the non-seizure-free group. Areas with stronger red color correspond to the most important (left panel) and most significantly different between the groups (right panel). Ipsi = ipsilateral (represents the side ipsilateral to the seizure onset). Reproduced from Gleichgerrcht and others, 2020, with permission. (B) In green are structural connectome links that were repeatedly chosen by the cross-validation model to have the highest ability to predict postsurgical seizure freedom. Yellow spheres represent the eight cortical regions of interest defined as pertaining to the temporal region. Patients who exhibited greater weights among these links were less likely to become seizure-free after surgery. Reproduced from Bonilha and others, 2015a, with permission.
Postsurgical Visual Field Deficits
In the temporal lobe, the optic radiations project from the lateral geniculate of the thalamus, anteriorly and laterally over the temporal horn of the lateral ventricles before coursing posteriorly toward the occipital pole. During ATL, the anterior portion of the ventral visual pathway (i.e., Meyer’s loop) is removed, producing a visual field defect (VFD; typically an incomplete [medial sector] quadrantanopia) in a majority of patients. VFDs can preclude patients from driving in some countries and states, significantly affecting their quality of life and independence (Gilliam and others 1997). Converging evidence has demonstrated that the extent of degeneration along or transection of temporo-occipital fiber tracts predicts the severity of postoperative visual field defects following ATL (Chen and others 2009; Powell and others 2005; Taoka and others 2005; Wieshmann and others 1999; for review see Piper and others 2014) (Table 4 and Fig. 7). In fact, Winston and others (2014) found that no patient failed to meet visual criteria for driving as a result of ATL resection when visualizing the optic radiations with tractography in comparison to 13% of controls who did not undergo tractography. As a result of these promising findings, preoperative dMRI has been proposed as a viable method for minimizing risk for VFDs. However, the optic radiations disperse broadly in the temporal lobe and can prove difficult to track, with differences in data acquisition and tractography algorithms leading to limited reproducibility of these fibers. Therefore, advanced dMRI models and tractography approaches are needed to augment the ability of dMRI tractography to minimize VFDs associated with ATL.
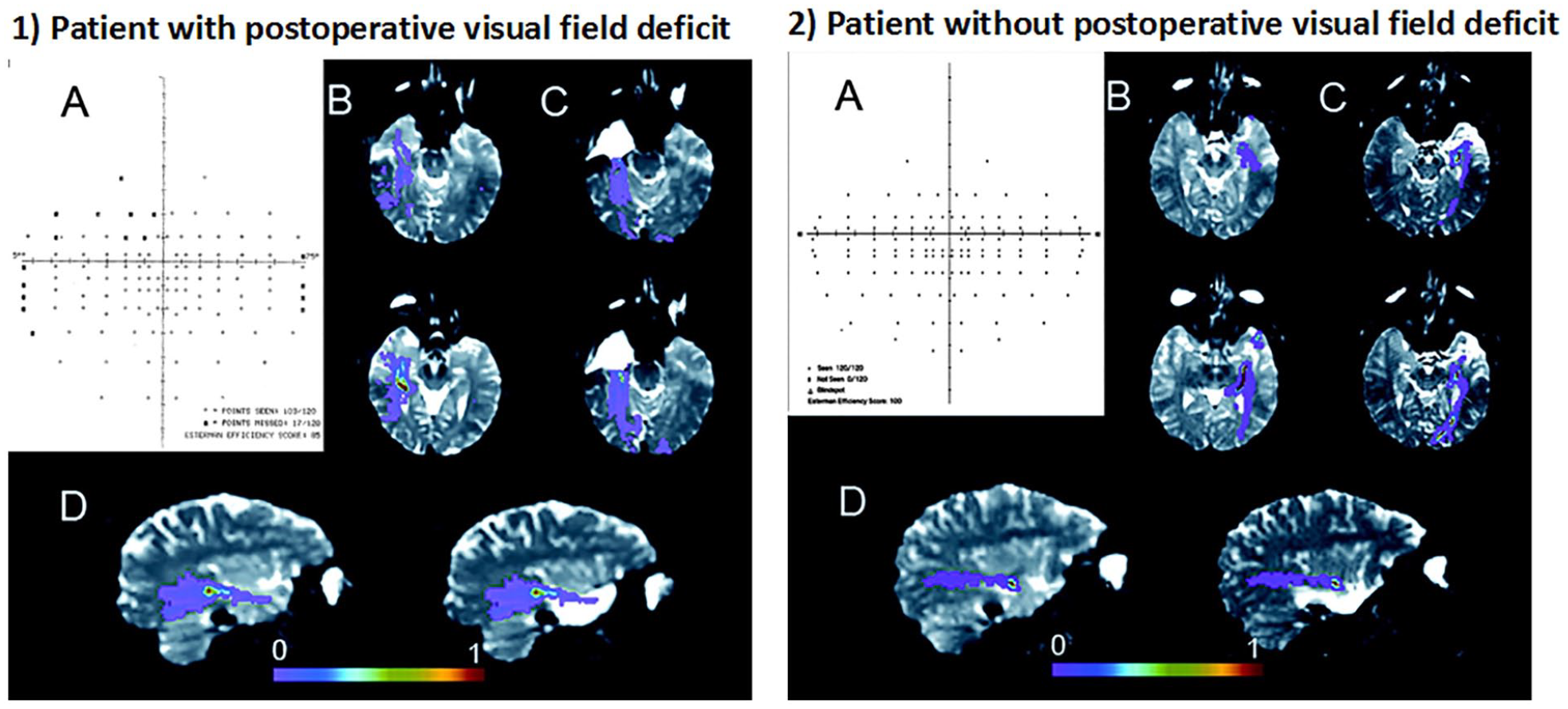
Tractography of optic radiations decreases risk for postsurgical visual field deficits. (1) Patient 1 demonstrated a postoperative visual field deficit (VFD) after anterior temporal lobectomy, manifesting as a superior quadrantanopia (A). This patient experienced a surgical disruption of the anterior segment of the Meyer’s loop (C). The preoperative right optic radiation tracts overlap with the resected anterior temporal lobe (D). (2) In contrast, Patient 2 did not experience a postoperative VFD (A). The anterior border of the Meyer’s loop remained intact (C), and the tracts do not overlap with the resected anterior temporal lobe (D). Thus, preoperative DTI tractography allows for identification of those patients at greatest risk of VFDs. The color bar represents a measure of connection probability to the starting point. Reproduced from Powell and others, 2005, with permission.
Advanced Diffusion Techniques
Although WM microstructural abnormalities are commonly observed in epilepsy using conventional dMRI and have been validated against histopathological measures of WM pathology (Concha and others 2010), the full extent of neuropathological alterations in epilepsy requires more sensitive measures of microstructural properties. In particular, it is increasingly appreciated that FA and MD are nonspecific measures of cerebral microstructure that are influenced by a number of tissue-related factors. In addition to axonal loss and demyelination, decreases in FA obtained from the basic tensor model may reflect the presence of crossing fibers or increases in extracellular diffusion due to edema or inflammation. Given the role that inflammation may play in the pathogenesis of some forms of epilepsy, a better understanding of the neurobiology behind decreased FA could help guide treatments in patients with different epilepsy syndromes.
Advancements in dMRI data acquisition (i.e., scanning parameters) and postprocessing techniques have enabled more sensitive and/or specific measures of cerebral pathology in epilepsy. Studies in diffusion kurtosis imaging (DKI), a statistical method that uses multiple diffusion weightings (i.e., b-values) to probe non-Gaussian diffusion and estimate diffusion heterogeneity in tissue, have found that kurtosis measures reveal a broader and more robust pattern of microstructural abnormalities in TLE compared to conventional DTI (Bonilha and others 2015b; Lee and others 2014). This may reflect a greater sensitivity of DKI to multiple pathologic factors including cell loss, inflammation, and axonal and dendritic reorganization.
In addition, diffusion spectrum imaging (DSI), a high-angular diffusion imaging (HARDI) technique, has been combined with the neurite orientation dispersion and density imaging (NODDI) model, a multicompartment diffusion model, to estimate structural connectivity and network properties in TLE (Lemkaddem and others 2014). Restriction spectrum imaging (RSI) is another multicompartment (multi b-value) model well-positioned to evaluate whether decreases in FA are better explained by decreased axonal/neurite density, crossing fibers, and/or increases in extracellular diffusion (e.g., cerebrospinal fluid–filled spaces; inflammation), all within a clinically feasible (4–6 minutes) time frame. This method has demonstrated that measures of WM pathology obtained with RSI are greater in magnitude, more lateralized to the epileptogenic hemisphere, and broader than those obtained with conventional DTI (Loi and others 2016;– Fig. 8).
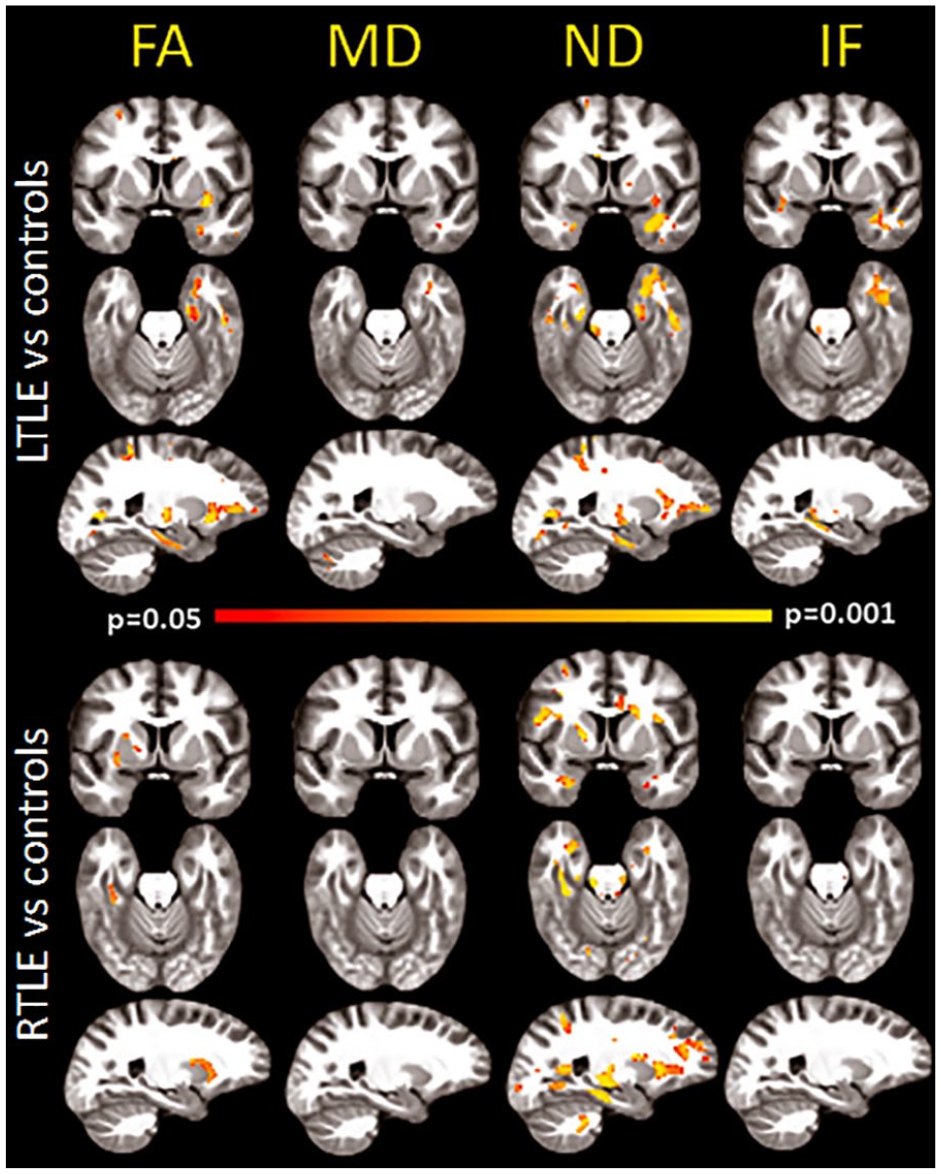
Restriction spectrum imaging (RSI) provides a more robust measure of white matter injury in TLE relative to DTI. Voxel-based analysis of group comparisons between patients with right TLE (RTLE) and left TLE (LTLE) and age-matched controls. Areas of red-yellow represent decreased fractional anisotropy (FA) and neurite density (ND), and increased mean diffusivity (MD) and isotropic free (IF) water diffusion in patients compared with controls. Compared with FA, ND maps revealed a broader and more robust pattern of decreases of white matter integrity in TLE, with strong lateralization to the left hemisphere in LTLE, and to the right hemisphere in RTLE. Decreases were noted primarily in the anterior temporal lobe, with additional decreases in the inferior prefrontal white matter. Thus, neurite density using RSI may provide a more specific measure of WM pathology than standard DTI, distinguishing regions primarily affected by axonal/myelin loss from those where crossing fibers and increases in extracellular water also play a role. Adapted from Loi and others, 2016, with permission.
Recent advancements in scanner hardware, such as stronger gradients and multiband acceleration methods, greatly reduce the practical difficulties of scanning with very high b-values (i.e., b = 4000 or 5000) and a large number of diffusion directions. This allows for improved measurements, further improving the quality of tractography, as well as further separating intra-axonal from extracellular signals in epilepsy (Bryant and others 2021). These studies suggest that advanced diffusion techniques may provide more sensitive measures of network pathology in TLE, greatly increase the specificity of connectome imaging, and further the identification of epilepsy-specific network abnormalities.
Summary and Future Directions
Over the past several decades, dMRI has greatly advanced our understanding of WM network disruption within and across epilepsy syndromes and revealed the critical role of WM disconnection in cognitive, psychiatric, and clinical outcomes in epilepsy. In particular, there is clear evidence demonstrating a link between memory and language impairments in epilepsy and disruption to bilateral medial temporal and frontotemporal WM, respectively. Associations with executive dysfunction are less clear, but may be secondary to injury within frontotemporal (i.e., uncinate fasciculus) and fronto-striatal pathways. Similarly, psychiatric comorbidities are more likely to emerge in patients with distributed temporo-limbic WM pathology. With respect to clinical outcomes, there is strong evidence that WM patterns can facilitate lateralization of the seizure focus in TLE, and that the presence of extratemporal pathology increases risk for drug resistance as well as poor seizure outcomes following surgical resection (e.g., ATL). Finally, studies using dMRI tractography have demonstrated that visualization of the optic radiations can lead to improved visual field outcomes following surgery.
Despite these advances, future work is needed to replicate these findings in larger samples, expand to epilepsy syndromes beyond TLE, increase generalizability of findings by including more diverse populations, and utilize advanced analytical techniques. For instance, machine learning is well suited for WM analyses in epilepsy for two main reasons: (1) predictive models generated by training data can be tested in external samples and thus permit the evaluation of the generalizability of the results and (2) machine learning allows for abridging complex data into variables that can be identified as relevant or discarded as noncrucial, as well as reducing data into fewer dimensions. Conventional machine learning approaches such as support vector machine and random-forest, among others, have been applied to WM in epilepsy and are excellent strategies for the identification of complex patterns and out-of-sample testing. Moreover, feed-forward neural networks or convolutional neural networks are also well-suited to abridge and test information, with convolutional neural networks being particularly relevant for 2D or 3D image-pattern detections, which can be derived from connectome-based matrices.
In addition, many unanswered questions remain regarding the origin and evolution of WM disruption in epilepsy, including the following:
Does WM disruption lead to the development of seizures or do recurrent seizures result in progressive WM damage?
What is the functional relevance and temporal course of FA changes (i.e., does increased FA or connectivity early in disease represent pathologically enhanced signal flow that occurs prior to white matter degradation)?
How do WM networks reorganize after surgery and what is the time course of reorganization?
Does the trajectory of WM recovery or re-organization correspond with cognitive or psychiatric improvements?
How do patterns of reorganization within WM networks relate to functional reorganization?
In addition, it is challenging to study direct associations between any single clinical seizure variable (e.g., age of seizure onset, seizure duration, drug resistance) and WM injury given the high interdependence of these variables. Many of these questions can be addressed with longitudinal studies of patients with new-onset epilepsy and at multiple time points following surgery. Recent studies have developed nomograms, or easy-to-use risk stratification models that allow clinicians to estimate the probability of cognitive, emotional, or seizure outcomes in adults considering epilepsy surgery (e.g., Busch and others 2018; Doherty and others 2021; Jehi and others 2015). These studies have included clinical and demographic variables (e.g., side of seizure onset, education, cognitive score) as predictors of decline. Given new data suggesting that dMRI may add to the prediction of cognitive and seizure outcomes, future nomograms may benefit from the addition of markers of WM microstructure. In addition, no studies have used baseline WM integrity to risk-stratify patients with regard to cognitive or seizure outcomes following new surgical interventions that mostly spare collateral WM (e.g., laser ablation). Such studies may provide a more definitive answer as to the importance of WM integrity to a range of postsurgical outcomes.
As the field moves toward an understanding of epilepsy as a network disorder, there is an increased usage of neurostimulation to treat refractory epilepsy. The responsive neurostimulation system delivers responsive stimulation to halt seizures, and also provides long-term neuromodulation. Similarly, deep brain stimulation of thalamic nuclei and vagus nerve stimulation of the peripheral part of the cranial nerve are neuromodulatory treatments for seizures that could affect WM connectivity. With regard to vagus nerve stimulation, increased volume of WM microstructure in the vagus afferent network has been associated with increased treatment efficacy (Mithani and others 2019). Further research examining WM changes following each of these neuromodulation treatments could improve patient selection and increase our understanding of WM neuroplasticity in epilepsy.
Last, there is a need to understand the impact of racial and ethnic health disparities on integrity of WM networks in epilepsy. Literature outside of epilepsy suggests a strong link between health disparities and brain and cognitive health. For instance, poorer WM integrity has been associated with fewer years of schooling, lower household income (Gianaros and others 2013), and lower socioeconomic status (Shaked and others 2019), which may lead to an increased risk for age-related and disease-related cognitive decline. However, minimal research exists on the additive impact of epilepsy and social determinants of health on WM integrity. Deeper phenotyping of our patients and efforts aimed at increasing the sociocultural, ethnic, and racial characterization of our samples would enhance the generalizability of these findings and lead to a more enriched understanding of the causes and consequences of WM injury in epilepsy.
Footnotes
Acknowledgements
We thank Erik Kaestner, Donatello Arienzo, and Adam Schadler for helpful comments and discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CRM was supported by NINDS R01NS120976, R01NS124585, R01122827, and R21NS107739. AS was supported by a Ruth L. Kirschtein Postdoctoral Individual National Research Service Award from NINDS (F32 NS119285-01A1).
