Abstract
Objectives
The cancer knowledge gap represents a significant disparity in awareness and understanding of cancer-related information across different demographic groups. Leveraging Artificial Intelligence-Generated Content (AIGC) offers a promising approach to personalize health education and potentially bridge this gap. This study aimed to evaluate the potential of AIGC to bridge the cancer knowledge gap, assessing the roles of health self-efficacy as a mediator and educational level as a moderator in this relationship.
Methods
A 6-month longitudinal study was conducted using online surveys distributed to undergraduate students in non-medical disciplines at one university and graduate students in medical specialties at another university in China. The study assessed the frequency of AIGC use, health self-efficacy, and cancer knowledge at two time points.
Results
The results indicated that AIGC use significantly enhanced cancer knowledge levels and health self-efficacy over time. Educational level notably moderated the effects of AIGC use, with non-medical undergraduate students showing greater gains in knowledge and self-efficacy. Additionally, health self-efficacy mediated the relationship between AIGC use and cancer knowledge, underscoring the importance of health self-efficacy. The study confirms the efficacy of AIGC in narrowing the cancer knowledge gap and enhancing health self-efficacy, particularly among students with lower initial medical knowledge.
Conclusion
These findings highlight the potential of integrating AIGC tools in cancer education and public health interventions, particularly for individuals at different educational levels. By tailoring digital health resources to varying educational needs, these interventions could enhance cancer knowledge acquisition, improve health self-efficacy, and contribute to better cancer prevention and control outcomes.
Plain language summary
This study explores whether artificial intelligence-generated content (AIGC) tools, such as ChatGPT, can reduce disparities in cancer knowledge across different education levels. The research focused on university students in China over six months, with one group studying computer science and another in medical fields. Participants used AI tools to learn about cancer prevention, early detection, and treatment. The study found that frequent use of AIGC tools significantly improved participants' understanding of cancer and their confidence in managing their health (health self-efficacy). Non-medical students benefited the most, as they started with less prior knowledge compared to medical students. Additionally, students who felt more confident in their health knowledge were more likely to understand and retain cancer-related information. This research shows that AIGC tools can bridge knowledge gaps by providing personalized and accessible content. The findings suggest that such tools can play a vital role in public health campaigns and educational programs, especially for people with limited access to traditional healthcare education. These insights highlight the potential for AI technology to improve cancer awareness and health decision-making, ultimately contributing to better prevention and treatment outcomes.
Introduction
The concept of a “knowledge gap” refers to disparities in access to information among different segments of society, often due to variations in socioeconomic status, educational background, and other demographic factors. 1 In health care, the “cancer knowledge gap” highlights the uneven distribution of critical information related to cancer prevention, diagnosis, and treatment, 2 which significantly affects public health by influencing individual outcomes and the success of national cancer control programs. 3 Addressing this gap is crucial, as equitable access to cancer-related information can enhance early detection, improve treatment outcomes, and reduce the disproportionate burden of cancer on underserved populations. 4
Historically, research on the cancer knowledge gap has examined how demographic variables such as age, race, and gender shape individuals’ understanding of cancer-related information.5-7 Studies have also emphasized the role of media exposure in shaping public perceptions and promoting awareness of cancer prevention and treatment. 8 However, while targeted information campaigns have been somewhat successful, more personalized approaches are necessary to disseminate cancer knowledge effectively across diverse populations and tackle persistent disparities. 9
As technology evolves, Artificial Intelligence-Generated Content (AIGC) offers a transformative opportunity to address disparities in cancer knowledge and outcomes. AIGC uses sophisticated algorithms to generate customized, user-specific content tailored to the informational needs of various demographics, 10 making it particularly effective in bridging gaps caused by differences in education, cultural backgrounds, and access to health care resources. For instance, AIGC can provide underserved populations with accessible explanations of cancer prevention strategies, promote early screening, and empower individuals to make informed health care decisions. 11 By enabling equitable access to critical health information, AIGC has the potential to reduce disparities in cancer prevention and treatment outcomes, contributing to more effective cancer control on both individual and societal levels. Despite this promise, research on AIGC’s ability to directly address cancer-related disparities remains limited.
This study employs the knowledge gap hypothesis to conduct a 6-month longitudinal investigation among graduate students in medical fields and undergraduate students in non-medical disciplines. It examines how AIGC influences cancer knowledge, with health self-efficacy as a mediator and educational level as a moderator, providing insights into AIGC’s role in reducing knowledge disparities and enhancing cancer prevention and control.
Literature Review
Theoretical Foundation: Knowledge Gap Hypothesis
Knowledge gap hypothesis, first introduced in the 1970s by Tichenor et al, 12 posits that information in a social system tends to be unevenly distributed among various segments of the population. This distribution often aligns with socioeconomic status, whereby individuals with higher socioeconomic status tend to have better access to information and are quicker at integrating this information compared to those from lower socioeconomic strata. 13 The hypothesis emphasizes that as the infusion of media and information into a society increases, these gaps in knowledge tend to widen rather than diminish, unless specific measures are taken to address this imbalance. 14
In the context of health care, and specifically cancer, the “cancer knowledge gap” refers to the disparity in awareness, understanding, and information regarding cancer prevention, diagnosis, treatment, and management among different population groups. 15 Factors contributing to this gap include educational levels, access to health care resources, and communication effectiveness. Those with higher education levels or better access to health resources typically have a more profound understanding of cancer-related issues, which can lead to earlier detection and more effective management of the disease. Conversely, individuals in lower socioeconomic groups often experience barriers to accessing and comprehending essential cancer information, which can delay diagnosis and treatment, ultimately affecting health outcomes. 16
Relationship Between the Frequency of AIGC Use and Cancer Knowledge Level
Based on the knowledge gap hypothesis, assuming equal access to technology, tools like AIGC have the potential to mitigate these disparities by providing tailored information that addresses specific needs and gaps in understanding. This personalized approach is designed to enhance the baseline knowledge of underserved populations, thereby helping to narrow the knowledge gap. 17
Several studies have explored the impact of digital tools on health literacy, providing insights that can be applied to AIGC. For instance, Luo et al 18 highlighted that internet use among diverse demographic groups leads to improved health knowledge, suggesting that more frequent digital engagement could enhance understanding. In the context of AIGC, tools like chatbots and personalized health dashboards have been shown to effectively engage users and increase comprehension of complex medical topics. 19 These systems adapt their outputs based on user interactions, which can include assessments of prior knowledge and preferences. 20 Moreover, research by Boucher on digital health interventions has demonstrated that personalized content delivered through AI significantly increases the efficacy of health communication. This personalization is critical in addressing individual differences in baseline knowledge, learning styles, and information retention capacities. 21 Additionally, a study by Chow found that users who engaged with an AI-powered educational platform on a regular basis showed significant improvements in their understanding of cancer screening and prevention techniques compared to less frequent users. 22 This indicates not only an increase in knowledge but also suggests that sustained interactions with AIGC tools could lead to deeper and more durable learning, particularly in the context of cancer prevention and control.
Given the growing body of evidence supporting the efficacy of personalized, AI-driven health interventions, it is clear that AIGC can play a crucial role in improving cancer-related health literacy. This is particularly important for underserved populations who may have limited access to traditional forms of health education. Moreover, the use of AI tools can help overcome barriers to understanding complex medical information, empowering individuals to make more informed decisions about cancer prevention, early detection, and treatment. Therefore, we propose:
Mediating Role of Health Self-Efficacy
Health self-efficacy, defined as an individual’s belief in their capability to manage their health successfully, is a pivotal element in health behavior change. Empirical research has shown that personalized feedback and interactive content, which are hallmarks of AIGC systems, 23 can enhance an individual’s confidence in understanding complex health information and making informed health decisions. For instance, a study by Shah et al 24 demonstrated that users who regularly interacted with an AI-driven health advisor reported higher levels of health self-efficacy compared to those who did not. This increase in self-efficacy was attributed to the system’s ability to provide immediate, relevant, and understandable feedback based on the user’s interactions and queries. Moreover, AIGC tools are designed to assess the user’s initial knowledge and adapt their informational output accordingly, thereby reducing the cognitive load and enhancing the learning process. 25 This adaptive learning environment supports the development of self-efficacy by ensuring that users are neither overwhelmed by complexity nor under-challenged by simplicity. By continually adjusting the difficulty and relevance of the information, AIGC can foster a sense of mastery and control over health-related knowledge, which is essential for building health self-efficacy. 26 Research by Cañamero and Lewis examined “Robin,” a robot designed as a toddler with “robot diabetes,” to support children with this condition. Through engaging and realistic interactions, Robin enhances children’s self-efficacy and emotional well-being, helping them feel more in control of their diabetes management. 27 A systematic review and meta-analysis conducted by Bendotti et al 28 explored the effectiveness of conversational AI interventions, such as chatbots, in supporting smoking cessation. The study suggests that conversational AI can effectively personalize support and increase engagement, thereby enhancing users’ self-efficacy in quitting smoking.
These empirical findings highlight AIGC’s potential in supporting individual health management through its unique capability to generate and tailor content based on specific user needs. Extending this capability to the context of cancer knowledge, it is logical to hypothesize that frequent use of AIGC may positively correlate with health self-efficacy.
Health self-efficacy plays a critical role in how individuals gather, process, and use health information. It is particularly vital in the context of cancer, where accurate knowledge can significantly influence prevention, early detection, and effective management of the disease. 29 High health self-efficacy encourages individuals to actively seek out, comprehend, and utilize cancer-related information, thereby facilitating better health outcomes. 30 Several studies underscore the correlation between health self-efficacy and cancer knowledge. For example, a study by Tiraki explored this relationship in breast cancer patients and found that those with higher health self-efficacy showed greater knowledge about their treatment options and side effects management. This enhanced knowledge was not only crucial for their treatment decisions but also improved their ability to cope with the disease. 31 Further, in a review of AIGC-based interventions aimed at increasing cancer screening knowledge, Herts et al 32 observed that interventions designed to boost self-efficacy were particularly effective at enhancing participants’ understanding of cancer screenings. This suggests that health self-efficacy facilitates not just knowledge acquisition but also the ability to comprehend complex medical information. Merluzzi et al in their meta-analysis on cancer patients, demonstrated that individuals with higher self-efficacy had a better grasp of complex treatment information, which empowered them to actively participate in treatment decisions. 33
These findings suggest that individuals with higher health self-efficacy are more likely to actively seek out and engage with health-related information, including cancer-related content. This proactive engagement may enhance their ability to understand, interpret, and apply cancer knowledge effectively, particularly in areas such as cancer prevention, early detection, and treatment. A stronger sense of self-efficacy is hypothesized to support individuals in navigating the complexities of cancer-related information, leading to more informed and confident health care decisions. Additionally, higher health self-efficacy may encourage greater participation in cancer screening programs, adherence to preventive behaviors, and better management of treatment, all of which are crucial to improving cancer outcomes. While the exact mechanisms linking self-efficacy to cancer knowledge levels remain to be fully explored, existing evidence suggests that self-efficacy plays a key role in enhancing cancer-related knowledge. This underscores the potential for digital tools, such as AIGC, to positively influence both cancer knowledge and associated health behaviors, contributing to better cancer prevention, earlier detection, and more effective treatment strategies.
Based on the empirical research presented, there appears to be a potential positive correlation between the frequency of AIGC use and health self-efficacy. Furthermore, evidence suggests a possible positive relationship between health self-efficacy and cancer knowledge levels. These observations imply that individuals who frequently use AIGC tools might experience increased self-efficacy, which could in turn enhance their knowledge about cancer. To rigorously examine these relationships, we propose the following hypothesis:
Moderating Role of Educational Level
Drawing from the knowledge gap hypothesis, this study examines whether educational level can moderate the effectiveness of AIGC in bridging the cancer knowledge gap. Education, particularly medical knowledge, may significantly influence an individual’s ability to effectively utilize digital tools like AIGC, which in turn can affect their cancer knowledge and self-efficacy.
Educational level has consistently been identified as a crucial moderator affecting the knowledge gap, particularly in the context of new technology interventions. On one hand, the relationship between the use of new technologies and knowledge levels can be significantly influenced by one’s educational attainment. Some studies suggest that individuals with higher educational levels are able to leverage new technologies more effectively, enhancing their access to and comprehension of complex information. 34 Conversely, other research indicates that without adequate educational support, new technologies may exacerbate existing knowledge disparities, as those with lower educational levels struggle to benefit equally from these advancements. 35 Regardless, these findings collectively underline the potential moderating role of education, especially within health contexts, where the disparity in knowledge can have direct implications on patient outcomes and public health initiatives. 36
On the other hand, educational level is also likely to influence the relationship between new technology use and self-efficacy. Similar to its impact on knowledge acquisition, some studies argue that higher education equips individuals with the confidence and skills necessary to interact with and benefit from digital health technologies, thereby enhancing their self-efficacy. 37 However, there is also evidence suggesting that without sufficient educational background, individuals may not experience the full self-efficacy benefits that these technologies are designed to offer, potentially leading to underutilization and ineffective engagement. 38 This dichotomy in findings highlights the essential role of educational level in moderating the effects of technology on self-efficacy, particularly in health domains where effective management of conditions often requires confident decision-making supported by reliable information. 39
However, AIGC, with its capability to generate personalized content, offers the potential to cater to users across various educational and knowledge levels. This unique feature of AIGC raises the question: Could it possibly bridge the cancer knowledge gap? This aspect has not been extensively discussed in previous research. Empirical studies consistently show that educational level remains a significant moderating variable. If AIGC can tailor its content to the specific needs of individuals based on their educational background, it may be particularly effective in addressing disparities in cancer knowledge, ultimately contributing to more equitable cancer prevention, early detection, and treatment outcomes. By customizing information delivery, AIGC could support the improvement of cancer-related health literacy, especially in underserved populations. Thus, we propose:
Method
This study was conducted from August 15, 2023, to April 15, 2024, and collected data by distributing online surveys in the WeChat course groups at two universities. The study was approved by the Institutional Review Board at the authors’ institution (Approval number: B2022328I), and the participation of all subjects was based on informed consent. Participants who completed all two surveys were compensated with 10 yuan each, via WeChat Pay or Alipay. The reporting of this study conforms to STROBE guidelines. 40
Participants and Procedure
The sample for this study originated from two universities in China. The first university, which has established a robust foundation in AIGC usage, provided undergraduate students from the computer science department. Previously, the institution had equipped these students with individual AIGC accounts to foster familiarity with the technology. 41 The second university is renowned for its prestigious medical program, particularly its clinical medicine specialty, which ranks first in the country and produces a significant number of medical graduates annually. 42 Participants from this university included graduate students from various medical specializations, though not specifically from oncology. This selection aimed to explore the impact of AIGC across fundamentally different educational levels and academic backgrounds: non-medical undergraduates from the first university (undergraduate students in non-medical majors with a medical background, such as those whose family’s primary income comes from operating a clinic, were also excluded) and medical graduate students from the second.
Before the survey, a manipulation check was conducted to validate the appropriateness of our sample for studying the cancer knowledge gap. A standardized cancer knowledge test, developed by three oncology professors and comprising 50 questions, was administered, with final scores normalized on a scale from 1 to 5. The initial test showed that non-medical undergraduate students had an average cancer knowledge level of 1.572, whereas medical graduate students scored an average of 4.288, confirming a significant knowledge gap between the two groups.
To ensure the reliability of the frequency of AIGC use data and its relevance to cancer knowledge search, after the initial cancer knowledge assessment, we assigned homework on cancer topics at both universities, advising students to use ChatGPT for information retrieval. The first set of data (T1) was collected on September 15, 2023, one month after the homework assignment. Six months later, on April 15, 2024, the second set of data (T2) was gathered, with monthly cancer knowledge assignments given in the interim to maintain engagement.
Based on relevant literature,3,5,43 we set the parameters for the power analysis as follows: a small to medium effect size (|ρ| = 0.2), a significance level of α = 0.05, and a power level of 95% (1-β = 0.95). Using G*Power (Version 3.1.9.2), we conducted an a priori power analysis to determine the required sample size under these conditions. The analysis indicated that a minimum of 262 participants would be required for two-tailed testing. Considering potential dropout, we initially recruited 870 participants, ensuring robust data for our longitudinal analysis.
The sample size was initially 870 participants at T1, comprising 533 undergraduates and 337 graduate students. At T2, the sample included 486 undergraduates and 301 graduates, totaling 787 participants. The retention rate between T1 and T2 was 90.46%. The decrease in participant numbers was primarily due to reduced engagement in the WeChat course groups after the courses ended, posing challenges in data collection. Despite this drop, the relatively high retention rate provided robust longitudinal data for analysis.
Statistical Analysis
To handle the missing data, we first conducted a Little’s Missing Completely at Random (MCAR) test. The results indicated that the missing values were randomly distributed (χ2 = 15.362, P = 0.224). Subsequently, to address the issue of missing data in our longitudinal study, we employed the Full Information Maximum Likelihood (FIML) method, which is particularly suited for handling missingness in complex, multi-wave data sets like ours. 44 FIML works under the assumption that the data are missing at random, which is a reasonable assumption in our context given that the primary reasons for dropout were the low engagement of some students following the conclusion of the courses, rather than factors related to the study variables. By utilizing FIML, all available data from participants, regardless of whether they completed both phases of the study, were incorporated into the model parameter estimations. 45
For statistical analysis, SPSS 26.0 was used to calculate descriptive statistics, including means, standard deviations, and correlations. AMOS 25.0 was employed for multi-group confirmatory factor analysis (CFA) to validate the measurement model and assess the fit of constructs such as health self-efficacy, cancer knowledge, and AIGC use, using indices like χ2/df, CFI, TLI, and RMSEA. Cross-lagged panel modeling was conducted to examine the directional relationships between AIGC use, health self-efficacy, and cancer knowledge over time. 46
Measures
The Frequency of AIGC Use
Participants’ frequency of AIGC use, including AI-generated text and voice, was assessed with the question: “What is your frequency of use of AIGC in the forms of AI-generated text and AI-generated voice?” Response options ranged from 1 (“Less than once a week”) to 5 (“Multiple times a day”). Participants who selected “never” or used versions other than ChatGPT 4.0 were excluded.
A strong correlation between text and voice use was observed across both waves (Pearson correlation >0.66, P < 0.05), attributed to the alignment between text recognition and voice output in AI-generated voice. Therefore, the mean frequency of both forms was combined into a single score for analysis. The internal consistency of this measure was high, with Cronbach’s alphas of 0.905 (T1) and 0.898 (T2), indicating strong reliability over time.
Cancer Knowledge Level
To accurately assess the cancer knowledge level among participants, we utilized a standardized test specifically designed for this study. For each of the two waves of data collection, we collaborated with three distinguished professors of oncology in China to develop a set of cancer knowledge questions. Each wave included a unique set of 50 multiple-choice questions. Participants received one point for each correct answer, with no point for incorrect responses.
To facilitate comparison and analysis across different participant groups, we normalized the total scores to a scale of 1 to 5, rounding decimal points using the round-half-up method to assign scores within this range. This normalization involved converting the raw scores, which ranged from zero to 50, into a scaled score that reflects a participant’s knowledge level on a graduated scale where 1 represents minimal knowledge and 5 indicates expert-level understanding of cancer-related topics. This approach ensured that the scores were uniformly interpretable, providing a clear quantification of the participants’ cancer knowledge that could be statistically analyzed and compared over time and between different groups within the study.
Health Self-Efficacy
Health self-efficacy was evaluated using a scale adapted from Lee. 47 Participants were asked to reflect on their capacity to influence and control their health status. They were to rate their level of agreement with statements concerning their confidence in having a positive impact on their health, their goal-setting behavior for health improvement, their ability to meet health goals, their active efforts towards health improvement, and their perceived control over learning about their health. Responses were recorded using a 5-point Likert scale, with 1 indicating “Strongly Disagree” and 5 indicating “Strongly Agree.” The mean score of these items was calculated to generate an overall measure of health self-efficacy, with higher scores representing a greater sense of self-efficacy related to health.
The internal consistency of the health self-efficacy scale was robust, with Cronbach’s alphas of 0.828 (T1) and 0.801 (T2), and the CFA confirmed good model fit, with χ2/df ranging from 2.47 to 2.70, CFI from 0.96 to 0.98, TLI from 0.95 to 0.97, and RMSEA from 0.02 to 0.03 across both waves.
Educational Level
In this study, educational level was operationalized distinctly by differentiating between undergraduate students in non-medical majors and graduate students in medical fields. Statistical analyses will employ educational level as a categorical variable to test its moderating effects in the proposed models.
Results
Descriptive Statistics and Correlations
The results of Descriptive Statistics and Correlations (N = 870).

Notes: Gender: zero = Female, 1 = Male; AIUF = The frequency of AIGC use; HSE = Health self-efficacy; CKL = Cancer knowledge level; EL = Educational level, zero = Undergraduate students in non-medical majors, 1 = Graduate students in medical fields; M = Mean; SD = Standard deviation; T1 = Time 1; T2 = Time 2.
*P < 0.05, **P < 0.01, ***P < 0.001.
Regarding the age of the participants, the average was 23.891 years with a standard deviation of 1.608. In terms of gender distribution, coded as zero for females and 1 for males, the mean was 0.578 with a standard deviation of 0.125, indicating a relatively balanced gender distribution in the sample.
Across two waves, the frequency of AIGC use, health self-efficacy, cancer knowledge level, and educational level showed considerable consistency over time, as evidenced by the significant correlations between the same constructs measured at different time points.
Based on the provided data from Table 1, the positive correlations between the frequency of AIGC use at Time 1 (T1) and Time 2 (T2) and health self-efficacy at both time points suggest that participants who engaged more frequently with AIGC tools reported higher levels of self-efficacy. Specifically, the frequency of AIGC use at T1 showed significant positive associations with health self-efficacy at T1 (r = 0.260, P < 0.001) and at T2 (r = 0.241, P < 0.001), and these associations remained strong at T2 for the frequency of AIGC use (r = 0.235, P < 0.001 for T1 and r = 0.266, P < 0.001 for T2).
Furthermore, there were significant positive correlations between the frequency of AIGC use and cancer knowledge level at both T1 and T2, indicating that higher engagement with AIGC was associated with greater knowledge about cancer. The frequency of AIGC use at T1 correlated with cancer knowledge level at T1 (r = 0.218, P < 0.001) and cancer knowledge level at T2 (r = 0.200, P < 0.001).
Health self-efficacy at T1 showed a significant positive correlation with cancer knowledge level at T2, highlighting that earlier self-efficacy is a predictor of later cancer knowledge. Specifically, health self-efficacy at T1 had a correlation of r = 0.340, P < 0.001 with cancer knowledge level at T2.
Additionally, a noteworthy finding is the correlation between educational level at T1 and the subsequent measurements of other variables at T2. Educational level at T1 showed significant positive associations with health self-efficacy and cancer knowledge level at T2 (r = 0.372, P < 0.001 for health self-efficacy and r = 0.395, P < 0.001 for cancer knowledge level), implying that educational background plays a critical role in both self-efficacy and cancer knowledge acquisition over time.
Cross Lagged Analysis of the Frequency of AIGC Use, Health Self-Efficacy, and Cancer Knowledge Level
In statistics, correlational analysis in descriptive statistics can only indicate that there is some statistical relationship between two variables but cannot determine the directionality of this relationship. Cross-lagged analysis helps researchers explore potential causal relationships between variables, identifying which variable occurs first and which follows (Zyphur et al, 2020). Therefore, following our descriptive analysis, we conducted a cross-lagged analysis. We constructed a cross-lagged model with the frequency of AIGC use serving as the independent variable, health self-efficacy acting as the mediator, and cancer knowledge level being the dependent variable. The model and standardized path coefficients are depicted in Figure 1. Two-wave cross-lagged model of the frequency of AIGC use, health self-efficacy, and cancer knowledge level. The measurement error and non-significant paths were omitted to enhance the clarity of the figure. Only the main paths of interest for this study are retained.
The cross-lagged paths indicate that the frequency of AIGC use at T1 positively influences health self-efficacy at T2 (β = 0.229, P < 0.001), and similarly, health self-efficacy at T1 has a positive impact on cancer knowledge level at T2 (β = 0.322, P < 0.001). These results highlight the frequency of AIGC use influencing subsequent health self-efficacy, which in turn enhances cancer knowledge level. The model also shows direct paths from the frequency of AIGC use at T1 to cancer knowledge level at T2 (β = 0.179, P < 0.05), reinforcing the direct impact of the frequency of AIGC use on cancer knowledge level over time.
The findings from this cross-lagged analysis underscore the sequential influence of AIGC use on health self-efficacy and cancer knowledge level. AIGC use not only directly promotes better understanding of cancer-related information but also indirectly supports this learning through enhancements in self-efficacy. Hypotheses 1 and 2 were supported.
Longitudinal Analysis of Educational Level as a Moderator
Regression Analysis results.
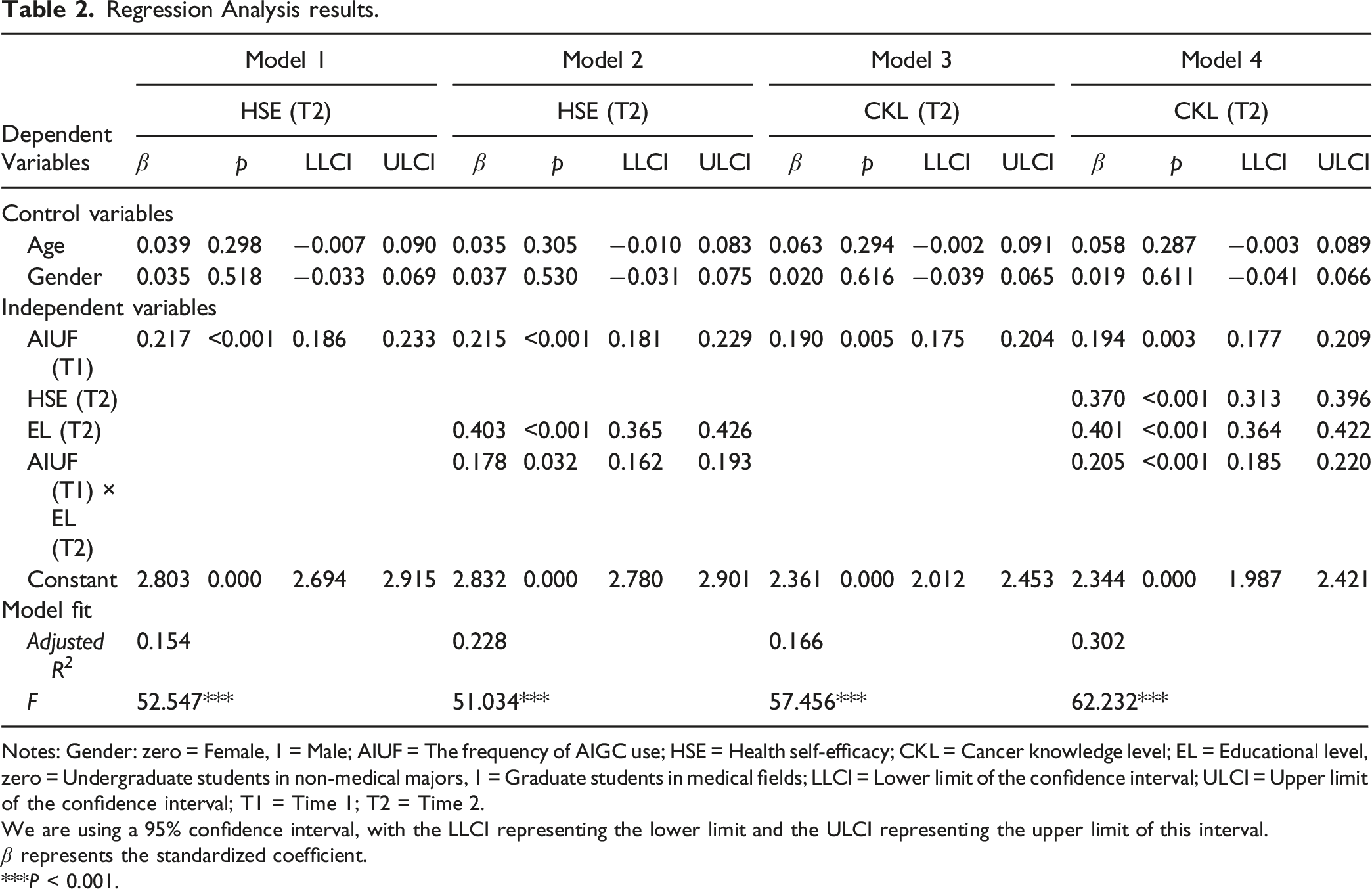
Notes: Gender: zero = Female, 1 = Male; AIUF = The frequency of AIGC use; HSE = Health self-efficacy; CKL = Cancer knowledge level; EL = Educational level, zero = Undergraduate students in non-medical majors, 1 = Graduate students in medical fields; LLCI = Lower limit of the confidence interval; ULCI = Upper limit of the confidence interval; T1 = Time 1; T2 = Time 2.
We are using a 95% confidence interval, with the LLCI representing the lower limit and the ULCI representing the upper limit of this interval.
β represents the standardized coefficient.
***P < 0.001.
Examination of the Moderating Effect on the Direct Effect and Mediating Effect.

Notes: EL = Educational level, zero = Undergraduate students in non-medical majors, 1 = Graduate students in medical fields; SE = Standard error; LLCI = Lower limit of the confidence interval; ULCI = Upper limit of the confidence interval; T2 = Time 2.
We are using a 95% confidence interval, with the LLCI representing the lower limit and the ULCI representing the upper limit of this interval.
The simple slopes graph further reveals the moderating role of educational level. As depicted in Figure 2, within the context of educational level’s moderation on the direct effect, the dashed line representing non-medical undergraduate students (lower educational level) shows a more pronounced positive slope compared to the solid line representing medical graduate students (higher educational level). This indicates that as the frequency of AIGC use increases, non-medical undergraduate students exhibit a greater increase in cancer knowledge level compared to medical graduate students. The moderating effect of educational level on the relationship between the frequency of AIGC use and cancer knowledge level.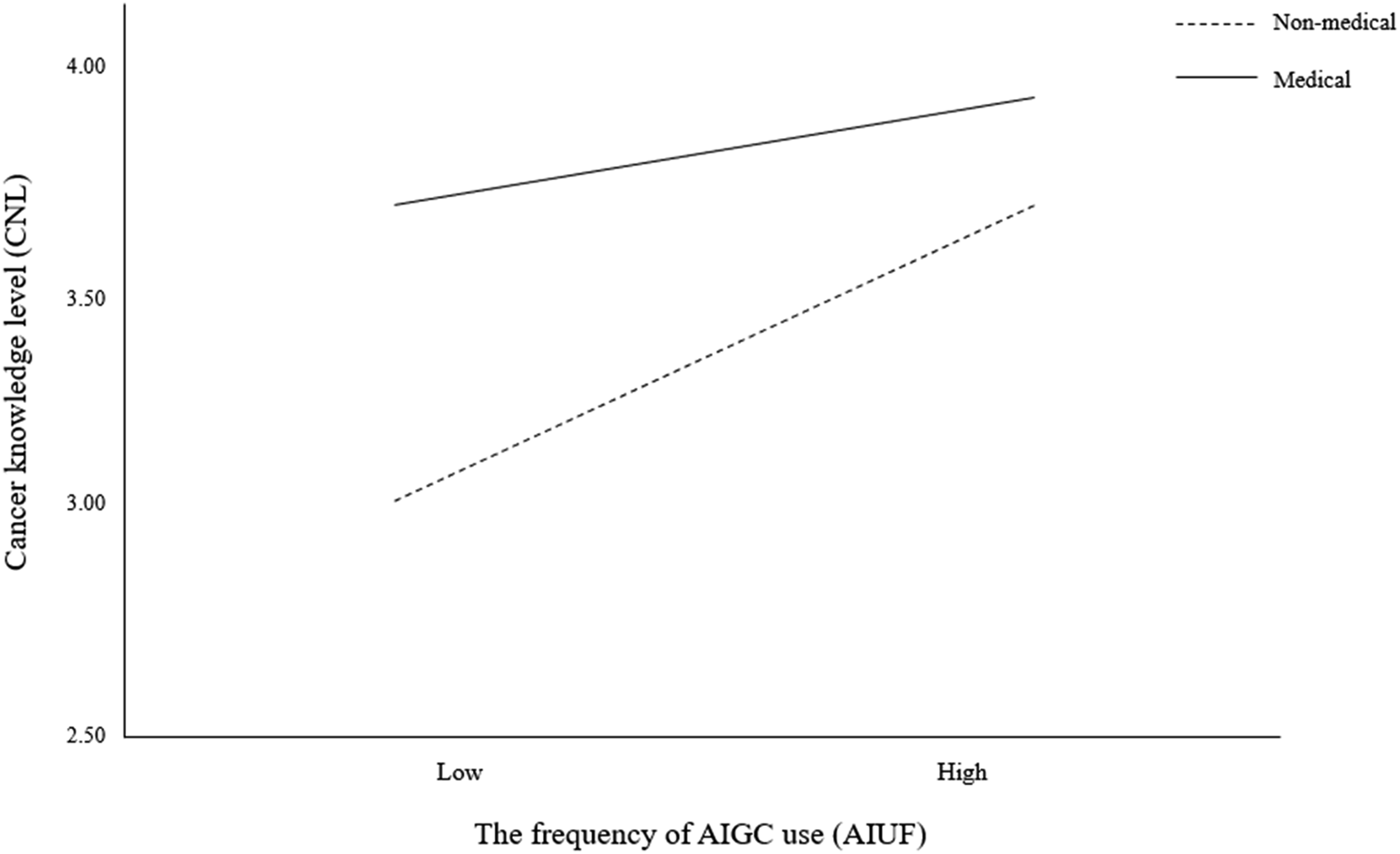
As shown in Figure 3, in the moderation of the mediating effect of educational level on health self-efficacy, the dashed line representing non-medical undergraduate students once again displays a more pronounced positive slope compared to the solid line representing medical graduate students. This graphically illustrates that non-medical students, with a lower educational level, experience a more significant increase in health self-efficacy as the frequency of AIGC use rises. The moderating effect of educational level on the relationship between the frequency of AIGC use and health self-efficacy.
Discussion
Firstly, our study highlights the critical role of educational level as a moderator in shaping the relationship between the use of AIGC, health self-efficacy, and cancer knowledge level. Traditionally, knowledge gap hypothesis suggests that as media and information influx increases in a society, knowledge disparities tend to widen rather than close. 50 However, our findings introduce a nuanced perspective by demonstrating that under conditions of equal access to technologies like AIGC, the personalized nature of the content generated can effectively address and reduce the cancer knowledge gap.
In our analysis, educational level moderated both the direct effects of AIGC use on cancer knowledge and its mediated impact through health self-efficacy. Notably, the influence of AIGC was more pronounced among non-medical undergraduate students compared to medical graduate students. This can be interpreted through the lens of the initial knowledge base and the novelty effect. Non-medical undergraduate students, likely starting with a lower baseline of specific medical knowledge, may experience a more significant relative increase in knowledge when exposed to specialized content delivered via AIGC. This is in line with the findings of prior research, such as Le 51 and Kurashige, 52 who suggested that the acquisition of knowledge could be more substantial among those less familiar with the content area, thereby rapidly decreasing their knowledge gap compared to those who are already well-versed. Moreover, the less steep increase in cancer knowledge among medical graduate students suggests a ceiling effect, where additional information does not translate into significant knowledge gains due to their already high baseline understanding. This observation supports the arguments by Armougum, 53 who noted that higher baseline knowledge might lead to diminishing returns in knowledge acquisition from new information sources. These findings suggest that AIGC has the potential to reduce cancer disparities by providing more significant benefits to populations with lower initial levels of cancer knowledge. Additionally, the difference in the increase of health self-efficacy between educational groups underscores the importance of contextual and cognitive factors in AIGC-based interventions using digital tools. It suggests that while AIGC tools have the potential to enhance self-efficacy broadly, the extent of this enhancement may depend on an individual’s ability to relate the AI-provided content to their pre-existing knowledge and skills.
These findings highlight the nuanced dynamics of the knowledge gap hypothesis, demonstrating AIGC’s potential to address disparities in cancer knowledge across educational groups. For non-medical undergraduates, AIGC effectively bridges foundational gaps by providing personalized, accessible content that caters to their specific needs in understanding cancer. This adaptability underscores AIGC’s role in making complex cancer-related topics more approachable for individuals with limited medical knowledge. In contrast, medical graduate students exhibited smaller relative gains, likely due to their already advanced baseline knowledge, which reduces the perceived novelty and utility of generalized content. To engage this group, AIGC must evolve to deliver more specialized, in-depth materials, such as interactive case studies or advanced simulations that encourage critical thinking about cancer. Additionally, differences in perceptions of AIGC may play a role—non-medical students likely view it as an innovative tool for learning about cancer, while medical students may consider it supplementary or redundant. Improved cancer knowledge through AIGC can also have tangible effects on cancer control outcomes, influencing health-related behaviors. For example, a deeper understanding of cancer prevention, early detection strategies, and treatment options may lead to greater participation in screening programs, earlier diagnoses, and improved adherence to treatment regimens.
Secondly, the relationship between the frequency of AIGC use and cancer knowledge level further illuminates the transformative potential of digital tools in enhancing cancer education. Our study findings underscore that frequent interaction with AIGC significantly enhances an individual’s understanding of cancer-related topics. The strong positive correlation between the frequency of AIGC use and cancer knowledge level across both time points suggests that consistent engagement with AI-driven educational content not only increases immediate knowledge but also contributes to sustained learning and retention of complex medical information. This effect can be attributed to the adaptive and interactive nature of AIGC systems, which tailor educational content to fit the user’s current knowledge state and learning pace. 54 By providing information that is both accessible and contextually relevant, AIGC tools help bridge the gap between complex cancer-related data and the user’s ability to understand and apply this information effectively. 55 This highlights the potential of AIGC to provide tailored cancer education that can empower underserved populations with actionable knowledge, ultimately narrowing disparities in cancer prevention and treatment outcomes.
Thirdly, the relationship between the frequency of AIGC use and health self-efficacy highlights the critical role of interactive and adaptive learning environments in enhancing self-regulatory capabilities. The data from our study show a significant positive correlation between the frequency of AIGC use and health self-efficacy, which suggests that regular use of AIGC tools can substantially boost an individual’s confidence in managing cancer-related health behaviors. This increase in self-efficacy is particularly important in the context of cancer education, where the ability to understand and act upon cancer information can directly influence health outcomes. This relationship can be explained by the supportive nature of AIGC systems, which not only provide personalized cancer-related information but also offer feedback and encouragement. 56 Such features are key components of effective learning environments, as they help individuals feel more competent and empowered to take action regarding cancer prevention, early detection, and treatment. 57 This is in line with Bandura’s social cognitive theory, which posits that learning is most effective when individuals are actively engaged and when they receive positive reinforcement for their learning behaviors. 58 Moreover, AIGC tools often incorporate elements of interactive content, which have been shown to increase motivation and engagement. 59 By engaging with these tools, users are likely to experience a sense of achievement and control over their cancer-related learning process, which further enhances their self-efficacy. 60 These findings support the notion that digital health interventions should not only focus on the delivery of cancer-related information but should also aim to foster a supportive learning environment that enhances the individual’s capacity to use that information effectively. By doing so, AIGC can play a pivotal role in closing the gap between cancer knowledge acquisition and practical application, leading to better cancer management and potentially improved health outcomes.
Fourthly, the relationship between health self-efficacy and cancer knowledge level provides compelling evidence for the integral role of self-efficacy in the effective assimilation and utilization of health information. Our study’s findings highlight that higher levels of self-efficacy significantly correlate with enhanced knowledge about cancer, reinforcing the theory that self-efficacy acts as a key facilitator in the process of acquiring cancer-related knowledge, particularly in the domain of cancer health literacy. This relationship can be explained through the lens of motivational and behavioral theories which posit that individuals with high self-efficacy are more likely to engage in and benefit from educational activities because they feel capable of successfully understanding and applying the information. 61 In the context of cancer education, individuals who believe in their ability to understand complex medical information are more likely to seek out, comprehend, and retain this information, thereby enhancing their overall knowledge levels. Moreover, self-efficacy affects not only the acquisition but also the application of knowledge in making health-related decisions. 62 High health self-efficacy empowers individuals to navigate through the vast amount of information available, discerning what is relevant and reliable, and applying this knowledge in decisions about their health care and lifestyle choices. These findings underscore how AIGC can play a vital role in enhancing self-efficacy, which in turn supports better health management and reduces barriers to equitable cancer education and care.
This study makes a significant contribution to the theoretical framework surrounding the knowledge gap hypothesis by integrating AIGC as a mediating technology. Our findings suggest that AIGC can effectively address disparities in cancer knowledge by providing personalized content that bridges the information gap across various educational levels. This adaptation of the knowledge gap hypothesis to include digital tools introduces a new perspective on how technology can enhance cancer education specifically. Additionally, by demonstrating the mediating role of health self-efficacy and the moderating role of educational level, this research enriches existing models of health communication and literacy in the context of cancer knowledge. It extends the application of Bandura’s social cognitive theory to digital environments, highlighting how self-efficacy influences the acquisition and application of cancer-related knowledge, particularly in an era where digital information exchange is increasingly central to learning.
From a practical standpoint, the study provides valuable insights for cancer education professionals, educators, and developers of health technologies. By showing the effectiveness of AIGC in improving cancer knowledge, our findings support the development of targeted digital interventions tailored to diverse educational backgrounds. These insights emphasize the importance of incorporating AIGC tools into cancer education initiatives focused on prevention, early detection, and treatment. This approach ensures that individuals from various educational backgrounds can equally benefit from advancements in cancer knowledge and digital health technology. Thus, this research not only advances theoretical understanding of cancer knowledge acquisition but also offers practical solutions for improving cancer education and health literacy through the strategic use of digital interventions.
Conclusion
This study explored the potential of AIGC to bridge the cancer knowledge gap, with a particular focus on the mediating role of health self-efficacy and the moderating role of educational level. The findings confirm that AIGC can significantly enhance cancer knowledge across different educational groups by providing personalized, adaptive content that caters to the specific needs and knowledge bases of users. Moreover, health self-efficacy was shown to play a crucial role in mediating the relationship between AIGC use and cancer knowledge acquisition, suggesting that increased confidence in managing health information can lead to better outcomes. The study also highlighted that educational level significantly influences the effectiveness of AIGC tools, with non-medical undergraduates benefiting more markedly than their medical graduate counterparts.
This study, while offering valuable insights, is not without limitations. Firstly, this study’s external validity is limited by the inclusion criteria, which focused on university students from two Chinese institutions. This was done to assess the impact of AIGC on cancer knowledge across varying educational levels, ensuring a controlled sample with comparable prior knowledge and digital tool exposure. However, this sample may not fully represent broader populations, especially those from non-academic backgrounds or lower socioeconomic statuses, who may have different levels of digital access, health literacy, and learning experiences. Secondly, the reliance on self-reported measures for assessing the frequency of AIGC use, health self-efficacy, and cancer knowledge introduces the possibility of response bias. While self-reported data provide useful insights into participants’ subjective experiences, they may not fully capture actual usage patterns or knowledge levels. Thirdly, the study focused solely on educational level as a moderator and health self-efficacy as a mediator. Examining additional factors such as digital literacy, socioeconomic status, or technological familiarity could provide a more nuanced understanding of the mechanisms underlying AIGC’s effects on health-related outcomes. These variables might reveal how AIGC interacts with broader systemic disparities to influence cancer knowledge and related behaviors. Finally, while this study acknowledges the potential benefits of AIGC, future research should incorporate objective measures, such as digital usage logs, pre/post-intervention knowledge assessments, or observed behavioral changes, to validate self-reported findings and strengthen the study’s conclusions.
To address these limitations, future studies should expand the participant pool to include diverse educational, socioeconomic, and cultural backgrounds, ideally across multiple regions or countries. This would improve the generalizability and applicability of the findings in a global health context. Moreover, integrating objective data collection methods, such as usage analytics from AIGC platforms and standardized cancer knowledge assessments, would enhance the precision and robustness of the findings. Finally, exploring additional moderators and mediators, such as digital literacy and socioeconomic factors, could provide deeper insights into how AIGC shapes health outcomes, informing more targeted and effective interventions in cancer education and control.
Footnotes
Acknowledgments
Thanks to all the participants in the survey.
Author Contributions
ZX: Research design, data collection and analysis, paper writing, and paper revision. RC: Research design, paper discussion, and paper revision. WD: Research design, paper discussion, and paper revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
