Abstract
Introduction
This study aimed to evaluate the independent associations of myopenia, myosteatosis, and frailty with survival outcomes in older patients with prostate cancer undergoing definitive radiotherapy.
Methods
A total of 124 patients aged ≥65 years who received definitive radiotherapy for localized or locally advanced prostate cancer between 2014 and 2023 were retrospectively analyzed. Myopenia and myosteatosis were quantified using the psoas muscle index (PMI) and Hounsfield unit average calculation (HUAC), respectively. Comorbidity-based frailty was assessed using the modified frailty index-11 (mFI-11). The cut-off points for the PMI and HUAC were determined using receiver operating characteristic (ROC) curve analysis. Univariate and parsimonious multivariable Cox regression analyses were used to examine the prognostic value of these markers for progression-free survival (PFS) and overall survival (OS).
Results
The median age of the patients was 72 years (range: 65–87). According to the mFI-11, 47.6% of the patients were classified as frail. The cut-off values for PMI and HUAC were 2.0 cm2/m2 and 37.7 HU, respectively. In univariate analysis, only PMI was significantly associated with both PFS (HR: 2.23, p = 0.02) and OS (HR: 2.45, p = 0.03). When entered separately into parsimonious multivariable Cox models adjusted for age and NCCN risk group, PMI remained an independent prognostic factor for both PFS (HR: 2.36, p = 0.01) and OS (HR: 2.40, p = 0.04), whereas mFI-11 and HUAC were not significantly associated with either endpoint.
Conclusion
Among the evaluated body composition and frailty-related markers, only PMI-defined myopenia remained independently associated with both PFS and OS in older patients receiving definitive radiotherapy for localized or locally advanced prostate cancer. As PMI can be obtained opportunistically from routine planning CT, it may have value for prognostic stratification, although confirmation in external prospective datasets is required.
Plain Language Summary
Prostate cancer is common in older men, who often have other health problems and may be physically weaker. Doctors need simple tools to estimate how long these patients are likely to live and how well they might do after radiotherapy. In this study, we examined 124 men aged 65 years or older who received radiotherapy for prostate cancer that had not spread to distant organs. Before treatment, all patients had routine planning CT scans. From these scans, we measured the size of a large muscle in the lower back (the psoas muscle) and calculated a value called the psoas muscle index, which reflects overall muscle mass. We also assessed muscle quality and a standard frailty score based on other illnesses and general condition. We found that men with lower muscle mass had a higher risk of cancer worsening and a higher risk of death. This link remained even after accounting for age and cancer risk group. Our results suggest that a simple muscle measurement from existing scans may help doctors better estimate prognosis and guide treatment decisions in older men receiving radiotherapy for prostate cancer.
Introduction
Prostate cancer is the most prevalent malignancy in men and ranks among the top five cancers with the highest mortality rates. 1 Localized and locally advanced prostate cancer is commonly risk-stratified using contemporary guideline-based systems that incorporate serum prostate-specific antigen (PSA), Gleason score, and clinical T stage. 2 Treatment modalities are determined using a multidisciplinary approach, tailored to the patient’s risk category and disease stage. 3 In addition, the performance status, physiological condition, and life expectancy of patients play a crucial role in guiding treatment decisions.
Sarcopenia is a muscle disease characterized by the generalized and accelerated loss of skeletal muscle mass, strength, and function, primarily associated with aging and compounded by factors such as chronic inflammation, malnutrition, reduced physical activity, and comorbidities including cancer or organ failure. 4 Although sarcopenia encompasses loss of both muscle mass and strength, many oncologic studies rely solely on imaging-based assessments of muscle quantity or quality. In such cases, the terms myopenia and myosteatosis more accurately describe the measured phenomena. Myopenia, defined as reduced skeletal muscle mass, and myosteatosis, characterized by increased fat infiltration within skeletal muscle, are frequently observed in older adults and cancer patients.5-8 McDonald et al reported in a pooled analysis of prostate radiotherapy trials that body composition parameters, including psoas-based measures, provided prognostic value for mortality. 9 Similarly, an earlier study showed that the L4–L5 psoas area predicted non-cancer mortality in men with localized prostate cancer treated with radiotherapy. 10 In another study, it was shown that decreased Hounsfield unit average calculation (HUAC) values in the psoas muscles, a marker of myosteatosis, predicted lower overall survival (OS) in patients with metastatic prostate cancer. 11
Frailty is a clinical syndrome defined by diminished physiological reserve and resilience to stressors, leading to an increased vulnerability to adverse health outcomes. Multiple models have been developed to operationalize frailty. The phenotypic model introduced by Fried et al defines frailty based on physical criteria such as weakness, slowness, and unintentional weight loss, while the Frailty Index is based on the accumulation of health deficits. 12 The modified frailty index-11 (mFI-11), adapted from the Canadian Study of Health and Aging Frailty Index, offers a simplified and pragmatic approach by relying on 10 comorbid conditions and 1 functional status variable. 13 Its main advantages include ease of use in retrospective studies and compatibility with electronic health records. However, unlike phenotype-based tools, the mFI-11 does not account for physical performance or muscle status, which may limit its ability to fully capture physiologic vulnerability in older cancer patients. Frailty, as defined by the mFI-11, is strongly associated with an increased risk of postoperative complications in patients undergoing urological surgery.14,15 Pan et al found that frailty, assessed using a distinct multimorbidity-based frailty index, was associated with increased mortality in older prostate cancer patients undergoing radiotherapy. 16
Numerous tools have been developed to facilitate the screening and assessment of myopenia, myosteatosis, and frailty.17-20 Recent literature supports the prognostic relevance of computed tomography (CT)-derived body composition and frailty-related parameters in prostate cancer. However, these measures have rarely been evaluated concurrently within the same cohort of older men treated exclusively with definitive radiotherapy, and their relative clinical utility in this setting remains unclear. It is uncertain whether a simulation-CT-derived marker of muscle quantity (PMI), a muscle quality marker (HUAC), or an mFI-11-based comorbidity frailty measure provides the most informative prognostic value. Therefore, this study aimed to evaluate the independent associations of PMI, HUAC, and mFI-11 with progression-free survival (PFS) and OS in older patients with localized or locally advanced prostate cancer undergoing definitive radiotherapy.
Materials and Methods
Patient Selection
Following institutional ethics board approval (Approval No: 99, Date: 08.02.2023), a retrospective analysis was conducted on 124 patients aged ≥65 years with localized or locally advanced prostate cancer who received definitive radiotherapy between January 2014 and January 2023. Patients with an Eastern Cooperative Oncology Group performance status (ECOG-PS) of 3–4, the absence of simulation CT images, a prior history of radical prostatectomy, or evidence of metastatic disease at the time of diagnosis were excluded from the study. The patient selection process is summarized in Figure 1. The reporting of this study conforms to relevant Equator guidelines.
21
Patient selection overview
Treatment
Patients were staged and risk-stratified according to the contemporaneous National Comprehensive Cancer Network (NCCN) guidelines applicable at the time of treatment. The indication for definitive external-beam radiotherapy (EBRT) was determined in accordance with these recommendations. Definitive-intent EBRT to a total dose of 70–76 Gy was delivered using either linac-based intensity-modulated radiotherapy (IMRT) (Elekta Synergy, Elekta, Sweden) or helical tomotherapy (TomoTherapy HDA, Accuray, USA). All patients received conventionally fractionated EBRT, with fraction sizes of 1.8–2.0 Gy. Daily image-guided radiotherapy (IGRT) was used in all patients, with cone-beam computed tomography (CBCT) for linac-based treatments and megavoltage computed tomography (MVCT) for helical tomotherapy.
In all patients, the prostate and proximal seminal vesicles were included in the high-dose clinical target volume (CTV), which received 70–76 Gy with corresponding planning target volume (PTV) margins of 5–7 mm. In patients with high-risk or very high-risk disease, the entire seminal vesicles were included in the target volume. Pelvic nodal irradiation was planned for patients with high-risk or very high-risk disease, as well as for those with PSMA PET-confirmed nodal involvement. In patients selected for pelvic nodal irradiation, the CTV included the common iliac, internal iliac, external iliac, presacral, and obturator nodal regions; 46 Gy was delivered to this volume with corresponding PTV margins of 4–6 mm. However, in a subset of high-risk patients without evidence of nodal metastasis on PSMA PET imaging, elective pelvic nodal irradiation was omitted based on individualized treatment strategy and treating physician discretion. Whenever dose escalation beyond the initial treatment volume was required, it was delivered exclusively as a sequential boost; a simultaneous integrated boost (SIB) technique was not used. No patients received a brachytherapy boost.
Throughout the study period, androgen deprivation therapy (ADT) was generally administered in accordance with NCCN-informed, risk-adapted clinical practice. Short-term ADT was typically used in intermediate-risk disease, whereas long-term ADT was generally administered in high-/very high-risk disease; however, a small subset of low-risk patients also received short-term ADT based on individualized clinical judgment. ADT was initiated either in the neoadjuvant setting or concurrently with radiotherapy. When given neoadjuvant, EBRT was typically started after approximately 3 months of ADT; otherwise, ADT was initiated concurrently with radiotherapy and continued in the adjuvant setting after completion of EBRT. Because of the retrospective, real-world nature of the cohort, ADT indication, timing, and duration were ultimately individualized according to disease characteristics, comorbidity profile, and treating physician discretion.
Data Collection
Patient characteristics such as age, height, ECOG-PS, PSA level, histopathology, Gleason score, and TNM stage were collected from hospital records. PMI and HUAC, used as markers for myopenia and myosteatosis, respectively, were derived from bilateral psoas muscle measurements obtained on a single axial slice at the intervertebral level between the fourth (L4) and fifth (L5) lumbar vertebrae.9,22 The L4–L5 level was selected because it was consistently captured on radiotherapy simulation CT scans in all patients, allowing a pragmatic and standardized assessment in this retrospective planning-CT–based cohort. Non-contrast radiotherapy simulation computed tomography (CT) scans acquired prior to treatment were used for all measurements. Image analysis was performed using the Monaco treatment planning system (Elekta, Sweden). To standardize skeletal muscle segmentation and exclude non-muscle tissue, voxels within the commonly used skeletal muscle attenuation range (–29 to +150 HU) were included. The psoas muscles were manually segmented by a radiation oncologist with 5 years of experience in radiotherapy-based image analysis. Segmentations were subsequently reviewed by a second radiation oncologist with over 20 years of experience. Final contours were established by consensus.
Measurements of PMI and HUAC in two sample patients are presented in Figure 2. Additionally, the formulas utilized for calculating PMI (Equation (1)) and HUAC (Equation (2)) are presented below. Measurements of psoas muscle index (PMI) and Hounsfield unit average calculation (HUAC) in two sample patients
The mFI-11, a comorbidity-based frailty index, was utilized to assess frailty in the patients. This index consists of 11 parameters, including diabetes mellitus, hypertension requiring medical treatment, history of chronic obstructive pulmonary disease or pneumonia, presence of congestive heart failure, history of myocardial infarction, percutaneous coronary intervention, stent or angina, a performance score of 2 or above, history of peripheral vascular disease or ischemic pain at rest, sensory impairment, history of cerebrovascular accident or transient ischemic attack, and history of cerebrovascular accident causing neurological deficit. Each parameter was assigned 1 point, resulting in mFI-11 scores ranging from 0 to 11 for the patients. In line with prior studies, 23 patients with an mFI-11 score of 0–1 were considered non-frail, whereas those with a score ≥2 were classified as frail.
The Glasgow Prognostic Score (GPS) was calculated based on the C-reactive protein (CRP) and albumin values. A CRP ≤ 10 mg/L was assigned 0 points, CRP > 10 mg/L and albumin ≥ 3.5 g/dL was assigned 1 point, and CRP > 10 mg/L and albumin < 3.5 g/dL was assigned 2 points. A score of 0 indicates a good prognosis, 1 represents an intermediate prognosis, and 2 indicates a poor prognosis. 24
Follow-Up and Outcome Assessment
All patients were followed from the completion date of radiotherapy until death or the date of last clinical follow-up. The final patient completed radiotherapy in January 2023, and all patients had a minimum follow-up duration of 12 months, with follow-up completed by January 2024. Biochemical recurrence was defined according to the Phoenix definition (PSA nadir + 2 ng/mL). 25 Locoregional progression and distant metastasis were determined based on follow-up imaging studies. PFS was defined as the time from the end of radiotherapy to the first documented event of biochemical recurrence, locoregional progression, distant metastasis, or death, whichever occurred first. OS was defined as the time from the end of radiotherapy to death from any cause. Outcome data were obtained from electronic medical records and independently verified by two radiation oncologists.
Statistical Analyses
Statistical analyses were conducted using IBM SPSS Statistics software, version 24.0 (IBM Corp., Armonk, NY, 2016), with p < 0.05 considered indicative of statistical significance. Descriptive statistics were conducted to summarize the baseline characteristics of the study population. Continuous variables were reported as mean ± standard deviation or median (range), while categorical variables were described using frequencies and percentages. Categorical variables were compared using the χ2 test.
Receiver operating characteristic (ROC) curve analysis was performed for PMI and HUAC using all-cause mortality status at last follow-up (dead vs alive) as the binary outcome. Cut-off values were selected at the point where sensitivity most closely approximated specificity. 26 Because these cut-off values were derived and subsequently applied within the same cohort, they were considered exploratory and cohort-specific, and hazard ratios (HRs) from dichotomized analyses were interpreted as potentially optimistic. To assess the stability of the ROC-derived cut-off values, nonparametric bootstrap resampling (5,000 samples) was performed using the same prespecified selection rule, and percentile-based 95% confidence intervals (CIs) were derived from the empirical distribution of bootstrap cut-off values. The Kaplan-Meier method was used to construct survival curves, and differences between them were analyzed using the log-rank test. Univariate Cox proportional hazards regression analyses were conducted to estimate HRs and 95% CIs for PFS and OS. For Cox regression analyses, the original four-category NCCN risk classification was dichotomized as high/very high versus other, with “other” representing the combined low- and intermediate-risk categories, to preserve model parsimony given the limited number of outcome events. Additionally, given the limited number of outcome events, parsimonious multivariable Cox regression models were constructed to reduce the risk of overfitting. Rather than including age, NCCN risk group, PMI, mFI-11, and HUAC in a single model, separate models were built for PMI, mFI-11, and HUAC, each adjusted for age and NCCN risk group. Because PMI and HUAC represent related CT-derived body composition domains and showed a significant association in baseline analyses, they were not entered simultaneously into the same multivariable model. The proportional hazards assumption was assessed using log–minus–log survival plots, and no major violations were observed.
Results
Patient and Tumor Characteristics of the Entire Cohort and Stratified by Psoas Muscle Index (PMI) Groups

Abbreviations: PMI, psoas muscle index; PSA, prostate-specific antigen; ADT, androgen deprivation therapy; ECOG-PS, eastern cooperative oncology group-performance status; mFI-11, modified frailty index-11; HUAC, Hounsfield unit average calculation.
The mean PMI was 2.2 ± 0.52 (range: 1.16–3.54), with a cut-off value of 2, sensitivity of 59.1%, specificity of 63.7%, and an area under the ROC curve of 0.61 (95% CI: 0.47–0.74). Bootstrap resampling yielded a 95% CI of 1.88–2.27 for the PMI cut-off. The ROC curve for PMI is presented in Figure 3. The mean HUAC was 36.6 ± 8.8 (range: 11.7–59.4), with a cut-off value of 37.7, sensitivity of 50.0%, specificity of 52.0%, and an area under the ROC curve of 0.53 (95% CI: 0.39–0.66). Bootstrap resampling yielded a 95% CI of 35.99–41.77 for the HUAC cut-off. Receiver operating characteristic (ROC) curve analysis for psoas muscle index (PMI)
Myopenia and myosteatosis were observed in 40.3% (n=50) and 51.6% (n=64) of patients, respectively. Based on the mFI-11, 47.6% of patients were classified as frail. There was no significant association between PMI groups and mFI-11 categories (χ2 test, p=0.65), nor between HUAC groups and mFI-11 categories (χ2 test, p=0.20). In contrast, a statistically significant association was found between PMI and HUAC groups, with lower PMI more frequently associated with lower HUAC (χ2 test, p=0.02).
The median follow-up duration was 48 months (range: 12–113). During follow-up, 13 patients (10.5%) experienced disease progression. Additionally, 20 patients (16.1%) died without any documented progression, accounting for 60.6% of all PFS events and indicating that the composite PFS endpoint was substantially influenced by non-progression deaths. Accordingly, 33 patients (26.6%) experienced a PFS event (progression or death, whichever occurred first). Overall, 22 patients (17.7%) died during follow-up. The 3-year and 5-year PFS rates were 90.0% and 75.0%, respectively, while the 3-year and 5-year OS rates were 94.7% and 81.5%.
Univariate Cox Regression Analysis of Prognostic Factors for Progression-Free Survival
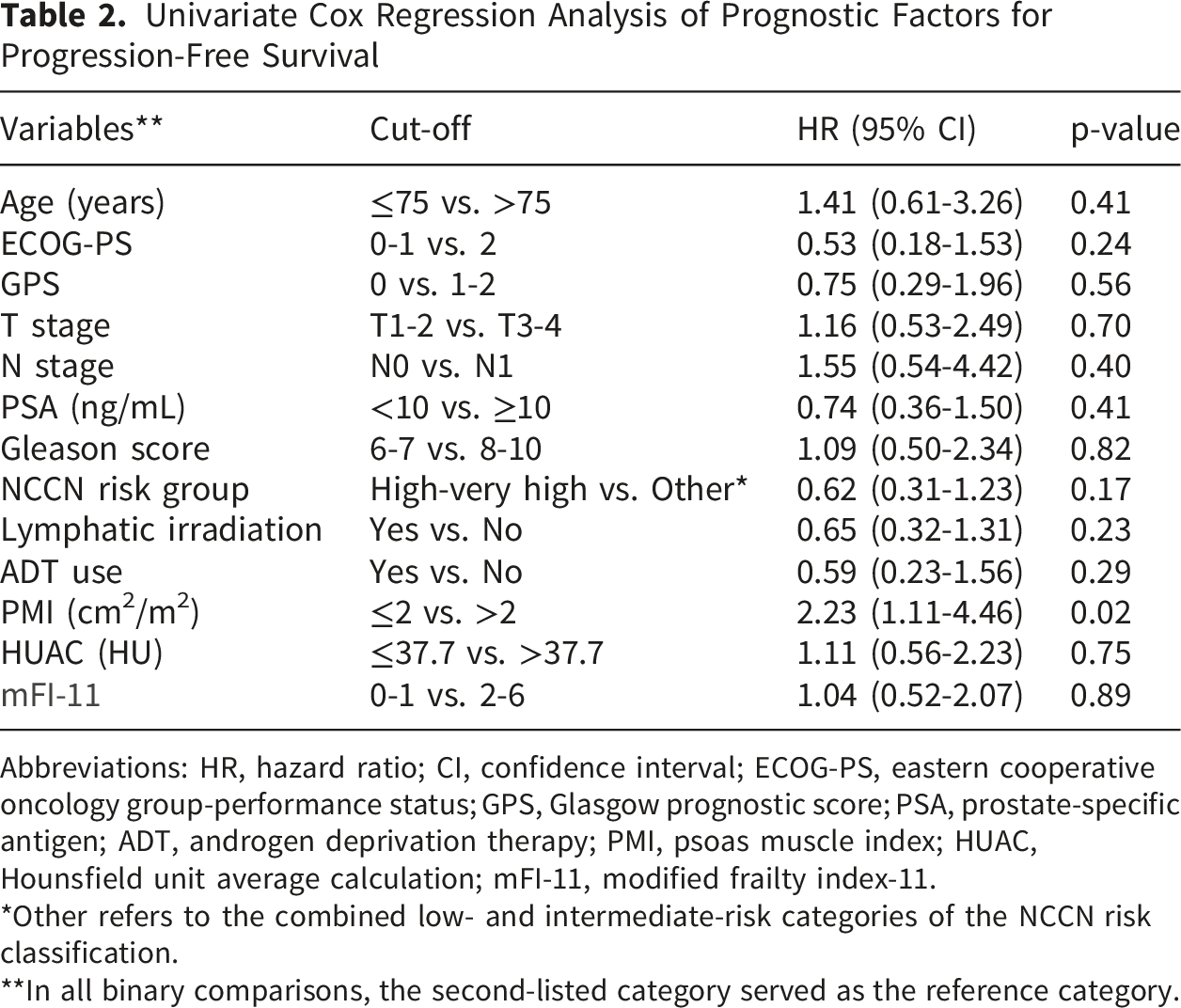
Abbreviations: HR, hazard ratio; CI, confidence interval; ECOG-PS, eastern cooperative oncology group-performance status; GPS, Glasgow prognostic score; PSA, prostate-specific antigen; ADT, androgen deprivation therapy; PMI, psoas muscle index; HUAC, Hounsfield unit average calculation; mFI-11, modified frailty index-11.
*Other refers to the combined low- and intermediate-risk categories of the NCCN risk classification.
**In all binary comparisons, the second-listed category served as the reference category.

Kaplan–Meier curves for progression-free survival (A) and overall survival (B) according to psoas muscle index (PMI) groups. P values were calculated using the log-rank test
Univariate Cox Regression Analysis of Prognostic Factors for Overall Survival

Abbreviations: HR, hazard ratio; CI, confidence interval; ECOG-PS, eastern cooperative oncology group-performance status; GPS, Glasgow prognostic score; PSA, prostate-specific antigen; ADT, androgen deprivation therapy; PMI, psoas muscle index; HUAC, Hounsfield unit average calculation; mFI-11, modified frailty index-11.
*Other refers to the combined low- and intermediate-risk categories of the NCCN risk classification.
**In all binary comparisons, the second-listed category served as the reference category.
Parsimonious Multivariable Cox Models for Progression-Free Survival and Overall Survival

Abbreviations: PFS, progression-free survival; OS, overall survival; HR, hazard ratio; CI, confidence interval; PMI, psoas muscle index; mFI-11, modified frailty index-11; HUAC, Hounsfield unit average calculation.
*Other refers to the combined low- and intermediate-risk categories of the NCCN risk classification.
**In all binary comparisons, the second-listed category served as the reference category.
Discussion
This study assessed the prognostic value of myopenia, myosteatosis, and a comorbidity-based frailty index for PFS and OS in patients with prostate cancer aged ≥65 years who were treated with definitive radiotherapy. Among these markers, the PMI was identified as a statistically significant prognostic factor for both PFS and OS. In parsimonious multivariable models adjusted for age and NCCN risk group, PMI remained independently associated with both PFS and OS. By contrast, HUAC and mFI-11 were not significantly associated with either endpoint in their respective adjusted models. This suggests that PMI-defined myopenia may provide prognostic information that was not captured as robustly by HUAC or mFI-11 in this cohort. These findings extend prior related studies on CT-derived body composition and frailty in prostate cancer. Our incremental contribution is the direct comparison of a simulation-CT-derived muscle quantity marker (PMI), a muscle quality marker (HUAC), and an mFI-11-based comorbidity frailty measure within the same treatment-homogeneous cohort of patients aged ≥65 years with localized or locally advanced prostate cancer receiving definitive radiotherapy. Interpretation of the PFS finding warrants caution. In our cohort, 20 of the 33 PFS events were deaths without documented progression; therefore, the composite PFS endpoint likely reflected overall health vulnerability and competing mortality at least as much as prostate cancer progression. Accordingly, the association between low PMI and shorter PFS should not be interpreted as evidence of impaired cancer control alone.
Myopenia has been associated with adverse clinical outcomes, including increased mortality, in patients with various malignancies. This condition may occur independently due to aging or develop secondarily because of cancer or other comorbidities. Several studies have demonstrated that quantitative muscle depletion, often assessed by imaging-based markers such as the PMI, can predict OS in older cancer patients.27,28 For instance, a study in patients aged ≥80 years with early-stage gastric cancer reported that low PMI was significantly associated with shorter OS. 29 Similarly, another investigation involving patients aged ≥70 years with Diffuse Large B-Cell Lymphoma treated with R-CHOP found that reduced PMI predicted poorer OS. 30
Summary of Previous Studies Evaluating Muscle-Related Prognostic Markers in Older Patients With Prostate Cancer

Abbreviations: D, design; HR, hazard ratio; PMI, psoas muscle index; R, retrospective; PSMA-RLT, prostate-specific membrane antigen-directed radioligand therapy; OS, overall survival, Ra-223, radium 223 dichloride; CSS, cancer specific survival; ALT, alanine aminotransferase; SMI, skeletal muscle index; NCS, non-cancer-specific survival; AMI, attenuation metabolic index; FFS, failure-free survival; ADT, androgen deprivation therapy; ARAT, androgen receptor axis targeted; P, prospective; PMID, psoas muscle index density.
The prognostic significance of HUAC, a marker of myosteatosis examined in our study, has been explored in various types of cancer. A study involving patients who underwent major surgery for pancreatic cancer demonstrated that low HUAC levels were associated with increased postoperative mortality and complications. 41 In patients with gastric cancer who underwent radical gastrectomy, HUAC has been found to be a significant predictor of both OS and cancer-specific survival (CSS). 42 Similarly, in a study involving 59 metastatic prostate cancer patients, higher HUAC levels were found to be associated with improved OS. 11 In contrast, our study found that HUAC was not a significant predictor of either PFS or OS in older patients with prostate cancer receiving radiotherapy. While several studies in the literature report the prognostic significance of PMI and HUAC in various cancers, our findings indicate that PMI is a more reliable prognostic factor than HUAC in older patients with prostate cancer undergoing radiotherapy.
Frailty is a multidimensional condition that can be measured using different instruments. Some tools focus on physical performance, whereas others are based mainly on accumulated health deficits and comorbidity burden.43-45 In the present study, frailty was assessed using the mFI-11, a comorbidity-based deficit-accumulation index that is practical for retrospective analyses. In patients with endometrial cancer, a preoperative mFI-11 value greater than 3 has been shown to negatively impact OS, with statistical significance observed in both univariate and multivariate analyses. 46 Another study demonstrated that an elevated mFI-5 score negatively impacted OS in bladder cancer patients who underwent radical cystectomy. 47 In contrast, the mFI-11 was not found to be a significant prognostic factor of either PFS or OS in our patient population. This finding should not be interpreted as evidence against the prognostic relevance of frailty in older patients receiving radiotherapy. Instead, it suggests that the mFI-11 may not fully reflect the physical and functional aspects of frailty that are most relevant to radiotherapy outcomes.
This study has several important limitations that should be acknowledged. First, the sample size was modest, which may limit the statistical power and the ability to detect weaker associations. For instance, conventional clinical indicators such as T stage, N stage, and NCCN risk category did not demonstrate statistically significant associations with PFS or OS in this cohort. This may be attributable to the limited number of patients and relatively short follow-up duration. In addition, the cut-off values for PMI and HUAC were derived from ROC analyses within the same study cohort. Although their stability was further assessed by bootstrap resampling, these cut-off values should still be considered exploratory and cohort-specific rather than universally applicable. Moreover, the limited number of outcome events may reduce the precision of multivariable estimates and support interpreting these findings as hypothesis-generating rather than definitive. Second, although the use of the psoas muscle at the L4–L5 level is pragmatic in the radiotherapy setting because this region is consistently captured on planning CT, it does not correspond to the more commonly used whole-muscle surrogate at the L3 level in body composition research. In addition, psoas-based measurements may not uniformly reflect global myopenia or myosteatosis and may therefore incompletely represent overall muscle quantity and quality. Third, although the mFI-11 is practical and suitable for retrospective studies, it primarily reflects comorbidity burden rather than multidimensional geriatric frailty. In this respect, the comparison between mFI-11-defined frailty and myopenia should be interpreted with caution, as these measures do not capture equivalent biological and functional domains. In particular, the mFI-11 does not directly assess muscle strength, mobility, or physical performance, which may be more relevant to radiotherapy tolerance and survival in older adults. Future studies may benefit from incorporating broader geriatric assessment tools, such as the Geriatric-8, Vulnerable Elders Survey-13, or frailty phenotype–based approaches, to provide a more comprehensive evaluation of frailty in this setting.
Conclusion
Among CT-derived body composition measures and mFI-11-defined frailty, PMI-defined myopenia was the only variable independently associated with both PFS and OS in this cohort of older patients treated with definitive radiotherapy for localized or locally advanced prostate cancer. These findings support the potential role of opportunistic PMI assessment from routine simulation CT as a clinically accessible prognostic marker. Nevertheless, the results remain hypothesis-generating and should be interpreted cautiously given the retrospective design, limited event rate, and cohort-specific cut-off derivation. Further prospective multicenter studies are needed to validate these findings and to establish more generalizable cut-off values for clinical use.
Footnotes
Acknowledgements
This study was presented at the 6th Geriatric Oncology Congress Annual Meeting held in Turkey, November 08-10, 2024. This study is the extension, and the final manuscript of “Sarcopenia and frailty: Can they predict survival in older patients with prostate cancer undergoing definitive radiotherapy?”. Generative artificial intelligence tools were used solely for language editing and clarity improvement; no scientific content or data were generated or modified.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and approved by the Akdeniz University Faculty of Medicine Clinical Research Ethics Committee (Approval No: 99, Date: 08.02.2023).
Consent to Participate
Informed consent was waived due to the retrospective design of the study and the de-identification of patient data.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


