Abstract
Background
Despite the relatively low breast cancer incidence in Estonia, mortality remains high, and participation in mammography screening is below the recommended 70%. The objective of this register-based study was to evaluate incidence-based (IB) breast cancer mortality before and after the introduction of organized mammography screening in 2004.
Methods
Breast cancer deaths individually linked to breast cancer diagnosis were obtained from the Estonian Cancer Registry and used for calculating IB mortality. We compared age-specific IB mortality rates across 5-year birth cohorts and 5-year periods. Poisson regression was used to compare IB mortality for one age group invited to screening (50-63) and three age groups not invited to screening (30-49, 65-69, and 70+) during two periods before and after screening initiation (1993-2003 and 2004-2014). Joinpoint regression was used for age-standardized incidence and IB mortality trends.
Results
Age-standardized IB mortality has been decreasing since 1997. Age-specific IB mortality for birth cohorts never exposed to screening showed a continuous increase with age, while in cohorts exposed to organized screening the mortality curve flattened or declined after the age of first invitation. Significant decreases in mortality from 1993-2003 to 2004-2014 were seen in the 30-49 (age-adjusted rate ratio 0.51, 95% CI 90.42-0.63) and 50-63 (0.65, 95% CI 0.56-0.74) age groups, while no decline was seen in the 65-69 and 70+ age groups.
Conclusions
The age specific IB mortality curves in birth cohorts exposed to screening and the significant mortality decline in the target age group after the initiation of the organized program suggest a beneficial effect of screening. Improved treatment without screening has not reduced mortality in older age groups. Our results support raising the upper screening age limit to 74 years.
Introduction
Breast cancer (BC) is the most frequent malignant neoplasm and cause of death among women in Estonia and elsewhere.1-4 The estimated age-standardized (World) incidence of BC in Estonia was 62/100,000 in 2022, the third lowest in Europe, compared with 83/100,000 in European Union (EU-27) and over 100/100,000 in countries with the highest incidence. 5 However, the estimated age-standardized mortality in Estonia (13/100,000) was close to that in EU-27 (15/100,000). Relative survival from BC remains about 10% units lower in Estonia than that seen in Northern or Western Europe. 6
The total population of Estonia is around 1.3 million, of which women comprise 700,000. In 2021, about 800 new BC cases were diagnosed, with almost half in the 50-69 age group. 7 The total number of BC deaths in 2021 was 250.
In 2003, the European Commission issued a recommendation for organized population-based mammography screening for BC in women aged 50 to 69 as a means of lowering the cancer burden. 8 With screening procedures, the cancer can be detected at early stages when the treatment is more efficient, which consequently should decrease BC mortality. In Estonia, organized population-based mammography screening started in 2004, 9 with several pilot studies conducted in late 1990s, in 2002 and 2003. 10 From 2004 to 2020, mammography screening was organized based on stationary units in hospitals and two mobile units. A third mobile unit was added in 2021. During 2004-2018, the target population was limited to women aged 50-62 (63 for some birth cohorts) invited every two years. The average number of invited women was about 66,000. From 2018, women up to the age of 68 were gradually included, increasing the number of invited women to around 86,000. A considerable shortcoming of the screening program was the exclusion of women without health insurance until 2021 (about 95% of the working age population in Estonia are covered with insurance). The participation rate has remained well below the recommended 70%, being around 51% in 2006 and 56% in 2015 and 2016.11,12 Unemployed women, lower educated women and those not adhering to health behavior recommendations (eg, smokers) were shown to be less likely to have had a mammography. 13 In 2015-2018, 20% of all BC cases in Estonia and 49% of those diagnosed at 50-64 were detected at screening. 14
Previous studies of BC trends in Estonia have indicated that in parallel with continuous incidence increase, mortality started to slowly decrease in the second half of the 1990s. 15 This was accompanied by an increase in relative survival.16,17 Declining mortality together with consistent survival increases in Europe have been contributed to increasing proportions of women being diagnosed at an earlier stage, enabling more effective treatment. 18 The decrease in mortality in Estonia, which started before the introduction of population-based screening, suggests a combination of different factors, mainly earlier diagnosis because of rising public awareness and better accessibility of high-quality mammography but also new treatments becoming increasingly available. 16 There have been no studies looking specifically at the effect of screening on BC mortality in Estonia. Previous studies showing decreasing death-certificate-based mortality trends have not considered the time when BC was diagnosed and whether cancer may have been detected at screening. 14
The use of incidence-based (IB) mortality has been recommended as a valuable method to evaluate the impact of any intervention, including organized cancer screening programs.19-23 When using IB mortality, only BC deaths occurring in women diagnosed with BC during a specified time period will be taken into account, excluding other causes of death unrelated to early detection and/or BC treatment and BC deaths due to cancers diagnosed outside the specified time window (eg, before the introduction of screening). The resulting trends should show the combined effect of anticancer measures directed solely at BC.20,21 The additional advantage of IB mortality is the opportunity to associate cancer deaths with the period of and age at cancer diagnosis.
The Estonian Cancer Registry has collected population-based cancer incidence data linked to causes of death since 1968, enabling a long period of IB mortality analysis in relation to the initiation of organized mammography screening in Estonia in 2004. The aim of the study was to investigate IB mortality in birth cohorts invited and not invited to screening and compare IB mortality by age groups before and after the introduction of organized BC screening in Estonia.
Materials and Methods
Data Definitions
For this retrospective register-based study, pseudonymized data on incident cases of invasive BC (International Classification of Diseases, version 10 (ICD-10) code C50) diagnosed in women in Estonia in 1974-2019 and individually linked to causes of death were obtained from the Estonian Cancer Registry. IB deaths were defined as BC deaths (the underlying cause of death ICD-10 C50) occurring in 1974-2019 in women who had a first-time invasive BC diagnosis during 1974-2019. IB mortality trend is shown for 1979-2019 to allow for a 5-year burn-in period. BC trends were modeled with Joinpoint regression to obtain overall age-standardized incidence and IB mortality with annual percentage change (APC) and 95% confidence intervals (CI).
To compare the IB mortality rate across different birth cohorts (born during 1909-1978), we used the number of IB deaths and population counts for 5-year age groups and 5-year periods at the time of death. Subsequently, we applied derived rates per 100,000 person-years to corresponding cohorts. IB mortality trend is expressed per 100,000 person-years, using the LOESS (locally estimated scatterplot smoothing) technique for smoothing. We compared IB mortality in birth cohorts invited to organized screening during the study period (years of birth 1943-1969) with birth cohorts not invited to organized screening during the study period (years of birth 1909-1942 and 1970-1978), to provide background data on BC mortality curves in the absence of screening.
Additionally, we compared IB mortality during equal intervals of 1993-2003 and 2004-2014 (before and after the introduction of screening, respectively) in different age groups. During 2004-2014, only women aged 50-62/63 were invited to screening (Figure 1). Therefore, we divided the study population into four age groups (30-49, 50-63, 65-69, and 70+), to compare the group covered by screening (50-63) with those not covered by screening (30-49, 65-69, and 70+). Women aged 64 were excluded as they were invited in some years but not consistently, which would have complicated the interpretation of the results. IB deaths included in this analysis were restricted to those where the diagnosis of and death from BC for the individual occurred in the same period and within the same age group, minimizing the lead time and overdiagnosis bias. Birth cohorts invited to organized mammography screening, Estonia 2004-2019. The circles indicate the age of the birth cohort at the time of invitation. The year of invitation is indicated on the x-axis.
IB deaths were summed for respective age groups and time periods with national population figures of corresponding age and year used as the population at risk in stratified Poisson regression analysis to calculate corresponding IB mortality rates and risk ratios with 95% confidence intervals.
Statistical Analysis
Incidence and mortality rates were age-standardized to Segi’s World standard population. 24 The Joinpoint Regression Program (version 4.9.1.0) from the Surveillance Research Program of the US National Cancer Institute (https://surveillance.cancer.gov/joinpoint/) was used for joinpoint regression. Trend smoothing, Poisson regression analysis, and graphs were performed with Stata 17.0 (StataCorp. 2021. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC).
The reporting of this study conforms to STROBE guidelines. 25
Ethics Approval
The study protocol was approved by the Tallinn Medical Research Ethics Committee (Decision no 2636, February 14, 2019). The requirement to obtain informed consent was waived by the Tallinn Medical Research Ethics Committee for this register-based study.
Results
Joinpoint analysis of age-standardized rates for 1974-2019 showed a continuously increasing incidence trend with a small but statistically significant deceleration occurring around 1998 (from APC 2.3% to APC 1.5%) (Figure 2). For IB mortality (1979-2021), the modeled trend showed a significant increase until 1997 (APC 2.9%), after which the trend changed to a significant decline (APC -2.1%) until the end of the study period. Observed (dotted line) and modelled (solid line) age-standardized incidence (1974-2019) and incidence-based (IB) mortality rates (1979-2021) of breast cancer with annual percentage change (APC) and 95% confidence intervals, Estonia. *APC is significantly different from zero at alpha = 0.05.
Age-specific IB mortality rates for 5-year birth cohorts are presented in Figure 3. The IB mortality curves for women never exposed to organized screening (birth cohorts born in 1909-1942) show a continuous increase with age. IB mortality curves for birth cohorts exposed to organized screening display a distinctly different pattern showing stabilization or decrease immediately after the age of first invitation to screening (birth cohorts born in 1949-1953, 1964-1968) or later. Incidence-based (IB) mortality by birth cohorts and age, Estonia 1974-2019. Dots indicate age at first screening invitation.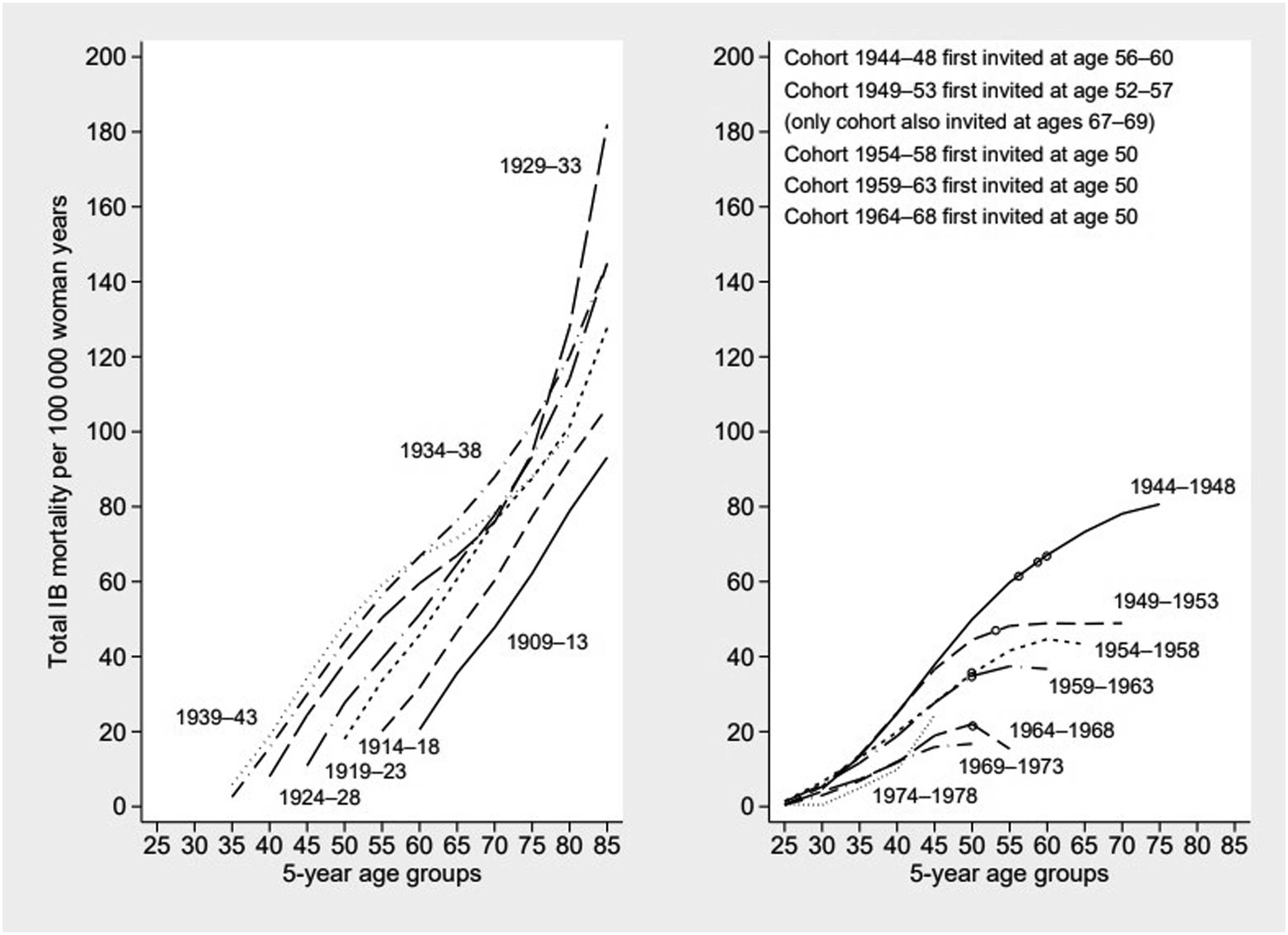
Breast Cancer Incidence-Based (IB) Mortality and Rate Ratios by Age Group in 1993-2003 and 2004-2014, Estonia.

Abbreviations: IB – incidence-based; CI – confidence interval.
Discussion
This population based IB mortality analysis showed that overall BC mortality started to decrease in Estonia in the late 1990s, several years before the introduction of organized mammography screening in 2004. However, birth cohort analysis revealed that age-specific BC mortality curve flattened or started to decline after entering screening age in birth cohorts invited to screening, while in earlier birth cohorts, mortality continued to increase with age. When comparing IB mortality before and after the introduction of screening, a significant decline was observed in young women not yet invited to screening (mortality decline 49%) and women aged 50-63 years who were invited to screening (mortality drop 35%). No mortality decline was seen in women 65 years and older who were not invited to screening during that period.
In Estonia, the organized screening program has not met all the requirements outlined in the recommendations of European Commission, being limited to women aged 50-62/63 (until 2018) and those with valid health insurance (until 2021). Furthermore, screening attendance has remained low, and increasing only from 41% to 58% during 2004-2014 with an average annual change of 1.7%. 26 In comparison, in the neighboring Nordic countries, the attendance in Finland has been consistently above 80%, with Denmark and Norway closely following. 26
The long-term trend of BC incidence and mortality in Estonia showed that from 1974 to 2019, the age-standardized BC incidence climbed steadily with slight variation in pace and without any evident screening-induced surge around or after 2004. A previous study on BC trends indicated a sudden increase in incidence in 2004 for the age group 50-59, limited to one year. 15 The trend of total IB mortality showed no apparent impact of screening, as IB mortality started declining before the 2000s. 14 However, our birth cohort analysis showed that in cohorts subject to multiple screening invitations, the IB mortality curve flattened with increasing age in some birth cohorts immediately after entering screening age, as opposed to the more upright increase in earlier cohorts not exposed to screening invitation. Previous research on the Dutch screening program, which began in 1989, showed that uninvited cohorts experienced a continuous increase in mortality with age, while invited cohorts presented a diverging trend within five years after screening invitation. 27 Afterward, mortality rates started to rise again, although at a lower rate than in uninvited cohorts. 27 In Lithuania, the pattern of BC mortality is very similar to Estonia, showing a major decrease since 1996. 28 The Lithuanian BC screening program was initiated in 2006 for women aged 50-69, and an age-period-cohort analysis showed cohort effects possibly associated with risk factor changes, particularly affecting younger generations, but also period effects suggesting the beneficial effect of increased mammography testing, as well as general improvements in early detection and new treatments. 28
The comparison of IB mortality between two periods before and after the introduction of screening in our study including only BC deaths that occurred within respective age groups and periods showed a reduction in IB mortality for younger women and those in the screening age group. The largest change occurred in the age group 30-49 not yet invited to screening with an almost 50% reduction, while in the screening age group 50-63, the reduction was smaller, though still amounting to 35%. Mortality reduction in the youngest age group can be partly attributed to earlier diagnosis through better awareness, which may have been an indirect consequence of extensive screening-related BC communication measures. 29 Early detection has become increasingly available for women outside screening age group with several large hospitals opening breast health offices that patients may visit without a referral and there have been initiatives to increase BC awareness among family doctors. In addition, there is evidence that major treatment improvements may favor younger age groups and thus, the effect of treatment may differ across age. 30 At the same time, mortality decline was not apparent in age groups 65-69 or 70+. This may be partly associated with increasing incidence as women aged 60-69 and 70+ have experienced the most rapid incidence rise, 15 that may be associated with background BC risk due to increasing age at first birth, lower parity, and increased use of contraception and hormone replacement therapy.31,32 Our results also suggest that women older than screening age group have benefitted less from both early detection and new treatments. We have shown previously that the proportion of stage I cases was the lowest in age group 70+ and survival gain the smallest in women aged 60 and older. 17 In particular, stage-specific survival was considerably lower in older patients with non-localized cancers, especially T4 tumors. 17
A recent meta-analysis of cohort studies using IB mortality as the outcome estimated the effect of screening invitation on mortality for women aged 50 years and older to be 20%. After adjustment for self-selection, the effect increased to 26%. 23 An earlier review of IB mortality studies estimated that the impact of mammography screening for invited women who were followed for a maximum of 11 years was most likely around 26%. 21 In their study, Møller et al 20 hypothesized that the BC treatment effect could be around 16%. Given the 35% decline in IB mortality in the age group 50-63 before and after screening initiation in Estonia, we estimate the possible screening effect to be about 19%. The lack of mortality reduction in age group 65-69 compared to the 35% reduction in age group 50-63 supports the role of screening. A recent study using simulation models and observational and clinical trial data on screening and treatment of stage I to III BC and of metastatic BC estimated that 25% of the 58% reduction in BC mortality observed in the United States between 1975 and 2019 was due to screening. 33 However, the authors also pointed out that BC cases diagnosed without screening may produce poorer outcomes that modern treatments are unable to improve. 33 It has been suggested that given the later adoption of mammography screening in Eastern Europe, it has the potential to prevent 23% of BC deaths.34,35
The study’s main strength was the possibility to use IB mortality data collected over a long period, and obtaining data from a high-quality nation-wide population-based cancer registry with individually linked cause-of-death data. Using data on diagnosis and death during the same period minimized the possibility of lead-time bias and eliminated problems related to competing causes of death. The main limitation was the inability to identify women who were invited to or participated in screening during our study period, as this information has been collected only since 2015 and the period 2015-2019 was too short to provide meaningful results in a subgroup analysis. This analysis is planned in the future. Misclassification of the underlying cause of death is more likely among older patients with multiple comorbidities, 36 which may have masked the effect of improved treatment in age group 70+.
Conclusions
The results of this study contribute to real-world evidence regarding the beneficial effect of BC screening on mortality. Even though screening attendance in Estonia has consistently remained below the recommended 70%, the age specific IB mortality curves in birth cohorts exposed to screening invitation distinctly differed from those seen in younger and older cohorts. When comparing 11-year periods before and after the introduction of screening, IB mortality showed a 49% reduction in women younger than the screening age and a 35% reduction in the age group invited to screening. These findings suggest a strong combined effect of improved treatment and early diagnosis. In contrast, no decline in IB mortality was seen in older age groups. Hopefully, expanding of the screening invitation to the age group 65-69 in 2018 will reduce BC mortality among older women in the coming years. The results of this study also support the recent national decision to gradually extend the upper age limit of the screening age group to 74 in 2024, which is in accordance with the new recommendation of the European Commission. Our results do not currently support lowering of the screening age limit in Estonia. Further monitoring of BC trends by age and stage at diagnosis is warranted. Efforts should continue to increase screening attendance in all age groups.
Supplemental Material
Supplemental Material - Incidence-Based Breast Cancer Mortality Trends in Estonia Before and After the Introduction of Organized Mammography Screening: A Register-Based Study
Supplemental Material for Incidence-Based Breast Cancer Mortality Trends in Estonia Before and After the Introduction of Organized Mammography Screening: A Register-Based Study by Aleksei Baburin, Piret Veerus, Katrin Lang, and Kaire Innos in Cancer Control
Footnotes
Acknowledgments
The authors thank Dr Margit Mägi and Mrs. Pille Härmaorg from the ECR.
Author Contributions
AB and KI designed the study, AB and KI acquired the data, AB and KI performed statistical analysis, AB drafted the manuscript, AB, PV, KL and KI interpreted the results. All authors critically revised the manuscript for intellectual content and approved the final version to be submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Estonian Research Council (Grant No. PRG722).
Ethical Statement
Data Availability Statement
The data will be available on request pending approval by the scientific committee of the relevant institutes.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
