Abstract
Immunotherapy has substantial attention in oncology due to the success of CTLA-4 and PD-1 inhibitors in the treatment of melanoma, lung cancer, head and neck cancer, renal cell carcinoma, and Hodgkin’s lymphoma. A deeper understanding of interaction of tumor with its environment and the immune system provides best guide for oncology research. Recent studies in oncology have explained how a tumor alters antigen presentation, avoids detection, and activation of the host immune system to live and develop. Understanding the connections between the tumor and the immune system has resulted in several innovative therapy options. The extensive field of gene therapy has provided a number of cutting-edge medicines that are expected to play an important role in lowering cancer-related mortality. This article explains the history, important breakthroughs, and future prospects for three separate gene therapy treatment modalities: immunotherapy, oncolytic virotherapy, and gene transfer. Immunotherapies have completely changed how cancer is treated, especially for individuals whose condition was previously thought to be incurable. Examples include ACT (adoptive cell therapy) and ICB (immune checkpoint blockade). This review article will discuss the relationship between the immune response to cancer and the mechanisms of immunotherapy resistance. It will cover combination drugs authorized by the US Food and Drug Administration and provide a thorough overview of how these drugs are doing clinically right now. Cytokines, vaccines, and other soluble immunoregulatory agents, innate immune modifiers, ACT, virotherapy, and other treatment modalities will all be covered in detail.
Introduction
Cancer immunotherapy has replaced traditional cancer treatments. These medicinal agents have shown clinical activity as monotherapies in a variety of tumors. 1 Further advancements in the efficacy of immunotherapies against cancer demand the use of the anti-tumor immune action at various points that are achieved by the combination of different techniques. 2 The present landscape of cancer immunotherapy will be discussed in this review and selection of dose and practice techniques, clinical pharmacotherapeutics, combinations in clinical development and regulatory issues will also be discussed. 3 Immunotherapy has revolutionized oncology that how different tumors are treated over the last 5 years by utilizing the immune system to target tumors, primarily by reducing the protein of programmed cell death and the protein associated with cytotoxic T lymphocytes (CTLA-4). 4 This methodology is functional and efficient in head and neck cancer, lung tumors of non-small cells, melanoma, carcinoma of renal cells, and lymphoma of Hodgkin’s. Most cases show that PD-1 and CTLA-4 inhibitors have made significant advances in medication procedures, with durable remissions and response rates ranging between 10 and 61%, but this treatment is not effective for all patients, leaving many patients in need of other treatments. 5 More advanced immunotherapies have been achieved by understanding the interactions of the immune system with tumors. The tumor microenvironment contains colony stimulating factor, which prevents dendritic cell maturation and results in defective antigen and T-cell allergy cell presentation. TGF-beta and IL-10 from regulatory T cells reduced this, as done by the chemokine CCL2 from TAMs. 6 Abscission, chemo, radiation, and combination therapy are the mainstays for treatment of cancer currently. Gene therapy based on viral vectors has subsequently been shown to be an effective method. This is evidenced by outstanding results attained with cytotherapy that is mediated by viruses to cure conditions due to genetic defects, which is a major field of concern for the methodologies that are based on gene therapy. 7 For example, metachromatic leukodystrophy (MLD) is treated by lentiviral vector-based HSCS cytotherapy and eczema–thrombocytopenia–immunodeficiency syndrome is cured by the methodologies that are based on viral vectors. In such techniques, the expression of genes that are introduced by viral vectors is stopped at sufficient stages for remedial results, and this process of gene expression may be going on for various months or even longer. 8 There are less chances for the evaluation of side effects due to both the insufficiency of cases and the period of observation, and this is a major restriction of these types of studies. However, considering the restrictions and benefits, gene therapy is the best technique for genetic disorders. 9 Several gene therapy techniques are used for many disorders. This is not limited to genetic defects. Non-viral vectors are safe but ineffective sometimes. On the other hand, viral vectors are effective, but the problem is that they are immunogenic, pathogenic, and somewhat carcinogenic. 10
Vaccines Based Therapies
Vaccines, particularly those targeting dendritic cells, are providing another way to activate an anti-tumor T-cell response. 11 Due to their immaturity, dendritic cells in malignancies are assumed to have less ability to present antigens to T lymphocytes. 12 Dendritic cells are activated by dendritic cell-based vaccines coupled to GM-CSF or by cyclic dinucleotides that initiate the STING pathway activator. Vaccines that use dendritic cells to present antigen for T cells to respond have been studied in a variety of cancers for decades. 13 For cell cultures and murine models, they showed improved immunogenic response but translation for significant therapeutic effect is restricted except for sipuleucel-T, which is a vaccine created by using a person’s own monocytes ex vivo, combined with antigens, having prostatic acid phosphatase and GM-CSF and then administered for metastatic prostate cancer.11,14 Moreover, sipuleucel-T provided a 4.1-month survival benefit over placebo in a phase III clinical trial. With a better insight into tumor immunology, a variety of applications for vaccines based on dendritic cells are currently being researched, including glioblastomas, neoadjuvant HER2 + carcinoma, adjuvant melanoma, and some others.15,16 Vaccines have proven to be a difficult therapy to continue pursuing because of the long time of manufacturing. 17 As a result, they are restricted to patients that do not have a chance of moving ahead when waiting for a cure, and have the classic waiting period is from 28 to 42 days. Vaccines based on combinations with inhibitors of checkpoints are being researched. 11
The idea of therapeutic vaccines is based on tumor-associated antigens, which are hypothesized to be present in cancer cells but missing or in low quantities in healthy cells. Therapeutic vaccinations may help the immune system acquire the ability to identify and respond to these antigens as well as destroy cancer cells that carry them. There are 3 basic methods for creating cancer vaccines. Your body’s tumor cells are what cause them to develop. This indicates that they are specifically designed to elicit an immune response against characteristics particular to your tumor. They might be produced using tumor-associated antigens that are present on the cancer cells of many patients with a certain kind of cancer. Any patient whose tumor generates that antigen may have an immunological response after receiving such a vaccination. This vaccine is presently undergoing testing. They could be created from your own dendritic cells, which are immunological cells. Your immune system is made to react to an antigen on tumor cells via dendritic cell vaccines. An FDA-approved dendritic cell vaccination called sipuleucel-T is used to treat a subset of men with advanced prostate cancer. Cancer vaccinations are another name for oncolytic viral therapy, a kind of cancer therapy. It works by utilizing an oncolytic virus, which attacks and destroys cancer cells while sparing healthy ones. The first oncolytic viral treatment to get FDA approval is talimogene laherparepvec. Its foundation is type 1 of the herpes simplex virus. This virus can infect both cancer cells and healthy cells, but cancer cells are unable to eradicate it. A tumor receives a direct injection of T-VEC. The virus causes cancer cells to rupture and die while it continues to replicate itself. New viruses and other chemicals that are produced by dying cells have the potential to trigger an immune response against cancer cells throughout the body.
Viral Vector–Based Therapies
Oncolytic viruses are another type of agent used to stimulate an immune response against a tumor. Originally generated from HSV, they are the viruses that are modified to induce toxicity as well as replication in cancer cells. HSV elicits a massive innate and humoral immune response by activating innate cells and initiating complement protein, cytokine cascades, and immunoglobulin formation, respectively. Oncolytic viruses activate tumor cell immunity when introduced into the tumor microenvironment. 18 The FDA has cleared Talimogene laherparepvec for intralesional therapeutic use as the first oncolytic virus. In a phase III trial comparing the 2, 436 individuals with stage 3B and C melanoma and stage 4 melanoma with different specific sites of metastasis were assigned to dendritic cell–based vaccine GM-CSF or oncolytic virus T-VEC. The ORR was 40% in 249 individuals when dealing with the oncolytic virus T-VEC and 2.3% when dealing with the vaccine GM-CSF, with 27 patients achieving CR. In the phase 1 clinical trial, when dealing with the combination of 34 T-VEC and ipilimumab, they showed an ORR of about 50%, with an outstanding 44% of patients achieving about 18 months of CR and PFS. 19 Oncolytic virus T-VEC is being tested by combining it with inhibitors of checkpoints like PD-1. Oncolytic viral therapy for malignant gliomas has also been a hot topic of investigation. Six of nine patients with malignant gliomas exhibited illness or a PR when treated with modified G207 and HSV1, accompanied by radiation. 20 However, no survival advantage was observed. The FDA has approved PVSRIPO, a more successful recombinant virus, as a revolutionary treatment for heavy gliomas. 11 Having its internal ribosomal entry site exchanged with the entry site of human rhinovirus 1, PVSRIPO prevents itself from replicating and damaging neuronal cells but can cause growth in glioma malignant cells and have an infective response to them. When dealing with PVSRIPO, individuals with malignant gliomas of stage 4 show an OS of about 12.6 months and, more importantly, 23% of individuals have 2-year survival in contrast with 10% of individuals in the control group. Some other viruses are also being researched for virus-based oncolytic therapy right now.21,22
Viral vectors account for 60% or more of the delivery mechanisms and 100% of the approved gene therapies in gene therapy research, including oncolytic virotherapy. We can identify the areas where vector particles may be most useful thanks to this collection of knowledge. The use of therapeutic viral vectors will grow as a result of recent success and rapid technological breakthroughs in the industry. When paired with complementary radiation, chemotherapy, and immunotherapies like CAR-T and checkpoint inhibitors, combination therapy, in particular, offers a lot of promise. Modular vector genome design allows for the incorporation of cutting-edge biotherapeutics like BiTEs, cytokines, and CRISPR/Cas into the genetic payload to broaden the repertoire of anti-tumor effectiveness. In addition to the development of innovative vectors, T-VEC and other approved viral medications are being used to treat many more solid tumors than only melanoma. The whole spectrum of the aforementioned vector platforms, including oncolytic viruses and non-replicative vectors, may be employed in prime-boost cancer vaccination regimens.
Immune Response Activation
Boosting the immunity that is specific for tumor agonist antibodies has been done for the checkpoints of activation and also with the help of increasing the presentation of antigen for T-cell processes to T-cells by using dendritic cell vaccines and oncolytic viral vectors. This is a useful technique for future research.
Innate Immunity
TLRs and generalized stimulation of innate immunity by the STING pathway can produce anti-tumor immune responses. Introducing a mouse tumor model with brain gliomas, an agonist of the STING pathway, c-di-GMP, showed improved survival and exhibited increased T-cell infiltrates in the tumors. 23 Furthermore, globally administering the dendritic cell growth factor FLT3L into melanoma-bearing mice and intratumoral poly I:C doses and PD-1, that is, programmed death 1 inhibitor or BRAF inhibitor-based treatment, resulted in better responses than PD-1 or BRAF inhibitors alone.24,25 Patients who have failed to respond to PD-1 or CTLA-4 inhibitors alone may benefit from a combination of dendritic cell activating drugs and checkpoint inhibitors. TLRs bind antigens and boost cytokine release, which triggers immunological reactions in stimulated LCs, that is, Langerhans, lymphocytes, and macrophages. TLRs by using protein kinase B initiate division in T cells.26,27 In preclinical mouse models, TLR agonists have been found to reduce tumor growth by counteracting suppression of the immune system or its response in the tumor microenvironment. The FDA has approved imiquimod as a topical TLR agonist for condylomata acuminata, actinic keratosis, and cutaneous basal cell carcinoma. 28
Immune Suppression Inhibition
In their microenvironment, tumors can also inhibit immune responses by using pathways of checkpoints like PD-1 or by using Tregs and MDSCs like suppressive cells. By inhibiting the relevant suppressive signals, immune surveillance can be improved, and an anticancer T-cell response can be mounted.29,30
Inhibitory Checkpoints
In the tumor microenvironment, checkpoint molecules that are present on both regulatory and CD8+ T cells can be inhibited, like TIM-3, TIGIT, PD-1, BTLA, LAG-3, and CTLA-4. This is beneficial for CTLA-4 inhibitors and PD-1 inhibitors. LAG-3 inhibitors have a functional role in melanoma cancer and ovarian cancer anti-tumor reactions.31,32 In mouse model trials, inhibiting TIM-3 with a LAG-3 inhibitor and a PD-1 inhibitor resulted in an extended immune response and regulatory T cells specific for tumors, as well as a decrease in melanoma tumors, particularly those that recurred.CTLA-4 inhibitors and PD-1 inhibitors have been studied in a variety of cancers.33,34 Preclinical and clinical trials are also looking into other combinations of checkpoint-targeting drugs. Indoleamine 2,3-dioxygenase 1 is a special and effective enzyme that affects tryptophan metabolism and is generated in macrophages within the tumor microenvironment. It is an immunosuppressive molecule that has been investigated for therapy. By counteracting interferon gamma, IDO1 reduces T-cell proliferation and enhances neovascularization. 35
Therapy Based on Adoptive T-Cell
It is an immunotherapy that uses the extraction and modification of autologous T cells for the detection of tumor antigens, and then to focus on the tumor, selectively reintroduces T cells into the patient. Such T cells possess a membrane receptor called the chimeric antigen receptor. This receptor targets cancerous antigens. T-cells also possess a unique intracellular component, and this component stimulates the T-cell when it is exposed to the antigen. T-cells with CAR have recently integrated signals like CD137 and CD28 for long-term immunity responses and to keep stimulated T-cells alive. These cells can be programmed to produce CARs, which detect a variety of tumor antigens and initiate the formation of cytokines or interleukins upon binding.
36
These can also be used to change tumor-suppressing immune system pathways like the PD-1 inhibitor pathway. To invert the suppression of the immune response in tumors, the receptors, that is, CARs, use the PD-1 pathway. When PD-1 is delivered to T-cells and binds to them, it stimulates T-cells and signaling pathways, as shown in Figure 1.
37
Many cancer types and antigens are undergoing clinical testing. Hematologic malignancies have had the most effective treatment to date, with numerous permanent full remissions in all tumors, including chronic lymphocytic leukemia and lymphomas. Nonetheless, clinical trials against solid tumor antigens such as CEA antigen, VEGFR2 antigen, ERBB2 antigen, and a variety of other antigens are being investigated.37,38 The Adoptive T Cell Immunotherapy Technique. T cells (TILs, tumor-infiltrating lymphocytes) are separated from peripheral blood or tumors (PBLs, peripheral blood lymphocytes). TILs can be expanded without being cultivated, particularly since they are primarily tumor-specific before being cultured. PBLs, on the other hand, must be genetically changed or exposed to antigen-specific growth to gain tumor selectivity. After a few weeks of growth in vitro, tumor-specific T lymphocytes can be reinfused into the cancer patient.
Combination of Immunotherapies and Significance
Oncology Immunotherapy Agent Classes.

Biomarkers
In early clinical trials, biomarker data can help to guide dose and regimen selection. The occupancy of receptors found in the blood and their corresponding time outline for PD-1 inhibitors circulating on CD3+ T cells in refractory solid tumors were investigated as potential PD indicators and evaluated across several nivolumab dosage regimens. Shorter dosage intervals were extrapolated from the data to enhance occupancy and penetration into tissues, which will be investigated further in the future. 42 Cytokine levels in peripheral blood act as a PD marker to check the risk and safety assessment, and it also helps in dose recommendation. To do this, after 1 week of treatment, the dose is increased to decrease the potential for the release of cytokines in individuals with non-Hodgkin’s B-cell lymphoma. The exposure-dose or response relationship for the depletion of B-cells found in peripheral blood was also modeled or examined for the change in tumor size. The difference between the exposure-response curves of any 2 has demonstrated the translation of B cell response in peripheral blood to tumor response as well as tumor accessibility to the drug.43,44 As previously stated, the increase in the production of PD-L1 inhibitors in baseline tumors is related to improved efficacy in a variety of tumor types. As a result, Keytruda pembrolizumab companion testing for 2L non-small cell lung cancer was approved. Moreover, it’s also helpful to include PD-L1 production in the baseline and its status in exposure or dose response analyses. 45 Combination medication development could use similar biomarker-informed dose selection approaches with the addition of a component that allows the interaction between the agents that are used in combined form in combination therapy. Blocking therapies of program death 1 inhibitor and program death ligand 1 inhibitor are used in combination with check point inhibitors. Check point inhibitors such as LAG-3, TIM-3, and CTLA-4, for example, have a greater impact on the process of reverse T-cell depletion and CD8+ function restoration. 46 Costimulatory receptors present on T cells, including CD137 and CD27, influence activation of T-cells and activate proliferation of T cells, migration of T cells, and the formation of memory T-cells. Therefore, a thorough understanding of the basic biology of each therapy’s mode of action and its function in the pathway or mechanism of cancer immunity will be required to identify combined effects of the drugs when they are used in combination and biomarkers of the drugs that are associated with them. 47
Clinical Pharmacology Considerations
List of Some Combination Immunotherapies in Clinical Development.

Hematologic Malignancies Immunotherapy Development
The creation of biomarkers that are immunologic and represent the related MOA of the medication is a difficult process or provocation for many immunotherapies. 54 Such studies must preferably be able to discriminate quantitatively between the essential aspects of robust and weak immune responses. As this allows helpful comparisons between groups, including those that are given with the same vaccine but have different adjuvants and use different schedules and doses. 55 These responses are sometimes required, although they are not satisfactory all the time for the eradication of tumors or for clinical responses. Such parameters show a higher NPV until they are investigated across a population of patients. In any case, validating a prospective biomarker still starts with clinical linkages, necessitates enough “clinical responders.” 56 That is a restriction that has hampered numerous immunotherapy studies in patients with solid tumors. As previously stated, clinical responses that are mediated by immunity show ubiquity in malignancies of blood-forming tissues like leukemia, lymphoma, and multiple myeloma. Furthermore, among many cases of such malignancies, quantification of systemic, molecular changes in tumor load, like size, shape, and amount of tumor, is conceivable. 57 As a result, in the case of MRD, real-time assessment of clinical responses and immunotherapy efficacy is possible. Despite these possibilities, the relationship between clinical response and immunological responses and reactions to a variety of LAA has not been thoroughly investigated. 58
The findings from different phases of clinical trials of vaccination and observational studies conducted longitudinally with hard endpoints about the extent and composition or habit of immune responses in different patients against a broad spectrum of LAA will need to be incorporated into the next phase of leukemia immunology research.59,60 Early efforts at standardization have been made as there is no globally agreed-upon set of guidelines for extensively documenting and comparing immune responses between individuals. Antigen-specific responses in patients vs controls can be measured on basis of antigen expression, disease or tumor load over time. 61 It is also possible to measure the linkage of responses that are mediated by specified situations, such as donor lymphocyte infusion in chronic myeloid leukemia and chronic lymphocytic leukemia. While more accurate measurements of T cell functions and T cell density or frequency with specificities have obvious scientific appeal. Apparently, hypersensitivity of the delayed type has effects on leukemia cells that deal with radiation and frequently produce more effective therapeutic outcomes. 62 This is because, rather than any single component, their overall response is shown by them. Moreover, since chemotherapy of hematologic malignancies disrupts the balance of lymphocyte homeostasis, all types of lymphocytes are measured regardless of their specificity. 63 Finally, bone marrow SAGE demonstrates trends in patient response and reaction, influencing disease control and progression, that are relevant to evaluating systemic alterations in the tumor and immune response modification. 64
When it comes to quantifying individual antigen-specific responses, there are significant differences in the approaches commonly used for analyzing cytotoxic and cellular immunity vs humoral responses. 65 Traditional immunological tests for T and NK function usually rely on functional abilities, for example, production of cytokines, proliferation, cytotoxic killing, intracellular cytokine production, and enzyme-linked immunosorbent assays. 66 Antibody responses, on the other hand, can be measured solely by binding to the antigen, as in ELISA. Unfortunately, immune responses to cancer antigens are poor in people with hemological malignancies, and such approaches are likely to underestimate abnormalities in the functional ability of potentially responding cells. To properly analyze the entire range of effective responses of T cells against cancer antigens such as leukemia, it may be necessary to use assays more similar to those used to identify antibody responses. 67 It also includes practically insensitive ones that become insensitive due to energy, fatigue, and immunological tolerance, especially peripheral tolerance. When comparing the number, function, and density of these cell populations in the blood and in the marrow, significant variations arise. CD8+ T lymphocytes against LAA, for example, have been detected in the bone marrow although they are not detected in the peripheral blood of individuals with myeloid leukemia. 68
Effector Immunity Induction
Direct effector immunity induction and its effects, and immunity amplification vs modification of immunity against a response, are the 2 major, complementary, and overlapping investigational immunotherapy methods for hematological malignancies, perhaps within a permissive host milieu. 69 Both kinds are likely to be present in any successful immunotherapeutic strategy. 70 Conventional clinical vaccination procedures used in the prevention of infectious diseases are analogous to the best-known direct effector immunity induction by short-lived effector cells. 71 Nonetheless, stimulating immunity against antigens that are present in the body of an individual before vaccination and stimulating immunity against antigens for which recognition of endogenous nucleic acid by the innate immune system is occurring differ from the mechanism of preventive vaccination for the host who has never been exposed to the antigen before. 72 As mentioned in the earlier section, many currently occurring efforts generate effector immunity by introducing vaccination based procedures on recognized antigens of cancer. In an adjuvant preparation, subcutaneous injection of a peptide like montanide and often concomitant GM-CSF have been used in these experiments normally. This broad method exhibits some success with early promise, employing WT-1 and PR-1 epitopes, but subsequent investigations have demonstrated that it is inadequate to satisfy consistent high avidity LAA-specific responses when it is employed alone. 73 Vaccination approaches have been conducted using the possible neoantigen produced using a protein unique sequence of Philadelphia chromosomes Bcr-Abl. Unfortunately, immune response in individuals having hematological malignancies naturally has inadequate, and such approaches are likely to underestimate abnormalities in the functional ability of potentially responding cells. 74 It may be essential to use techniques more like those used to identify antibody responses to appropriately evaluate the whole range of possible T cell reactions for antigens of leukemia that also include operationally inactive ones. 75
Strategies for Immunomodulation
The cancer and immunity interactions have been known for at least a decade, having evolved through lengthy stages of equilibrium in nature. The tumor microenvironment suppresses immune effectors by forming IDO, MDSCs, and Tregs and releasing cytokines like TGF and VEGF. 76 These changes seem to well describe the antigenicity changes due to immunity pressure. Clinical evidence suggests that stimulation of effector immunity that is specific for cancer can be achieved solely through vaccination methods based on peptides that are productive, increase immunity tolerance, and increase T regulatory cells, while also eliminating a greater portion of the range of T-cells that are specific for antigen. 77 Disease-specific changes occur in the dominant resistance mechanism(s) for the formation of effector cells that produce effector immunity, the maintenance of effector cells, and the implementation of effector function. 78 The pathways mentioned here have been observed in individuals having malignancies of blood tissues to varying degrees. To get through these barriers, investigations are attempted, and it will be critical to figure out which of the various (but limited) resistance mechanisms the tumor is using to avoid immune elimination.70,79
TMB (Tumor Mutation Burden) and its Relationship to Neoantigens
Somatic mutations present in the DNA of tumor cells result in neoantigens and antigens that are produced by mutations, which the immune system recognizes and targets, particularly following therapy with T-cell activators. 80 Transcription and translation of these mutations can occur, and peptides containing neoantigens are handled through the apparatus of antigen processing. They show their expression on the surface of the cell by deposition on MHC molecules.81,82 Moreover, mutations do not always result in the formation of neoantigens. Only a small percentage of mutations result in peptides that, when correctly processed, are placed on or deposited on MHC molecules, and even fewer mutations are recognized by T cells. As a result, not all neopeptides that are present on the surface of cells are immunogenic.83,84 Significantly, the more neoantigens it is expected to produce by the tumor, the more somatic mutations it has, and TMB can be used to estimate the burden of neoantigens. It’s vital to remember that immunogenic neoantigens aren’t the sole element that determines the ability of T cells to recognize and kill cancerous cells.84,85 Some mutations are inactivated, and if they can develop in the pathway for presentation of antigen, they affect the ability of cells to exhibit peptides for activation and stimulation of the immune system. 86 The Janus kinases 1, Janus kinase 2, beta-2 microglobulins, and Serine Threonine kinase 11, or LKB1, are immunologically important genes that can be altered in malignancies, and the presence of these changes can change how tumor mutation burden affects ICB. 87
TMB Against Different Types of Tumors
Many studies have been conducted to determine the degree of variation in TMB and types of tumors. Previously, numerous researchers conducted studies to describe variations of TMB against disease pathologies. 88 They explain that TMB is highest in melanoma, slightly lower in NSCLCs, and lowest in leukemia and pediatric malignancies. Breast cancer, kidney cancer, and ovarian cancer all have moderate degrees of mutational load. 89 TMB levels vary significantly between tumor types, but there is also a considerable TMB range that is found in even the same type of tumor. 90 When comparing gastric cancer and breast cancer with non-small cell lung carcinoma, a great difference in TMB has been found in individuals who are smokers. Stratton et al identified mutational markers that give information about the exact location of mutations in tumors.90,91 Ultraviolet radiation and cigarette components, for example, have proved to be the two most common causes of mutations in melanoma cancer and non-small cell lung cancer, respectively. Subclonal mutations appear later in tumor evolution and have been linked to APOBEC in a variety of tumor types; they can also be found in resistant arising subclones following cytotoxic chemotherapy. 92 The neoantigens can produce clones and have been linked with the priming of T cells and the reaction of immune checkpoint inhibitors. Because intratumoral heterogeneity (ITH) varies by tumor type and can influence anti-tumor immunity, ITH should be considered while assessing TMB. 93
TMB (tumor Mutation Burden) and ICB (Treatment With immune Checkpoint Blockade)
Prior to the discovery of TMB’s function as a biomarker, PD-L1 inhibitor production in the tumor microenvironment was investigated as a biomarker and showed some promise in screening individuals who would benefit most from treatment. 94 PD-L1 quantification has been approved as a complementary analytic to pembrolizumab in NSCLC. Because of the heterogeneous production of PD-L1 inhibitor in the surrounding tumor, there is a lack of standardization and assay interpretation everywhere. PD-L1, when acting as a biomarker, indicates an irregular response to ICB, necessitating the development of some other biomarker. TMB also shows the same issues.95,96 Furthermore, studies imply that TMB may complement rather than replace chemotherapy. Tumor mutation burden and programmed death ligand 1 inhibitor are independent, and they are not related to each other even though they both show predictive variability on numerous occasions. The Checkmate 026 trial, which compared nivolumab to SOC in first-line NSCLC, found no increase in PFS in individuals with 5% expression of PD-L1. Individuals having a greater TMB, that is, tumors based on more than 243 mutations, had significantly better PFS when treated with nivolumab than with chemotherapy, which has an HR of about .62.97,98 The fact that all individuals who underwent TMB testing also had 1% PD-L1 expression limits the validity of this conclusion. Furthermore, patients with fewer than 2/3 mutations below 243 exhibited a bad PFS when treated with nivolumab and showed an HR of about 1.82 when treated with SOC, which emphasized the role of lower TMB as a negative indicator of benefit in the context of successful SOC alternatives. 99 Likewise, in IMvigor211, when atezolizumab is compared with SOC treatment, there is no OS improvement, and there is also no improvement for PD-L1 selection. A study revealed a statistically insignificant but quantitatively enhanced OS, with a HR of .68. With atezolizumab medication, the individuals having elevated TMB plus elevated PD-L1 show a HR of about .50, which is less than the former. Even though tumor mutational burden and PD-L1 inhibitor are not related to each other in many of the studies, the relatively high mutation burden and expression of PD-L1 inhibitor are persistently linked to better effect with a single agent, showing that these separate biomarkers can be utilized in tandem.100,101 Due to the strong response rates shown in early solid tumor studies when PD-L1 and CTLA-4 blockade were combined, it was thought that individuals with a lower mutation burden would get a good effect from the introduction of anti-CTLA-4 and anti-PD-1 antibodies against inhibitors, respectively. In NSCLC patients with a TMB limit of 200 mutations, the efficacy of combination ICB was independent of PD-L1 production.102,103 When nivolumab was introduced in conjunction with ipilimumab in small cell lung cancer, it showed a comparable advantage, which is related to mutation burden but does not depend upon PD-L1. These findings are supported by a non-small cell lung cancer phase III clinical trial, which found that in both individuals with positive PD-L1 and individuals with negative PD-L1, PFS was significantly improved in high TMB vs SOC. 104 These findings imply that elevated TMB is immune sensitive, but T-cell stimulation is regulated in a pathway that depends on CTLA-4 inhibitors. Effector T cells that are activated again can increase the expression of PD-1, although this increase in PD-L1 overexpression is in reaction to CTLA-4 blocking or in reaction to regulatory T cell elimination mediated by anti-CTLA-4, which indicates the interaction with PD-1 blocking. 97 Overall, the current data with TMB and combined immunotherapy refutes the earlier hypothesis that individuals with positive PD-L1 respond to immune checkpoint blockade with inflammatory cancers while those with negative PD-L1 and non-inflammatory cancers do not. TMB has demonstrated its predictive ability in immunotherapy modalities apart from immune checkpoint blockade treatment, which is interesting. 105 It was reported that for people with melanoma who were given adoptive T cells, the mutation burden and load of neoantigen indicated better progression-free survival and overall survival. This study shows that tumors with more potential neoantigens may have a stronger clinical response to TILs that have been grown and reinfused. 106
Gene Therapy
Gene therapy is a therapeutic procedure for correcting or eliminating disease symptoms by introducing exogenous genes into an individual’s cells that can replace or change a malfunctioning gene and cause cell death. Gene therapy uses four basic strategies: gene replacement, gene augmentation, gene modification, and gene blockade. 107 More than 2200 gene therapy clinical trials had been performed and approved worldwide by July 2015. More than 60% of these trials are linked to cancer gene therapy, suggesting that gene therapy is not restricted to genetic diseases but can also be utilized for acquired diseases like cancer, and it has already been proven to be a viable therapy option. Anti-angiogenic cancer therapies that use gene augmentation and gene blockade, pro-apoptotic cancer therapies, immunomodulatory cancer treatments that modify the immune response, siRNA treatments, and therapy based on suicide genes have all been developed in recent years as cancer gene therapy techniques.108,109 Furthermore, because cancer has a complex process involving several signaling channels and mechanisms, and because a single gene can often elicit multiple biological processes and activate multiple signaling pathways, there hasn’t always been a clear distinction between these gene treatments. For example, the tumor protein gene p53 has been shown in multiple studies to have anti-angiogenic properties in addition to triggering apoptotic actions in tumor cells.110,111 As a result, gene therapy based on the p53 gene can be classified as anti-angiogenic and pro-apoptotic. 112 In general, whether gene therapy can be successfully implemented depends on two factors. One of them is that an appropriate gene must be recognized to alleviate disease symptoms, and the second is that this gene must always be directed to the correct place for gene expression products for the treatment of the disease without causing side effects. 113 Because gene therapy has a specific and sensitive treatment approach at the molecular level, it also has many technical challenges to be addressed. One of them is the capacity to build an appropriate gene therapy delivery system.
Delivery Systems for Gene Therapy
The foundation for gene therapy is the development of an effective, safe, and precise delivery system. 108 A good gene delivery method should have some important characteristics that are: It has a wide range of insertion ability, with greater rates of success and a non-invasive administration technique. 114 It enables long-term gene expression, and it has a large target specialized selectivity for the tumor type. It also has safety mechanisms like biocompatibility, stability, and non-immunogenicity, and it should be readily available. 115 A variety of vectors are currently in use. In general, gene therapy delivery technologies can be divided into two categories: viral and non-viral vector systems. 116
Gene Transfer Efficiency of Non-viral Vectors
Non-viral transfection methods include microinjection, magnetic microparticle electroporation with a high voltage pulse, hydrodynamic gene delivery systems, and chemical-based treatments. 117 Nucleofection can effectively and efficiently introduce DNA into cells and has good potential, so it is perhaps a reliable and approved non-viral method for transferring genes into cells. Researches regarding the administration of interfering RNA has aided in the optimization of the stem cell system. 118 RNAi also helps in regulating non-viral vehicle systems of gene transfer for gene combining. 119 Sophisticated techniques are encouraging. Nucleofection facilitates gene transfer with greater efficacy than further techniques, showing varying resistance. Furthermore, numerous RNA delivery mechanisms, like siRNAs, are still used for RNA interference. 120 The method for delivery of RNA was developed by using the traditional concept of gene transfer. 121 Similarly, some studies claimed that plasmid size was indeed critical for transfusion efficacy with non-viral vector structure proving to be the most important factor. Non-viral vectors bind more electrostatically with cytoplasmic membranes having negative charges, which speeds up receptor-mediated endocytosis. 122 As a result, increasing cellular absorption is the most popular way to improve siRNA delivery efficiency. The contrast between viral-based vectors and non-viral vectors in terms of efficiency for transferring genes is remarkable. Non-viral vector efficiency is 1/10th and the efficiency of viral vectors is 1/1000th. For example, for in vitro expression of genes using adenovirus as a vector for delivery, a non-viral vector can also be used, such as lipofectamine plus. 123 Lipofectamine plus has low productivity as compared to viral vectors, whose productivity is 1000 times greater. Varga et al 124 tested PEI and adenovirus vectors to discover that adenovirus is more successful, even 10 to 1000 times. This shows that non-viral vectors are not appropriate for gene therapy, and they have very low efficiency. Vectors containing ligands are used to improve cellular absorption. 125 In the two trials stated above, the researchers identified the steps when non-viral vectors performed significantly worse than viral vectors in terms of gene expression. They determined that non-viral vectors had superior DNA absorption into cells compared to viral vectors. 126 Adenovirus, on the other hand, outperformed non-viral vectors in later steps, such as DNA discharge into the cytoplasm and entrance into the nucleus. The effectiveness of the processes between DNA absorption and transcription will become a focus of non-viral vector development in the future. 127 A comparison of non-viral vector efficiency vs viral vector efficiency can aid in the refinement of non-viral techniques. Learning how viruses attain high efficacy aids the development of non-viral vectors. 128 Instead of utilizing viruses, it may be possible to create reagents with virus-like features to increase gene transfer efficiency. The lower pH, for example, facilitates fusion of viruses with lipid membranes when DNA is transported towards the cytosol using viral vectors from endosomes. 129 Sato et al 130 were inspired by this phenomenon and created a multipurpose envelope-type nanodevice that was 100 times more effective than standard nanostructure tools by using nanotechnology. Production of nanomedicines of the next generation is under consideration, which has underlined the importance of lipid nanoparticle size for optimal tumor tissue penetration. Using the similar endogenous cellular activities that viruses use to improve gene transfer success is a comparable strategy. 131 Endocytosis is the process by which most viruses, like adenoviruses, enter cells. A segment of Del1—which is an extracellular matrix protein—has been identified to improve endocytosis and gene transfer. Non-viral gene therapy has been performed using this function. 132 Because the proteins should never be poisonous or immunogenic, introducing segments and whole proteins that are endogenous to the host is a non-harmful strategy and can be employed again. 133
Gene Therapy Against Tumors
There has been no published research demonstrating more effective gene therapy for treatment using vectors that are not based on viruses. Non-viral gene therapy, on the other hand, seems to have the potential to revolutionised the way cancer patients are treated.
134
However, further work is needed to expand the therapeutic potential of non-viral gene therapy for cancer. The fundamental limitation of non-viral vectors, namely, a poor gene transfer rate, appears to be extremely significant in terms of cancer cell elimination.
135
Furthermore, functional evaluation of non-viral delivery methods like nanoparticles, liposomes, and dendrimers is critical.
136
Clinical results with non-viral vectors for cancer gene therapy seem to be a long way off. We looked through the research and found only 17 papers adopting vectors that are non-viral in cancer animal models.
137
There was some research that showed a significant increase in survival among these reports. Understanding what these techniques have in common could lead to more successful use of non-viral vectors, as shown in Figure 2. Only nine papers have been found in which non-viral vector treatment improved the prognosis of animal models.
138
Klutz et al
139
presented three of the nine investigations that looked at treating neuroblastoma and HCC with a common method. Animals with explanted tumors were given DNA expressing a NIS via non-viral vector administration for research purposes. Then, intravenously, radioactive iodine was injected. The prognosis improved dramatically after repeated treatment. Each of these investigations used a different sort of non-viral vector. It’s worth noting that there was no difference in outcomes between treatments with a pseudodendritic OEI vector and treatments with an EGFR vector. When there are no target-directing elements like the EGFR peptide, greater permeability and persistence of tumor blood capillaries might result in selective DNA accumulation in a tumor. Radiation exposure is a drawback of this method.
140
The findings are significant, showing the feasibility of the introduction of vectors other than virus-based vectors. Sharma et al employed a PLGA-containing nanoparticle to introduce a p53-deficient mouse with a p53 gene to treat explanted prostatic cancer. The tumor protein p53 gene produces the tumor suppressor protein p53.
141
Furthermore, p53 is involved in a variety of signaling cascades that include the cell cycle and stimulate the signaling cascade for proliferation, differentiation, and the growth of cells.
142
Systemic delivery by intravenous injection was also tested, which improved life expectancy significantly but was not as successful as a local injection. EGFR peptide and other target-directing components were not detected, but specific DNA aggregation was. Sun et al
143
investigated the possible preventative impacts of treatment targeted for the prevention of lung metastases through testing the impact produced by p27 on pulmonary metastatic cancers using a non-viral gene transfer approach. Using the p27kip gene, they investigated the impact produced by administration through veins in tumor cells of the colon. They used cisplatin and non-viral gene therapy in numerous cycles of directed combination therapy.
144
Although target-directing elements like the EGFR peptide were still not included with the vector in the Sharma et al
140
investigation, preferential build-up of the DNA complex inside the tumor was found. When paired with cisplatin treatment, gene therapy proved successful. DNA was administered locally in the following investigations. Finoccharo et al used a combination of chemotherapy, gene therapy, and cytotherapy to treat dogs with spontaneous melanoma.
144
The thymidine kinase gene was injected into a tumor using the lipoplex technique, which resulted in a better prognosis. In combination therapy, ganciclovir, interleukin-2, granulocyte macrophage colony stimulating factor (GM-CSF) cell treatment, and gene therapy were found to be highly effective. They have had promising outcomes when treating spontaneous sarcoma in dogs by injecting ganciclovir and interferon into the tumor of the TK gene.
145
The prognosis improved dramatically because of this treatment. By gene transfer, Casey et al
146
used bare DNA to express B7-1 and GM-CSF to treat a fibrosarcoma model animal by gene transfer. Both treatment approaches enhanced prognosis significantly, implying that tumor development is suppressed therapeutically. Fewell et al
147
developed an ovarian cancer peritoneal spreading model. The interleukin-12 gene was consistently given intraperitoneally utilizing polyethylene glycol (PEG) and PEI. This showed a higher prognosis while also causing low harm and increasing survival rates. Combining this strategy with Taxol improved the prospects even more. Kitano et al
148
employed a multifunctional fragment of Del1 that is an extracellular matrix protein, and triggers apoptosis. Constrained to the extracellular matrix, it also improves non-viral vector gene transfer efficiency. The researchers used jet-PEI to deliver cDNA intratumorally in cells with squamous cell carcinoma that are explanted once a week.
149
The prognosis improved dramatically because of this treatment. Examining the similarities between these techniques can be interesting. The non-viral gene therapy targets in this research were mostly refractory malignancies, including malignant melanoma, neuroblastoma, sarcoma, and metastatic malignancy, or peritoneal dissemination.
150
Surgical therapy, chemotherapy, and radiation therapy are the standard cancer treatments. For early stage cancer, surgery plus radiation therapy is often effective, so gene therapy is not required in these circumstances. Therefore, cancer gene therapy should currently be reserved for patients who are not candidates for surgery due to metastasis, chemotherapy due to drug resistance, and radiation therapy because of the limits of radiation.
151
It’s important to talk about the DNA delivery methods used in these experiments. According to Sharma et al,
140
the local injection method of a non-viral vector was more successful than systemic treatment. The concept of customized gene expression regulated by a tumor-specific promoter and certain transcription factors can stimulate the expression of therapeutic genes in cancer cells. Normal cells, on the other hand, are unable to express the therapeutic gene due to a lack of transcription factors.
Given the low gene transfer efficacy of non-viral vectors, injection of them into a sealed lesion, like intraperitoneal or intratumoral injection, increases the concentration of the DNA complex, which appears to be beneficial. There may be signs of gene therapy after a local injection. For example, local ethanol infusion or percutaneous laser therapy are used to treat liver cancer with intrahepatic metastases. Alternatives to these treatments could include local gene therapy. Furthermore, gene therapy could be used to treat disseminated cancer in the peritoneal or thoracic cavity. Furthermore, individuals with obstructive malignancies of the airways or gastrointestinal system who have previously received the highest dose of radiation therapy may benefit from endoscopic DNA injection. We found some occasions when non-viral vectors were administered systemically and were effective. It’s intriguing that DNA complexes develop in tumors without target-directed ligands, likely because of tumor vasculature’s increased permeability and retention. The addition of PEG further into the vector improves the persistence of the DNA complex in the blood for intravenous injection. PEG improves permeability and retention, but it also lowers cell uptake of the DNA complex. This is called the “PEG paradox.” 152 A peptide-based interconnect that connects a phospholipid and PEG and can be destroyed in the tumor by something like a matrix metalloproteinase has indeed been designed to address this. The complete vector can approach a tumor via PEG, and the processed vector can then enter cells by this method. Following systemic delivery, gene expression levels have improved thanks to the production of vectors with complicated functions. However, given the pharmacokinetic variations among systemic and local administration, it may be beneficial to create alternative vectors for systemic and local administration. Multiple cycles of gene therapy are one of the most straightforward methods for enhancing transgene expression. 153 This is a practical technique to take advantage of the benefits of non-viral vectors, such as safety, including both local and systemic injections. Regular intravenous injection of a small fraction of a DNA complex could increase tumor formation without the side effects associated with gene transmission and transfection to healthy tissue. A strategy that leads to the accumulation of proteins that are expressed in the extracellular matrix was previously shown to be successful. Fusion of a therapeutic protein with a “deposition domain” from that of an extracellular matrix protein results in the fusion protein’s posttranslational deposition into the extracellular matrix. 154 In circumstances where the expression level is low, this technique can raise the natural concentration of protein and lengthen the therapeutic period. It can also reduce the protein’s effects on non-target tissues. The selection of genes to be expressed ultimately determines the effectiveness of cancer gene treatments. It is impossible to employ non-viral vectors to transfer DNA into cancer cells. When it comes to non-viral vectors, treatments with autonomously active “anti-tumor genes” like p53 or TK may not be the best approach to take. To compensate for the drawbacks highlighted for non-viral vectors, new ideas are needed to advance the research. In the research mentioned above, proteins derived by transfected cells seem to have beneficial therapeutic effects on untransfected cells. 154 Successful non-viral vector research frequently chooses proteins that can alter the cancer microenvironment. Cancer stroma has recently been discovered to have a crucial role in cancer proliferation and metastasis. Cancer cells release factors that stimulate cells in the stroma, which in turn promote cancer cells to grow and move. As a result, altering the cancer microenvironment may be a viable treatment option. Approaches targeted at modulating stroma, such as the injection of IL2, IL12, or GM-CSF, seem to contribute to the efficacy of non-viral vector-based gene therapy. These cytokines’ biological characteristics are likely to play an important role. Cytokines, for example, can act in very low quantities (in the pmol/L range). 155 This compensates for the non-viral vectors’ low transfection effectiveness and small amounts of protein expression. A Del1 component can likewise be used to manipulate the microenvironment in a similar fashion. Radiation emitted by radioactive iodine that accumulates in cells that contain the sodium iodide symporter can harm nearby cells.
Nanoparticles-Based Gene Therapy
The aim of gene therapy is to replace a non-functional or damaged gene with a normal, functional one. For successful gene transfer, extracellular and intracellular barriers must be overcome. 156 By electroporation, sonoporation, or laser irradiation, the lipid bilayer membrane is routinely penetrated to enable the systematic transport of bare pDNA. The immune system is deliberately avoided or repressed. Minicircles that are immunologically inert are also used for effective systemic gene delivery. 157 Traditional single-function systems have been surpassed by a simple combination of multifunctional envelope-type nanodevices (MENDs) as non-viral gene therapy delivery devices. Several promising nanoparticles for gene delivery and immunomodulation for cancer therapy are undergoing clinical studies. Fakhri and colleagues 158 used a hybrid silver–gold (Ag–Au) bimetallic nanoparticles system on the human BC cell line MCF-7, which showed effective antiproliferative effects. A magnetic field can accelerate gene editing in vivo, as suggested by Zhu et al 159 for CRISPR-Cas9 therapeutic systems. In tumor-bearing mouse models, they employed recombinant baculovirus vectors combined with nanomagnets to increase transduction in target cells and precise tissue delivery while limiting genotoxicity and maximizing effectiveness. 160 Gene therapy is combined with various diagnostic systems to identify, treat, and evaluate the therapeutic response as part of a diagnostic and therapeutic strategy. Nanoparticle-mediated siRNA therapy modulates gene expression by interfering with RNA, and it offers a lot of promise in cancer treatment. 161 For non-invasive real-time imaging of siRNA delivery to tumors using MRI, the siRNA delivery system is tagged with imaging agents such as dextran-coated superparamagnetic nanoparticles. The use of additional imaging agents to label siRNA delivery systems could aid in therapy monitoring and perhaps predict therapeutic outcomes. 162 Biodegradable DNA scaffolds like DNA dumbbells, DNA nanoribbons, and spherical nucleic acids represent a safe, non-immunogenic alternative for accurately inserting targeting ligands, siRNA medicines, and chemotherapeutic medications. 163 To serve as imaging and diagnostic platforms, self-assembling DNA nanostructures are also modified using biocompatible organic and inorganic fluorescent probes. 164 DNA nanostructures also appear to be flexible carriers for Cas9/sgRNA delivery for CRISPR-based targeted gene editing. 165 The visual, mechanical, electrical, and physicochemical properties of graphene, a 2D nanomaterial comprised of monocarbon atoms arranged in a honeycomb lattice, are all intriguing. Scientists are interested in its possible application in biomedicine because of its ease of functionalization. Lipophilic and hydrophilic agents have high drug loading ratios due to their wide surface area, delocalized electrons, and chemically polar groups on their surfaces. Graphene-based nanomaterials (GBNs) comprise graphene derivatives like graphene oxide, reduced graphene oxide, nanographene oxide, and graphene QDs, which are used in cancer theranostics in a synergistic manner. Drug loading ratios with GBNs can reach 200 wt%, which is far higher than in any other pharmaceutical nanocarrier technology.166,167 In tumor cells, transferrin receptors are overexpressed. Patients were given a targeted nanoparticle formulation of siRNA (CALAA-01), which included a four-component self-assembled composition of cyclodextrin-containing polymer, PEG, steric stabilizing reagent, and human transferrin as a specific focus for binding to receptors. 168 This is a clinical translation of self-assembling cyclodextrin polymer-based nanoparticles for siRNA delivery. The most frequently mutated gene in aggressive TNBC is TP53, although it is not druggable. 169 POLR2A is a TP53 neighborhood gene and a potential collateral target. Most of the TNBCs with TP53 copy number reduction also have hemizygous loss of POLR2A, making cancer cells particularly vulnerable to additional reduction or blocking of this gene. TNBC has a partial removal of this critical gene, which could lead to a targeted therapy approach. A new low pH prompted nano-bomb attacking POLR2A was recently developed and patented. 170 When this POLR2A inhibitor-containing nano-bomb was dropped inside a TNBC cell, it exploded 100 times its original size, killing only the cancer cells by a controlled release. Most potential nanoparticles now in clinical studies contain heavy metals like cadmium and selenium, which elude renal and hepatic elimination and pose a toxicity risk. Other diagnostic testing and imaging approaches may be hampered by the prolonged particle retention in the body. As a result, developing nanoparticles with optimum clearance or biodegradability is a must before clinical application. 171
Nanoparticle-Mediated Targeted Death-Induced Gene Therapy for Breast Cancer (BC)
With only a few FDA-approved gene therapy medications and many more potential therapies in the pipeline, gene therapy has sparked a lot of interest in the academic and clinical arenas. Several genetic mutations and abnormalities discovered in BC ideal prompt the use of gene therapy, in which a nucleic acid-based medication is administered to cancer cells to rectify genetic faults or eradicate tumors. Recent advances in BC gene therapy include RNA interference techniques and DNA immunization against overexpressed tumor antigens. 172 However, successful DNA delivery is a bottleneck since most nucleic acid-based medications are susceptible to circulating “nuclease,” which can damage them as they reach their target. 173 A variety of viral and non-viral techniques have proven successful. We address death-induced gene therapy in British Columbia using nanotechnology. The ideal transfection vector seems to be a critical hurdle in gene therapy, and nanoparticles appear to be a promising candidate for BC gene treatment. 174
Anti-tumor Targets for Gene Editing
To optimize efficacy while minimizing damage, selecting a good gene target is important for successful CRISPR-based anticancer treatment. The treatment efficacy of CRISPR/Cas-based systems will be influenced by complex interactions among tumor, host, and environment when considering therapeutic targeted tumor therapy.175,176
Tumor Microenvironment-Associated Gene Targets
Representative Therapeutic Targets in Current Studies.

Immunosuppressive Gene Therapy
Cancer cells are recognized for their inability to trigger an immunological response, limiting the immune systems ability to detect and eliminate cancer cells. Cancer cells have low immunogenicity due to low expression of important histocompatibility complex antigens in cell membranes.
185
These antigens cause T-cell activation, which is the first step in producing an immune response. Furthermore, cancer cells release immunosuppressive chemicals such as IL-10 and TGF in a variety of tumor forms, reducing the likelihood of an immune response to them. Figure 3 depicts a variety of techniques used to elicit an immune response against cancer cells. Clinical trials, for example, have used systemic administration of cytokines like interferons (IFNs) and interleukins (ILs) to increase immunity to cancer cells, leading to tumor regression but with significant limitations that limited their success.
186
Cytokines are those proteins that degrade quickly in the bloodstream after being administered systemically, reducing their ability to generate an immune response against tumor cells. To counteract this systemic breakdown, they must be given in large dosages to guarantee that enough cytokines reach the tumor location. This strategy, however, is not feasible because cytokines have been shown to be lethal at high concentrations. Local or targeted delivery of cytokines has been sought to overcome the limits of systemic administration with better outcomes.
187
To retain their efficacy in boosting immunity, cytokine or protein injections must be given regularly. Immunomodulation gene therapy, in which cytokine-encoding genes are administered into the patient instead of proteins to encourage the continuous secretion of cytokines from the cells, is one method for avoiding the need for recurrent injections. Immunomodulatory genes are frequently delivered using one of two methods. First, a vector (typically viral) is utilized to transfer an appropriate gene to the tumor location, where it increases the genes cytokine release. As a result, T cells are activated against tumor antigens, leading to the establishment of an immunological response against tumor cells and their eradication through the immune system. The second method, known as cancer vaccination, involves inserting the gene encoding the desired cytokines into excised cancer cells and reinjecting them into the patient, where the antigens released by the injected cells trigger an immune response against the cancer cells. Many studies have employed genes that encode unique prostate antigens to trigger an immune response that is only aimed at prostate cancer cells. Xiao et al employed lentivirus to introduce PSCA to prostate cancer. After injecting the medication into mice, they noticed a large rise in CD8+ and CD4+ T cells.186,188 CRISPR technology use in several facets of cancer therapy.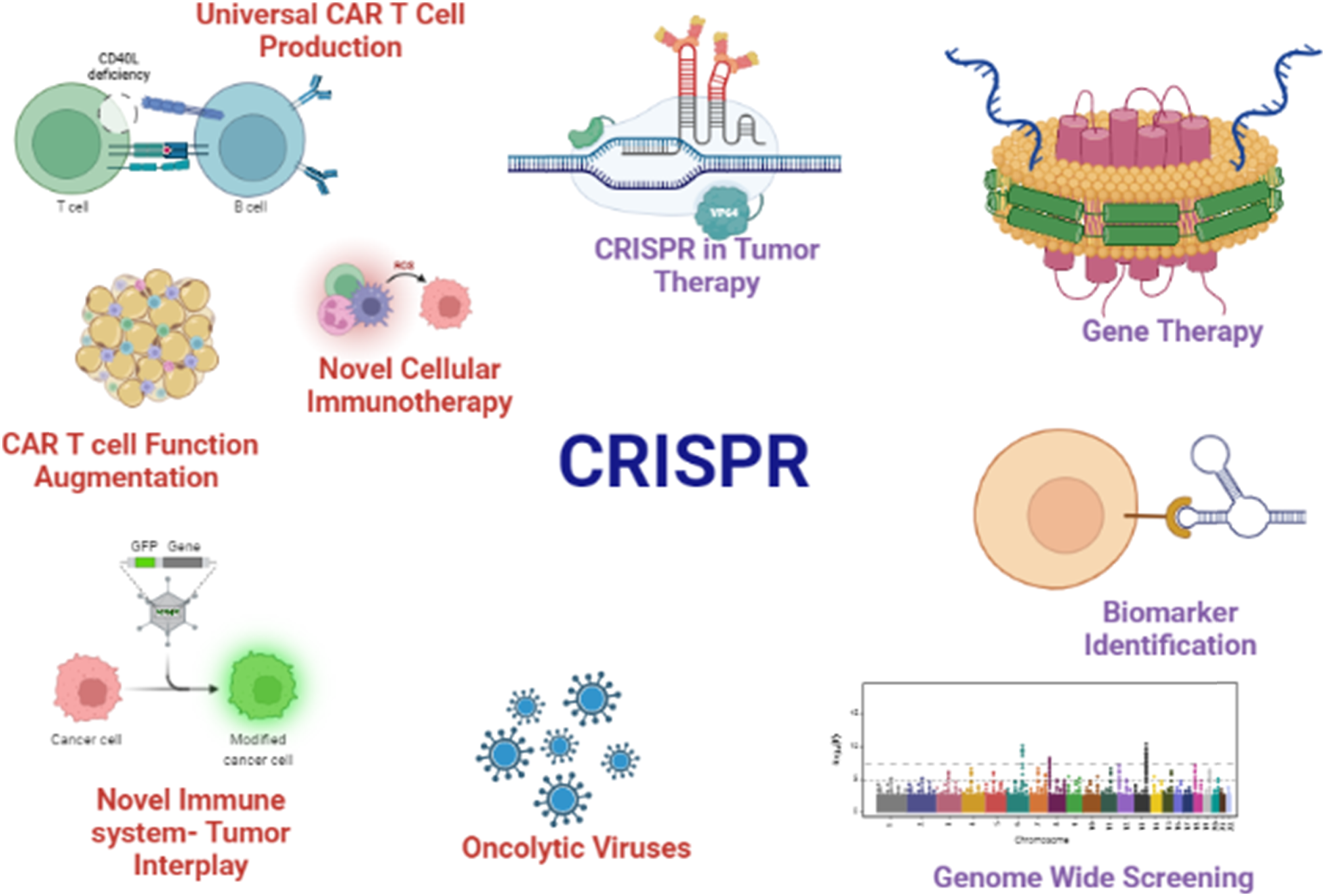
This technique may also have a preventative effect on tumor growth and a protective effect against metastasis. Similarly, Maurer et al 188 investigated the immune-stimulatory effects of introducing the cytosine–phosphorothioate–guanine gene to dendritic cells in the bone marrow. This resulted in a considerable secretion of IL6 and IL12, indicating that they could be used as an ex vivo adjuvant. Other researchers have improved immunoresponse by creating ex vivo prostate cancer cells to have cytokines and/or cytokine-receptor genes, allowing them to respond more effectively to cancer cells and give cancer immunity. Morris et al 189 utilized TRAMP-C2 murine prostate cancer cells to express IL15 and its receptor IL15 R, and then used them to vaccine animals. Their findings revealed a high level of CD8+ T cells and natural killer cells. Hull et al developed an in situ vaccination for the RM9 prostate cancer model in mice. An adenovirus carried a mixture of two genes that encode IL-12 and B7-1 in the vaccination. In vivo studies following in situ injections revealed considerable tumor growth inhibition. 190
Suicide Gene Therapy
Suicide gene therapy is among the most important methods in gene therapy, with the goal of selectively killing cancer cells while leaving healthy cells alone. It works by transferring a particular gene that codes for an enzyme that can transform prodrugs into cytotoxins in cancer cells that kill the host cells. 191 The Escherichia coli cytosine deaminase (CD), which converts the prodrug 5-fluorocytosine (5FC) to 5-fluorouracil (5FU), and the herpes simplex virus thymidine kinase gene (HSV TK), which also converts the prodrug Gcv to Gcv mono-or triphosphate, have both been extensively studied in prostate cancer suicide therapy. 192 The bystander effect, that is the potential of the activated prodrug to stimulate cytotoxic activity in the target tissue and other surrounding cancer cells, generating a killing zone all around cells introduced with the suicide gene, is the main benefit of this therapeutic approach. This effect could be caused by passive transport and gap junctions, or activation in the tumor’s microenvironment. 193 This bystander effect was discovered in the HSV-TK-GCV suicide gene therapy system and is thought to be caused by intracellular diffusion of the active prodrug Gcv triphosphate across gap junctions between neighboring cells. 194
The HSV TK-GCV system, on the other hand, has a much smaller bystander effect than CD-5FU. A strong bystander impact has been discovered in the CD-5FU suicide gene therapy system. It diffuses easily through the tumor site without the need for cell-cell adhesion, which has been attributed to this system’s ability to engage the immune system against cancer cells by activating natural killer cells such as CD8 and CD4, making it more effective in treating metastasized cancers. 195 The combo of the two suicide therapy systems is said to increase prostate cancer treatment outcomes. Yoshimura et al 196 used a cationic lipid as a vector to study the therapeutic potential of HSV TK-Gcv and CD-5FC both alone and in combination with the LNCaP prostate cancer cell line.193,196 Each system reduced cell viability by roughly 60% on its own, but when combined, they killed 77% of LNCaP cells. Similarly, in the pIRES plasmid, the HSV TK and CD genes were combined, with the Cytomegalovirus promoter replaced by PSMA for better targeting of prostate cancer cells. In vitro studies indicated growth suppression of 70% and 60% in LNCaP and PC3 cell lines, respectively. These results outperformed those seen with either therapy alone.
For suicide gene therapy, many delivery and effector methods are presently in use. Each of them has been found to be effective against certain cancer cell lines. Combining various systems has been found to improve anti-tumor efficacy. In tumor models with significant tumor vascularization, combining interventional techniques has also demonstrated superior tumor control in less time than a single modality therapy. Novel chemicals are being developed for non-viral vectors in order to maximize DNA loading capacity while minimizing toxicity. Along with novel promoters of overexpressed genes, a variety of innovative approaches to suicide gene therapy are being investigated. Suicide gene therapy has shown that the major lesion can be adequately managed even when the pharmaceutical formulation is administered to a region far from the underlying sickness. The same is found for the control of distant metastases. Suicide gene therapy strengthens the immune system both locally and remotely to metastatic locations.
Conclusion
Cancer treatment has been revolutionized by cancer immunotherapy, which mainly targets proteins 1 (PD-1) and 4 (CTLA-4). Vaccines that target dendritic cells for T-cell responses have also been used, but they have proven difficult to manufacture and are limited for patients. Oncolytic viruses activate immunity for tumor cells. T-VEC showed a much higher ORR of 50% when used in combination with inhibitors of checkpoints, for example, PD-1 and PVSRIPO, and has more potential for heavy gliomas. When combined with PD-1 and a BRAF inhibitor, the dendritic cell growth factor FLT3L produces significantly better results than when used alone. CARs on T cells detect tumor antigen and stimulate the formation of cytokines and interleukins. As a result, various antigens such as VEGFR2, ERBB2, and others are being studied. Combination therapies are more effective than single therapies and have more PFS and OS. Nivolumab and ipilimumab, bevacizumab and I-F, BRM and Avastin, CTLA-4 and PD-1 are more effective in cancer treatment. Biologic use for cancer treatment should be safe according to pharmacokinetic variables like PK, ADA, and DDI. TMB also shows predictability in immunotherapy modalities. Gene therapy is used for genetic and acquired tumors and uses four techniques: gene replacement, gene augmentation, gene modification, and blockade. Anti-angiogenic cancer therapy uses gene augmentation, gene blockade, immunomodulatory and pro-apoptotic cancer treatments, suicidal gene treatment, and siRNA treatment. Viral vectors for cancer treatment have more efficacy than non-viral vectors. Nanodevices have also been used for gene delivery. Gene therapy is highly effective when used in combination with other therapies like chemo and cytotherapies. MENDS have also been used for gene delivery. Death-inducing gene therapy using nanotechnology is much more effective for breast cancer. For the treatment with gene therapy and CRISPER/Cas, the main target has to be the best target. For immunomodulation, genes that are involved in the production of cytokines are induced in the body by viral or vaccine methods, and they are very effective against tumors. Like other combination therapies, suicidal gene therapies are more effective than single suicidal gene therapies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
