Abstract
Introduction
Quitting smoking is challenging even with existing pharmacotherapy. Thus, discovery of new cessation medications is imperative. Memantine, a well-tolerated Alzheimer’s disease drug, partially antagonizes glutamate at the N-methyl-D-Aspartate receptor (NMDAR), modulating dopamine release in addiction pathways. Memantine may interrupt nicotine reward and promote smoking cessation.
Materials and Methods
At 23 community oncology practices nationwide, we recruited 130 breast, prostate, lung, or colorectal cancer survivors ≥ six months beyond definitive treatment who currently smoked at least 10 cigarettes daily and wanted to quit. In a double-blind fashion, participants were randomized to take either memantine (10 mg) or a matching placebo twice daily for 12 weeks (65 per arm). Toxicity, nicotine dependence, and past-week abstinence were recorded at 2, 4-, 6-, 9-, and 12-weeks post-randomization. The primary endpoint was feasibility and preliminary estimation of 12-week self-reported past-week smoking abstinence.
Results
There were no significant differences in abstinence rates or nicotine dependence between the two groups at 12 weeks. Twelve-week completion of therapy was low, but lower in memantine than control participants (42% vs 63%, respectively; P = .01). Memantine participants reported trends of less anxiety, craving, and hunger. No significant differences in toxicity were observed between groups. Serious adverse events (3 in memantine arm, 1 in control arm) occurred; none considered possibly or probably related to study medication.
Conclusion
Memantine did not improve 12-week smoking abstinence rates in cancer survivors. While other NMDAR antagonists might deserve evaluation, this study suggests memantine is not efficacious for smoking cessation in a cancer survivor subpopulation.
Trial registration number
NCT01535040 - February 17, 2012.
Plain Language Summary
Quitting smoking is hard, even with current medicines, so finding new ones is crucial. Memantine, a drug used for Alzheimer’s, affects the brain chemical glutamate, which is involved in addiction. It might help people stop smoking by changing how the brain reacts to nicotine. In this study, 130 cancer survivors who smoked at least 10 cigarettes a day wanted to quit. They were split into two groups: one took memantine pills, the other took placebo pills (which have no medicine). Neither the participants nor the doctors knew who got which pills, making it a fair test. Researchers checked in with them over 12 weeks to see if they were smoking less. They found that memantine didn’t help more people quit than the placebo. Also, fewer people in the memantine group finished the whole 12 weeks of pills compared to the placebo group. Some people in both groups had side effects, but they weren’t serious. In conclusion, memantine didn’t increase the number of people who quit smoking among cancer survivors in this study. Drugs like memantine should still be studied. But this research shows memantine might not be the best choice to help cancer survivors quit smoking.
Introduction
Smoking is the leading cause of United States (US) preventable mortality and a major contributor to many chronic diseases. 1 Yet even in motivated smokers, quitting is difficult. In patients with cancer, for example, a high proportion continue to smoke after their diagnosis, with highest rates among those with tobacco-related malignancies.2,3 For example, up to 57% 4 (or more 3 ) of lung cancer patients are current smokers. Oncology patients who stop smoking have improved outcomes,3,5 yet often exhibit higher levels of nicotine dependence, craving and relapse than patients without cancer. 5 Even with intensive counseling and pharmacotherapy, many oncology patients who smoke struggle to quit.5,6 High rates of smoking are found among cancer patients at diagnosis. For example, among patients with head and neck cancer, 45-75% smoked a year prior to diagnosis. While smoking cessation rates after a cancer diagnosis range from 42-86%, there is a high rate or relapse with 41% smoking 4 months after diagnosis. Even after treatment, 20% of these patients remain smoking. 7 Smoking cessation pharmacotherapies available to cancer patients and survivors are the same as for non-cancer patients—e.g. varenicline, bupropion and nicotine replacement therapy (NRT). However, these medications often do not work well as the sole intervention. In a recent meta-analysis of 19 RCT and 20 observational studies 6 evaluating smoking cessation interventions among cancer patients, behavioral interventions alone (OR 1.33, 95% C.I.: 1.02-1.74, P = .03) and interventions combining behavioral therapy with pharmacotherapy (OR 1.67, 95% C.I.: 1.24-2.26, P = .0008) significantly increased smoking abstinence whereas pharmacotherapy alone did not (OR 1.11; 95% C.I.: 0.69-1.78, P = .66). Therefore, the development of more stop-smoking medications for cancer patients is highly desirable, particularly those that would work without extensive behavioral counseling. This is especially true since confidence in smoking cessation counseling by oncology providers varies widely. 8
When nicotine is administered, it attaches to nicotinic acetylcholine receptors (NAchR) throughout the brain’s reward pathways of the mesolimbic system. In the ventral tegmental area (VTA), nicotine’s activation of NAchRs α4β2 and α7 leads, respectively, to the release of dopamine into the nucleus accumbens and glutamate into the VTA, The release of glutamate into the VTA allows activation of glutamatergic receptors on dopamine neurons further increasing the release of dopamine into the nucleus accumbens. 9 Thus, both nicotinic and glutamatergic transmission increase the release of dopamine into the nucleus accumbens, which is the final common pathway of drug addiction. 10
Glutamate is the major excitatory amino acid in the brain. It regulates both normal physiologic functions (e.g., learning and motivation); as well as pathophysiologic processes (e.g., pain, addiction and excitotoxicity in Alzheimer’s and Parkinson Diseases).9,11–14 The N-methyl-D-Aspartate receptor (NMDAR), when activated, releases glutamate. Among other actions, glutamate release effectuates downstream transmission of dopamine in the brain’s reward pathways. The surface of the NMDAR also houses the excitatory nicotinic acetylcholine receptors α7 NAchR and α4β2 NAchR. During smoking, nicotine activates these two NAchR, then the NMDAR itself activates, releasing glutamate and dopamine throughout the reward pathway. This nicotine-induced glutamatergic transmission points to new stop-smoking medications. 13 Indeed, in laboratory animals, NMDAR antagonism by the drug memantine reduces VTA dopaminergic transmission and decreases systemic nicotine self-administration.13–16
Memantine is a well-tolerated noncompetitive low-affinity, selective open-channel partial antagonist of the NMDAR used in Alzheimer’s Disease which attenuates excessive but not normal neuronal glutamatergic excitation. 9 Memantine is unlike the successful smoking cessation drug, varenicline, which is a partial agonist at the α4β2 nicotinic receptor. First while varenicline is a partial agonist at that receptor, memantine acts as a full antagonist at the α4β2 NAchR,17–22 the receptor so crucial to nicotine addiction.22–24 Furthermore, unlike memantine, varenicline has no agonist, partial agonist or agonists/antagonist activity at the NMDAR. Memantine is unlike the successful smoking cessation drug, varenicline, which benefits from being a partial agonist at the α4β2 nicotinic receptor. First, it does not act at that nicotinic receptor. Second, it has no agonist, partial agonist or agonists/antagonist activity at the NMDAR. Since the off-rate of memantine on the NMDAR is rapid, memantine does not accumulate in the channel pore, minimizing effects on normal synaptic transmission. Nonetheless, memantine preferentially blocks “excessively activated” NMDAR because those receptors are “excessively open”—hence the nomenclature of “open channel blocker.” 25 Agonist effects of glutamate on NMDAR synaptic transmission increases dopamine receptor activity in both the striatum and hippocampus26,27; as well as in the VTA and nucleus accumbens. 9 By contrast, memantine’s antagonism of the NMDAR decreases dopamine receptor transmission, a process that reduces rat self-administration of nicotine.15,16 Blockade of glutamatergic transmission also inhibits nicotine reward.12,13 Thus, memantine—by blocking glutamate transmission via the NMDAR and its downstream dopaminergic effects on nicotine reward and self-administration could theoretically reduce the dopaminergic rewarding effects of nicotine, making it useful in smoking cessation.13,15,16
To our knowledge, only three studies have examined memantine in human smoking.12,28–30 In two of those, memantine reversed pleasant subjective effects of smoking at daily doses of 40 mg12 and 20 mg. 29 In the third study, researchers hypothesized that memantine would alter reconsolidation of substance use cue memories, 28 a process involving the NMDAR.31–33 This study enrolled smokers motivated to quit who were randomized in a double-blinded fashion to one of three groups: those receiving 10 mg daily of memantine with or without associative cue-smoking memory reactivation; or those receiving placebo and cue-smoking memory reactivation. Results from this trial showed possible worsening of cessation outcomes with memantine: Participants on active drug relapsed to smoking in a median of 1 day; yet on placebo participants relapsed in a median of 5 days (P > .1). Since a low dose of memantine was used (half the therapeutic dose of 10 mg twice daily), it was unclear if the low dosage or if the memantine itself insufficiently sustained NMDAR blockade giving null results. 28
To expand on limited human evidence, we conducted a pilot study assessing the potential smoking cessation efficacy of memantine 10 mg twice daily among 130 cancer survivors. To our knowledge, this is the first study to test memantine for smoking cessation among cancer survivors—a group with marked difficulty in quitting smoking and remaining quit.
Materials and Methods
The goals of this randomized, placebo controlled, double-blinded pilot study were: (1) to estimate accrual, retention, adherence, and participation rates, (2) to obtain a preliminary estimate of the treatment effect (difference in abstinence rates in the two groups), (3) to estimate the variability of and treatment effect on nicotine dependence, nicotine withdrawal symptoms, and quality of life, and (4) to estimate the rates of toxicity and adverse events associated with memantine. Adherence was calculated (1) as the number of tablets taken divided by the intended number of tablets that could have been taken while on study (mean percent intended dose); and (2) as the number of tablets taken divided by the number of tablets that could be taken if the participant completed the study (mean percent maximum possible dose). The mean percent maximum possible dose calculated any missing patient diaries as missing the dose. Accrual is the number of participants randomized to the study divided by the number of months the study was open to enrollment. Retention is the proportion of patients who complete 12-week treatment period. Participation is the number of participants enrolled and randomized out of those who met eligibility criteria. The abstinence rate was defined as self-reported one-week abstinence at 12 weeks post-randomization. (A “No.” response to the question “Have you smoked in the last week?”.) 34 Biological confirmation of smoking status was not obtained. Nicotine withdrawal was assessed using the Wisconsin Smoking Withdrawal Scale, 34 a 28 item questionnaire with seven subscales, each consisting of 3-5 questions all answered on a 0-4 scale, with higher scores indicating more severe withdrawal symptoms (Supplemental Material 1). Nicotine dependence was assessed using the Fagerstrom Nicotine Dependence Scale 35 which consists of 8 questions, each of which is scored on a 0 to 1 or 0 to 2 scale. The total score ranges from 0 to 11, with higher scores representing greater dependence. For participants not abstinent of smoking in the past week, we assessed the effect of memantine on the number of cigarettes smoked. The reporting of this study conforms to the CONSORT statement. 36
Sample and Setting
Participants were recruited among survivors of breast, lung, prostate or colorectal cancers six months beyond definitive treatment at the Wake Forest Baptist Comprehensive Cancer Center and 22 community clinical oncology programs (CCOPs) affiliated with the National Cancer Institute (NCI) Wake Forest Community Oncology Research Program (NCORP). These sites exist across 20 different states and represent diverse demographic populations. 37
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the Helsinki Declaration or comparable ethical standards. The study was approved by the Wake Forest Institutional Review Board on 05/07/2012 (Protocol: IRB00020412). Informed consent was obtained from all individual participants included in the study. This trial is registered at the U.S. National Institutes of Health on ClinicalTrials.gov (Identifier: NCT01535040).
Inclusion and Exclusion Criteria
Complete inclusion and exclusion criteria are found in Supplemental Material 2. Below are summaries of the criteria.
Inclusion Criteria
Women and men who were over the age of 18; were survivors of breast, lung, prostate or colorectal cancers six months beyond definitive treatment; smoked 100 cigarettes over lifetime and at time of first interview smoked 10 or more cigarettes per day on most days over the past month; and had no period of smoking abstinence longer than 3 months in the past year were eligible for the study. All participants were screened using a combination of a telephone interview and two in-person visits. These cancers were chosen because improved longevity rates enhanced the long-term impact of smoking cessation and probable association of these cancers with cigarette smoking. 38
Exclusion Criteria
Patients were excluded if they were receiving specific concomitant medications (anticonvulsant, antiparkinsonian, hypnotic, anxiolytic, neuroleptic, or cholinomimetic agents, or any other investigational compounds). Participants who had used in the previous month or intended to use medications that might interfere with study medication evaluation (e.g., nicotine replacement, nortriptyline, clonidine, bupropion or varenicline) were also excluded. Female participants of childbearing potential were required to have a negative serum pregnancy test at baseline and agreed to use effective birth control during the treatment phase of the study and for 30 days thereafter.
Safety Considerations
A medical exam with history and physical and serum pregnancy testing was carried out at baseline. No hematological or serum lab tests were ordered during the study. Participants were excluded if they had a history of renal and/or hepatic disease; had uncontrolled hypertension > 160/90; had other significant, uncontrolled medical conditions (e.g., serious psychiatric illness, unstable angina, myocardial infarction, TIA or CVA) within the past three months; or had any medical illness which, in the opinion of the investigator, would prevent participation in the study. Participants were also excluded if they reported allergic reactions to memantine; excessive alcohol use (more than 5 drinks per day for men and 4 drinks per day for women); current use of illegal drugs; or misuse of prescription medication.
At the time of the study, memantine was well tolerated, with known side effects occurring in > 10% of patients. Less likely side effects occurred in < 10% of patients. These included, but were not limited to: dizziness, headaches, confusion, extreme tiredness, shortness of breath and elevated blood pressure. Rare but serious side effects known at the time of the study included, but were not limited to: stroke, seizures, severe skin reactions and liver or kidney failure. Participants had in-person visits at weeks 2, 4, 6, 9 and 12, at which time vital signs were taken, and they were queried about known and/or expected adverse events and unexpected adverse events. Reported toxicities were followed for one month afterward.
Study Medication
Participants received active memantine or identical placebo as follows: 10 mg of study drug per day (5 mg twice daily) days 1-3, then 20 mg per day (10 mg twice daily) thereafter. This dosing schedule was patterned after Thuerauf et al. 29 Although different from the titration schedule used in Alzheimer’s disease studies (e.g., titration from 10 mg once per day to 10 mg twice daily over 2 to 4 weeks), 39 Thuerauf et al found this regimen to be well-tolerated. 29 Memantine was purchased from Forest Laboratories; NY, NY. Drug and placebo were over-encapsulated and distributed to the study sites by Biologics Inc., Raleigh, NC.
Behavioral Intervention
All participants received brief cessation advice based on the Centers for Disease Control and Prevention Smoking Web site at each visit. 40 Information was adapted to this study’s timelines (e.g., setting quit date for day 8). Research staff were trained in the use of this website and quit information, which focuses on the five “A’s” of smoking cessation (Ask, Advise, Assess, Assist, and Arrange) and tips to help a smoker quit smoking.
Statistical Considerations
Participants were stratified by gender and assigned within strata to receive memantine or a matched placebo with equal probability using variably sized permuted block randomization; block sizes were chosen randomly to ensure that future assignments could not be inferred from previous assignments. The sample size was determined to estimate the difference in quit rates to within ± 20% with 95% confidence (assuming quit rates of 50% in both groups). The hypothesized rate of 50% was set to provide the largest possible sample size for a 20% difference. Anticipating that 25% of the participants would drop out, our total required sample size was 130 participants.
The one-week abstinence rates were measured every two weeks for the 12 weeks of the trial. Self-reported smoking cessation rate at 12 weeks was calculated as the number abstinent divided by the number evaluable. Additionally, worst-case rates assuming dropouts also did not quit smoking were reported. Exact 95% CIs were calculated for the abstinence rate in each arm and an approximate, large-sample 95% CI was calculated for the difference in abstinence rates (providing a preliminary estimate of the treatment effect). Estimates of treatment efficacy were obtained using the intent to treat approach.
Repeated measures longitudinal models were used to assess changes over time in the number of cigarettes smoked and nicotine dependence, and to assess the effect of the intervention on these changes. Unstructured covariance matrices were used in the repeated measures models to model the within-patient correlations over time, while adjusting for the sex. All statistical analyses were performed using SAS 9.3 software (SAS Institute, Cary, NC), and test results were considered significant at the two-sided 5% level.
Results
Participant Characteristics
Participant Characteristics.

*Some missing data for the smoking history variables.
**Eastern Cooperative Oncology Group (ECOG) Performance Status describes a patient’s level of functioning in terms of their ability to care for themself, daily activity, and physical ability (walking, working, etc.). 0 is fully active with no restrictions, 1 is restricted in physically strenuous activity but can do light or sedentary work.
The median age at which participants began smoking was 16 years (range: 7-38), and the median number of years smoked was 40 (range: 3-75). The mean number of cigarettes smoked per day was 34.0 (SD 32.6). Participant characteristics did not differ significantly between treatment groups.
Feasibility
One hundred thirty participants from 23 participating sites enrolled between September 24, 2012 and November 5, 2013 (13 months), corresponding to an accrual rate of 9.7 participants per month (Figure 1, Supplemental Material 3). Sites enrolled a median of 3 (range: 1-24) participants to the study. Patients who refused therapy, but continued to provide outcome data were considered off therapy but on study. Overall, the retention rate of study completion at 12 weeks was 61%, lower than the 75% anticipated; 14% dropped out in the first 2 weeks (11 memantine, 7 control). The control arm had slightly higher retention (68% vs 54%, P = .09). Twenty-seven memantine patients (42%) and 41 control patients (63%) completed 12 weeks of therapy (P = .01). CONSORT Diagram.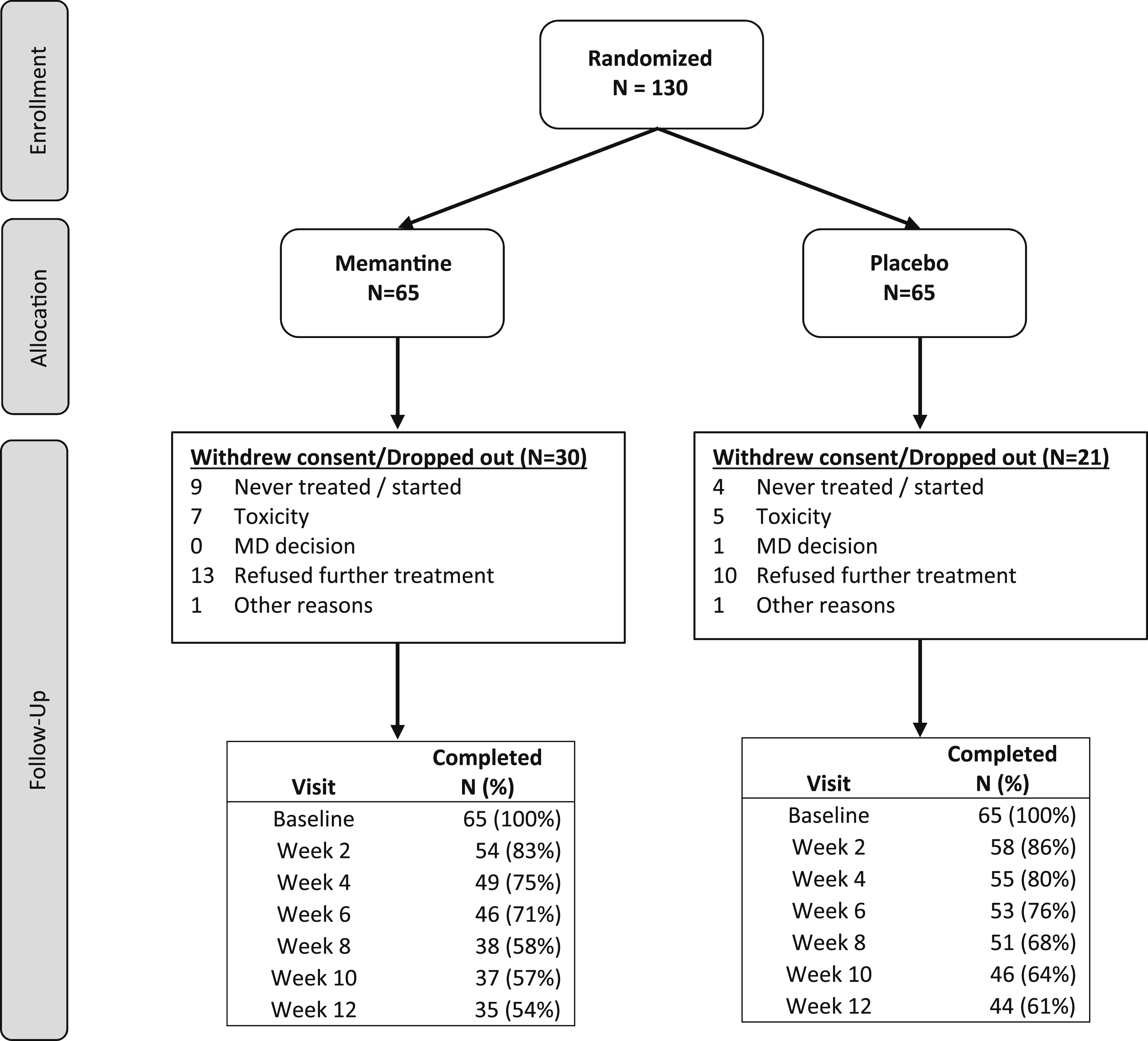
Adherence
One hundred participants (76.9%) completed all or some pill diaries. The mean compliance from percent intended dose was 86% (range 0%-100%) for Memantine and 93% (range: 10%-100%) for control (P = .1). The mean percentage of the maximum possible dose (which assumes non-recorded days were missed doses) was 45.9% (range: 0-100%) for memantine and 62.7% (range: 0-100%) for control (P = .02).
Cessation and Addiction Outcomes
Self-Reported Past Seven-Day Smoking Abstinence, Cigarettes Smoked per Day and Fagerstrom Nicotine Dependence Scale Scores Over Time.

**P-values are from 2-sided Fisher’s exact tests for past seven-day smoking abstinence.

Average Cigarettes Smoked per Day Least Square Mean Estimates by Group at Each Week +/− Standard Error From a Mixed Effects Repeated Measures Model to Assess Treatment Differences Over Time. An Unstructured Covariance Matrix was Used to Model the Within Patient Correlations. The Stratification Variable (Sex) was Included in this Model but no Other Covariates. The Number of Cigarettes Smoked per Week did not Change Significantly Between 2 and 12 weeks (P = .136). There Were no Significant Group Differences at Any Time Point (all P-values Were > 0.05).
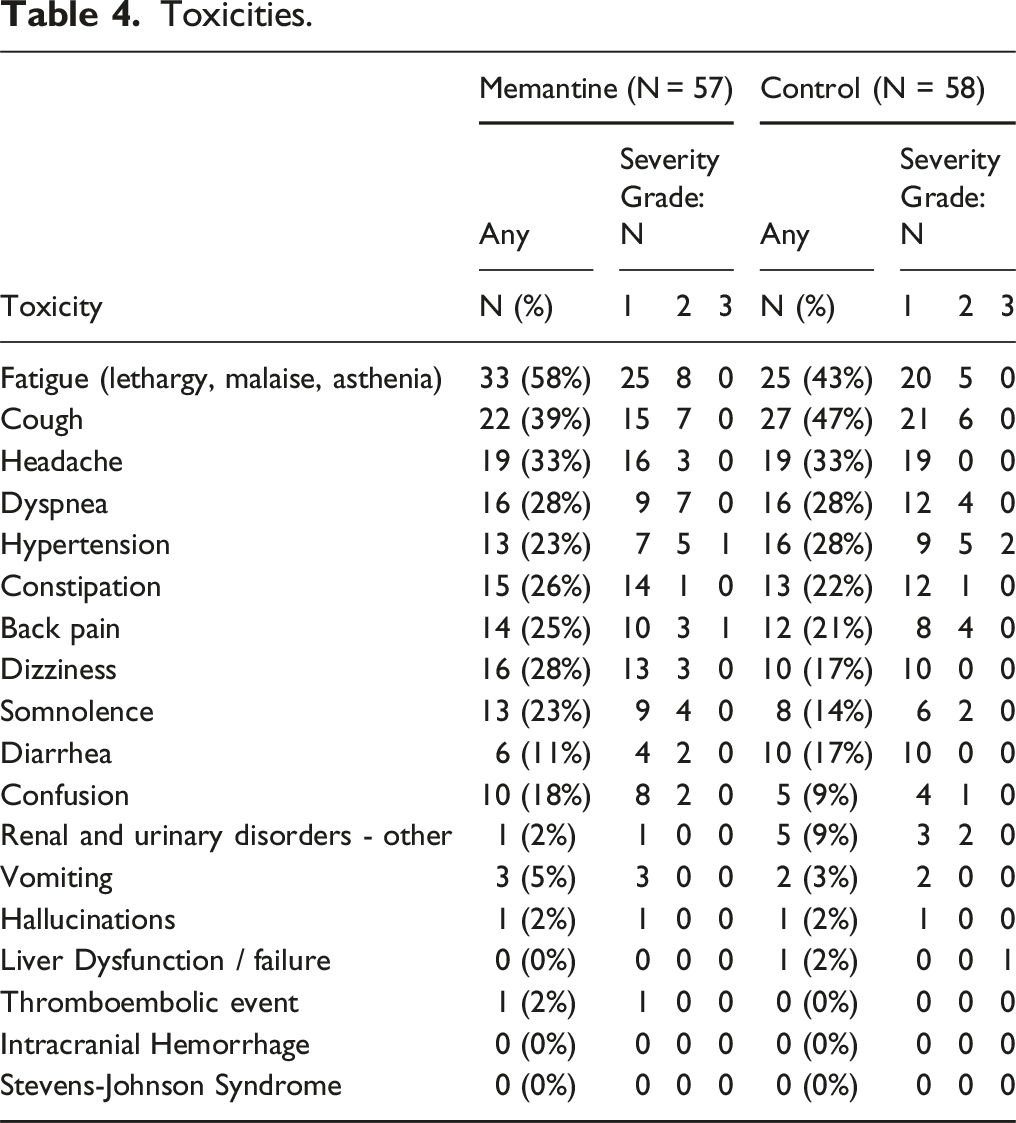
Fagerstrom Nicotine Dependence Scale least squares means and standard errors are shown in Figure 3. There was a significant decrease in nicotine dependence after baseline (P = .009) which did not differ significantly between groups (P = .45). At week 12, memantine participants had non-significantly worse Fagerstrom scores (5.29 + 0.3 vs 4.84 + 0.28, P = 0.2) On the Wisconsin Smoking Withdrawal Scale (Table 3), overall memantine participants scored non-significantly lower (better) on anxiety, craving, and hunger subscales than control participants. Fagerstrom Nicotine Dependence Scale* Least Square Mean Estimates by Group at Each Week +/− Standard Error From a Mixed Effects Repeated Measures Model to Assess Treatment Differences Over Time. An Unstructured Covariance Matrix was Used to Model the Within Patient Correlations. The Stratification Variable (Sex) was Included in this Model but no Other Covariates. There was a Significant Decrease in Dependence after Baseline (P = 0.09), which did not Differ Between Groups (P = 0.447). There Were no Significant Group Differences at Any Time Point (all P-values Were > 0.05). Fagerstrom Nicotine Dependence Scale: * 1-2 = Low Dependence; 3-4 = Low to Moderate Dependence; 5-7 = Moderate Dependence; 8+ = Severe Dependence. Least Squares Means and Standard Errors for Each Wisconsin Inventory of Smoking Dependence Motive and Estimated Treatment Difference at 12 Weeks.

Toxicities
Toxicities.
Conclusions
When nicotine attaches to NAchR in the VTA, it stimulates the release of dopamine in the nucleus accumbens and glutamate in the VTA. This glutamate further increases dopamine release into the nucleus accumbens. This dopamine increase in the nucleus accumbens is responsible for the rewarding effects of nicotine use.9,12–14 In this study, we hypothesized that blockade of glutamatergic NMDAR in the VTA by memantine could aid in smoking cessation by attenuating dopamine’s release into the nucleus accumbens. Our data do not support that hypothesis among participants surviving cancer. While randomization was well balanced, retention was lower than expected (54% in the memantine arm and 61% in the control group). Moreover, there was no significant difference in complete abstinence rates between the two groups. Studies in laboratory animals have suggested the importance of glutamate transmission in addiction, supporting the idea that NMDAR blockade might assist in tobacco cessation.12–15 Despite such evidence, our pilot study found participants on memantine did (non-significantly) worse than participants on placebo in cessation (0% vs 5% abstinent at 12 weeks, P = .499). In Das et al.’s study, 28 participants on memantine relapsed non-significantly more quickly than those on placebo. Thus, both studies suggested worsened smoking cessation outcomes with memantine.
Discussion
Memantine has numerous actions in the CNS and by itself might not be sufficient to aid in smoking cessation. For example, it acts as a full antagonist at α7 NAchR. This nicotinic acetylcholine receptor is involved in the rewarding effect of extracellular outflow of dopamine into the nucleus accumbens. Indeed this drug has a higher affinity for this receptor than for the NMDAR. 42 Although the α7 NAchR desensitizes rapidly after stimulation by nicotine, blockade of this receptor in the nucleus accumbens increases nicotine self-administration in rats, while agonist activation of this receptor reduces such activity. 43 Thus, to some degree, memantine’s antagonist activity at α7 NAchR might actually increase nicotine intake, albeit briefly given this receptor’s rapid desensitization, contributing to the imbalance of higher study discontinuation rates among memantine participants. . Indeed, Natarajan et al 15 found in rats that acute administration of memantine over minutes increases nicotine self-administration, while chronic administration over days to weeks non-significantly decreases nicotine self-administration. To complicate things further, NMDARs physically interact with dopamine D1 and D2 receptors; and both groups of receptors are made up of multiple classes of subunits. 44 Therefore, various combinations of NMDAR subunits interact with various combinations of dopamine receptor subunits leading to complex but finely balanced circuitry. 44 Moreover, regulatory presynaptic NMDARs known as heteroreceptors stimulate the release of a number of neurotransmitters, including norepinephrine, dopamine, and certain peptide neurotransmitters. This regulatory function is blocked by memantine. 45 In sum, memantine (1) shows a biphasic effect on nicotine self-administration in rats 15 ; (2) blocks the α7 NAchR 43 ; (3) must accommodate NMDAR and dopamine receptor subunit heterogeneity 44 ; and (4) blocks the NMDA heteroreceptor release of several neurotransmitters. 45 Clearly, this drug’s activity is multifaceted. Much of this research came after completion of this trial. From this perspective, to have expected a single response to this drug—smoking cessation—might have been not only unrealistic but also simplistic.
Low retention limited the power of this study, which could be partially due to missed safety events since hematological or serum labs were not obtained. Poor retention could be due to a number or issues including the high nicotine dependence of our sample of cancer survivors compared to the general population; and the fact that blockade of the α7 NAchR acutely increases nicotine self-administration in laboratory animals.14,40 Additionally, this study’s use of cancer survivors as participants limits its generalizability. While this limitation is obvious, there are unique reasons cancer survivors have more difficulty in quitting smoking. As Lowry et al note, “People with cancer who smoke are often highly nicotine-dependent, and the presence of depression, pain, anxiety, fatalism, or treatment side effects may influence their motivation and ability to quit smoking and maintain long-term smoking abstinence.” 46 Sample size was calculated assuming a 50% overall quit rate; while this rate was most likely not realistic or justified from previous studies, the choice of 50% was simply to ensure adequate power by utilizing the largest possible sample size needed to detect a 20% improvement. Date of start of treatment from date of randomization was not systematically collected as part of the database, but for those with available information, most started within the first month post-randomization. Moreover, cotinine was not analyzed, although numbers that quit were so small as perhaps not to be helpful. Nonetheless, cotinine levels would have provided a more definitive measure of reduced smoking and would have substantiated no difference in biologically confirmed smoking rates between arms. Adherence by patient diary and self-report is inherently difficult to collect and can have some level of recall bias. The percent intended dose calculates across those with received diary information, most likely overestimating the adherence, while the percent maximum tolerated dose assumes missing diaries were missed doses and underestimates adherence. The true adherence estimate lies somewhere between those two metrics. Cotinine levels also might have identified gradations of reduced smoking in some memantine treated patients. Further, this study was completed a decade ago. Much has happened in this field since then, particularly as it applies to glutamate transmission and nicotine addiction.
Had the study been more recent, we might have benefited from insights emerging from these areas of inquiry. Moreover, we do not have adequate screening data from the multiple sites to evaluate participation rates. We do not know, for example, how many individuals were contacted, how many were screened for participation, reasons for not participating or not being randomized, etc. While research on other NMDAR modulating compounds might discover smoking cessation efficacy, given a previous negative study 28 and the low number of participants who quit in this study, it seems unlikely that additional research of memantine would yield different results.
Supplemental Material
Supplemental Material - Randomized Placebo-Controlled Trial of Memantine for Smoking Cessation (CCCWFU 99311)
Supplemental Material for Randomized Placebo-Controlled Trial of Memantine for Smoking Cessation (CCCWFU 99311) by John Spangler, Elizabeth Skidmore, Emily V. Dressler, Kathryn E. Weaver, Glenn J. Lesser, Gary Burton, Benjamin Esparaz, Brooke Gillett, and Edward G. Shaw in Cancer Control
Footnotes
Acknowledgements
This project was funded by National Cancer Institute Grants U10 CA081851 (PI: Shaw) & UG1 CA189824 (PIs: Lesser & Weaver). It is registered with the U.S. National Institutes of Health on ![]() (identifier: NCT01535040). We would like to acknowledge the following CCOP sites: Meharry Medical College Minority-Based Community Clinical Oncology Program (MBCCOP), Ozark Health Ventures, LLC dba Cancer Research for the Ozarks, Delaware/ Christiana Care (CCHS) Community Oncology Research Program, Medical College of Georgia MB-CCOP, Louisiana State University Health Sciences Center in New Orleans (LSUHSC) MB-CCOP, Heartland Cancer Research CCOP, Iowa Oncology Research Association CCOP, Metro-Minnesota CCOP, Michigan Cancer Research Consortium NCORP, Southeast Cancer Control Consortium CCOP (SCCC), Comprehensive Cancer Center of Wake Forest CCOP Research Base, Wichita CCOP, Gundersen Lutheran Health System CCOP, Upstate Carolina CCOP, and St. Vincent Hospital Regional Cancer Center CCOP, Anthony J. Jaslowski. Additionally, we would like to thank Wake Forest NCORP Research Base staff members, Karen Craver, Julie Turner, Kimberly Blish, and Cheyenne Wagi for their efforts on behalf of this study and study participants
(identifier: NCT01535040). We would like to acknowledge the following CCOP sites: Meharry Medical College Minority-Based Community Clinical Oncology Program (MBCCOP), Ozark Health Ventures, LLC dba Cancer Research for the Ozarks, Delaware/ Christiana Care (CCHS) Community Oncology Research Program, Medical College of Georgia MB-CCOP, Louisiana State University Health Sciences Center in New Orleans (LSUHSC) MB-CCOP, Heartland Cancer Research CCOP, Iowa Oncology Research Association CCOP, Metro-Minnesota CCOP, Michigan Cancer Research Consortium NCORP, Southeast Cancer Control Consortium CCOP (SCCC), Comprehensive Cancer Center of Wake Forest CCOP Research Base, Wichita CCOP, Gundersen Lutheran Health System CCOP, Upstate Carolina CCOP, and St. Vincent Hospital Regional Cancer Center CCOP, Anthony J. Jaslowski. Additionally, we would like to thank Wake Forest NCORP Research Base staff members, Karen Craver, Julie Turner, Kimberly Blish, and Cheyenne Wagi for their efforts on behalf of this study and study participants
Statements and Declarations
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Cancer Institute Grants U10 CA081851 (PI: Shaw) & UG1 CA189824 (Wake Forest NCORP Research Base, PIs: Lesser & Weaver). It is registered with the U.S. National Institutes of Health on ClinicalTrials.gov (identifier: NCT01535040). ![]()
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author, JS.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
