Abstract
Introduction
Cisplatin is particularly recognized for its adverse effects, most notably its impact on chemosensory perception.
Methods
This cross-sectional study enrolled 98 cancer patients undergoing cisplatin therapy from April 2022 to December 2023. Patients were administered the Chemotherapy-Induced Taste Alteration Scale (CITAS) and the Rhodes Index of Nausea, Vomiting, and Retching (RINVR).
Results
A multiple linear regression model was employed to determine the effect of taste changes among cancer patients undergoing cisplatin therapy. The study of the model coefficients for the structural model (χ2/df = 1.338, P = .004, GFI = 0.955, CFI = 0.986, RMSEA = 0.063, SRMR = 0.079) demonstrated a robust match. This model, formed to evaluate the effects of taste alteration, appetite loss, nausea, vomiting, age, quality of life, and body surface area, was statistically significant and explained 76.4% of the variation. Multiple linear regression evaluations determined significant factors affecting taste alteration associated with nausea-vomiting [(95% CI: 0.111-0.244); P = 0.042], loss of appetite [(95% CI: 0.102-0.254); P = 0.000], age [(95% CI: −0.827-0.424); P = 0.001], quality of life [(95% CI: −0.908-0.489); P = 0.000], and body surface area [(95% CI: 0.024-0.098); P = 0.021].
Conclusion
Taste alteration significantly influenced symptoms of appetite loss, nausea, vomiting, age, and body surface area. These results provide valuable insights for developing effective intervention strategies to alleviate the relational effects of symptom clusters on patients’ quality of life and symptoms, particularly for cancer patients receiving cisplatin therapy.
Introduction
Many organs, including the testis, stomach, ovary, and cervix, use Cisplatin as a drug to manage solid tumors. Inducing apoptosis in the cells of these organs through DNA alkylation provides anticancer effects. Cisplatin is an organic platinum derivative that structurally differs from other antineoplastic agents. It cross-links the double-stranded DNA of cells. 1 At the genomic level, DNA represents the principal target of cisplatin, and the creation of cisplatin complexes affects various DNA-dependent cellular activities, including the suppression of replication and transcription, cell cycle arrest, and DNA damage, ultimately resulting in cell death and apoptosis. Cisplatin, although a frequently used chemotherapeutic drug, is associated with several adverse effects. The most notable of these adverse consequences are alterations in chemosensory perception. 2 Chemosensory adjustments encompass reduced taste sensitivity, total loss of taste, distortion of normal taste, and taste perception without an external stimulation. Taste disturbances start within 1-3 weeks after chemotherapy and can continue throughout treatment. It is estimated that more than 75% of patients receiving chemotherapy reported that their food was too sweet, sour, salty, bitter, or tasteless, or even tasted like cardboard, metal, or sandpaper. Additionally, numerous clinical issues such as dysphagia and nausea are frequently noted. 3 Post-chemotherapy, alterations in chemosensors significantly affect patients’ nutritional status and quality of life throughout the illness. 4 The reduction and alterations in taste sensitivity correlate positively with decreased appetite, resulting in insufficient energy and nutrient consumption, weight loss, and ultimately, compromised nutritional status.5,6
Taste alteration following chemotherapy is recognized as an essential matter, prompting more research into its impact on those receiving treatment and their eating habits. Taste alterations among cancer patients might affect their eating habits and appetite, resulting in reduced calorie intake and potential deficits in vital vitamins and minerals. 7 Numerous studies highlight the relevance of taste variation, conceivably as a side effect in patients with cancer, leading to deficits in macronutrients and micronutrients.8,9
In Turkey, although several studies have examined chemotherapy-induced nausea, vomiting, and changes in appetite, which often lead to inadequate energy and nutrient intake, weight loss, diminished nutritional status, and alterations in taste during treatment, none have evaluated these factors simultaneously. Moreover, there are no studies specifically addressing taste abnormalities in cancer patients undergoing cisplatin therapy. While prior research has assessed the impact of chemotherapy side effects on patients’ overall quality of life, it is particularly important to focus on specific symptoms such as taste changes, anorexia, and gastrointestinal discomfort in patients treated with cisplatin. Looking at how chemotherapy drugs affect taste changes, appetite, and nausea and vomiting, and giving specific advice, could greatly help both research and medical practice.
This study aims to address the following research questions: 1. What are the effects of chemotherapy-induced taste changes, nausea, vomiting, and anorexia in cancer patients receiving cisplatin treatment? 2. What are the significant predictors of these symptoms when analyzed together using a logistic regression model? 3. What are the significant predictors of these symptoms when analyzed together using a linear regression model?
Methodology
This study is a cross-sectional descriptive study.
Participants
The participants consisted of cancer patients receiving cisplatin treatment at least once between April 1, 2022, and December 31, 2023, in the Oncology Training and Research Hospital. Prior to the study, approval had been obtained from the Ethical Committee of Dr Abdurrahman Yurtaslan Ankara Oncology Education and Research Hospital (Ethical Committee Protocol Approval No: 2022-03/60). The reporting of this study conforms to STROBE guidelines. 10
The study sample comprised patients aged 18 to 65 who were literate, communicative, and cooperative. Participants were told about their diagnosis, had received cisplatin treatment at least once, did not have any stomach, brain, or metabolic issues, were not pregnant or diabetic, had not used non-drug methods for taste changes and nausea or vomiting, had no history of smoking or drinking, and agreed to take part in the study willingly. Patients whose treatment regimen or medication was changed or terminated were excluded from the study. The size of the collected data for the analysis of multiple regressions was around 5-10 times the number of independent variables. The study comprised 9 independent factors, such as age, gender, level of education, and income. In this study, an anticipated follow-up loss rate of 20% was considered acceptable due to expected participant refusals and errors in completing the questionnaire. Accordingly, an initial sample size range of 50 to 95 was proposed, based on preliminary evaluations accounting for various scenarios involving differences in variance and effect size to maintain adequate statistical power.
Sample size estimation was conducted using the G*Power software (version 3.1.9.6). Based on data obtained from a pilot study involving 5 participants, a power analysis was performed using simple linear regression. Assuming a moderate effect size (f2 = 0.15), a Type I error rate of 0.05, and a statistical power of 95%, the minimum required sample size was calculated to be 80.
This calculated sample size is also deemed sufficient for more complex analyses such as multiple regression and structural equation modeling, as similar assumptions regarding effect size and statistical power apply. Taking into account the anticipated 20% loss rate, the final target sample size was set at 98, and data collection was carried out accordingly.
Data Collection
The nausea-vomiting and taste alteration scales had items that pertain to broader experiences rather than specific time intervals. The nausea and vomiting scale evaluated anticipatory symptoms (ie, occurring before treatment) and acute symptoms after chemotherapy delivery. The taste alteration scale pertains to alterations experienced during the chemotherapy cycle, rather than at a particular instance. Therefore, in the current investigation, data have been collected from cancer patients who had undergone a minimum of 1 cycle of cisplatin-based chemotherapy during their treatment regimen. When patients who received at least one course of cisplatin treatment came to the clinic again to receive medication, data were collected about the symptoms they felt. Before collecting information, subjects were acquainted with the study and provided informed consent. Data were collected from the Patient Information Form, EORTC QLQ-C30 Quality of Life Index, Chemotherapy-Induced Taste Alteration Scale (CITAS), and the Rhodes Index of Nausea, Vomiting, and Retching (RINVR), which were given to all participants in the study.
The EORTC QLQ-C30 Quality-of-Life Index was developed to assess a range of cancer-specific quality-of-life issues relevant to a wide sample of cancer patients. This questionnaire consisted of 30 cancer-specific questions employing multiple-point scales, including a global health status/QoL scale, 5 functional scales, and 9 symptom scales. Multiple-point scales were transformed into standardized scores spanning from 0 to 100. Increased scores on global health status/QoL and functional scales indicated a good quality of life, but heightened scores on the symptom scales reflected more severe symptoms. 11
The Chemotherapy Induced Taste Alteration Scale (CITAS) is a five-point Likert scale developed to evaluate individual taste alterations. CITAS is a measurement system of 4 dimensions classified under 3 subcategories. The first sub-heading discusses flavor changes, the second sub-heading concerns undesirable taste adjustments, and the concluding sub-heading encompasses serious grievances or issues. The rise in scores on the scale signifies the extent of the individual’s change in taste and the associated discomfort arising from this alteration. 12
Genç assessed the validity and reliability of the Rhodes Index of Nausea-Vomiting-Retching (RINVR) in a 2010 study in Turkey. The scale evaluates nausea, vomiting, and retching following chemotherapy. The incidence of nausea, vomiting, and retching in patients is assessed using a Likert-type scale. This scale consists of 8 questions: “0” indicates negligible distress, whereas “4” represents extreme distress. It includes 3 subdimensions: symptom experience, symptom occurrence, and symptom distress. 13
Data Analysis
Data collected from patients were analyzed using the IBM Statistical Package for the Social Sciences (SPSS) for macOS, version 29.0 (IBM Corp., Armonk, NY, USA). Normality of data distribution was assessed using skewness and kurtosis values, with values between − 1.0 and + 1.0 considered indicative of normal distribution. Additionally, the Shapiro-Wilk test was applied; a P-value > .05 was interpreted as evidence of normality. Descriptive statistics, including means and standard deviations, were calculated for continuous variables such as age, while categorical variables (eg, gender, education level) were presented as frequencies and percentages.
Pearson correlation analysis was conducted to explore relationships among continuous variables and scale scores. One-way ANOVA and multinomial logistic regression analyses were used to examine group differences and associations with the Chemotherapy-Induced Taste Alteration Scale (CITAS) and the Rhodes Index of Nausea, Vomiting, and Retching (RINVR). Variables that exhibited multicollinearity were excluded from regression models. Subsequently, backward stepwise linear regression analysis was performed to identify the most parsimonious set of predictors. Multiple linear regression was used to examine the influencing factors of nausea and vomiting.
For the structural model analysis, Mplus version 7.4 was used. Model fit was evaluated based on several indices: χ2/df = 1.338, P = .004, GFI = 0.955, CFI = 0.986, RMSEA = 0.063, and SRMR = 0.079, indicating an acceptable to good model fit. A P-value of <0.05 was considered statistically significant for all analyses.
Results
Demographic Characteristics of Patients
Distribution of Demographic Characteristics of Patients (n = 98)

SD = Standard Deviation.
Correlation Between the Sub-Dimensions of the Taste Scale and Demographic Characteristics of Patients
Correlation Between the Sub-dimensions of the Taste Scale and Demographic Characteristics of Patients

aSD = Standard Deviation; r: Pearson correlation test t: independent sample t test F: One Way Anova test *:P < 0,05.
Correlation With Taste Scale and Nausea-Vomiting-Retching
Correlation Between Taste Scale Subscales and Nausea-Vomiting-Retching Subscales
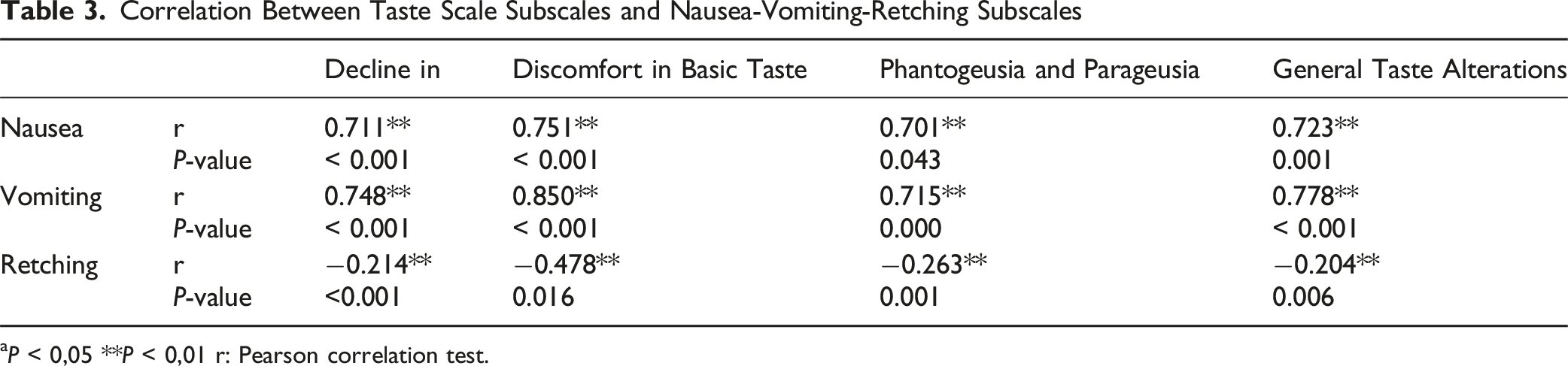
aP < 0,05 **P < 0,01 r: Pearson correlation test.
Multiple linear regression of taste alteration risk factors
In the process of regression analysis, following the elimination of factors responsible for multicollinearity, all variables were subsequently reintroduced into the model to derive the most appropriate model for path analysis (Figure 1). Only the most statistically significant predictors were retained in the final model. In this regression analysis, the dependent variable is the one that the model aims to explain or whose variation is being studied. The dependent variable is “taste alteration”, as the effects of other variables on taste alteration were analyzed. Analysis of the model coefficients for fit for the structural model indicated that the model had a strong correspond, as stated in Table 4. The regression model developed to assess the impact of taste alteration, loss of appetite, nausea, vomiting, age, general state of life, and body surface area was statistically significant and accounted for 76.4% of the variance. Analysis of Table 4 indicates that a 1-unit variation in taste alteration resulted in a 0.411-unit change in nausea-vomiting, a 0.651-unit change in loss of appetite, and a 0.323-unit change in body surface area. They found that each unit increase in taste alteration corresponds to a decrease in quality of life (0.464-unit change) and age (0.374-unit change). Path Model of Multiple Indicators on the RINVR, CITAS, and QLQ-C30. Abbreviation: RINVR = Rhodes Index of Nausea, Vomiting, and Retching; CITAS = Chemotherapy Induced Taste Alteration Scale; QLQ-C30 = EORTC QLQ-C30 Life Quality. Index Note: Model fitness index: x2 /sd=1,338 p=0,004 GFI=0,955 CFI=0,986 RMSEA=0,063 SRMR=0,079 *P < 0.05, **P < 0.01, ***P < 0.001 BSA Multiple Linear Regression Analysis of Risk Factors Affecting Taste Alterations Dependent Variable: R2: 0.764; P < 0.001

χ 2 /df = 1.338,
P = 0.004,
Goodness of Fit Index (GFI) = 0.955,
Comparative Fit Index (CFI) = 0.986,
Root Mean Square Error of Approximation (RMSEA) = 0.063, Standardized Root Mean Square Residual (SRMR) = 0.079.
These values indicate a high level of model fit, meaning that the proposed model corresponds well with the observed data. Specifically, CFI and GFI values above 0.95, along with RMSEA and SRMR values within acceptable thresholds, reflect that the model structure is statistically robust and conceptually sound.
Discussion
This study is the first to model the relationships between taste alterations, chemotherapy-induced nausea and vomiting, and appetite using path analysis in cancer patients who have undergone at least 1 cycle of chemotherapy. Although various correlations among these symptoms have been acknowledged, the specific methodological approach and the demographic combination of the analyzed sample provide an original update to the literature. Targeting nausea may indirectly improve taste alterations and reduce appetite loss. Chemotherapy-induced taste changes are directly linked to nausea and vomiting resulting from the treatment. In this context, addressing nausea could alleviate the effects of taste changes, thereby contributing to the prevention of appetite loss. Furthermore, directly addressing taste alterations can enhance quality of life. Specifically, loss of taste and changes in taste perception can negatively affect patients’ eating habits, potentially leading to nutritional deficiencies. Direct interventions aimed at these changes (eg, treatments to ease taste alterations or nutritional support) could improve patients’ overall health and enhance their quality of life. In particular, symptoms such as the metallic taste induced by platinum-based therapies can be managed through specific dietary and treatment interventions, making the treatment process more tolerable.
Chemotherapy can increase sensitivity or alter taste, leading to intensified metallic, bitter, or sour flavors that heighten the sensation of nausea or vomiting. Chemotherapy-induced changes in taste were correlated with the patients’ sociodemographic variables. Our study found that age, body surface area, gender, and educational status correlated with a degradation in basic taste, discomfort in basic taste, phantogeusia, and parageusia, as well as overall taste change. No investigation was reported in the literature reviews that assessed a combined association between taste alterations, nausea, vomiting, and appetite loss concerning aging; existing research focused on correlations for only 1 particular symptom with age. A study evaluated the correlations between nutritional status and various age groups. It was utilized with a modified model accounting for genders, educational attainment, and BMI. In the 65-74 age group, nutritional status was not substantially associated with it; however, in the 75 + age group, at-risk dietary status was significantly correlated. 14 Considering advancing age, gastrointestinal function declines and hunger diminishes, frequently due to sensory alterations, which may result in insufficient nutrient intake and an increased risk of malnutrition. 15 Considering that taste change is related to saliva amount and odor variations in how taste is perceived are associated with saliva production and olfactory stimuli, revealing discrepancies in findings. 16 Patients with testicular cancer aged 18 to 60 years had 2.47 cycles ± 0.5 of cisplatin treatment for odor identification and discrimination. The Sniffin’ Sticks test showed no significant alterations, although the odor threshold score drastically declined during the treatment cycle. 17 Subsequent studies revealed no substantial disparity in psychophysically assessed olfactory threshold scores between cancer patients undergoing cisplatin-based chemotherapy regimens. 18 In a study with 576 participants, the overall prevalence of undernutrition was 24.5%, and being 80 years or older was significantly associated with undernutrition. 19 The regression model used in our study for evaluating the impact of taste alteration, appetite loss, nausea, vomiting, age, quality of life, and body surface area was statistically significant and accounted for 76.4% of the variance to be analyzed.
The number of chemotherapy cycles is a critical risk factor for taste change severity. The stage of chemotherapy could further add complexity to the reported alterations. Prior research has demonstrated a gradual escalation in the intensity of taste impairment correlating with prolonged exposure to chemotherapeutic drugs. 20 A study assessing risk factors for taste change after chemotherapy revealed that a BMI of up to 1.5 m2 was inversely correlated with dysgeusia, 13 in the same study stated that the impact of tumor-specific chemotherapy protocols on the incidence of dysgeusia cisplatin, the most common chemotherapy regimen for head and neck neoplasms, has been directly linked to grade 1 and 2 dysgeusia. Furthermore, when pharmacological treatment was modified for head and neck radiotherapy, only cisplatin was directly linked to grade 2 dysgeusia. 13 Various guides identify platinum-based treatments as a primary cause of metallic taste as an adverse reaction. 21 Metallic taste is reported by 9.7% to 78% of chemotherapy-treated cancer patients. 22 In the other study, a statistically significant relationship was identified between chemotherapy type and the incidence of taste alteration. Antimetabolite therapy combined with platinum derivatives was linked to an increased incidence of taste change (77%). The latest research using a Gustometer test to analyze taste behavior, transcriptome, and apoptosis was carried out to examine cisplatin-induced taste alteration. This study showed that Cisplatin suppressed proliferation and enhanced apoptosis in the circumvallate papilla, resulting in considerable impairment of taste function and receptor cell production. The transcriptional profile of genes related to the cell cycle, metabolic processes, and inflammatory responses was markedly altered after cisplatin treatment.22,23 The multivariate logistic regression analysis in our study indicated that taste alteration, nausea-vomiting, and loss of appetite were more prevalent in patients with a BMI of 1.76 ± 0.15. Our study findings coincide with prior research, indicating that symptom intensity intensifies with higher cumulative dosage.
Oltong et al. demonstrated in a prospective cohort research that alterations in taste are cyclical and intermittent, peaking at the initial part of the third chemotherapy cycle 24 and reverting to baseline taste capacity 8 weeks post-final cycle. 25 In our study, the multiple linear regression model results show that the mean values for taste alterations, nausea, vomiting, and loss of appetite reach 3.3 ± 0.8 (minimum: 2, maximum: 4) at most throughout the third cycle, consistent with the literature.
When the literature related to education level and gender is examined, female sex was a major risk factor linked to an investigation’s twofold increase in the prevalence of taste change. 13 Women exhibit greater susceptibility to neurosensory issues involving the head and neck, and the existence of sex hormone receptors in the cranial nerve V complex partially clarifies these disparities, as these receptors contribute to the hypoactivity of the ganglionic inhibitory system. 26 A study of 89 cancer patients undergoing chemotherapy, found that 32% had moderate alterations, 20% had severe alterations, and dysgeusia was more frequent in women. 27 Breast cancer patients are associated with the highest rate of taste alterations compared to other treatments. 28 As in another study 29 dysgeusia was more frequent in women than men. While in the univariate analysis, fewer years of education were associated with taste changes, 30 none of the demographic characteristics remained significant in the other multivariable study model. 25 Our study performed multivariate logistic regression, revealing that education diploma influences taste alterations, nausea, vomiting, and anorexia. The 2 clinical characteristics associated with taste alterations in the multivariable model were chemotherapy-induced nausea and vomiting and appetite.
A study evaluating the significance of taste alterations and nausea and vomiting, which activate 5-HT1b, 5-HT2b, and 5-HT2c receptors, has an impact on suppressing eating behaviors.31,32 The injection of particular agonists for the 5-HT1 b, 5-HT2 b, and 5-HT2c receptors leads to substantial reductions in suffering in foundational research studies involving normal rats. The genes for the 5-HT1 b and 5-HT2c receptors, which modulate the brain and spinal cord, are elevated during fasting and diminished post-feeding. 33 Post-cisplatin administration, concentrations of 5-HT and its metabolite 5-hydroxyindole-acetic acid have been reported to increase in the hypothalamus, hippocampus, and medulla oblongata. 34 The activation of the 5-HT3 receptor is essential for the onset of vomiting and nausea following cisplatin administration. Nausea and emesis arise from the stimulation of the vomiting zone located in the nucleus of the solitary tract within the medulla oblongata. This stimulation originates from the activation of the vagus nerve or the chemoreceptor trigger zone in the medulla oblongata, induced by the stimulation of the 5-HT3 receptor. 34 Our study addressed these taste alterations, nausea-vomiting, and loss of appetite symptoms using linear regression and conducted path analysis, presenting the path analysis of the model constructed following these findings.
Conclusion
This study evaluated the effects of cisplatin on taste changes. Our findings have significant implications for developing alternative methods to restore taste function in individuals undergoing chemotherapy. The correlation among nausea, vomiting, taste issues, and appetite loss highlights the need for a comprehensive strategy to manage chemotherapy side effects. Addressing one symptom may help alleviate others, thereby enhancing the patient’s overall quality of life. Interventions aimed at reducing nausea could also lessen taste alterations and improve appetite, providing a holistic approach to symptom management. The study emphasizes that nausea, vomiting, taste changes, and appetite affect cancer patients receiving cisplatin therapy in profound ways. Consistently addressing these symptoms could enhance treatment outcomes and quality of life.
Limitation
This study indicates that taste alteration affects appetite loss; nonetheless, numerous methodological decisions demand criticism. This constraint of our study pertains to the subjectivity of the assessment approach. Our assessment of taste ability is based on patients’ self-reports, thereby indicating a subjective perception of change rather than actual objective chemosensory dysfunction. The study’s other limitations encompassed the absence of data regarding smoking and alcohol intake, as all patients reported neither smoking nor drinking alcohol. This hindered the assessment of any correlation between these 2 parameters and the development of taste changes. Future study efforts will be directed to overcome such limitations. Additionally, this study was conducted in a single facility in Turkey, which may introduce racial bias and limit generalizability. The path analysis suggests a theoretical direction, but it is not possible to definitively establish causality. This study, therefore, cannot confirm cause-and-effect relationships between taste alterations, nausea, vomiting, and appetite loss. Additional longitudinal studies are needed to clarify the relationships between variables and enhance our understanding of how these symptoms change over time.
Footnotes
Acknowledgments
The authors thank all the patients who participated in the study. Many thanks are also due to sci-editing services for English language editing and the statistics center for statistical analysis.
Ethical Approval
This research was carried out according to the ethical standards outlined in the Declaration of Helsinki. Prior to the study, approval had been obtained from the Ethical Committee of Dr Abdurrahman Yurtaslan Ankara Oncology Education and Research Hospital, and written consent was obtained by the relevant department administration (Ethical Committee Protocol Approval No: 2022-03/60). The research data were assured complete confidentiality. All participants in the study were informed of its objectives and importance and were enrolled after providing both written and verbal consent. The patients were informed that their participation was voluntary and that they might withdraw from the study at any time. Participants who declined to participate were excluded from the study.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
