Abstract
Introduction
This study aimed to describe the course of patients with lung cancer from treatment initiation to end-of-life.
Methods
This retrospective cohort study used Claims Data from the National Health Insurance and Advanced Elderly Medical Service System. We analyzed data from patients newly diagnosed with lung cancer between April 2013 and March 2021 who had been hospitalized at our University Hospital in urban area. We evaluated (1) treatment courses of these patients, and (2) end-of-life care and clinical factors related to end-of-life care.
Results
A total of 818 patients who were diagnosed with lung cancer and received lung cancer treatment were included, of whom 200 were assessed for end-of-life. During the study period, 464 patients underwent surgery, while 308 received chemotherapy without undergoing surgery. The patients generally received lung cancer treatment within 2 years. The median time from initial treatment to death was 13 months for deceased patients. Patients in the palliative care unit (PCU) constituted 9% at 30 days before death, 25% at 7 days before death, and 40.5% on the day of death, whereas only 15% died at home. The prevalence of end-of-life care in the home/PCU was elevated among patients receiving molecular targeted drugs and in the female group.
Conclusion
This study highlighted the patterns of end-of-life care following lung cancer treatment by using Claims Data. PCU utilization was concentrated in the period shortly before death, with fewer patients passing away at home. End-of-life care may be influenced by clinical factors, including the type of lung cancer treatment received, which may change the place of care.
Introduction
Advances in lung cancer treatment have improved survival rates, especially in non-small cell lung cancer (NSCLC) and non-elderly patients.1,2 However, lung cancer remains the leading cause of cancer death, annually claiming the lives of 1.8 million people worldwide. 3 Among those with lung cancer who pass away, only 21% achieve their quality of life goals, 4 and 30-50% of Japanese patients with cancer receive insufficient physical pain management. 5 Therefore, it is crucial to consider and enhance end-of-life care for patients with lung cancer.
For patients with cancer, dying in their preferred place constitutes one of their major hopes. 6 More than 50% of the general public prefers home as the place of death. 7 The quality of death for patients with terminal cancer is considerably higher at home than at other places. 8 However, only 21.4% of Japanese patients with cancer die at home, while 70.8% die in hospitals. 9
The palliative care unit (PCU) is another preferred place; over 60% of the bereaved families of patients who had died in a PCU preferred it as the place of death, 7 with 37.6% felt that the PCU was like home. 10 However, half of the bereaved families considered the timing of referral to the PCU as late or too late. 11 Although the number of PCU beds is increasing in Japan, fewer than 10 000 beds are available, 12 which remains inadequate for the whole cancer patient deaths (380 000 deaths annually). 9
Aggressive treatment at the end-of-life is associated with poor quality of care,13,14 however, such treatment is still delivered.15,16 Previous reports about end-of-life care focused only on the terminal phase, but preparation for end-of-life care requires earlier attention. To evaluate end-of-life care for lung cancer, it is important to follow-up the patient from the beginning of treatment until death.
To provide a time series of a patients’ treatment progress, we used Claims Data with the characteristics of follow-up across facilities, which is not possible with a facility’s medical record. Claims Data are medical billing data issued monthly by medical institutions to bill insurers for medical expenses.17-19
The primary objective of this study was to follow-up the patients across medical institutions using Claims Data and assess the course of patients with lung cancer from treatment initiation to end-of-life. Additionally, we aimed to show the association between patient characteristics or lung cancer treatment and end-of-life care.
Patients and Methods
Study Design and the Data Source
This retrospective cohort study used Claims Data in Kyoto Prefecture between April 2013 and March 2021. Kyoto Prefecture, with a population of approximately 2.6 million, is a microcosm of Japan, with urban and rural areas. Most (63%) of the prefecture’s population is concentrated in the Kyoto-Otokuni Medical Area (urban), which has several advanced medical hospitals, including the University Hospital Kyoto Prefectural University of Medicine (KPUM).
The database contains Claims Data for approximately 1 million patients annually, covering approximately 40% of the residents of Kyoto Prefecture (Table S1). Two major insurance systems are in the database: National Health Insurance and Advanced Elderly Medical Service System. The former covers self-employed persons, retirees, and their families, and the latter covers mainly those aged ≥75 years. Clinical information in the database includes patient characteristics such as sex, age, and area of residence; date of medical examination; name of medical institution; disease information such as ICD-10 codes and the date of medical treatment; all medical treatment carried out; death; and nursing home services. The database includes medical information across hospitals, even if a patient has visited several hospitals.
Study Population and Endpoints
Patients with new-onset lung cancer who received cancer treatments were included in this study. First, we identified patients with lung cancer (C34 in the ICD-10 code) and extracted the hospital admission history during the disease course. Second, we selected the patients receiving lung cancer treatment after September 2013. Using clinical information, we divided these selected patients into three groups as follows: (1) OP(+), those who underwent surgery for lung cancer (including those who underwent surgery after chemotherapy or radiation) assumed primarily early-stage lung cancer; (2) OP(−)CT(+), those who received chemotherapy without surgery for lung cancer assumed primarily advanced lung cancer; and (3) “others,” those who received only radiation (curative and/or palliative radiation) as lung cancer treatment (Figure 1, Table S2). This grouping is an alternative classification for the database that does not contain information on lung cancer stage. Patient flowchart. OP(+): patients who underwent surgery for lung cancer; OP(–)CT(+): patients who received chemotherapy without surgery for lung cancer; others: patients who receive only radiation (curative and/or palliative radiation) as a lung cancer treatment. The database does not allow for distinguishing between curative and palliative radiation.
The endpoints of this study are (1) to provide an overview of the entire treatment process for patients with lung cancer from treatment initiation to death, and (2) to assess end-of-life care and its association with clinical factors.
Data Collection
The index date was defined as the date the first lung cancer treatment was recorded. Patients’ ages were calculated in the first month with a definite disease name of lung cancer.
We collected medical details from all medical institutions in Kyoto prefecture throughout the treatment period. The medical details contained medical treatment for lung cancer treatment and end-of-life care, place of care, and death.
Overviews of the Entire Treatment
Transition of Treatment over the Entire Patient Journey
Lung cancer treatment consisted of surgery, radiation therapy, and chemotherapy (Tables S3–S5). We reviewed the three main treatment combinations every 3 months from the treatment initiation. The period during which patients did not undergo lung cancer treatment was defined as “no treatment.”
Changes in the Place of Care from Treatment Initiation
We evaluated the place of care monthly from treatment initiation, using the following categories: outpatient/home visit treatment, PCU, general ward (KPUM), general ward (other hospital), nursing home, and home/day care.
Assessments of the End-Of-Life
Deaths were identified using “death” outcomes in the database, medical claims for death certificate issuance and terminal care, and post-discharge outcomes from nursing care receipts. We evaluated end-of-life care for patients who died since April 2017, for whom daily data were available.
Medical Treatment at the End-Of-Life
End-of-life care, covering the 30 days prior to death, was categorized into aggressive treatment and palliative treatment. We defined aggressive treatment as follows: surgery, radiation therapy, chemotherapy, and molecular targeted drugs for driver gene mutations (MTD); thoracentesis; cardiopulmonary resuscitation; and intensive care unit (ICU) admission. This treatment indicates poor-quality end-of-life care.13,14 We identified palliative treatment as follows: oxygen administration, narcotic use, PCU admission, and palliative care team intervention. The palliative care team consists of physicians, nurses, pharmacists, and other professionals to manage and support physical and mental symptoms. The team intervention is covered by insurance and is actively promoted by the Ministry of Health, Labor, and Welfare in Japan. 11 Once patients received care with narcotic and palliative care team intervention during 30 days of end-of-life care, we assumed the care continued until death.
Place of End-Of-Life Care
We counted the places of care using the following categories: general ward, PCU, nursing home, and home. We evaluated the place of care daily in the 30 days before death.
Statistical Analysis
We used the Sankey diagram to illustrate the transition of treatment and place of care. This tool visualizes flow/transition over time effectively, 20 and is well suited to present a whole picture of real-world data, which may vary from patient to patient. We created the diagrams using SankeyMATIC. 21
All statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). More precisely, it is a modified version of R commander designed to add statistical functions frequently used in biostatistics. 22 The Kaplan–Meier method was performed to estimate overall survival. The stratified log-rank test was performed to assess between-group differences in overall survival. The Cochran-Armitage test was performed to test for trends in the transition of treatment places. The chi-square test was performed to compare clinical factors related to the place of death, and multivariate analysis was performed using logistic regression analysis. Statistical significance was determined as a P-value below 0.05.
We did not calculate the sample size because this was an exploratory study examining clinical factors related to end-of-life care.
Ethics
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of KPUM (approval number: ERB-C-2960; approval date: October 10, 2023). The reporting of this study conforms to STROBE guidelines. 23 All personal information in the database was anonymized and stored on a secure independent site; thus, the need for informed consent to participate and publish was waived.
Results
Patient Characteristics
Baseline Characteristics of all Enrolled Patients.
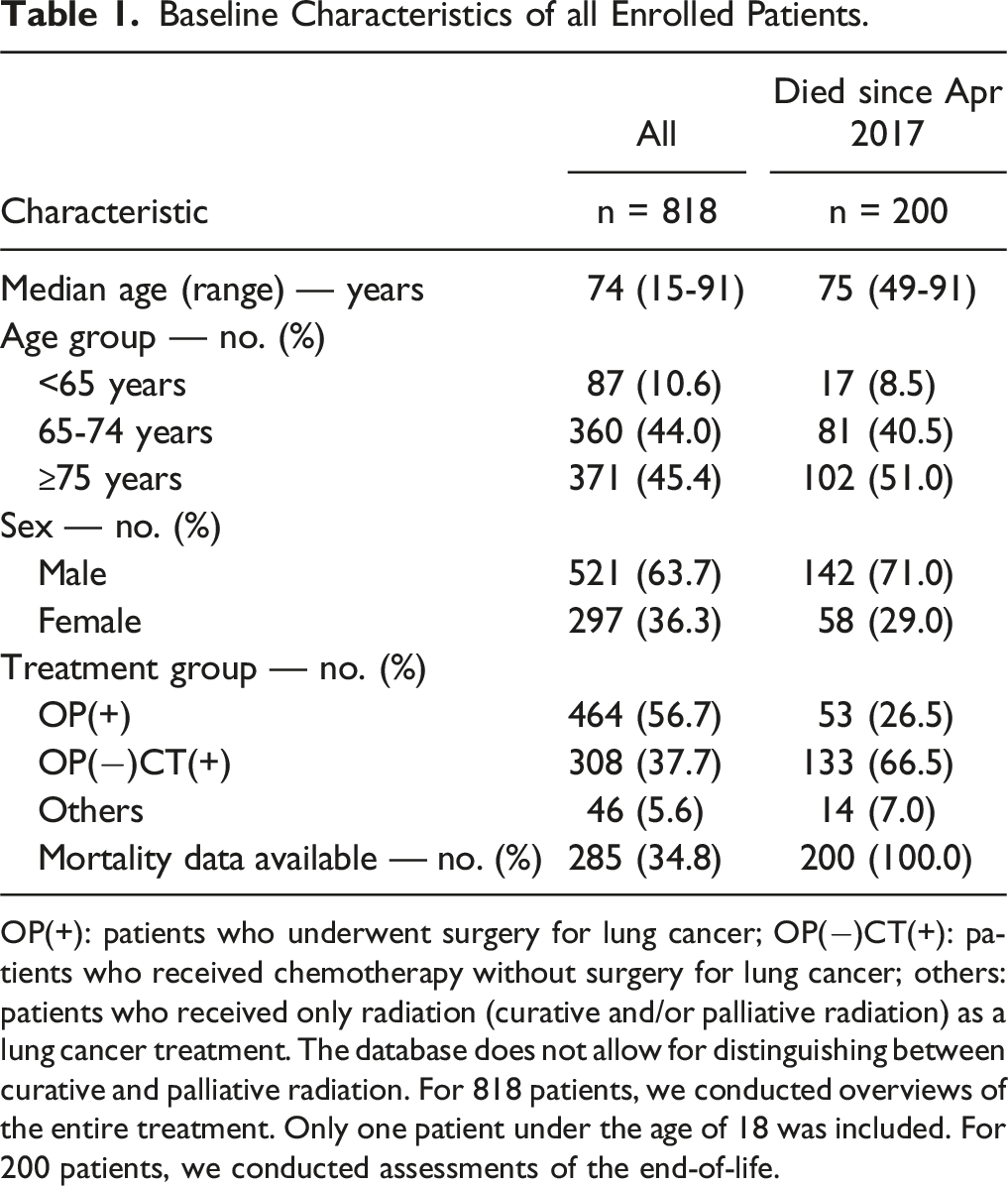
OP(+): patients who underwent surgery for lung cancer; OP(−)CT(+): patients who received chemotherapy without surgery for lung cancer; others: patients who received only radiation (curative and/or palliative radiation) as a lung cancer treatment. The database does not allow for distinguishing between curative and palliative radiation. For 818 patients, we conducted overviews of the entire treatment. Only one patient under the age of 18 was included. For 200 patients, we conducted assessments of the end-of-life.

Overall survival. The median overall survival for all patients was 51 months (95% confidence interval, 42–75). The 5-year survival rate for all patients was 48.3%.
Of the 285 patients who died, we evaluated end-of-life care for 200 who died after April 2017, when daily data were available. Of the 200 patients, 66.5% were in the OP(−)CT(+) group (Table 1).
The majority of censoring was due to database termination; of the 533 patients who were censored, 416 (78.0%) were censored when the database ended.
1) Overviews of the Entire Treatment
1–a) Transition of Treatment over the Entire Patient Journey
In the OP(+) group, 67.2% were treatment-free for the second 3 months, compared to 13.6% in the OP(−)CT(+) group (Figure 3(a)). In the OP(+) group, 37.1% received chemotherapy. For all patients, lung cancer treatment was generally undergone within 2 years. Most patients in the “no treatment” category were from the OP(+) group. Patient flow showing patterns of treatment and place The thickness of the lines indicates the number of patients belonging to them. (a) Transition of treatment content every 3 months over the entire treatment period. (b) TRAnsition of place by month from the start of treatment to the 30th month. PCU: palliative care unit; OP(+): patients who underwent surgery for lung cancer; OP(−)CT(+): patients who received chemotherapy without surgery for lung cancer. The majority of censoring was due to database termination; of the 533 patients who were censored, 416 (78.0%) were censored within 3 months before the database ended (after December 2020). (a) Lung cancer treatment was generally administered within 2 years. Most patients in the “no treatment” category were from the OP(+) group. In the OP(+) group, 47.7% of patients who received chemotherapy were treated with the UFT (Tegafur–Uracil) regimen, which is used for adjuvant treatment of NSCLC with stage IAc-IIA in Japan. (b) In the OP(+) group, hospitalization in the general ward was 39.2% at 1 month after treatment initiation, while in the OP(−)CT(+) group the length of general ward stay was relatively prolonged.
Until the fourth year, the number of deaths increased continuously, then stabilized in both the OP(+) and OP(−)CT(+) groups. The median survival time among deceased patients was 13 months from initial treatment (24.5 months in the OP(+) group and 13 months in the OP(−)CT(+) group).
1–b) Changes in the Place of Care from Treatment Initiation
In the OP(+) group, the percentage of patients staying in the general ward declined rapidly, with 39.2% at 1 month, 19.0% at 2 months, and 13.6% at 3 months after treatment initiation (Figure 3(b)). In contrast, the length of hospital stay in the general ward was relatively prolonged in the OP(−)CT(+) group: 72.7% at 1 month, 37.3% at 2 months, and 25.6% at 3 months after treatment initiation.
2) Assessments of the End-Of-Life
2–a) Medical Treatment at the End-Of-Life
There were 144 patients (72.0%) admitted to the general ward at least once during the 30 days before death.
Percentage of Aggressive Treatment Given in the 30 days Before Death.

MTD, molecular targeted drugs for driver gene mutations; ICU, intensive care unit. The database does not allow for distinguishing between curative and palliative radiation.
Regarding palliative treatment, 83 (41.5%) patients were admitted to the PCU at least once. Once hospitalized in the PCU, most patients remained there until death (81 patients, 94.0%), and their median length of stay in the PCU was 11 days. The percentage admitted to the PCU increased from 9.0% at 30 days before death to 40.5% on the day of death, with a considerable increase from approximately 2 weeks before death (Figure 4). The percentage of treatment at 30, 7, and 0 days before death was 9.0%, 25.0%, and 29.0% for oxygen; 8.0%, 36.0%, and 42.5% for narcotics; and 2.0%, 18.5%, and 22.0% for palliative care team intervention, respectively. Eighty-five (42.5%) patients died without receiving comprehensive palliative care, such as PCU admission or palliative care team intervention in the 30 days before death. Palliative treatment and hospitalization during the 30 days before death. PCU: palliative care unit. Narcotic use, oxygen administration, and palliative care team intervention were counted before admission to the PCU. Those palliative treatments cannot be extracted in the database after patients are admitted to the PCU. Patients received comprehensive palliative care during their stay in the PCU. The percentage of patients in PCU increased from 9.0% at 30 days before death to 40.5% on the day of death.
2–b) Place of End-Of-Life Care
At 30, 7, and 0 days before death, the percentage of patients who were at home decreased progressively, with 112 (56.0%), 58 (29.0%), and 30 (15.0%) patients, respectively (P for trend <0.001) (Figure 5). In contrast, the number of patients in the PCU increased at these time points: 18 (9.0%), 50 (25.0%), and 81 (40.5%), respectively (P for trend <0.001). The general ward accommodated 68 (34.0%), 92 (46.0%), and 89 (44.5%) patients, whereas 2 (1.0%), 0 (0%), and 0 (0%) were in a nursing home. A total of 44 (75.9%) women were at home/PCU on the day of death compared with 67 (47.2%) men, with an odds ratio of 2.55 (95% CI, 1.18-5.52) (Figure 5, Table S6). Similarly, the odds ratio for those in the urban area to those in the rural area was 3.94 (95% CI, 1.77-8.78); the odds ratio for the MTD(+) group who received treatment with MTD to the MTD(−) group was 3.56 (95% CI, 1.26-10.1) (Figure S2). In the MTD(+) group, fewer patients were admitted to the general ward as the time of death approached (34.2%, 28.9%, and 15.8% at 30, 7, and 0 days before death, respectively), and the number of patients in the home/PCU gradually increased. Place of care at 30 days before death, 7 days before death, and on the day of death (a) all patients, (b) classified according to sex, (c) classified according to MTD use. PCU: palliative care unit; MTD: molecular targeted drugs for driver gene mutations. On the day of death, only 30 patients (15.0%) were at home. Patients at home decreased as death approached (P for trend <0.001), whereas those in the PCU increased as death approached (P for trend <0.001). Comparisons of clinical factors related to place on the day of death are presented in Supplemental Table S6.
Discussion
We described the course of patients with lung cancer from treatment initiation to end-of-life care across medical institutions using Claims Data. We showed that treatment (e.g., MTD) and patient characteristics (e.g., sex) are associated with end-of-life care preparation, which influences the place of care and the timing of transfer.
The median time from initial treatment to death was only 13 months, leaving limited time for end-of-life planning.
Only 15.0% of deaths occurred at home, suggesting that systems and functions to support home care at end-of-life might be inadequate. The percentage of patients in PCU increased substantially just before death, from 9.0% at 30 days before death to 40.5% on the day of death. This study observed less aggressive treatment, with 15.0% and 2.5% of the patients receiving chemotherapy and ICU admission during the 30 days before death. However, 72.0% of the patients underwent general ward hospitalization during the 30 days before death, and 44.5% died in a general ward, highlighting the need for better end-of-life care settings.
The median length of stay in the PCU before death was only 11 days. Notably, 42.5% of the deceased patients died without receiving comprehensive palliative care during the 30 days before death. Patients may be insufficiently prepared for palliative care introduction, potentially influenced by the shortage of PCU beds in Japan 12 and a system that promotes shorter stays within 30 days from PCU admission due to fee-setting. It is desirable to increase the number of PCU beds and create an environment that enables patients to receive home care. Enhancing appropriate palliative care in general wards is also essential for patients who cannot spend their end-of-life at home or in the PCU.
We identified that 44.5% and 40.5% of all-cause deaths occurred in general wards and PCUs, respectively. This was similar to the results of a previous report from Australia, demonstrating that 42% of patients died from NSCLC in acute care hospitals and 42% in hospice settings. 24 In contrast, a report from France showed that the percentage of patients admitted to acute care wards increased markedly toward the end-of-life, with 72% being admitted at the time of death and 32% admitted 28 days before death. 25 The place and system of end-of-life care vary across countries. In another report, the proportion of lung cancer deaths at home ranged between 12.5% and 57.1%, whereas that in hospitals ranged between 27.5% and 86.5%, depending on the country. 26 The 15.0% home death rate in the present study was comparable to the 21.4% shown in Japanese government statistics 9 and could reflect the end-of-life care system in Japan. The current study predominantly comprised older adults, with 89.4% being aged ≥65 years and 45.4% aged ≥75 years, as compared to 80% aged ≥60 years in the report from Australia 24 and 76.4% aged ≥60 years with 42.6% aged ≥70 years in the report from France. 25 However, in Japan, patients in their 70s with lung cancer were the largest age group, 27 making the study population likely representative.
Relatively more patients among females, MTD(+) group, and urban area died in the preferred place, home/PCU. A report on causes of death among 5-year survivors of lung cancer stated that male patients were more likely to die from causes other than lung cancer. 28 For this reason, male patients in the present study may have died more frequently in the general ward. MTD(+) patients appeared to be more prepared for the end-of-life, with general ward admissions decreasing as they became closer to death. The MTD(+) group had a relatively long prognosis, 29 granting more time to plan their end-of-life care planning. General wards were more common as the place of end-of-life care outside of urban areas, thought to be influenced by regional disparities in the medical system. Physicians should plan end-of-life care considering local medical resources.
This study is strengthened by its ability to follow-up patients across medical institutions from the initial treatment to end-of-life care. Furthermore, palliative care outside the PCU, including narcotic use and palliative care team intervention, could be evaluated. This study is one of the few to demonstrate that physicians could improve preparations for their patients’ end-of-life care.
Limitations
Our study has some limitations. First, the stage of lung cancer and the presence of distant metastases were unknown in the database used; instead, we grouped patients based on their treatment to infer their stage. The 5-year survival rates were comparable to the measured survival rates in the cancer registries (41.9% overall, 73.8% in stage I, 47.5% in stage II, 26.7% in stage III, and 7.2% in stage IV), 30 suggesting that the grouping based on treatment was reasonable. Second, the accuracy of death records and diagnosis codes in the database remains a concern. Patients who were hospitalized or diagnosed with cancer exhibit a high sensitivity and specificity for death31,32; thus, mortality accuracy was not a disadvantage in the present population. Diagnosis codes used for insurance claims should be combined with medical data to enhance their quality33,34; we extracted the diagnostic codes along with information on lung cancer treatment. Third, medical administrative data have limited validity in determining disease status.33,34 Cases already receiving treatment before the database initiation are misidentified as “first treatments.” 35 We excluded patients whose first lung cancer treatment began less than 6 months after the start of data collection. Fourth, the database was censored due to insurance changes. We addressed this by combining the insured person’s number for long-term care insurance, which remained constant throughout life, including birth month, sex, place of residence, disease name, treatment details, and medical institution. Fifth, this study’s target population was limited, with a bias toward older and urban residents. The age demographics of patients with lung cancer in Japan 27 and the population in this study were similar. Due to selection bias, national statistics are difficult to infer from the data in this study. This was an exploratory study, so we did not calculate the sample size accordingly.
Conclusion
Using Claims Data, we were able to follow-up patients with lung cancer from the initial treatment to death over an extended period. Few patients received aggressive treatment shortly before death. The percentage of patients who died at home was low. Approximately 40% of the patients used PCUs, although there is a bias toward the period shortly before death, likely due to a shortage of PCU beds. End-of-life care preparation may relate to clinical factors, including lung cancer treatment, which influences the place of care. Physicians may be able to prepare better end-of-life care for their patients based on patient characteristics and clinical factors.
Supplemental Material
Supplemental Material - Patients’ Trajectory With Lung Cancer From Treatment Initiation to End-Of-Life: A Retrospective Cohort Study Using Claims Data in Japan
Supplemental Material for Patients’ Trajectory With Lung Cancer From Treatment Initiation to End-Of-Life: A Retrospective Cohort Study Using Claims Data in Japan by Yuri Ogura, Masahiro Iwasaku, Mami Ishida, Yuki Katayama, Naoya Nishioka, Kenji Morimoto, Chie Yamamoto, Shinsaku Tokuda, Yoshiko Kaneko, Tadaaki Yamada, Hideya Yamazaki, Masayoshi Inoue, Hiroshi Ikai, and Koichi Takayama in Cancer Control.
Footnotes
Acknowledgments
We thank the contributor of this study (Jun Okumura).
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Japan Society for the Promotion of Science KAKENHI (23K16334).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
