Abstract
Background
People living with HIV (PLWH) have a worse prognosis than the general population. Locally advanced or metastatic bladder cancer (BCa) in PLWH has gradually been increasing in recent years. Immune checkpoint inhibitors can improve antitumor activity in the general population, but relevant data in PLWH are unknown. We thus evaluated the efficacy and safety of tislelizumab in PLWH with locally advanced or metastatic BCa.
Methods
This retrospective study included 24 patients with locally advanced or metastatic BCa, both HIV positive or negative who underwent tislelizumab treatment (200 mg i.v. every 3 weeks, Q3W) from the multi-centers between December 2019 and March 2022. Demographic details, clinical data, and cancer status were collected. The overall survival (OS), progression-free survival (PFS), overall response rate (ORR), disease control rate (DCR), clinical benefit rate (CBR), and treatment-related adverse events (TRAEs) were recorded and evaluated.
Results
A total of 24 individuals were chosen for this study, 10 had HIV and the other 14 did not. The median OS in the HIV-negative group was 62.3 (95% CI, 52.6 to 72.2) was no longer than that of the PLWH group 41.9 (95% CI, 32.9 to 51.0) weeks (HR .7, [95% CI, .17 to 3.30], P = .70). Furthermore, the median PFS in the HIV-negative group was 50.0 (95% CI, 36.2 to 63.9) was also no longer than that of the PLWH group 35.9 (95% CI, 25.5 to 46.3) (HR, 1.34, [95% CI, .38 to 4.69], P = .63). Of 24 patients, treatment-related adverse events, grade 3 or 4 occurred in 2 in the PLWH group and 3 in the HIV-negative group.
Conclusion
This retrospective multi-center study suggested that tislelizumab may provide encouraging antitumor activity and could be generally well tolerated. In this retrospective analysis of patients with locally advanced or metastatic BCa, it seems that PLWH may have similar overall and progression-free survival compared to HIV-negative cases.
Background
Bladder cancer (BCa) is among the most common cancers worldwide, with approximately 550,000 new cases annually. 1 Despite local therapy, approximately one-third of patients will relapse and develop metastatic diseases. 2 Additionally, about 4% of patients have distant metastases at initial diagnosis. 3 With the same disease stage and treatment, people living with HIV (PLWH) with cancers may have a worse prognosis than the normal population. 4
The prognosis for patients with locally advanced or metastatic BCa is dismal. For decades, the mainstay of treatment has been cisplatin-based therapy. Gemcitabine plus cisplatin (GC) chemotherapy regimens have been a standard option for patients with locally advanced or metastatic bladder cancer, but unfortunately the median overall survival (OS) was approximately 13 to 15 months in patients with locally advanced or metastatic BCa.3,5-7 Thus, there is a need for additional regimes that improve the prognosis. Immune checkpoint inhibitors (ICIs) have deeply changed the oncology field and become the hotspot of bladder cancer treatment.8,9 BCa is considered immunogenic with a high PD-L1 expression level and high somatic mutation burden, providing a theoretical basis for immunotherapy.10,11 Pembrolizumab, nivolumab, and avelumab have revolutionized urothelial carcinoma care after the failure of platinum-based chemotherapy, with 17%–21.1% of patients achieving an objective response and a higher survival time.8,12-14 Most clinical trials with ICIs have excluded patients with chronic infections, including HIV. However, anti-PD-1/PD-L1 therapy has been proven safety for PLWH in some clinical trials, and the safety of pembrolizumab and durvalumab in patients with HIV and advanced cancer has been partially proven in recently published research.15-17 The tumor incidence in PLWH has an increasing trend, but immunotherapy is rarely used in this population. The evidence for using ICIs in PLWH with locally advanced or metastatic BCa is still lacking.
Tislelizumab, a humanized IgG4 monoclonal antibody with high affinity and binding specificity for PD-1, can minimize binding to FcγRs macrophages and reduce antibody-dependent phagocytosis which is a potential mechanism of T-cell clearance and resistance to anti-PD-1 therapy.18,19 Tislelizumab has also been shown to produce meaningful clinical efficacy and has a manageable safety profile in general patients with UC.20-22 HIV infections causing immune deficiency may accelerate the progression of cancers, but what about the great control of HIV viral load? However, are the efficacy and safety of tislelizumab in PLWH similar to the general population? In the present study, we investigated the efficacy and safety of tislelizumab for PLWH with locally advanced or metastatic bladder cancer in multi-centers.
Methods
Study Design and Patient
This is a retrospective study. Patients who were diagnosed with locally advanced or metastatic bladder cancer at our centers (Beijing Youan Hospital, Beijing Ditan Hospital, and Beijing Friendship Hospital) from December 2019 to August 2022 were screened in this study. The clinical and laboratory data were retrospectively retrieved by telephone and hospital medical case records. The selected criteria were as follows: Patients (1) had controlled HIV viral load, (2) had no central nervous system metastases, (3) had complete data, including general laboratory and radiological data, and related oncology treatment data; (4) had an Eastern Cooperative Oncology Group (ECOG) performance score of ≤ 1; and (5) must undergo radical surgery or maximal TURBT. The included patients may have experienced chemotherapy or were unable to tolerate chemotherapy. The included cases were split into a PLWH group and an HIV-negative group. Then they received tislelizumab (200 mg i.v. every 3 weeks, Q3W). The end of treatment occurred in confirmed disease progression, duration of treatment reaching 1 year, unacceptable toxicity, and patients’ disagreement. The PD-L1 was centrally tested utilizing archival or fresh tumor tissues by PD-L1 IHC 22C3 pharmDx test (Dako, Carpinteria, California, USA). PD-L1 expression was calculated as a Combined Positive Score (CPS), defined as the number of PD-L1 staining cells (tumor cells, lymphocytes, and macrophages) out of the total number of tumor cells, multiplied by 100.
The reporting of this study conforms to the STROBE guidelines. 23 All procedures followed the Clinical Practice guidelines. We have de-identified all patient details. The patients were selected consecutively. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Beijing Friendship Hospital affiliated with Capital Medical University (No. 2023-P2-066-01) on 7th, March 2023. The Ethics Committees of Beijing Youan Hospital and Beijing Ditan Hospital also passed the record. The follow-up period was up to November 2022.
Endpoints and Assessments
The primary endpoint was to assess clinical efficacy. The objective progression-free survival (PFS), overall survival (OS), overall response rate (ORR), disease control rate (DCR), and clinical benefit rate (CBR, response up to 183 days) were recorded and reviewed. Tumor responses were assessed by the investigator, at baseline and then every 3 cycles, according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, by CT from our hospital or local hospitals. Complete response (CR) and partial response (PR) were confirmed with a repeat scan at least 4 weeks after the initial response. The secondary endpoint was treatment-related adverse events (TRAEs). TRAEs were monitored for safety assessments. AEs were assessed and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) (V.4.0).
Statistical Analysis
We estimated the sample size using two-proportion testing, 24 with a set of power .9 and a difference of .5, and 22 patients need to be included. The sample size software was PASS v.20. Baseline disease characteristics and AEs were summarized using descriptive statistics. The comparisons between the 2 groups were conducted using the chi-square test. For ORR, DCR, CBR, and 95% CIs were constructed using the exact method. The Kaplan–Meier method was used to estimate the PFS and OS for each group. Statistical analyses were performed in SPSS 22.0 (IBM, Armonk, NY, USA) and R for macOS v4.2.1 (www.r-project.org). Differences were considered statistically significant at P < .05.
Results
General Characteristics
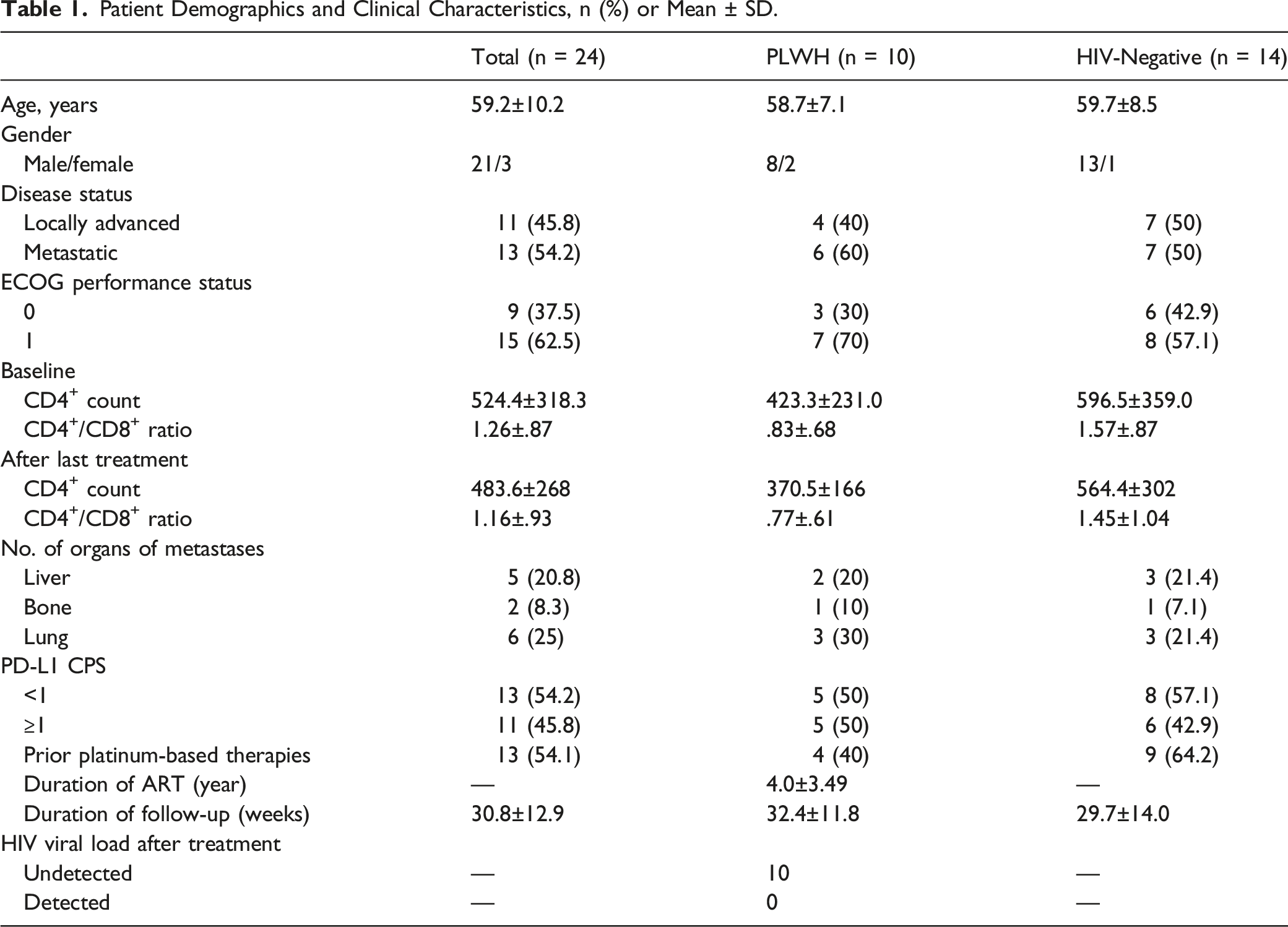
A retrospective analysis of 24 patients with locally advanced or metastatic bladder cancer was enrolled in the study, including 10 PLWH and 14 HIV-negative patients. The flow chart of patient selection is shown in Figure 1. The baseline characteristics between the 2 groups are shown in Table 1. There were no differences between the 2 groups. In the PLWH group, no change in viral load was detected after tislelizumab treatment, and HIV RNA was not detected in any of the cases. Flow chart of patients’ selection. Patient Demographics and Clinical Characteristics, n (%) or Mean ± SD.
Antitumor Activity
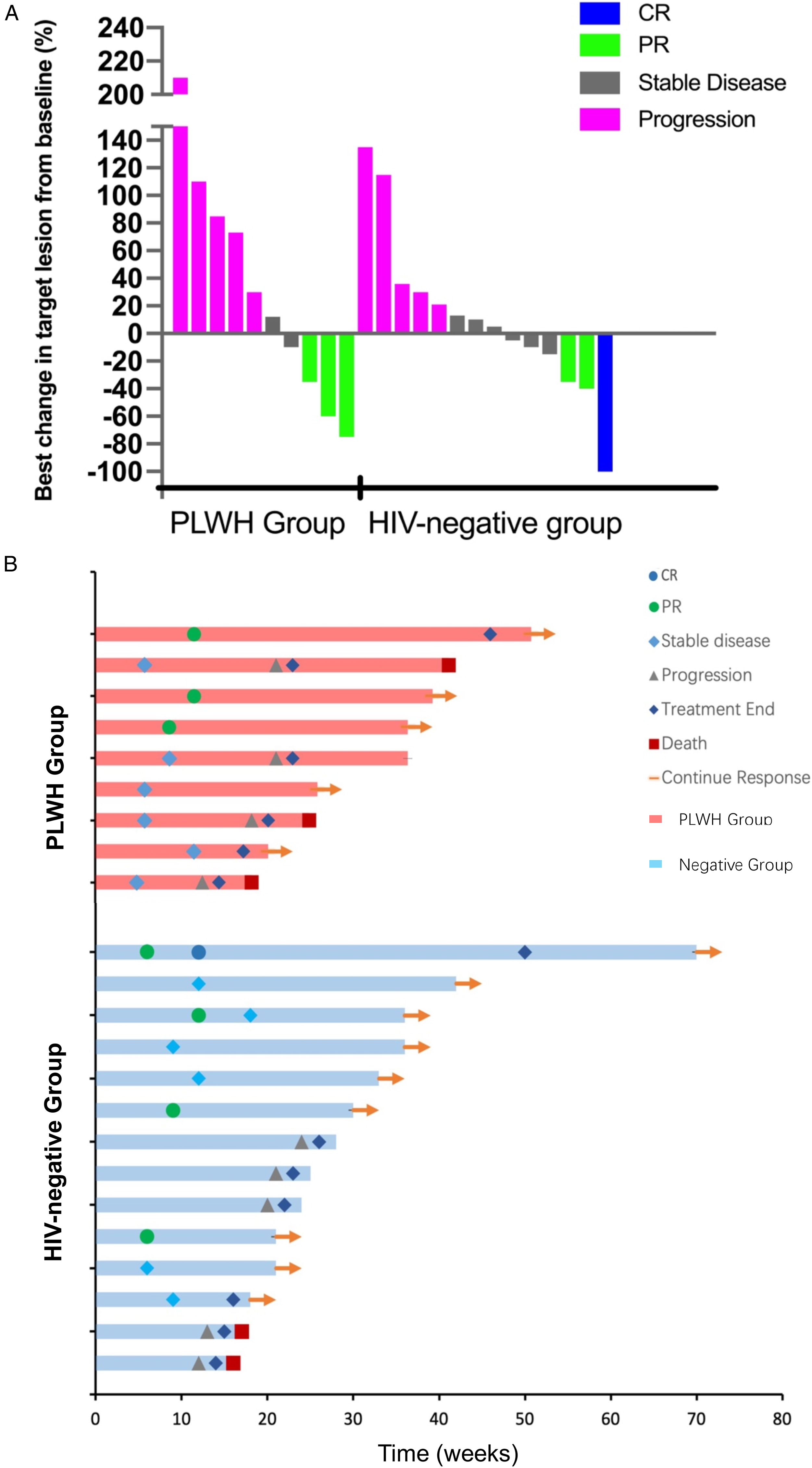
As of November 30th, 2022, all 24 patients were included in the efficacy evaluable set. The median survival follow-up was 32.4 (18 to 53) weeks in the PLWH group and 29.7 (16 to 70) weeks in the HIV-negative group. The median OS in the HIV-negative group was 62.3 (95% CI, 52.6 to 72.2) was no longer than that of the PLWH group 41.9 (95% CI, 32.9 to 51.0) weeks (HR .7, [95% CI, .17 to 3.30], P = .70). Furthermore, the median PFS in the HIV-negative group was 50.0 (95% CI, 36.2 to 63.9) was also no longer than that of the PLWH group 35.9 (95% CI, 25.5 to 46.3) (HR, 1.34, [95% CI, .38 to 4.69], P = .63) (Figure 2). As shown in Figure 3A, 9 of 24 patients who had post-baseline lesion assessment showed tumor shrinkage. Of all patients, the confirmed objective responses were observed in 6 patients, including 3 PRs in the PLWH group, 1 CR and 2PRs in the HIV-negative group, 5 responses (50%) in the PLWH group, and 9 (64.2%) responses in the HIV-negative group ongoing (Figure 3B). Tumor responses in all 24 patients are summarized in Table 2. The ORR was achieved in 30% (95% CI, 6.6 to 65.2) in the PLWH group and 21.4% (95% CI, 4.6 to 50.7) in the HIV-negative group. The DCR was 50% (95% CI, 18.7 to 81.2) and 64.2% (95% CI, 35.1 to 87.2), respectively. The CBR was 30% (95% CI, 6.6 to 65.2) vs 35.7% (95% CI, 12.7 to 64.8). All the patients in our study underwent a PD-L1 expression test, 1 with PD-L1 <1 achieved PR in PLWH. However, no statistically significant difference was observed between the 2 groups (Table 2). Due to the small sample size, these data should be interpreted with caution. Kaplan–Meier analysis of overall and progression-free survival for the PLWH group (red line) and HIV-negative group (blue line). Shown are Kaplan–Meier estimates of overall survival (A) and progression-free survival (B) according to Response Evaluation Criteria in Solid Tumors, version 1.1. CI, confidence interval. Clinical activity. (A) Best change in target lesion from baseline in all evaluated patients; (B) tumor responses over time. Disease Response Per RECIST v1.1 RECIST v1.1, Response Evaluation Criteria in Solid Tumors version 1.1; PLWH, people living with HIV; CI, confidence interval.

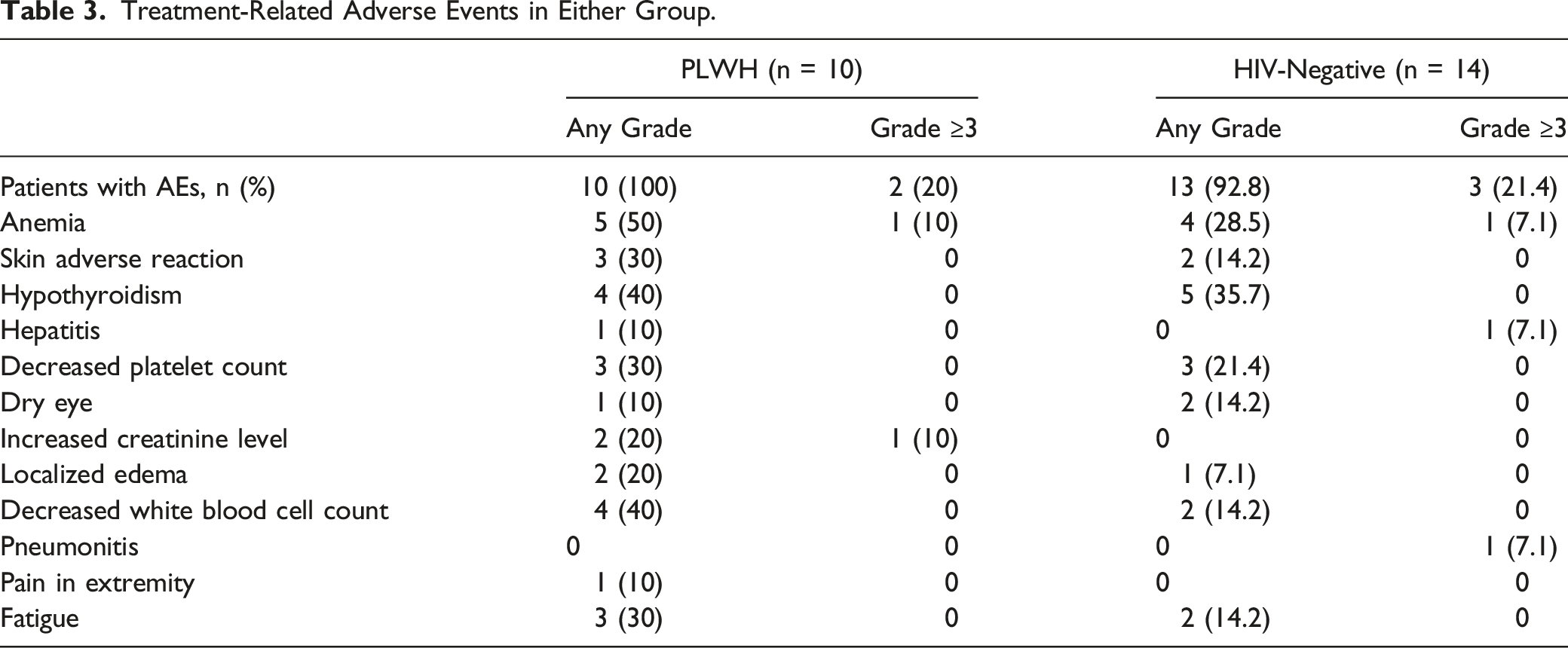
Safety
Treatment-Related Adverse Events in Either Group.
Discussion
Tislelizumab showed a similar clinically meaningful antitumor activity and a manageable safety/tolerability profile in PLWH with locally advanced or metastatic BCa. This retrospective study investigated the antitumor activity and safety of tislelizumab in PLWH with locally advanced or metastatic BCa compared with the HIV-negative population. As of the data cutoff date, this study showed similar PFS, OS, ORR, DCR, and CBR in the 2 groups. In addition, no significant differences were observed regarding safety and tolerability between PLWH and HIV-negative groups. The incidence of grade ≥3 TRAEs was not a significant difference in the 2 groups.
Immunotherapy has been shown in many clinical trials to be superior to chemotherapy in survival time and overall response rate, and the median PFS and OS in first-line chemotherapy typically observed can range from approximately 6.8 to 8.8 months and 11 to 15.3 months, respectively. 25 Some clinical trials showed the advantage of immunochemotherapy in metastatic UC. Pembrolizumab was tested in 370 patients with advanced UC or metastatic UC ineligible for cisplatin, showing an ORR of 29%. 26 Furthermore, pembrolizumab demonstrated a significant OS improvement in a phase III RCT, where 270 patients received pembrolizumab and the median OS was 10.3 months. 27 Atezolizumab was evaluated in the same patient population in a phase II trial (n = 119) showing an ORR of 23%. In IMvigor130 study, the median OS and PFS were 16 months and 8.2 months, respectively, that showed the addition of atezolizumab to platinum-based chemotherapy prolonged the PFS in patients with metastatic UC. 28 The Check-Mate 275 single-arm phase II trial showed the primary endpoint of ORR was 19.6% and OS was 8.7 months. 29 The phase 3 KEYNOTE_045 study 30 reported that patients with advanced UC who received pembrolizumab monotherapy had a similar median OS (10.1 months) and ORR (21%), as we report tislelizumab in this study. Recently, a phase 2 trial showed that OS (9.8 months), ORR (24%), and DCR (38%) after tislelizumab were found to be the same as our study. 22 As discussed above, the anti-tumor outcome of this study confirmed the results of the previous analysis despite only 24 cases. In the IMvigor211 phase III, atezolizumab did not improve the endpoint of OS (11.0 months) compared with the chemotherapy. 31 The endpoint of OS cannot reach in IMvigor211 trial may be due to patients not receiving bladder cytoreductive surgery. In our study, the clinical trial design excluded this factor. In our study, due to the small sample size, the subgroup analysis of PD-L1 expression cannot be assessed. In the single-arm phase 2 trial, tislelizumab demonstrated clinical benefits in high PD-L1 expression patients with metastatic UC. 22 However, whether the PD-L1 expression affects the prognosis of immunotherapy remains controversial. The CheckMate 274 trial showed nivolumab monotherapy has a longer disease-free survival among patients with high-risk muscle-invasive UC after radical surgery, despite the PD-L1 expression level. 8 In a multi-cohort phase 2 study, the antitumor responses were seen irrespective of PD-L1 expression, even with low PD-L1 expression, camrelizumab achieved an ORR of 31.6% in advanced or metastatic UC. 32
The 5-year survival rate can reach 94% but drops at 50% when the disease is muscle invasive. 11 The availability of specific biomarkers to predict the response to targeted therapy is desirable. To date, no biomarkers could predict therapy response exactly. As immunotherapy has been shown in many clinical trials to be superior to chemotherapy, predictive biomarkers like PD-L1 could be particularly helpful.13,25,29,30 In addition, the circulating tumor cells (CTCs) could be a potential prognostic marker for risk stratification in patients with non-muscle invasive bladder cancer (NMIBC). 33 The test of PIK3CA, FGFR3, and ERBB2 mutations may predict desirable treatment option in patients who were detected positive in these mutations. A panel of 9 cytokines measured in urine could improve the accuracy of 85.5% to predict recurrence after intravesical BCG. 34 Previous studies showed that BCG-unresponsive patients reported worse oncological outcomes. 35 Both previous studies and our study suggest the potential of tislelizumab as a PD-1 inhibitor in advanced uroepithelial carcinoma, and we hypothesize that whether its role in NMIBC could be extended to achieve recurrence prevention and whether it would be superior to that of BCG will require further experiments in the future to confirm. Meghani reported that intravesical pembrolizumab could be safe and feasible in BCa unresponsive to BCG, but with small sample size, further study needs to be performed. 36
There were no new TRAEs observed with the treatment of tislelizumab in PLWH, which was consistent with those of the previous tislelizumab studies in other advanced solid tumor trials. PLWH who are receiving ART have a near-normal life expectancy, and now cancer is one of the leading causes of mortality. 37 Despite growing data demonstrating the safety of anti-PD1/PD-L1 therapy in PLWH, such patients have routinely been excluded from ICB clinical trials. Only 5 of 107 evaluable trials (4.7%) allowed the enrollment of PLWH in a recent meta-analysis of published clinical trials. 38 One hesitation in testing anti-PD-1 therapy in patients would not have sufficient underlying T-cell immunity to benefit from therapy. In our study, the well-controlled PLWH inclusion may reduce the insufficient T-cell count. The safety of immunotherapy for multiple types of cancer associated with HIV has been demonstrated in some clinical studies.15-17,22 Maria 17 evaluated the feasibility and safety of durvalumab treatment in PLWH with advanced cancer, including 20 patients, but only 1 had bladder cancer. It showed that durvalumab treatment was feasible and safe in PLWH with cancer. Uldrick demonstrates 16 that pembrolizumab monotherapy does not appear to have a detrimental effect on CD4+ T-cell count. In our study, it was like that of previous studies, with no significant effect on CD4+ T-cell count. Whether the tumor prognosis of PLWH with well-controlled HIV is similar to that of the general population remains unclear. Our team’s previous studies have confirmed that the survival of the PLWH with stage I to III renal carcinomas after surgery is worse than that of the general population. 39 The PLWH with UC is rarely reported previously; however, encouragingly, our study provided preliminary evidence that tislelizumab in PLWH with locally advanced or metastatic BCa can achieve similar efficacy and safety comparable to those of people with the same stage BCa but no HIV.
Limitation
To our knowledge, this is the first study evaluating the safety of anti-PD-1 therapy in PLWH with locally advanced or metastatic BCa compared to the HIV-negative population. However, the sample size is relatively small. Although the current study’s design and sample size did not allow for a direct assessment and subgroup analysis of PD-L1 expression, the antitumor activity presented is like general population reports from other checkpoint inhibitors. Lastly, the study did not have enough participants with locally advanced or metastatic BCa, to accurately estimate response rates. A prospective, larger study with more participants should be conducted in great detail to assess the efficacy and safety of tislelizumab in PLWH with BCa.
Conclusion
This retrospective multi-center study suggested that tislelizumab may provide encouraging antitumor activity and could be generally well tolerated. In this retrospective analysis of patients with locally advanced or metastatic BCa, it seems that PLWH may have similar overall and progression-free survival compared to HIV-negative cases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of Beijing Friendship Hospital affiliated with Capital Medical University (No. 2023-P2-066-01) on 7, March 2023. The Ethics Committees of Beijing Youan Hospital and Beijing Ditan Hospital also passed the record.
