Abstract
Intraductal papillary mucinous neoplasms (IPMNs) are a very common incidental finding during patient radiological assessment. These lesions may progress from low-grade dysplasia (LGD) to high-grade dysplasia (HGD) and even pancreatic cancer. The IPMN progression risk grows with time, so discontinuation of surveillance is not recommended. It is very important to identify imaging features that suggest LGD of IPMNs, and thus, distinguish lesions that only require careful surveillance from those that need surgical resection. It is important to know the management guidelines and especially the indications for surgery, to be able to point out in the report the findings that suggest malignant degeneration. The imaging tools employed for diagnosis and risk assessment are Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) with contrast medium. According to the latest European guidelines, MRI is the method of choice for the diagnosis and follow-up of patients with IPMN since this tool has a highest sensitivity in detecting mural nodules and intra-cystic septa. It plays a key role in the diagnosis of worrisome features and high-risk stigmata, which are associated with IPMNs malignant degeneration. Nowadays, the main limit of diagnostic tools is the ability to identify the precursor of pancreatic cancer. In this context, increasing attention is being given to artificial intelligence (AI) and radiomics analysis. However, these tools remain in an exploratory phase, considering the limitations of currently published studies. Key limits include noncompliance with AI best practices, radiomics workflow standardization, and clear reporting of study methodology, including segmentation and data balancing. In the radiological report it is useful to note the type of IPMN so as the morphological features, size, rate growth, wall, septa and mural nodules, on which the indications for surveillance and surgery are based. These features should be reported so as the surveillance time should be suggested according to guidelines.
Keywords
Introduction
Intraductal papillary mucinous neoplasms (IPMNs) are a very common incidental finding on Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) performed for other reasons.
1
These lesions are more frequent in men than women, at an age over 65, and account for 21%-33% of all cystic lesions of the pancreas.
2
IPMNs are neoplasms of the pancreas caused by epithelial cell hyperplasia of the pancreatic ducts that produce mucin.
3
IPMNs, together with mucinous cystic neoplasm (MCN), are the cystic lesions with the highest risk of malignant degeneration.
4
Although in some cases an IPMN may remain stable throughout the patient’s life, these lesions may progress from low-grade dysplasia (LGD) to high-grade dysplasia (HGD) and even pancreatic cancer (Figure 1). The risk of IPMN progression growing with time, so discontinuation of surveillance is not recommended.
1
Therefore, it is very important to be able to identify imaging features that suggest malignant degeneration of IPMNs, and thus, distinguish lesions that only require careful surveillance from those that need surgical resection. Man with main duct degenerated IPMN. In A (T2-W sequence in coronal plane), the arrow shows mucin plug and thickened wall. In B (T2-W sequence with FS in axial plane), yellow arrow shows liver mucinous metastasis at VI hepatic segment.
In this paper, we would like to focus on the aspects and characteristics of IPMNs that are useful to radiologist diagnosis and patient risk assessment evaluation.
Guidelines of the Management of IPMN: What the Radiologist is Concerned With
It is important to know the management guidelines and especially the indications for surgery, to identify and to report the findings that suggest malignant degeneration. 5
The features correlated to HGD or invasive carcinoma (IC) in IPMN are identified as high-risk stigmata (HRS) and worrisome features (WF). The first are very strong prognosticators of HGD/IC but with low specificity. The guidelines suggest prudent surgical indication only considering HRS features since critical elements are clinical condition of patient, life expectancy, so as patient preference. Regarding to HRS, those include: (1) obstructive jaundice in a patient with head of the pancreas cystic lesion; (2) solid component or an enhancing mural nodule ≥5 mm; (3) main pancreatic duct (MPD)
Indications for Surgical Resection in IPMN as Outlined by Current Guidelines.
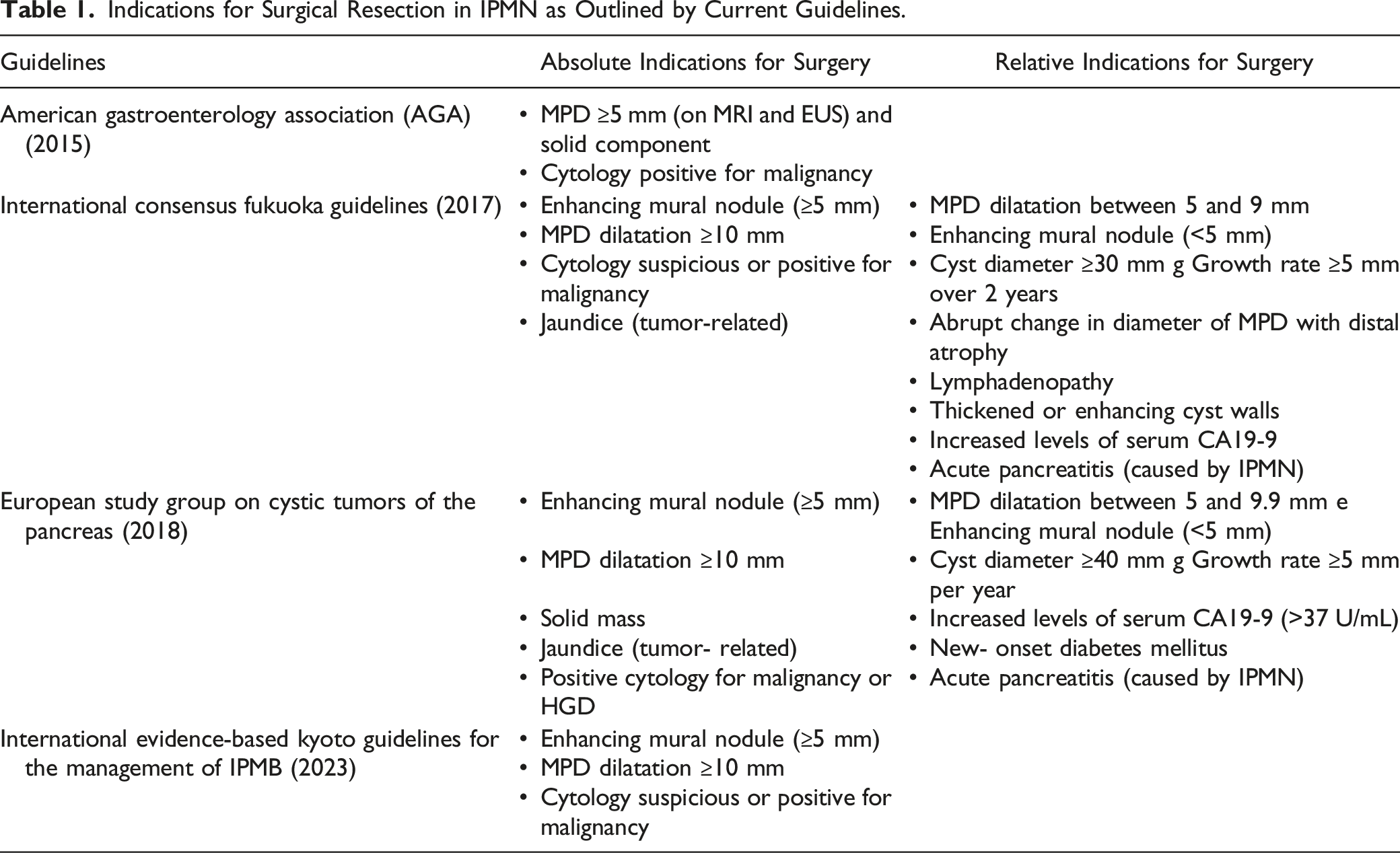
During this revision has been published the latest revised (2023) International evidence-based Kyoto guidelines for the management of IPMB.
9
In the revised edition 5 topics are critical: amendment of HRS and WF, follow-up of non-resected lesions, follow-up of resected lesions, amendment of pathological features so as the assessment of molecular markers in cyst fluid
Diagnosis: The role of MRI and magnetic resonance cholangiopancreatography
The imaging tools employed for diagnosis and risk assessment are EUS, computed tomography (CT) and Magnetic Resonance Imaging (MRI) with contrast medium. 10 According to the European guidelines, MRI is the method of choice for the diagnosis and follow-up of IPMN patients,8–11 since the diagnostic accuracy is 40%-95%. 8 Using cholangiopancreatography (MRCP) sequences, the pancreatic biliary system can be better detected so that it is easy to identify the communication branch with the MPD. 12 To increase the diameter of the pancreatic ducts and make them more evident, the use of the hormone secretin, Secretin-enhanced MRCP (S-MRCP)) may be useful, but this still has controversial indications in the detection of IPMN. 13 MRI, using in addition to conventional protocol MRCP sequences, has a higher soft-tissue contrast resolution than CT, allowing to detect single and multiple PCNs, including multifocal branch of the main duct (BD)-IPMN and mixed type (Montana-IPMN). 14 In addition, this tool has a high sensitivity in detecting mural nodules and intra-cystic septa. 1 It plays a key role in the diagnosis of worrisome features and high-risk stigmata, which are associated with malignant degeneration of IPMNs. MRI is less invasive and more tolerated by the patient than endoscopy ultrasound (EUS).11–15 Moreover, as it does not use radiation, it is preferred over CT for follow-up.16,17
With regard to MRI protocols for patient surveillance, there is no definitive agreement. 1 The main critical point is the use of contrast medium, 1 in fact, several abbreviated or short protocol (SP) have been proposed. 1 Several authors reserved conventional protocol at the first diagnosis and SP, including axial and coronal SSFSE T2-W sequences, 2D and 3D single-shot MRCP and 3 D T1-W spoiled gradient echo, for surveillance. 1 No consensus there is on DWI utility, although several researches suggested the use of DWI to early detection of pancreatic cancer. 1
EUS has been proposed as a problem solving in pancreatic cistyc neoplasm (PCN) with features that should be considered for surgical resection. 1 According to Fukuoka guidelines, EUS is suggested in patients with HRS and WF features. 7 While, the AGA guidelines suggests EUS-FNA only for cysts with 2 HRS or if significant changes develop during surveillance. 6 According to latest international evidence-based Kyoto guidelines, 9 EUS, including EUS-FNA and CE-EUS, is suggested for IPMN with high suspicious of HGD/IC and only if in the clinical setting it is available. Therefore, EUS-FNA to obtain cytological and molecular marker is an option, and should not be done when HRS are recognizable in others diagnostic tools. 9
The current pancreatic cancer screening programs are based on the use of diagnostic tests. A combination of EUS and MR/MRCP executed annually is suggested. 1 Computed tomography with dedicated protocol is suggested only if the patient cannot undergo EUS or MR/MRCP. 1
With regard to time of follow-up, according to the Fukuoka guidelines, high risk patients including those with HGD at the surgical margins, non-intestinal IPMN, or patients with PC family history should repeat imaging at least every 6 months. 7 For other IPMNs, is proposed follow-up every 6-12 months. The AGA guidelines proposed post surgical follow-up only for patients with HGD/IC using MRI every 2 years. 6
According to International evidence-based Kyoto guidelines, 9 surveillance should be adjusted considering the cyst size, the growth rate and the time to progression. For branch duct IPMN (BD-IPMN) misuring <20 mm, >20 mm and <30 mm, and >30 mm a follow-up time of 18, 12, and 6 months after an initial surveilance of 6 months is suggested. In patients with cysts <20 mm, without morphological changes and WF, after 5-year, surveillance may be discontinued. The discontinuation should not be appropriate for younger patients and for patients with familial or genetic risk. 9
Report:Wwhat the radiologist should consider
Radiological Report.

TYPES of IPMN
Depending on the morphology and the duct involved, they can be of 3 types: - of the MD (MD-IPMN), - of a branch of the MD (BD-IPMN) - and of a mixed type (Montana-IPMN).
MD-IPMNs are identified as a dilatation of MD (>5 mm), either segmental or along its entire length, without a concomitant cystic lesion or additional cause of ductal obstruction. It is the type that most frequently undergoes malignant degeneration (62%). 7
BD-IPMN is dilatation (>5 mm) of the branching ducts with uni- or multi-locular appearance (‘grape cluster’). 20 Typically, they are multifocal and located preferentially at the head (70%) or uncinate process of the pancreas.7,21,22 Communication with the pancreatic ducts is a specific aspect. However, failure to detect this does not exclude the diagnosis of BD-IPMN. Morphologically, BD-IPMNs can show several patterns: (a) uni-locular; (b) multi-locular with macro-cystic aspect and few septa; (c) micro-cystic characteristic defined a grape-fruit growth, in which multiple septa divide several fluid spaces; (d) pleomorphic, in with we can find a mixture of these types. The ability of these lesions to produce mucin can sometimes lead to mucin plugs or balls within the cyst or in the pancreatic duct. These often present as non-enhancing mural nodules on the dependent surface of the cyst and should not be confused with solid component. BD-IPMN has a lower mean risk of degeneration into invasive carcinoma or high-grade dysplasia than MD-IPMN (14%–48%). 7
MT-IPMN is shown as dilatation of the MD associated with 1 or more cyst-like growths. The risk of MT-IPMN cancer involvement is comparable to MD-IPMN. 23
Location
BD-IPMN are located preferentially at the head (70%) or uncinate process of the pancreas.7,21 But they can occur anywhere in the pancreatic paranchyma. Specifying the location of the lesion in the report is especially useful to facilitate localisation during EUS and for surgical planning.
Size and Rate Growth
Given the importance of cyst size, this must be reported and performed in a reproducible and consistently manner. The guidelines take into account only 1 measurement, calculated on the largest diameter of the formation. This should be measured in the plane in which it appears largest, which may also be oblique to the plane of the scan. It has been seen that the best plane in which to measure the cyst is the coronal plane in T2-weighted MRI sequences.24,25 Alternatively, the axial plane or MRCP sequences can be used. The measurement should be taken from outer wall to outer wall and the connection of the cyst to the main pancreatic duct must not be included in it. 25 In the case of multiple lesions, as is often in BD-IPMN, the largest cyst is chosen as the reference. To reduce inter-observational variability, it may be useful to indicate in the report the slice number, the plane and the sequence or scan in which the diameter was measured. This also facilitates the reproducibility of the measure in the follow-up comparison.
In a cyst with a diameter of less than 5 mm, the presence of a carcinoma is rare and does not require further diagnostic investigation. 24 A retrospective analysis by Das et al, however, recommends follow-up of cysts smaller than 5 mm. 24 Now, there is no agreement on the optimal interval for IPMNs surveillance. The 2017 IAP guidelines, identify a cyst ≥3 cm as a “WF”, which requires further investigation with EUS, and consider surgery for young patients.7,21 While the 2018 European guidelines, include in the relative criteria for resection of BD-IPMNs, a diameter ≥4 cm. 8 Size comparison with previous imaging examinations is imperative. Cyst growth rate >5 mm, in 2 years for IAP and in 1 year for European Society, is a relative indication for surgery, since it is correlated with IC in IPMN.
Wall and Septa
After defining the size of the cyst, its appearance should be assessed, based on its shape and the presence of septa. Presentation patterns may be: grape-like with cyst-by-cyst formation (with numerous thin septa or due to a group of clustered microcysts), macrocystic (which may be uni- or multi-locular with a few septa), finger-like (cylindrical dilatation of the side branches) or pleomorphic (mix of the other patterns).
26
Of the septa and wall, the thickness, enhancement after contrast medium administration (Figure 2) and irregularity should be analysed. The cut-off for defining thickened walls has not been defined, while for septa it is 2 mm. Woman with mixed degenerated IPMN. In A, cholangiopancreatography MRI, the arrow shows ‘grape cluster'. In B (T1-W after contrast medium during portal phase, in axial plane), the arrow shows enhanced cystic wall.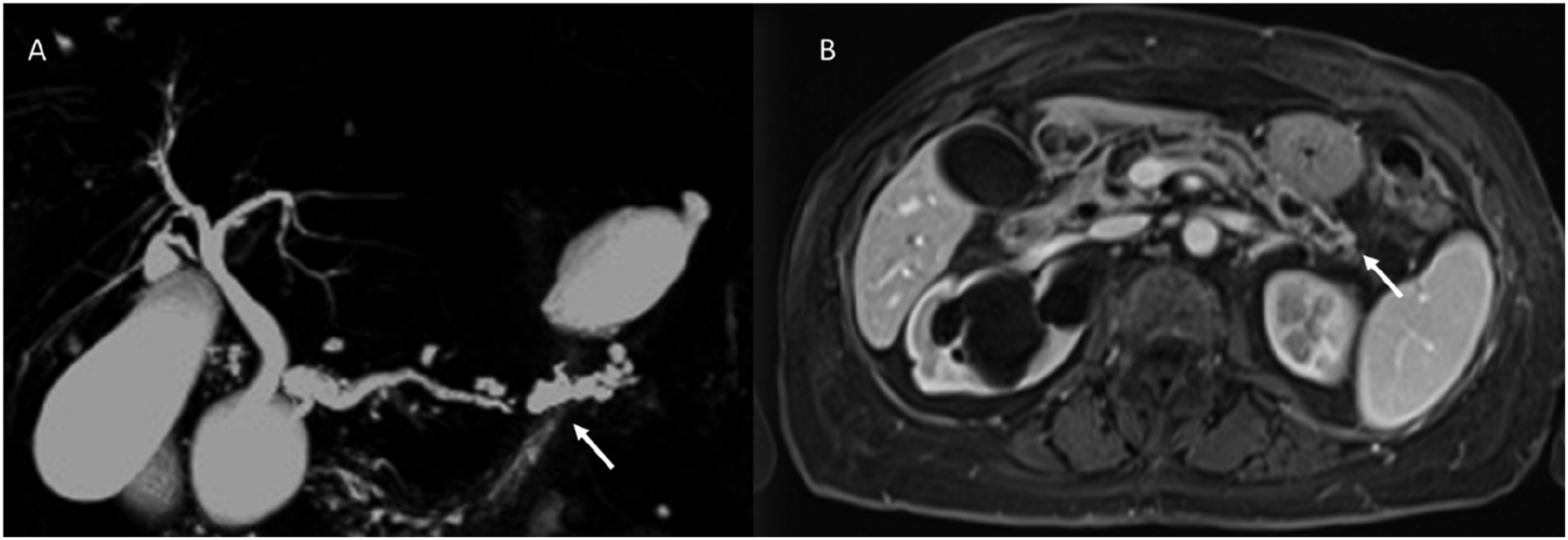
Kim et al demonstrated in a meta-analysis that the presence of septa >2 mm (Figure 3) correlates with the risk of malignant transformation.
27
Man with main duct degenerated IPMN, during MRI assessment (A: cholangiopancreatography MRI) In B, T2-W sequence in coronal plane, the arrow shows thickened septa which after contrast agent (C: T1-W sequence during portal phase in coronal plane) have contrast enhancement.
And thickened and enhanced cystic wall is a “worrisome feature”, and therefore requires further study with EUS. 7 However, these aspects should be considered together with the other alarming features to raise suspicion of degeneration. Especially in MRI, there may be limitations that can be misleading, such as low spatial resolution and artefacts. In particular, the presence of mural nodules should be investigated, as we will see below.
Mural Nodules
In all types of IPMN, it is important to identify wall outgrowths that underlie solid masses. When can we talk of a nodule? The definition of a mural nodule is a papillary outgrowth within the dilated main duct or cyst ≥3 mm.
21
Mural nodules, regardless of size, are suggestive of solid masses and the bigger ones are a significant predictor of malignancy (Figure 4). If this finding is < 5 mm then it requires further investigation with EUS, if it is ≥ 5 mm then surgical evaluation is recommended. Crucial is the evaluation of post-contrast CT and MRI images. Evaluation of solid nodules is performed on T1-weighted sequences after contrast medium, and venous phase is preferred. Indeed, the lack of enhancement should raise the suspicion that this is not a mural nodule but just mucin plug or a conglomerate of thin septa. Mucin plugs are generally more frequent than nodules and are due to dehydration of secreted fluid. these should be reported by radiologists, since they might grow and obstruct the lumen of the ducts resulting in possible pancreatitis. Even more recommended is to report the finding if in doubt between a conglomerate of thin septa or mural nodule. This is so because both entities require short-term follow-up or further diagnostic investigation by EUS and fluid sampling.6–8 Men with main duct degenerated IPMN, during MRI assessment. In T2-W sequence (A: axial Plane), the arrow shows mural nodule that after contrast agent (B: T1-W during portal phase in axial plane) has contrast enhancement.
Multifocal BD-IPMN
Cystic lesions in BD-IPMN are multiple in 41% of cases. 27 There is no consensus in the literature as either multiplicity predisposes to malignant degeneration or not. In multifocal BD-IPMN, each lesion should be assessed separately. 8 The most alarming cyst(s) should be described in detail in the report, selected on the basis of size and the presence of WF or HRS. In these cases, it is important to indicate whether they are distributed more in 1 portion of the pancreas (head, uncinate processus or tail) or not, to direct the eventual surgical approach (total or partial pancreasectomy).
Main duct dilation
MD-IPMNs are identified as a dilatation of the MD (>5 mm), focal or diffuse. The presence of MDP dilatation alone causes problems in differential diagnosis. The diagnosis of MD-IPMN is possible when other causes of dilatation of MPD as a stone or stenosis are excluded. In the absence of these findings, it also allows us to make the differential diagnosis between MD-IPMN and chronic pancreatitis, especially when this type of IPMN is associated with atrophy of the distal parenchyma. If the dilatation of the MDP is abruptly cut, in the absence of a history of pancreatitis, the presence of an invasive carcinoma causing ductal obstruction must be suspected. Furthermore, it should be remembered that a 5 mm dilatation of the isolated MDP with remaining sections of normal calibre (3 mm), may be completely normal, especially if already present at a previous check-up. 28
A calibre greater than 5 mm is an absolute indication for surgery for the American Gastroenterology Association (AGA), 6 while greater than 10 is for IAP 7 and European guidelines, 8 which consider the range between 5 and 9 a relative indication. Therefore, it is very important to be as accurate as possible when measuring. Because of its oblique course, it is useful to perform the measurement in MRCP sequences that allow the entire MDP to be visualised.
Radiomics
Radiomics and artificial intelligence (AI) are progressively expanding fields and, in recent years, are becoming a powerful tool for radiologists.29,30
Radiomics is the extraction of a high number of quantitative features from medical images, including several phases: image acquisition (eg, RX, US, CT, MRI, and ibrid imaging); segmentation (aumotatic, semi-automatic and manual); generation of features; development of database; analysis of database and radiomics signature. 10 Images used for radiomic analysis are collected from different hospitals or data centers; thus, these images are usually obtained using different parameters and protocols and reconstructed with different software. The differences may bring unexpected influences on the radiomic model. Segmentation is critical because the subsequent feature data are generated from the segmented volumes. It is challenging because many tumors have indistinct borders. Generation of features refers to the extraction of semantic features such as dimension, necrosis, margin, location or extraction of non semantic features such as shape, hystogram or texture. 10 In the past years, texture analysis has gained attention in medical applications and has been proved to be a significant computer-aided diagnostic tool 10]. There is not a strict definition of an image texture but it can be described as the spatial arrangement of patterns that provides the visual appearance of coarseness, randomness, smoothness, etc. Texture analysis describes a wide range of techniques for quantification of gray-level patterns and pixel inter-relationships within an image providing a measure of heterogeneity. It has been shown that different image areas exhibit different textural patterns that are sometimes imperceptible to the human eye. 10 Applications of texture analysis in medical imaging include classification and segmentation of tissues and lesions. Texture analysis applications involve a process that consists of 6 steps: image acquisition, region of interest (ROI) definition, ROI pre-processing, feature extraction, feature selection, and classification. None of these steps is specific, and the methods have to be chosen according to the application. 10 Manual definition of ROIs is still considered the gold standard in many applications, and it is the chosen option over automatic methods.
Radiomics can be performed with as few as 100 patients, although larger data sets provide more power. Radiomics could potentially aid cancer detection, diagnosis, assessment of prognosis, prediction of response to treatment, monitoring of disease status. 10 Radiomic is designed to be used in decision support of precision medicine, using standard of care images that are routinely acquired in clinical practice, it presents a cost-effective and highly feasible addition for clinical decision support. Also, this analysis non-invasively characterize the overall tumor accounting for heterogeneity, interrogating the entire tumor allows the expression of microscopic genomic and proteomics patterns in terms of macroscopic image-based features
In the recent literature, there are several studies on the application of radiomics to pancreatic tumors. 31 These involve the development of models applicable to both CT and MR.32–38 The application in clinical practice of these tools, can support radiologists in determining certain decisive characteristics in the workflow of PC patients, such as malignant degeneration, response to therapy, etc.39–42 In a large study of 408 IPMN patients, Tobaly et al evaluated the ability of CT-based radiomic analysis in predicting malignant IPMN degeneration. 37 In their study, from a retrospective analysis of preoperative CT images, they developed a radiomics model capable of distinguishing benign from malignant IPMNs with a sensitivity of .82 and specificity of 0,74. Radiomics was also applied to MR for the characterization of IPMNs. In a multicentre study, from the interaction of clinical data and radiomic analysis of pre-operative MR images of 202 patients, Cui et al developed a radiomic nomogram model capable of differentiating low-grade from high-grade IPMNs. 38 Recently, an Italian study evaluated the ability of radiomics to identify degenerated IPMNs before the appearance of radiological features of malignancy (Worrisome features and high-risk stigmata). 39 The constructed model obtained an AUC of more than.9 for the 4 MR sequences analysed (T1W, T1W post-contrast, T2W, and Apparent Diffusion Coefficient (ADC) map). The application of these quantitative analyses offers important support to the radiologist and other specialists in identifying the best treatment for patients with IPMN, with the ultimate aim of providing customised therapy.18,43–47
AI could have a critical effect on IPMN classification and patient risk assessment. Deep learning (DL) models could perform a critical role during the different phases of diagnostic work flow, including acquisition, reconstruction, and post-processing. Also, AI models may have the potential to automate the diagnosis and risk stratification. 1 In a 139 patients study, the addition of a DL algorithm to MRI data, permitted to find advanced neoplasia in IPMNs (sensitivity of 75%; specificity of 78%; and AUC .783). 48 However, this approach accuracy is similar to that obtained using the guidelines management, so additional and more sophisticated models are required.
There are several limits to MRI-based radiomic analyses that should be considered. First, several studies have assessed only internal dataset, and to make reproducible and to validate the results there is the necessity of external dataset. Second, MRI studies are lesser standardized than CT studies, it is due to the larger range of scanner assets, sequences and image settings. To improve this field and increase patient outcomes, the introduction of standardized protocols and multi-center datasets are critical.
Discussion
The proper management of patient at risk for pancreatic cancer is still debated.48–51 So as, analysing the guidelines,6–8 it is clear as the different working groups suggest different approach. The European group suggest observing all pancreatic cystic lesion, in asymptomatic as symptomatic patients. Otherwise for AGA guidelines the patient must be asymptomatic. While the Fukuoka guidelines are the main specific, since this group considers only patients with IPMN. Lacking accurate and rigorous studies comparing the different guidelines, it is not clear which is the best surveillance strategy in these patients. 1
Regarding to diagnostic management, dedicated study protocol, either in MRI/MRCP that in CT, showed similar accuracy in lesion characterization.48–53 However, MRI/MRCP has the higher accuracy either in the detection of the communication between a cystic lesion and the pancreatic duct system, than in detection of mural nodules and internal septa. 48 Multimodality imaging evaluation should be considered for tumor staging and pre-treatment evaluation.54–66
Regarding study protocol, considering the wide spread of published protocols, but the lack of comparative studies, there are no specific study protocols which could be suggested.66–81 Nowadays, the main limit of diagnostic tools is the ability to identify the precursor of pancreatic cancer (PC), and since PC remains the big killer, since the number of deaths almost equal to its incidence, 1 the chance of a surgical resection as soon as possible represents the only chance of a cure. 81 In this context, increasing attention is being given to AI and radiomics analysis.82–85
Latest technical progresses have led to the increased use of AI in radiological setting. Since, a computer can evaluate larger amounts of data respect to a human brain, this tool could resolve complicated tasks in oncological setting.74,86–92 Machine learning (ML), an AI sub-field, is a mathematical model that can realise detailed tasks through the replication of computations.66,93–98 The large quantity of ML models developed have made possible the introduction of a new approach for quantitative imaging assessment, named radiomics.44,99–106 Radiomics analysis allows to convert digital medical images into numbers called features, that can be analysed in a mathematical manner.29,46,87,107–111 These features can be correlated with clinical-pathological data and outcomes, and, hypothetically, open the way for a personalised approach, that considers that patient with that particular lesion.66,90,92,93,98,112–124 For several tumors, radiomic analysis has already provided a precise assessment of biology, allowing the identification of features that are correlated with outcomes.35,37,38,83,85,96,98–100,125–139 In the context of IPMN, AI and radiomics analisys remain in an exploratory phase, considering the limitations of currently published studies.31–42 Key limits include noncompliance with ML best practices, radiomics workflow standardization, and clear reporting of study methodology, including segmentation and data balancing. So the lack of quality and adequate sample sizes and external validation set remains critical points to resolve. 129
Conclusions
The management of the patient with IPMN requires a multidisciplinary approach. The radiologist plays a key role in the diagnosis of IPMN and the assessment of suspicious features for malignant degeneration. MRI is the method of choice and allows the identification of findings that represent absolute or relative indications for surgery. A structured radiological report in which the radiologist report the IPMN types and the HRS and WF features to allow a proper patient management either for surgical indication either for surveillance is required. Although AI and radiomics analysis represent an interesting field, however critical issues make these applications still limited to a theoretical field.
Footnotes
Acknowledgments
The authors are grateful to Alessandra Trocino, librarian at the National Cancer Institute of Naples, Italy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was suported by the Italian Ministry of Health “Ricerca Corrente” funds.
