Abstract
Background and Objectives
This study aimed to assess if extensive surgical resection enhances outcomes in wild-type Isocitrate Dehydrogenase (IDH) butterfly glioblastoma (B-GBM) patients, despite the presence of Telomerase Reverse Transcriptase (TERT) mutation and Epidermal Growth Factor Receptor (EGFR) amplification.
Methods
The study, retrospectively conducted from 2014 to 2022, involved 723 GBM patients, 41 of whom met the criteria for IDH wild-type B-GBM. Exclusion criteria comprised prior diagnoses or treatments for low-grade glial tumors. Surgeons, employing two approaches—partial and extensive surgery—categorized patients based on age, sex, tumor location, corpus callosum involvement, and genetic characteristics. The interval between initial surgery and tumor recurrence/tumor-free period (TR/TFP) and overall survival (OS) were recorded and compared between the partial and extensive resection groups, analyzing the impact of resection width on TR/TFP and OS. Preoperative assessments utilized thin-section cranial computed tomography (CT) and contrast-enhanced magnetic resonance imaging (MRI). Intraoperatively, tumor excision was guided by sodium fluorescein, and margins were delineated via neuronavigation. Genetic alterations (TERT mutations and EGFR amplifications) were correlated with surgical type, TR/TFP, and OS. Karnofsky Performance Scale (KPS) evaluations were performed pre- and post-operatively and at key intervals, comparing outcomes between surgical groups. Standard radiotherapy and chemotherapy regimens were administered to all patients.
Results
Extensive resection yielded significantly longer TR/TFP compared to partial resection, despite TERT gene mutation and EGFR amplification being linked to shorter TR/TFP and OS. Its impact on OS, however, was not significant. KPS scores indicated a superior quality of life after extensive resection, with sustained improvement upon recurrence.
Conclusions
Extensive resection of B-GBM, even in the presence of adverse genetic alterations, may prolong TR/TFP, offering patients a period of improved comfort with minimal distress.
Keywords
Introduction
Traditionally, the treatment of Butterfly Glioblastoma (B-GBM) has been fraught with difficulties due to the tumor’s aggressive nature, high recurrence rates, and overall poor prognosis. Historically, this has led most surgeons to opt for conservative treatment approaches over extensive surgery, primarily due to the high risks associated with aggressive resection in these cases. 1
Despite the historically favored conservative approach, there has been a growing demand for more radical surgical treatment methods for B-GBM.2–4 This shift is likely influenced by the limitations of conservative treatments in effectively managing these tumors and improving patient outcomes.
Improved imaging techniques allow for more precise surgical planning, enabling more extensive tumor resection while minimizing damage to healthy brain tissue. 5 Studies have shown that extensive resection surgery significantly enhances the effectiveness of recently initiated treatments, such as immunotherapy and chemotherapy, against genetic mutations, emphasizing that the smaller the remaining tumor mass, the greater the treatment efficacy.6,7
Advances in genetics have aided in more accurate diagnosis, classification of tumor types, and in formulating tailored treatment strategies based on individual patient genetic profiles.8,9
Recent studies have shown that EGFR amplification is predominantly found in high-grade gliomas, especially in IDH wildtype and TERT mutated gliomas. 10 Glioblastomas with EGFR amplification benefit from temozolomide-based therapies, highlighting the importance of determining EGFR status for personalized treatment approaches like immunotherapy. 11 TERT promoter mutations, found in majority of glioblastomas, are crucial for the limitless proliferation of cancer cells and are associated with poor prognosis due to increased TERT expression, contributing to their aggressive nature. 12 Understanding these genetic markers allows for the development of targeted therapies that can improve patient outcomes. Extensive surgical resection has been shown to enhance survival rates in the presence of these genetic alterations and underscores the importance of integrating genetic profiling into the treatment strategies for B-GBM to optimize therapeutic efficacy and extend patient survival.
Objectives and Prespecified Hypotheses
The primary aim was to assess if extensive surgical resection, compared to partial resection, improves outcomes in wild type Isocitrate Dehydrogenase (IDH) B-GBM patients, even in the presence of Telomerase Reverse Transcriptase (TERT) mutation and Epidermal Growth Factor Receptor (EGFR) amplification.
Additionally, the study aimed to evaluate post-operative quality of life using Karnofsky Performance Scale (KPS) scores and compare these scores between extensive and partial resection groups.
The core focus was to evaluate the outcomes of extensive surgical resection in managing B-GBM, exploring whether more radical surgical approaches, supported by advancements in diagnostic and genetic molecular science, could enhance outcomes for patients with B-GBM, given the aggressive nature of these tumors and their propensity for rapid recurrence.
Methods
This retrospective study, conducted between 2014 and 2022, involving 723 patients diagnosed with GBM, with 41 (5.6%) meeting the criteria for IDH wild-type B-GBM. This study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies. 13 Written informed consent was obtained from all study participants for the use and processing of their medical records for research purposes. All patient details have been de-identified to ensure that the identity of any individual cannot be ascertained in any way. Ethical approval was obtained from the Ordu University Non-Interventional Scientific Research Ethics Committee – Turkey (Decision No: 2024/03, Ethics Approval No: E−14647249-000-0986247, Approval date: 29.03.2024). The necessary permissions from the American Hospital’s management board to use and process patient files were also obtained. Patients with typical radiological patterns for B-GBM were included, while those previously diagnosed with low-grade glial tumors or with a history of glial tumor treatment were excluded. Only patients with IDH wild-type GBMs were included in the study, with IDH-mutant cases being excluded.
Surgical cases performed by surgeons with two different surgical approaches were categorized into two groups: partial and extensive surgery. Patients were classified according to age, sex, tumor location, affected regions of the corpus callosum (rostrum, genu, body, and splenium), and pathological findings.
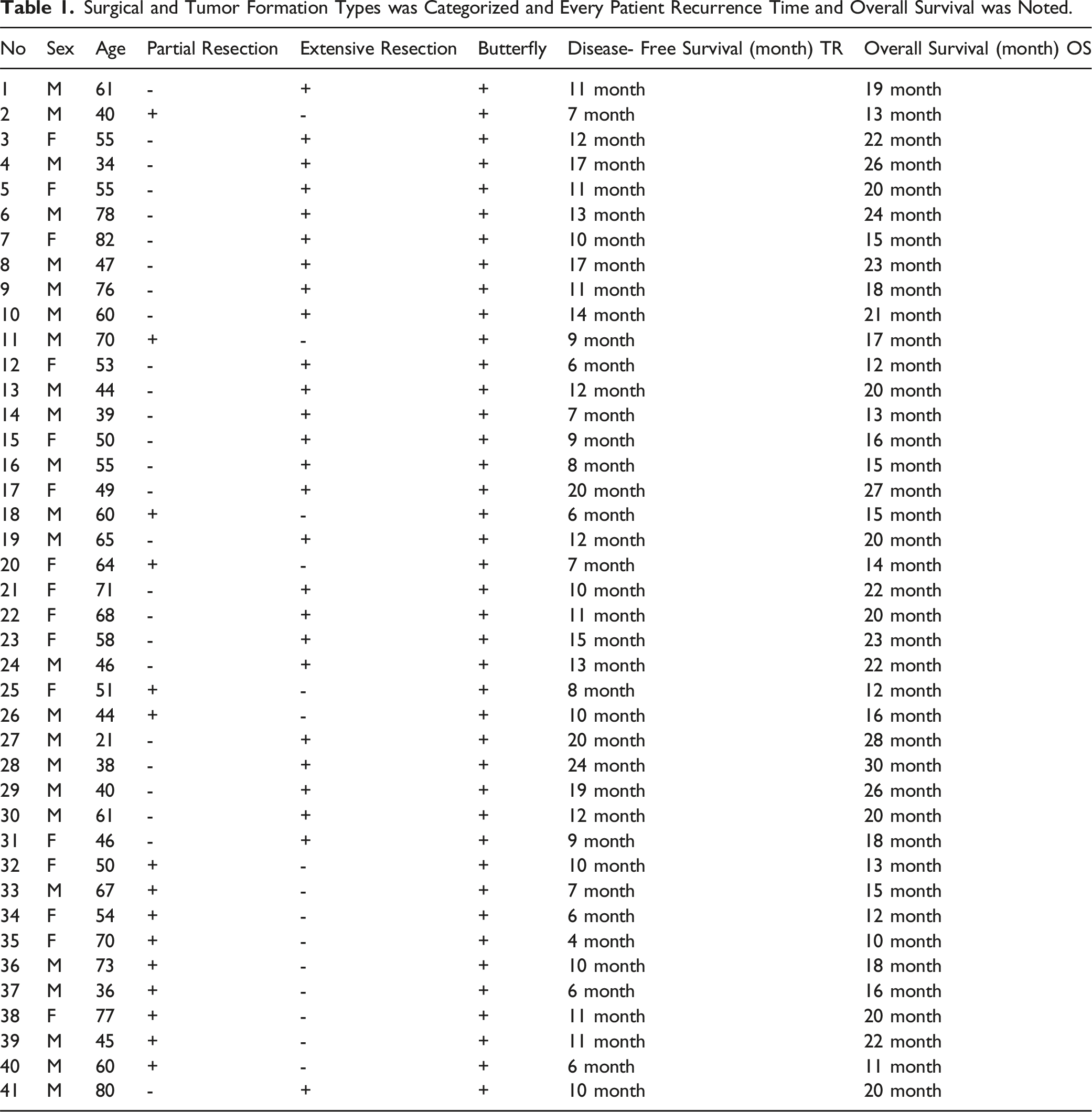
Surgical and Tumor Formation Types was Categorized and Every Patient Recurrence Time and Overall Survival was Noted.
Comparison of Variables by Resection. The Table Provides the Number of Individuals (n) in Each Resection Group and the Corresponding Percentages for Categorical Variables. Also the Table Provides the Mean and Standard Deviation (Ss) as Well as the Median and Range (Min-Max).
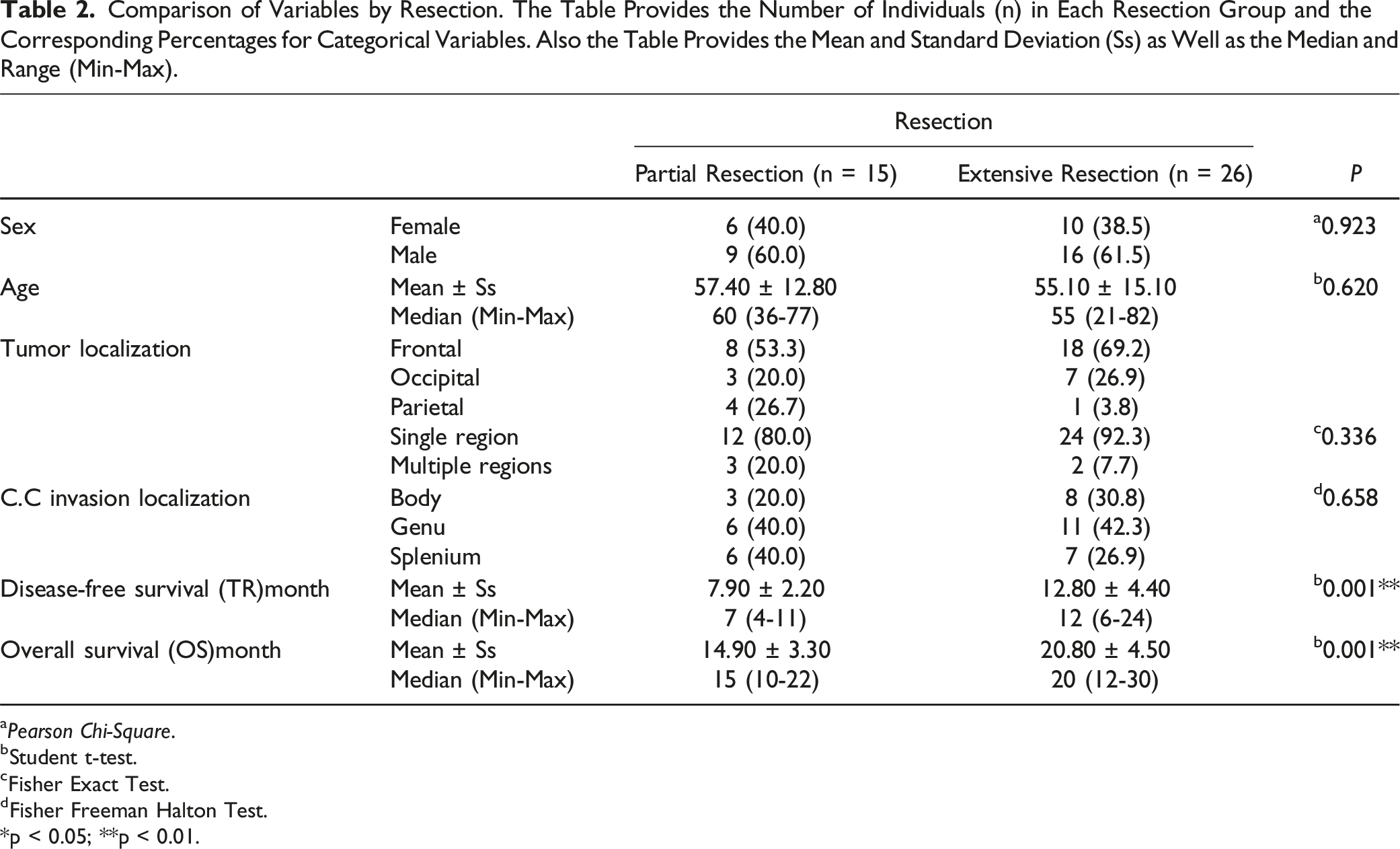
aPearson Chi-Square.
bStudent t-test.
cFisher Exact Test.
dFisher Freeman Halton Test.
*p < 0.05; **p < 0.01.
All patients underwent thin-section cranial computed tomography (CT) and contrast-enhanced T1, Fluid-Attenuated Inversion Recovery (FLAIR), and T2-weighted magnetic resonance imaging (MRI) using a neuronavigation device. Preoperative radiological findings were evaluated using a Stealth Station Surgical Navigation System (Medtronic, MN, USA), and resection margins were marked within safe limits.
To ensure maximum safe resection, possible tumor areas around the main mass were examined and marked on preoperative Contrast enhanced and FLAIR sequences. Sodium fluorescein was intravenously administered at the beginning of craniotomy. The yellow-stained tumor mass was excised, and the periphery was controlled with a neuronavigation device. Resection margins were extended to the FLAIR areas while avoiding damage to critical structures (Figure 1). All patients underwent MRI evaluation within 24 hours postoperatively, and the patients who had partial and extensive resections were identified. (A) preoperative FLAIR and contrast-enhanced axial MR images. (B) Postoperative FLAIR and contrast-enhanced axial MR images. To avoid damage to the eloquent area, maximum safe resection was made until FLAIR borders under the guidance of SFG microscope and neuronavigation.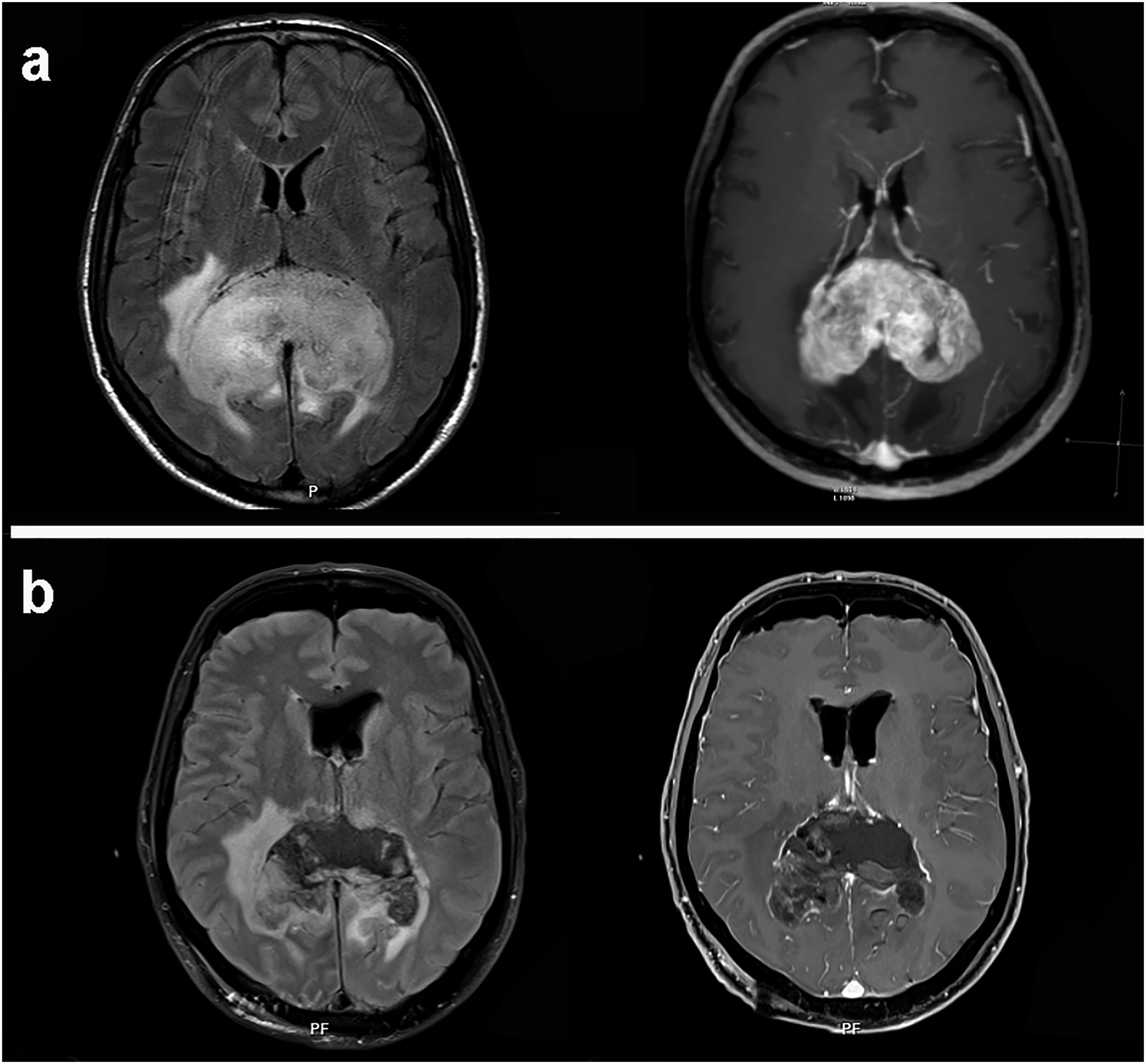
Genetic mutation for each patients.

Evaluation of TR/TFP and OS According to TERT and EGFR Genes in Partial Resection and Extensive Resection Groups.

Mann Whitney U test *P < 0.05; **P < 0.01.
Karnofsky Performance Scale Based on Pre and Post-Operative Follow-up.

All patients received standard radiotherapy and chemotherapy protocols as part of their treatment regimen.
Statistical Analysis
Number Cruncher Statistical System (NCSS) 2020 Statistical Software (NCSS LLC, Kaysville, Utah, USA) was used to perform the statistical analyses of the findings obtained in this investigation. When analyzing the study data, quantitative variables were represented by mean, standard deviation, median, and minimum and maximum values, while qualitative variables were shown using descriptive statistical methods such as frequency and percentage. To assess the normality of the data distribution, Shapiro-Wilk test and Box Plots were used. For quantitative two-group comparisons that showed normal distribution, the Student t-test was preferred, while the Mann-Whitney U test was employed to analyze variables that did not show normal distribution. To compare qualitative data, Pearson Chi-Square test, Fisher’s Exact test, and Fisher-Freeman Halton test were utilized. The results were evaluated at the 95% confidence interval and the significance level of P < 0.05.
Results
A total of 41 cases (5.6%) underwent surgery, comprising 16 females (39%) and 25 males (61%), with ages ranging from 21 to 82 years (mean age of 55.90 ± 14.20 years). Tumors were predominantly localized in the frontal region (63.4%), followed by the occipital (24.3%) and parietal (12.1%) regions. Corpus callosum invasion was distinctly observed, with 41.5% at the genu, 31.7% at the splenium, and 26.8% at the body in patients (Table 2).
Of those included in the study, partial resection was performed in 36.6% (n = 15) and extensive resection in 63.4% (n = 26). The mean TR/TFP for the partial resection group was 7.90 ± 2.20 months (median: 7 months) and for the extensive resection group was 12.80 ± 4.40 months (median: 12 months). The mean OS for the partial resection group was 14.90 ± 3.30 months (median: 15 months), while for the extensive resection group, it was 20.80 ± 4.50 months (median: 20 months). The TR/TFP and OS values for the extensive resection group were significantly higher than those for the partial resection group. (P = 0.001; P < 0.01) (Table 2).
TERT positive patients who had partial resection had shorter mean TR/TFP and OS durations (6.25 ± 1.17 and 13.13 ± 2.17 months) compared to TERT negative patients (9.71 ± 1.38 and 17.00 ± 3.37 months) (P < 0.01, P < 0.05). In the extensive resection group, TERT positive patients had shorter mean TR/TFP and OS (11.53 ± 4.47 and 19.47 ± 4.75 months) than TERT negative patients (15.22 ± 3.15 and 23.22 ± 2.77 months) (P < 0.01, P < 0.05). TERT positive patients who had extensive resection had significantly longer mean TR/TFP and OS times than those who had partial resection. (P = 0.001; P < 0.01) (Table 4).
In the partial resection group, EGFR amplification did not have a statistically significant impact on TR/TFP and OS between patients with or without amplification. However, in the extensive resection group, EGFR amplification was associated with shorter TR/TFP and OS durations compared to patients without amplification (P < 0.05). Patients with positive EGFR amplification who underwent extensive resection had a mean TR/TFP and OS of 10.85 ± 3.44 and 18.15 ± 3.95 months, respectively, while those who underwent partial resection and had EGFR amplification had a mean TR/TFP and OS of 7.33 ± 1.53 and 15.00 ± 2.00 months, respectively. The survival was longer in the extensive resection group, but the difference was not statistically significant due to the limited number of cases. (P > 0.05) (Table 4).
Comparison of Quality of Life Characteristics According to Resection.
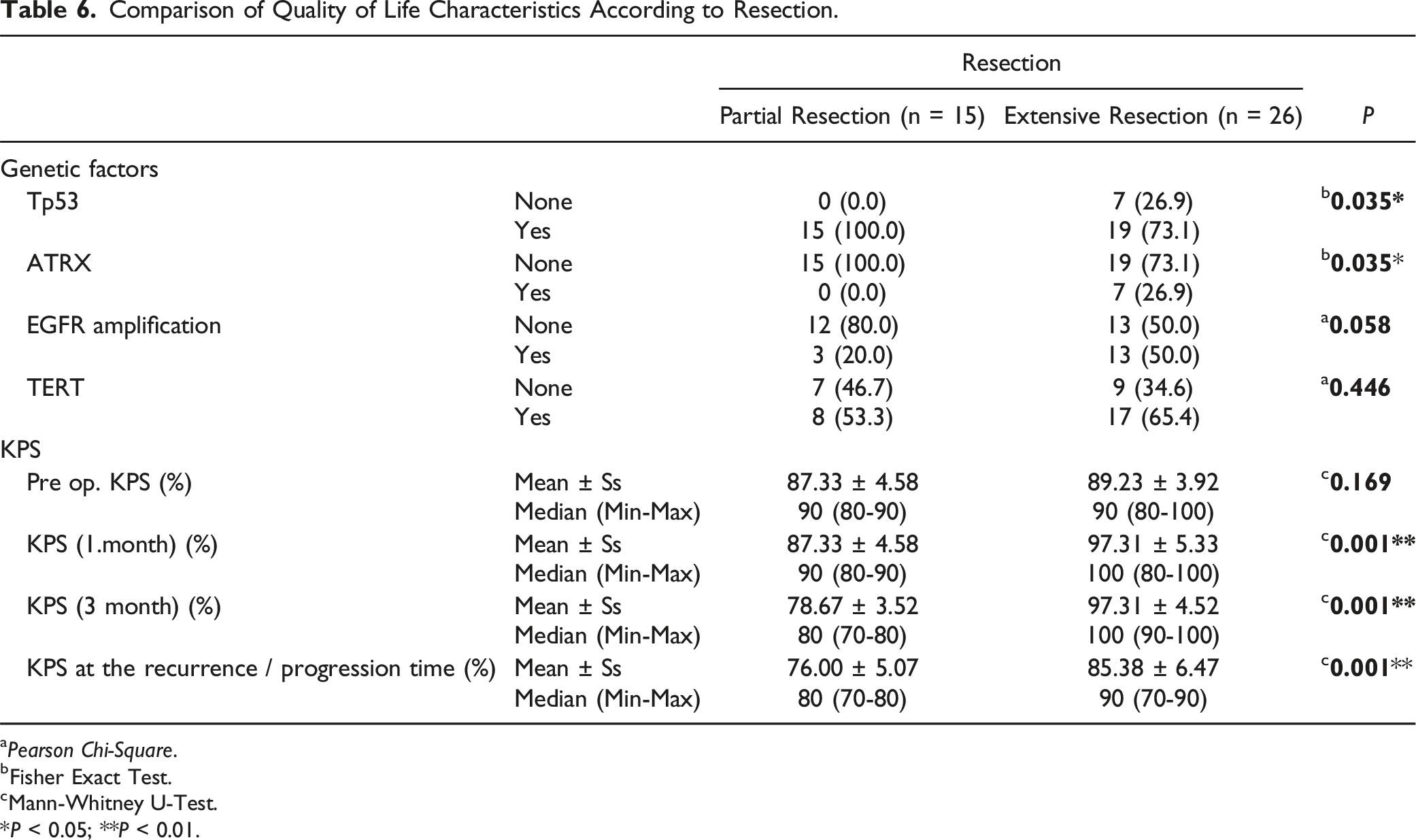
aPearson Chi-Square.
bFisher Exact Test.
cMann-Whitney U-Test.
*P < 0.05; **P < 0.01.

This figure demonstrates two distinct curves: one representing the extensive resection group and the other representing the partial resection group. The curve for the extensive resection group initially rises, indicating an improvement in KPS scores and quality of life, before declining as the disease recurs. Conversely, the curve for the partial resection group shows minimal improvement from the beginning and continues at a lower level. Ultimately, at the point of recurrence, the curves for both surgical types converge, indicating a prolonged period of wellness in the extensive resection group.
Discussion
Our study focuses on improving the quality of life for B-GBM patients during their survival. There is conflict about the preferred method of treating B-GBM as most authors believe that an extensive surgery will not be effective in extending patients’ lifespans, leading to non-surgical options being preferred.2,14,15
Current treatment options for newly diagnosed B-GBM are surgical resection or biopsy, followed by radiotherapy and/or chemotherapy. 16 However, there is uncertainty about the role of surgical resection in the treatment of B-GBM. Considering the limited life expectancy of patients, in previous years, they were considered inoperable and chemotherapy and radiotherapy were recommended after biopsy.
Although surgeons advocate biopsy as the preferred treatment option because of the unfavorable location and the low risk–benefit ratio of resection, increasing evidence shows the benefits of removing these tumors to achieve cytoreduction. 17
In our study, partial resection was chosen particularly in cases where the complex structure of Butterfly glioblastomas and their proximity to functionally significant areas posed a high risk for neurological damage if complete resection were attempted. Partial resection was deemed the most appropriate strategy to achieve the best possible outcome for the patient while preserving quality of life. This decision was not random but was made after careful consideration of the tumor’s location and the patient’s overall condition during preoperative multidisciplinary discussions.
Partial resection of glioblastoma often leaves residual tumor tissue, resulting in persistent peritumoral edema. This edema arises due to blood-brain barrier disruption by remaining tumor cells, fluid leakage into the surrounding brain tissue, and the inflammatory response induced by surgery. These processes increase intracranial pressure (ICP), compress brain structures, impair their function, and lead to symptoms and neurological deficits. These symptoms significantly reduce patient performance and quality of life. Increased ICP can also lower cerebral perfusion pressure, causing ischemic damage. 18 Severe edema can lead to brain herniation, a life-threatening condition, and patients may experience seizures due to increased pressure and irritation.19,20
Peritumoral edema also negatively impacts the efficacy of postoperative chemotherapy and radiotherapy. A larger residual tumor mass associated with edema creates a hypoxic environment, making tumor cells less sensitive to radiation and some chemotherapeutic agents. The irregular blood supply in edematous regions hinders chemotherapy delivery to the tumor cells. Additionally, edematous regions often contain isolated tumor cells with enhanced migratory and invasive capabilities, contributing to tumor regrowth and recurrence.19,20 Managing peritumoral edema and achieving maximum safe resection may be crucial for improving patient outcomes, alleviating symptoms, enhancing quality of life, and increasing the effectiveness of postoperative therapies.
Chojak et al. revealed that resection can significantly reduce the 6-month mortality rate compared with biopsy alone, though no significant difference was found in 12- and 18-month survival rates. 14 Similarly, Chawla et al. found that surgical resection, even when subtotal, was associated with better overall survival (OS) and positive survival expectations compared to biopsy, without a significant difference in postoperative complications. 21
Jamshidi et al stated that MRI-guided laser interstitial thermal therapy (LITT) is a safe and effective method in the treatment of deeply located brain tumors. In addition, they stated that bilateral LITT can be a safe and effective alternative to surgery for the treatment of B-GBM. 22
Their studies indicate that more extensive surgical approaches lead to a more favorable prognosis.1,15,23,24 However, the average tumor-free period is 1.5-6 months, with an overall survival period of approximately 12-18 months. Even with extensive resection, radiotherapy, and chemotherapy, tumor recurrence appears inevitable.25–27
Recently, the concept of supratotal resection has emerged to increase survival, based on the finding that most recurrences were observed in the periphery of resected tissues.28,29 The extension of resection beyond the contrast-enhanced portion into the MR hyperintense tissue may be a crucial aspect of tumor surgery, as it targets the regions most likely to harbor residual tumor cells. It is often visible on FLAIR images. Consecutively, it can positively affect the OS. 30
At present, several neurosurgical advances aim for a better prognosis, such as implementing intraoperative neuronavigation in combination with ultrasonography, fluorescence-guided surgery (FGS), intraoperative stimulation brain mapping, and even awake surgery in some neurosurgical centers. 31 Moreover, FGS with staining methods helps in resecting the tumor mass more extensively, but there are some limitations. Because of the insufficient diffusion of the dye in tissues near the tumor border, the surgeon may be unable to resect all afflicted areas. Otherwise, non-affected structures may be stained, and this may lead to the resection of critical locations. 32
In addition, some gene mutations and enzymatic changes may influence the operative outcomes. 9 The mutation of IDH enzymes plays a definite and important role in the response of these tumors to medical and radiological therapies. IDH1 and IDH2 mutations occur in most cases of low-grade and secondary high-grade gliomas, albeit not so favorable in IDH wild-type GBM. P53 and ATRX mutation may influence prognosis.31,33–35 TERT mutation and EGFR amplification may adversely affect the prognosis and OS of patients with GBM.36–38
We believe our study may be novel. While numerous genetic factors influencing glioblastoma prognosis have been studied, the literature may lack research specifically on B-GBM, combining an extensive surgical approach with the analysis of TERT and EGFR gene comparisons and their impact on outcomes.
As a result of our study, while no superior results were achieved in patients’ lifespan compared to the figures in the literature, a significant difference was observed in the extension of the disease-free period, even with adverse gene mutations. Patients experienced an extended period of wellness during this time. The extensive resection group exhibited significantly longer TFP/TR compared to the partial resection group, with recurrence occurring after a longer duration. Despite the longer OS associated with extensive procedures, it did not significantly increase the OS period known in B-GBM; patients succumbed after a certain period. Some disproportions in our results indicate that rapid recurrence occurred in certain patients following extensive surgical resection. The primary factor was the adverse influence of gene mutations on TR/TFP and OS, despite adequate surgical intervention. Nonetheless, the surgical approach demonstrates comparable efficacy to gene mutations in prognosis.
Although there were potential risks associated with surgical removal of the Corpus callosum, we did not encounter any permanent dysfunctions with extensive resection, except for attention and memory impairments that improved rapidly in the first month of our follow-up.3,17
Patients’ performance predominantly increased after extensive surgical resection during the 1st and 3rd months of follow-up. However, KPS scores decreased with tumor regrowth. At the time of recurrence, the KPS scores approached the values observed in the partial resection group as illustrated in Figure 2. Ultimately, at the point of recurrence, the curves for both surgical types converged. Nevertheless, the performance of patients in the extensive resection group remained better at the time of tumor recurrence, indicating a prolonged period of wellness, except for two who developed cerebral edema and had lower KPS levels than preoperative ones at the time of recurrence (Table 4).
This study has limitations that warrant consideration. Firstly, the mutations examined were limited to TERT and EGFR amplification, and other potential genetic factors impacting B-GBM prognosis were not explored. Secondly, the sample size, particularly the number of patients with IDH wild-type B-GBM, was relatively small, which may have limited the statistical power and generalizability of the results. Additionally, the retrospective nature of the study may have introduced biases related to data collection and patient selection. Since the study was conducted at a single center, the generalizability of the results to other populations or settings may have been limited.
Our research has shown most butterfly tumors traverse the corpus callosum, particularly from the genu region. Investing how this affects disease progression and treatment outcome is crucial and warrants further exploration. Age and gender significantly influence butterfly tumor development, though these topics has not been thoroughly researched in our study. Nonetheless, it is important to note that the majority of our patients were male.
Despite the favorable KPS scale results after extensive resection, the potential influence of age and gender factors should not be overlooked.
Briefly, in our view, efforts to find a cure for B-GBM should focus on developing methods capable of influencing gene mutations to alter the disease course . 31 B-GBM patients should not be left untreated, nor should biopsy be the sole option, considering the likelihood of rapid recurrence and poor prognosis.
Conclusions
Extensive surgical resection in patients diagnosed with B-GBM may result in significantly longer TR/TFP and OS compared to partial resection. Despite the presence of adverse gene mutations such as TERT gene mutation and EGFR amplification, extensive resection may show potential in improving the quality of life and extending TR/TFP. However, its impact on OS may not be statistically significant, although it notably augmented TR/TFP.
Supplemental Material
Supplemental Material - The Impact of Extensive Surgical Resection of Butterfly Glioblastomas on Outcomes in the Presence of TERT Mutation and EGFR Amplification: A Retrospective Cohort Study
Supplemental Material for The Impact of Extensive Surgical Resection of Butterfly Glioblastomas on Outcomes in the Presence of TERT Mutation and EGFR Amplification: A Retrospective Cohort Study by Mehdi Hekimoglu, Ahmet Tulgar Basak, Mehmet Yigit Akgun, Hıdır Ozer, Utku Ozgen, Raha Maleki, Dina Saban, Tunc Oktenoğlu, Ali Fahir Ozer, and Mehdi Sasani in Cancer Control.
Footnotes
Acknowledgments
We would like to acknowledge that this work has been previously published as a preprint in ResearchGate. I would like to extend my special thanks to Pediatrist Dr Ali Khajehdehi organization and editing of this paper.
Author Contributions
Mehdi Hekimoglu: Contributed to the conception, Investigation ,Methodology and design of the study, data acquisition, analysis and interpretation of data, drafting and writing and critical revision of the manuscript. Ahmet Tulgar Başak: Contributed to substantial contributions to the conception, methodology and design of the work Hidir Ozer: Contributed to Resources, Data curation and acquisition, investigation and critical revision of the manuscript. Mehmet Yigit Akgun: Contributed to data acquisition, analysis and interpretation of data, and critical revision of the manuscript. Utku Ozgen: Contributed to Investigation and acquisition. Raha Maleki: Contributed to investigation and formal analysis. Tunc Oktenoglu: Contributed to visualization and supervision and conceptualization ideas. Ali Fahir Ozer: Contributed to conceptualization ideas, supervision and visualization. Mehdi Sasani: Contributed to Conceptualization Ideas, Methodology, Writing - review & editing and revising critical issues. All authors have read and approved the final submitted manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
