Abstract
Introduction
The long-acting somatostatin analogues (LA-SSAs) octreotide LAR (OCT) and lanreotide (LAN) improve progression-free survival (PFS) in gastrointestinal neuroendocrine tumors (NETs), however, no head-to-head comparison exists. We compared treatment patterns and efficacy in a small bowel and pancreatic NET population-based cohort from British Columbia, Canada.
Methods
We identified 321 patients receiving either LAN or OCT for retrospective chart review. These somatostatin analogs were evaluated for impact on progression-free and overall survival.
Results
Age, sex, ECOG, and primary site did not differ by treatment, however, LAN was more commonly used in higher grade tumors (P = 0.019). PFS was longer for patients receiving LAN than OCT (Hazard Ratio (HR) 0.60, 95% CI 0.40-0.89, P = 0.011). Similarly, overall survival (OS) was longer for patients receiving LAN than OCT (HR 0.45, 95% CI 0.28-0.73, P = 0.016). Sensitivity analysis among patients diagnosed after both agents were reimbursed showed similar results for PFS (HR 0.50, 95% CI 0.28-0.90, P = 0.018). There was similar dose escalation with LAN vs OCT (OR: 0.80, CI 0.38-1.77, P = 0.70), with 29.4% of patients in the LAN group requiring LA-SSA dose escalation compared to 34.3% in the OCT group. There was numerically less short acting octreotide use in the LAN group (P = 0.087), with none of these patients requiring short acting octreotide, compared to 8.7% of the OCT group.
Conclusion
LAN was associated with longer time to cancer progression, as well as less use of short acting rescue octreotide in our population-based cohort. However, given the retrospective design and reimbursement-era differences, these findings should be interpreted cautiously and warrant confirmation in prospective or head-to-head studies.
Plain Language Summary
Comparing the outcomes from Lanreotide and Octreotide LAR in a subset of digestive system cancers using a British Columbia Cohort: This study looked at how two medications—Octreotide LAR and Lanreotide—are used to treat people with neuroendocrine tumors (NETs) in the small bowel and pancreas in British Columbia, Canada. These drugs help slow the growth of tumors, but no direct comparisons have been made between them in clinical trials. Using healthcare records, this study compared how often each drug was used, how well they worked, and how often doses were increased or switched. Lanreotide was more often used in more aggressive tumors and was linked to longer survival. Dose increases were similar between the two drugs, but fewer patients needed extra short-acting medication with Lanreotide. Most switches between medications were due to side effects.
Introduction
Neuroendocrine tumors (NETs) are malignant tumors that can arise anywhere in the body, and which originate from hormone-secreting cells. 1 Their ability to produce hormones can lead to symptoms such as diarrhea, flushing and wheezing.2-5 These cancers are a relatively rare, heterogenous group of tumors, accounting for 0.5% of all malignancies,6-8 with more than 50% of tumors arising from the pancreas or gastrointestinal system.2,9 In the last several decades, the incidence of neuroendocrine tumors has been increasing, potentially due to increasing recognition of this entity.5-8,10,11
Fortunately, patients diagnosed with NETs can live for a significant period of time due to the often-indolent clinical course of these tumors.1,2,12 For metastatic well-differentiated NETs, a variety of treatment modalities are available, including surgical debulking, cytotoxic therapy, somatostatin analogs, targeted agents and peptide receptor-targeted radionuclide therapy (PRRT). 13 The long-acting somatostatin analogs (LA-SSAs), OCT and LAN, have been found to improve the time to tumor progression (TTP) and progression-free survival (PFS) in gastrointestinal NETs in the PROMID and CLARINET studies, respectively, as compared with placebo,9,13 establishing the antitumor activity of OCT at a dose of 30 mg and LAN at a dose of 120 mg.7,9,13 For patients with hormone secreting tumors, often described as carcinoid syndrome when they secrete serotonin, these agents also reduce hormone secretion and improve symptom management.
These LA-SSAs are approved for use in metastatic NETs. For patients who have serotonin secreting tumors (carcinoid tumors), they are typically started on a first line LA-SSA, and continued life-long, even after progression on the first line. For patients without hormone-secreting tumors, but diagnosis of a well-differentiated NET, somatostatin analogues are usually the first treatment used in the metastatic setting for small bowel NETs, and are an option for first or subsequent lines of therapy in pancreatic NETs. Starting dose for LAN is 120 mg administered every 4 weeks by subcutaneous injection while OCT is administered 30 mg every 4 weeks intramuscularly and dose escalation and modification are at clinician discretion as this was a real-world study. Somatostatin exerts its physiological effects in the body by binding with high-affinity to at least 5 distinct G-protein-coupled somatostatin receptors, SSTR 1-5. Octreotide LAR and lanreotide are both synthetic cyclic octapeptides that bind with high affinity to the sstr2 and sstr5 subtypes, however studies have shown that lanreotide potentially exhibits more potent binding than octreotide to the sstr5 receptor. 14 Further to this, lanreotide is known to have a more rapid onset of absorption, while octreotide LAR has a more gradual absorption for the first dose. To date, there have been no head-to-head trials comparing the efficacy or overall survival (OS) in patients with gastrointestinal NETs treated with OCT vs LAN. We therefore completed a retrospective study within the province of British Columbia, Canada to compare the use of LA-SSAs, survival outcomes, dose escalation and breakthrough octreotide usage in patients with metastatic well-differentiated NETs of the pancreas and gastrointestinal system.
Methods
After the study was approved by the BC Cancer research ethics board (H18-01396), BC Cancer medical records and provincial pharmacy databases were queried for patients with well-differentiated small bowel and pancreatic NETs diagnosed between February 1990 and November 2020 who had received a LA-SSA. Informed consent requirements were waived due to the retrospective nature of the study. BC Cancer provides cancer care to the 5 million people of the province of British Columbia and is responsible for funding all publicly funded cancer medications in the province. All identified cases underwent expert pathology review to assess tumor differentiation, Ki67 status and mitotic index. 15 Data was collected from BC Cancer on patient demographics, treatments and survival and reporting was designed to follow STROBE guidelines. 16 Confirmed cases with metastatic well-differentiated small bowel and pancreatic NETs who had received a LA-SSA were included in the final analysis. Patients who received only short-acting SSA or in whom the date of commencement of LA-SSA was unknown were excluded from analysis. In British Columbia, Canada, LA-SSAs are used as first line treatment for well-differentiated neuroendocrine tumors. Of note, lanreotide was not reimbursed until after 2015, therefore a subgroup analysis was completed for patients starting a LA-SSA after December 14, 2014, when indications for LA-SSA reimbursement became identical in the province of British Columbia.
Outcomes included PFS (time from date of first long-acting somatostatin analogue to progression or death) and OS (time from first long-acting somatostatin analogue to date of death). The date of progression was assessed using radiological reports documenting progression and clinical notes indicating progressive disease resulted in a change in therapy. Thus, clinical progression was based on investigator assessment at the time of providing care to the patient, and did not follow RESIST criteria. Patients were censored if they had not had an event at the time of last follow-up, they switched LA-SSA (ie, octreotide LAR changed to lanreotide) or if they received local or other systemic therapies for a reason other than progression. The time to SSA switch was defined as the time from the start of the initial LA-SSA to the start of the subsequent LA-SSA.
Data was also collected regarding the frequency of LA-SSA dose escalation to a higher monthly dose and/or shorter dosing interval than the recommended anti-proliferative dose, the use of rescue short acting octreotide due to symptoms of hormone excess while on LA-SSA and LA-SSA switch and its reason if a switch occurred.
Statistical Analysis
Patient demographics were summarized as frequencies (%) for categorical variables and as medians with range for continuous variables. To compare groups, a Chi square test was used for analysis of categorical variables and a t-test for continuous variables. The Kaplan-Meier method was used to estimate PFS and OS and the log-rank test was used to compare survival between groups. Fisher’s exact test was used to compare differences in LA-SSA dose escalation and short acting octreotide use, Baptista-Pike was used to calculate odds ratios.
Age at diagnosis, sex, primary tumor location, grade, era (before 2007 or after) and type of somatostatin analog (Lanreotide vs Octreotide LAR) were evaluated for impact on progression-free and overall survival using a forward likelihood ratio multivariate model after all variables were shown to satisfy the proportional hazards assumption. Variables were chosen for inclusion if P < 0.05 and subsequently removed in a further step if P > 0.1. Patients were excluded if they switched between agents or had an unknown grade.
Statistical analysis was performed using IBM SPSS version 28 and GraphPad Prism 9. This study was a retrospective chart review so patient level data is not available in this manuscript, however it can be requested for future collaborations by contacting the corresponding author.
Results
Baseline Patient Demographics and Clinical Characteristics of Patients with Gastrointestinal Neuroendocrine Tumors Treated with Either Octreotide LAR or Lanreotide

Multivariate analysis for Progression-Free Survival

143 events, 237 events with all available data available for multivariate analysis (missing were unknown grade).
Era of SSA use and age not significant in the model and removed during model build.
Multivariate analysis for Overall Survival
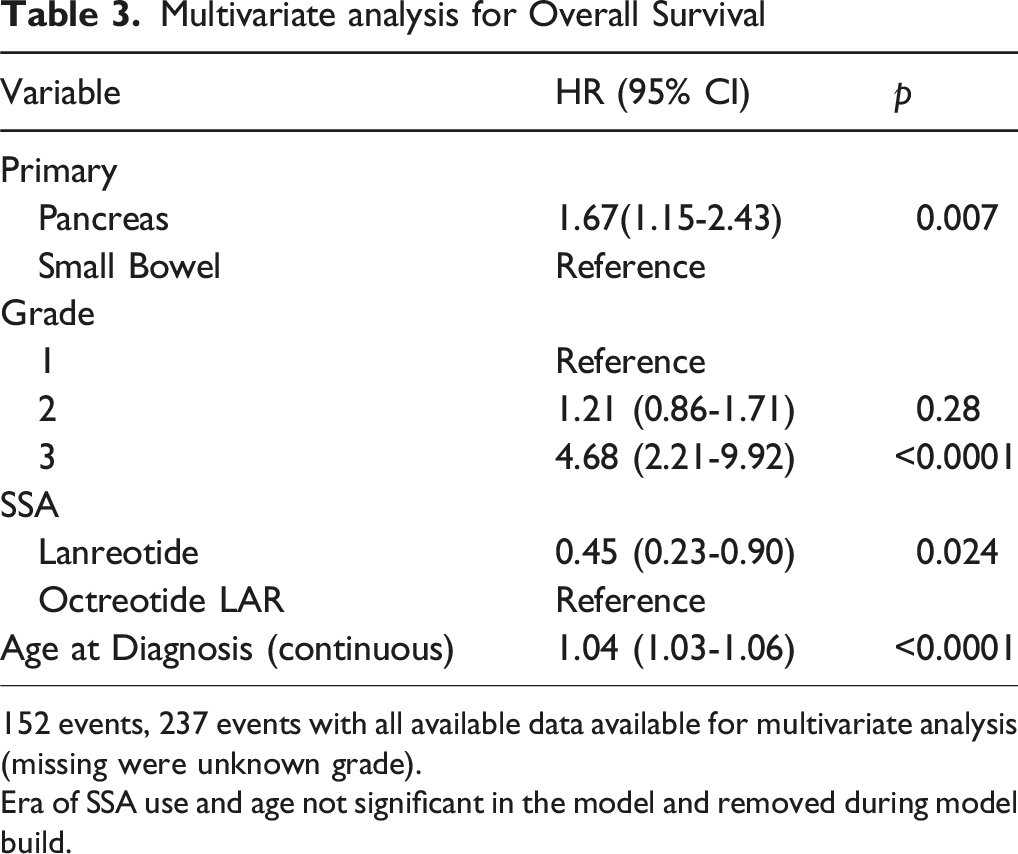
152 events, 237 events with all available data available for multivariate analysis (missing were unknown grade).
Era of SSA use and age not significant in the model and removed during model build.
Median PFS for the OCT group and LAN group were 13.7 months and 55.6 months respectively (HR: 0.60, 95% CI 0.40-0.89, P = 0.011, Figure 1). Similarly, OS was also longer for patients receiving LAN vs OCT (HR: 0.45, 95% CI 0.28-0.73, P = 0.016, Figure 1). Subgroup analysis was completed for patients starting LA-SSA after December 31, 2014, as patients could have used either agent with the same indications at this point and have their therapy reimbursed by BC Cancer (Figure 2). PFS remained longer for patients receiving LAN vs OCT in this era (HR: 0.50, 95% CI 0.28-0.90, P = 0.018). OS was no different for patients receiving LAN vs OCT (HR:0.69, 95% CI 0.32-1.50, P = 0.37). These findings suggest that the observed PFS benefit may persist beyond reimbursement-era effects, although residual confounding cannot be excluded. Comparison of progression-free survival and overall survival for patients receiving octreotide LAR or lanreotide for well-differentiated GI NETs
Studies have shown that patients who are censored at the time of treatment switch are at a higher risk of disease progression compared to the average patient population. Switch incidence and reasons for each SSA are described in Figure 4A & 4B.
17
Therefore, for the PFS analysis, a sensitivity analysis was also performed where patients who underwent a switch were excluded from the analysis as seen in Figure 3. Comparison of progression-free survival and overall survival in patients receiving octreotide LAR or lanreotide for well-differentiated GI NETs commencing respective treatments after Dec 31, 2014, when both agents were officially funded in British Columbia, Canada
There was no difference in dose escalation between LAN (OR: 0.80, 95% CI 0.38-1.77, P = 0.70) and OCT (Figure 4C). There was numerically less short acting octreotide use in the lanreotide group (OR: 0.0, 95% CI 0.0-1.16, P = 0.087), with none of the patients in the LAN group requiring short acting octreotide compared to 8.7% of the OCT group (Figure 4D).
Of the 321 patients included in the study, 45 patients switched from one LA-SSA to the other, with 91.9% switching from OCT to LAN and 8.9% switching from LAN to OCT. Data regarding the reason for switching long-acting somatostatin analogues were also obtained as seen in Figure 4B with the most frequent reasons seen being side effects, injection issues and diarrhea.
Discussion
This retrospective cohort study found that the PFS was longer for patients receiving LAN vs OCT for patients with metastatic well-differentiated pancreatic or small bowel neuroendocrine tumors. Very few real-world studies exist comparing LAN and OCT, however several of these studies have found both clinical and economic advantages of using LAN over OCT that match our findings.18,19 Some of these differences may be because patients receiving LAN are a more contemporary group, where changes in imaging and multidisciplinary care may contribute to improved outcomes. We did not see significant differences in baseline characteristics, and if anything, noted that patients receiving LAN had higher grade disease (Table 1). Subgroup analysis was completed for patients starting their LA-SSA after December 31, 2014, when the indications and reimbursement for these agents were identical in British Columbia and our results were unchanged, supporting that there may be a biologic underpinning to our results (Figure 2). Sensitivity analysis of progression-free survival with and without switch patients
Our study also examined the proportion of patients in each treatment who required LA-SSA dose escalation to a higher monthly dose and/or shorter dosing interval than the recommended anti-proliferative dose as well as the proportion who required rescue short acting octreotide due to symptoms of hormone excess. We found that numerically, octreotide LAR had more dose escalation and short acting octreotide use than the lanreotide, however these results were not statistically significant (Figure 4C and 4D). A recent study by Cheung et al found similar findings regarding less short acting octreotide use in patients receiving lanreotide which was greatest in the first month of treatment.
19
Interestingly, this study found that the likelihood of patients receiving an SSA dosage above the maximum recommended dose was higher for patients receiving LAN than OCT. The improved ability of lanreotide to control hormonal secretion may be because it can be absorbed in muscle or subcutaneous fat, while OCT is meant to be injected in muscle, but missed injections into subcutaneous tissue are not readily absorbed. A prior study showed that only around half of OCT intramuscular injections were administered to the appropriate location at a high-volume cancer center.
20
These missed injections may result in both worse hormonal control and may also explain some of the differences in anti-proliferative effect that we saw. Characterization of (A) patients undergoing a switch in somatostatin analogue type with (B) reason for switch and comparison of (C) need for dose escalation in somatostatin analogue or (D) use of short acting octreotide to help with carcinoid symptoms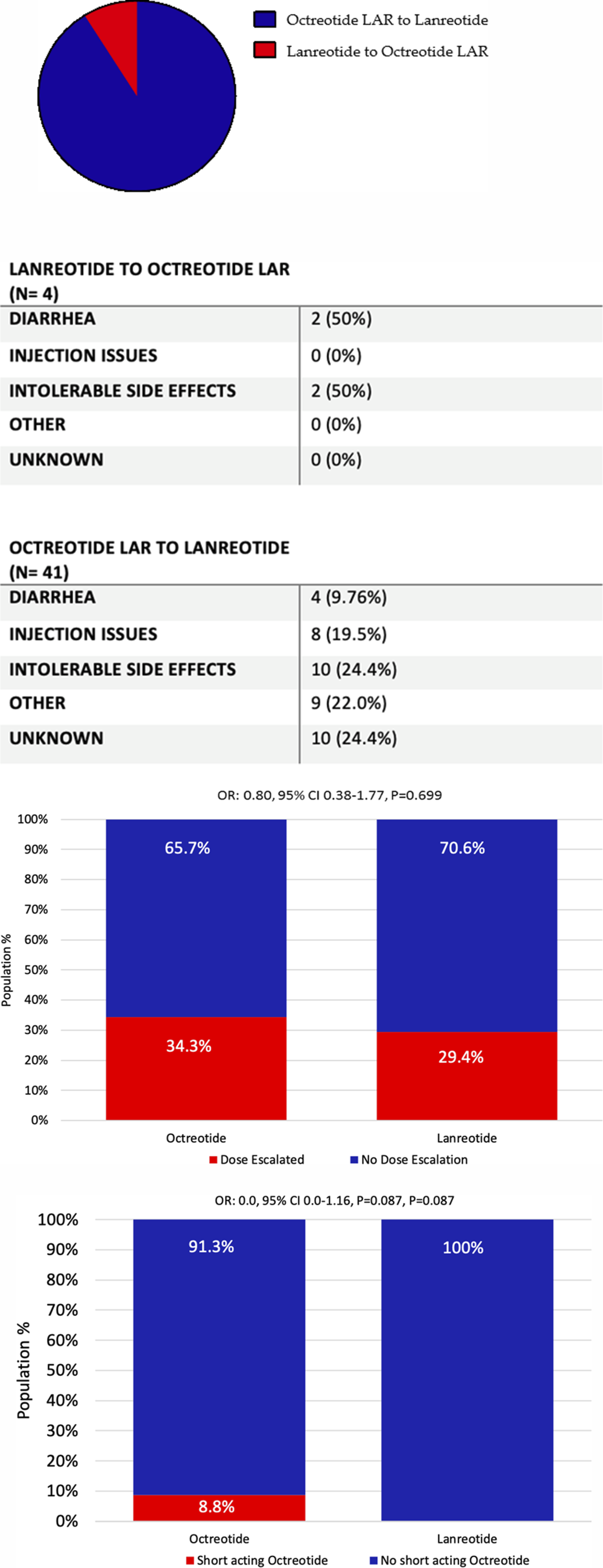
Data was also collected and analyzed on the proportion of patients who switched from one LA-SSA to another (Figure 4A). In total, 45 patients switched LA-SSA, with 41 patients switching from OCT and only 4 switching from OCT to LAN (P = 0.63). Charts were reviewed to determine the reasons for switching with diarrhea, painful injections and side effects as the most common rationale seen for change in long-acting somatostatin analogue (Figure 4B). When looking at the reason for switch being diarrhea, we further gathered data to see if these patients were prescribed pancreatic enzymes suggesting that the diarrhea was potentially felt to be secondary to pancreatic insufficiency from the somatostatin analogue rather than poor hormonal control. Of note, none of these patients were documented as having been prescribed pancreatic enzymes at the time of their switch to another long-acting somatostatin analog. Interestingly, a recent publication of a retrospective cohort by Prinzi et al showed that switching from one LA-SSA to another after progression of disease in patients with advanced well-differentiated gastropancreatic neuroendocrine tumors improved both PFS and OS. 21 It should be noted, however, that in the study by Prinzi et al, the two LA-SSA were being compared after patients had received PRRT, unlike in our study. 21 Despite this, this suggests that the act of switching from one LA-SSA to another may have greater utility than previously anticipated and warrants further investigation, alternatively this may also be explained if LAN is more efficacious than OCT. Interestingly, a recent study by Allaw MB et al, showed that there was no difference in the PFS between patients treated with octreotide LAR and lanreotide for patients with well-differentiated metastatic gastroenteropancreatic neuroendocrine tumors. 14 This may be due to the smaller cohort size of 105 lacking statistical power, for example for patients with G1 disease, they reported median PFS for the octreotide LAR and Lanreotide was 8.4 vs 32.4 months, respectively, yet the difference was not statistically significant (P = 0.159). This further highlights the need for investigation in the form of prospective studies.
Our current study has several limitations. As this is a retrospective study, our results were susceptible to both selection and recall bias. Additionally, pancreatic enzyme replacement is commonly used for patients receiving SSAs, however in BC, would be a prescription which would be filled at pharmacies outside the BC Cancer pharmacy system, and as such data capture would be incomplete and would require the clinicians to have explicitly stated they are starting this agent, given that we don’t have full data capture of the enzymes, it could not be included in this article. Furthermore, imaging was not reviewed and therefore PFS and OS were defined according to historical clinical notes and radiological reports. In our study there were also fewer patients who had received treatment with LAN than OCT. This could be secondary to the fact that marketing authorization for LAN in Canada was received in 2007 but had been received for OCT in 1999 and lanreotide only received reimbursement in BC in 2014. Hence, work is needed to further validate these results, however as discussed above, we did perform a subgroup analysis to attempt to control for this difference in the numbers of patients in each treatment group.
Conclusion
There are few real-world studies comparing LAN and OCT and our study included a significant time period resulting in a sizable sample of patients. Also, we have been able to collect information on the reasons that patients have been switched from one LA-SSA to another, which is an important event that occurs in the real world and warrants further characterization.
In summary, greater disease control was observed for LAN vs OCT as well as less long-acting dose escalation and short-acting octreotide use; however, these findings may be partially explained by a later date of reimbursement approval for LAN which could result in selection bias, whereby the populations receiving each treatment differs. Prospective comparative studies are warranted to further investigate if pharmacokinetic or pharmacodynamics differences are resulting in differing efficacy.
Footnotes
Ethical Considerations
The BC Cancer Research Ethics Board (H18-01396) approved this retrospective study. The need for informed consent was waived by the BC Cancer Research Ethics Board (H18-01396).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Ipsen who provided a research grant which paid for pathology review and by BC Cancer Foundation which provided other research support.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Loree has received research funding from Amgen, Ipsen, AstraZeneca and Foundation Medicine and consulting fees from Saga Diagnostic, Amgen, Eisai, Novartis, Ipsen, AAA, Pfizer and Bayer.
Data Availability Statement
This study was a retrospective chart review so patient level data is not available in this manuscript but can be requested for future collaborations by contacting the corresponding author.
