Abstract
Objectives
This study aimed to demonstrate potential translation of pre-clinical studies to a home-based exercise intervention in mediating inflammatory cytokine markers and tumor progression in men under active surveillance for prostate cancer.
Methods
A 2-arm randomized control parallel group design was used. The exercise intervention consisted of 24 weeks of an aerobic and resistance home-based exercise program and results were compared to a waitlist control group. Data were collected at baseline and end of study for eotaxin, interferon-γ (INF-γ), interleukin-12 (IL-12), interleukin-1α (IL-1α), interleukin-5 (IL-5), interleukin-6 (IL-6), tumor necrosis factor alpha (TNF-α), and vascular endothelial growth factor (VEGF), distanced walked during a 6-minute walk test (6MWT), body mass index, and health-related quality of life.
Results
Non-significant decreases were observed in all biomarkers, especially VEGF (pre: 125.16 ± 198.66, post: 80.29 ± 124.30, P = .06) and INF-γ (pre: 152.88 ± 312.71, post: 118.93 ± 158.79, P = .08), in the intervention group; only IL- α (pre: 332.15 ± 656.77, post: 255.12 ± 502.09, P = .20) decreased in the control group while all other biomarkers increased from baseline to end of study. A non-significant increase in 6MWT distance was observed in the intervention group, while a decrease was seen in the control group. Significant decreases in physical function, emotional wellbeing, and total composite scale on the FACIT-F were observed in the intervention group, possibly due to the isolation restrictions of COVID-19. Physical function on the SF-36 significantly increased in the control group.
Conclusions
Future studies with powered samples are needed to confirm the trends observed for inflammatory biomarkers and functional fitness.
Introduction
The National Cancer Institute estimates that 1 in 8 men will be diagnosed with prostate cancer (PCa) in their lifetime and there are currently an estimated 3.1 million men living who have already been diagnosed in the United States. 1 With a combined overall survival rate of 98% across all stages of the disease, 1 many PCa survivors are often left with lingering treatment side effects, like fatigue, that can significantly impact their quality of life (QOL). 2
Approximately half of all men with low-risk tumors will fall under active surveillance treatment protocols. 3 While beneficial for most, nearly 50% progress from active surveillance to active treatment within 5 years due to tumor progression. 4 PCa proliferation and metastasis is multifaceted and recent studies have investigated the role of inflammatory biomarkers, or cytokines, in this progression. Serum biomarkers including tumor necrosis factor-alpha (TNF-α), interleukins (IL), and vascular endothelial growth factor (VEGF), have been linked to tumor growth and metastasis in this population. These and other cytokines have been implicated in stimulating tumor progression,5,6 modulating the tumor microenvironment7,8 and developing treatment resistance. 7 Therefore, modalities to modulate these tumor proliferative cytokines are needed.
Recent literature has shown a growing focus on lifestyle interventions that target physical activity as a modality to delay disease progression. 9 Exercise has been shown to improve aerobic and muscular endurance, functional performance, quality of life, and increasing cancer-specific and overall survival in men with PCa.2,10,11 Work within our laboratory has demonstrated shifts in circulating cytokines in response to exercise in preclinical studies.(34) 12 Specifically, our group noted that up to 20 weeks of voluntary wheel running significantly reduced the progression to high-risk tumors in transgenic prostate tumor challenged mouse models. This reduction in tumor progression was associated with significant decreases in pro-inflammatory cytokines, such as eotaxin, interleukin (IL)-1a, IL-5, IL-12 (p40) and VEGF. While exercise interventions have demonstrated similar outcomes in clinical populations, 13 there is a paucity of longitudinal clinical trials that demonstrate exercise efficacy in modulating biomarkers associated with tumor progression in men with PCa. We aimed to demonstrate the translational potential of our running wheel intervention to a 6-month home-based modality developed around moderate physical activity in modulating cytokines associated with tumor progression.
The primary aim of this pilot study was to investigate the impact of a home-based exercise program on biomarkers associated with tumor progression in men with PCa under active surveillance. It was hypothesized that the home-based exercise program would result in decreased levels of inflammatory biomarkers. Secondary aims sought to determine the effects of the intervention on physical function, quality of life, and body mass index (BMI).
Methods
Ethics
Ethical approval was granted by the Institutional Review Board at the University of Texas Health Science Center at San Antonio (UT Health San Antonio), located in San Antonio, Texas, for the Audie L. Murphy Memorial Veterans Administration Hospital, and the Mays Cancer Center at UT Health San Antonio. Approval was obtained on February 14, 2017 under approval number 20160604HU. Participants were informed about the nature of the study, voluntary participation, potential benefits and risks of participating in an exercise study, the use of serum specimens, and confidentiality of participant data. Written informed consent was obtained prior to enrollment and participants were given a printed copy of the consent form. The study was registered at ClinicalTrials.gov under NCT03397030.
Randomization and Intervention Design
The reporting of this study conforms to the CONSORT statements.
14
This pilot study used a 2-arm randomized control parallel group design (Figure 1). Participants were randomized 1:1 into either the home exercise intervention group or the waitlist control group by the principal investigator. Recruitment took place August 2017 to November 2019 at the Mays Cancer Center and Audie L. Murphy Memorial Veterans Administration Hospital. Participants were screened for eligibility from patient records. Inclusion and exclusion criteria are found in Table 1. Study flow diagram. Inclusion and Exclusion Criteria.

Individuals meeting eligibility criteria were approached in-person during a regularly scheduled clinic visit to introduce the study. Once consented, screened, and medically cleared, baseline data were collected. Men were then randomly assigned to either the exercise intervention group or waitlist control group using a pre-specified randomization list. Participants assigned to the exercise intervention completed a 24-week, home-based exercise program while those assigned to the waitlist control group were asked to maintain normal activity with the option of obtaining the home-based exercise plan after end-of-study data collection. The frequency, interval, intensity and timing of the intervention was determined based on recommendations by the American College of Sports Medicine. 15 Due to the nature of exercise interventions, participants were not blinded to the intervention. The size of the research team necessitated investigators to assist in the intervention, data collection and analysis; thus, researchers were not blinded.
The exercise intervention group participated in a home-based exercise program, consisting of aerobic and bodyweight-based exercises. The aerobic portion of the intervention included 5 days per week of light-to-moderate intensity (40-60% of heart rate reserve) walking for 30 minutes, for a weekly accumulated total of 150 minutes. Heart rate reserve was calculated using the Karvonen formula: maximum heart rate = 220 – age (years). 16 Body-weight based resistance exercises focused on increasing strength of major body muscle groups. Squats, inclined push-ups, and hip thrusts were performed 3 times per week consisting of 3 sets of 15 reps for each exercise. A FitBit Flex activity tracker (Fitbit Inc, San Francisco, CA, USA) was issued to each exercise group participant to help track physical activity. Participants met with a research team member for an in-person orientation visit, where exercises were demonstrated, the FitBit account was setup and the participant were shown how to use the device, and participants were taught how to measure radial pulse to ensure they were in the target heart rate zone during exercise. Each participant in the intervention arm received a booklet with information on target heart rate for the 30-minute aerobic portion. Participants did not progress aerobic nor resistance intensity or frequency during the study. The remainder of the intervention was performed remotely at home until participants returned for the in-person final data collection. Participants were contacted if no FitBit activity had been reported for 1 week.
Cytokine Collection and Analysis
Blood samples were collected into 8-mL serum separating tubes from the antecubital space via venipuncture at baseline and end of study. End of study serum samples were collected within 7 days of participants completing the study. The samples were spun down at 1500XG for 15 minutes. Serum was placed into 600 μL aliquots and frozen at −80 °C until analysis. Serum cytokines (eotaxin, INF-γ, IL-12, IL-1α, IL-5, IL-6, TNF-α, and VEGF) were analyzed using an 8-protein multiplex assay (Millipore Sigma, Billerica, MA). Samples were assayed in duplicate and averaged.
BMI
Height (cm) and weight (kg) were collected at baseline and end of study to calculate BMI.
Functional Fitness Assessments
Functional fitness was assessed using a 6-minute walk test (6MWT) at baseline and end of study as previously described. 17 Participants were instructed to walk as quickly as possible between 2 markers on the floor placed 40 m apart for 6 minutes. Total distance was calculated in meters. A difference of 14-40 m has been suggested as a range for minimal clinically important difference in the 6MWT. 18
Health-Related Quality of Life Outcome Measurements
To measure health-related quality of life and fatigue, all participants completed the 36-Item Short Form Health Survey (SF-36) and Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) surveys at baseline and end-of-study. The SF-36 assessed 8 area of quality of life including physical functioning, role functioning, social functioning, physical pain, general health, mental health, and emotional health. The SF-36 has been previously validated and has a well-established reliability. 19 The FACIT-F served as a measurement of participant quality of life, encompassing domains of physical, social/family, emotional, functional well-being and fatigue. Reliability and validity for the FACIT-F have been previously established. 20
Statistical Analyses, Sample Size Determination and Data Transformations
Statistical analyses were performed using SPSS for Window version 28.0 (IBM Corporation). Participants lost to attrition were not replaced. The study data set is available upon request from the corresponding author. Sample size calculation was not conducted, however based on prior studies, it was determined that 15 participants in each arm would be feasible to complete this pilot investigation21,22 Due to small sample size, serum biomarker outliers were not excluded from analysis. Only participants with both baseline and final serum samples were included in biomarker analysis.
Psychosocial and fatigue measures were scored using standardized calculations from the respective organizations to create continuous composite domain scales for both the SF-36 and FACIT-F and a total composite score for the FACIT-F.19,20 Raw SF-36 item scores were re-coded on a scale from 0-100 and individual domains were created from averaging specific questions based the percentage of questions answered. 23 A higher scale score was associated with better reported quality of life. Similarly, raw FACIT-F items were re-coded based on developer’s guidelines and averaged to create individual scales based on the percentage of questions answered. Possible physical wellbeing, social/family wellbeing, and functional wellbeing scores ranged from 0-28, emotional wellbeing 0-24, fatigue subscale from 0-52, and composite scores from 0-160. Higher scores on FACIT-F scales and composite score indicated better quality of life. Scale scores within each study arm were analyzed using paired t-tests at baseline and end-of-study, while independent t-tests evaluated differences between the intervention and control groups for the same measures at the same time points. Missing questionnaire data were managed based on the percentage of missing responses per scale and recommendations based on the individual developers.21,22
Descriptive statistics were performed and are presented at mean ± standard deviation. Independent and paired sample t-tests were conducted to determine between group and within group comparisons for all outcome variables. Significance for all analyses was P ≤ .05.
Results
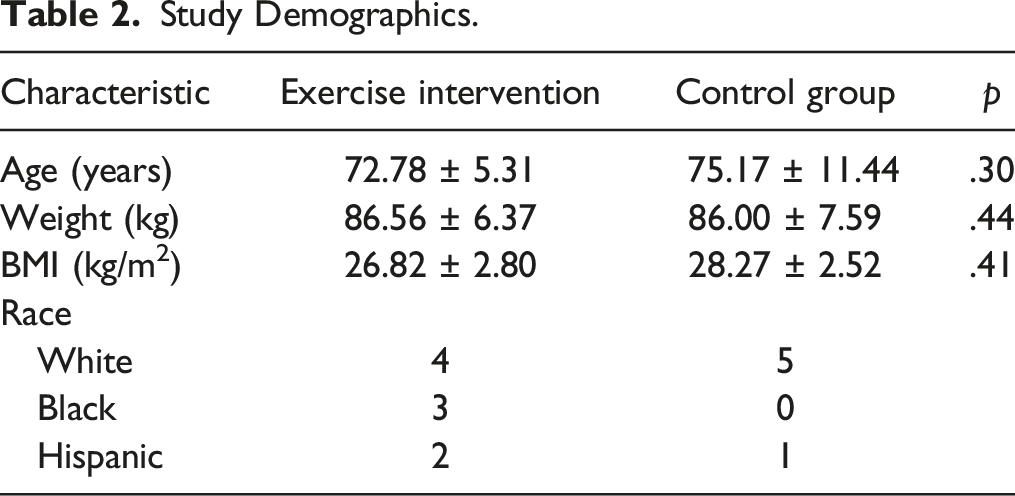
Study Demographics.
Cytokine Analysis
Inflammatory Cytokine Comparison.

INF-γ = interferon- γ, IL-12 = interleukin-12, IL-α = interleukin- α, IL-5 = interleukin-5, IL-6 = interleukin-6, TNF-α = tumor necrosis factor- α, VEGF = vascular endothelial growth factor.
Body Mass Index
No significant differences were observed in within group or between group comparisons. Differences in BMI for the intervention group at baseline (26.27 ± 2.80) and end-of-study (26.82 ± 3.42) were not statistically significant, (t(5) = −1.06, P = .17). The control group analyses returned non-significant results for BMI from baseline (27.10 ± 3.12) to end-of-study (27.43 ± 2.22), (t(2) = −.64, P = .29). No other significant correlations were found for BMI and serum biomarkers for either group.
Functional Fitness
No significant differences were observed within group or between groups. Though not significant, an increase in 6MWT distance was observed in the intervention group (baseline: 451.19 ± 95.95; final: 484.16 ± 78.59; P = .24; d = −.43) compared to a decrease in the control group (baseline: 414.06 ± 65.99; final: 411.77 ± 25.84; P = .47; d = .04 ). Further, between group comparisons at the end of study found the intervention group travelled farther than the control group (intervention: 414.06 ± 65.99; control: 411.77 ± 25.84; P = .47; d = 1.13 ).
Quality of Life and Fatigue
Health-Related Quality of Life Within Group Comparisons from Baseline to End of Study.

PWB = physical wellbeing, SWB = social/family wellbeing, EWB = emotional wellbeing, FWB = functional wellbeing. For SF36: PF = physical functioning, RLPH = role limitations-physical health, RLEH = role limitations-emotional health, EF = energy/fatigue, EMWB = emotional wellbeing, SFUN = social functioning, GH = general health. Higher scores on all scales indicate higher functioning.
Comparison of Exercise Intervention on Fatigue and Quality of Life.
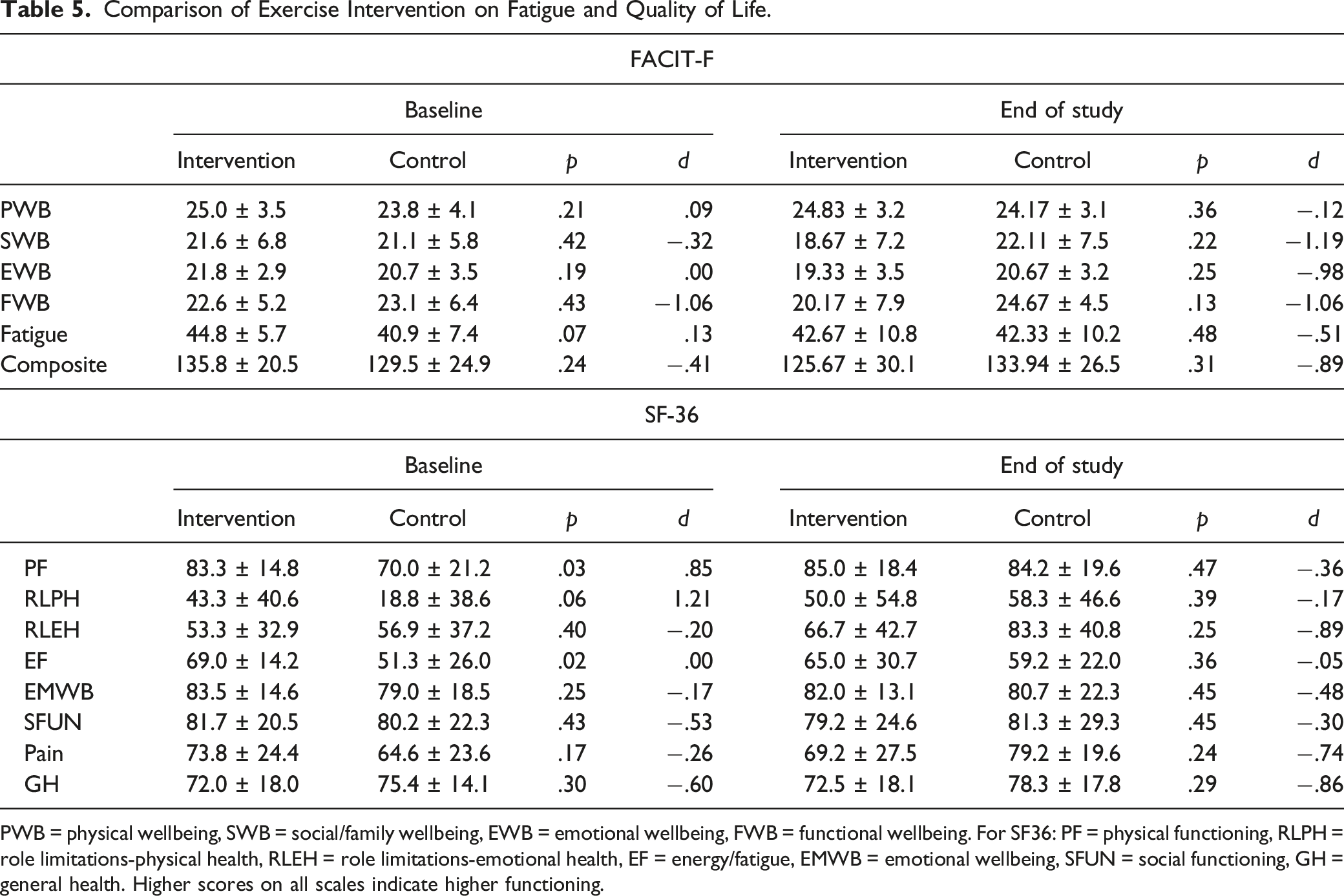
PWB = physical wellbeing, SWB = social/family wellbeing, EWB = emotional wellbeing, FWB = functional wellbeing. For SF36: PF = physical functioning, RLPH = role limitations-physical health, RLEH = role limitations-emotional health, EF = energy/fatigue, EMWB = emotional wellbeing, SFUN = social functioning, GH = general health. Higher scores on all scales indicate higher functioning.
Discussion
The primary purpose of this study was to determine the effects of a home-based exercise program on circulating cytokines associated with tumor progression in men with PCa under active surveillance. Despite challenges of COVID-19 restrictions, 9 participants in the intervention group and 6 participants in the control group completed the study. Although not statistically significant, a decrease across all biomarkers followed the same trend previously seen in the murine exercise study in our laboratory, 34 lending support for the translational nature of our intervention to the clinical population. Our results are similar to findings of a home-based exercise intervention by Hvid et al. 24 who observed a non-statistically significant decrease in mean IL-6 after 24 months; however, this study found an increase TNF-α within the same time parameters. This discrepancy may be due to differences in exercise prescription protocol. Our program utilized the minimum physical activity guidelines as prescribed by the ACSM, 15 with participants completing 150 minutes of activity over 5 days per week, for 24 weeks, whereas participants completed interval training 3 days per week for 24 months in Hvid et al study. 24
Body composition plays a major role in PCa risk and progression, due to the impact that adiposity has in manipulating androgen signaling and systemic inflammation.25,26 A review by Lopez et al. found that a combination of resistance and aerobic exercise was the most effective form of exercise intervention in reducing body fat. 27 Although our intervention included both resistance and aerobic components, we did not observe a change in BMI in either group. While our results do not agree with the literature, the increase in BMI seen in our studied mirrored the increase in BMI observed in the American population during the COVID-19 pandemic. 28 Local ordinances limiting access to gyms and recreational activities during the pandemic may have led to more sedentary time outside our intervention for our participants, resulting in the increased BMI seen in our study.
Impaired physical function and reduced physical activity status have been associated with increased risk for primary diagnosis of PCa or recurrence. 29 Our study evaluated the effectiveness of our home-based exercise prescription, mirroring the minimum physical activity guidelines recommended by the ACSM, 15 on 6MWT distance in men with PCa under active surveillance. The increase in mean distance walked seen in the intervention group fell within the minimal clinically importance difference range 18 ; this was not observed in the control group. The increase in functional exercise fitness observed is consistent with findings from Gaskin et al 30 and Villumsen et al 31 who both observed an increased distance walked during a 6MWT for community-based exercise interventions when compared to usual care. Together with the results from our study, these findings suggest that the use of a home-based intervention may have clinical relevance in preserving or improving physical function in prostate cancer during active surveillance.
Finally, quality of life and fatigue results contradict published studies. Physical wellbeing, emotional wellbeing, and overall composite scores on the FACIT-F decreased in the intervention group while only a small decrease in emotional wellbeing was observed in the control group from baseline to end-of-study. These findings contrast with results from a systematic review and meta-analysis by Vashistha et al 32 that found exercise significantly improved fatigue symptoms and increased quality of life in men with PCa. Similarly, our results were different from Kang et al 33 who found an increase in general quality of life during a high-intensity interval training program for men under active surveillance for prostate cancer. When evaluating the subscales, the largest decreases in the intervention group were social and emotional wellbeing on the FACIT-F. Similarly, emotional wellbeing and social functioning were also 2 scales on the SF-36 that decreased in the intervention group but not in the control group. As this study took place during the COVID-19 pandemic, the isolation and breakdown of social activities could help explain some variance, although the same downward trend was not seen in the social wellbeing scale for the control group.
Interpretation of our study outcomes need to be evaluated in the context of the limitations of this study. Due to the COVID-19 pandemic, we experienced a higher-than-expected attrition due to changes in participant availability and local guidelines limiting in-person study visits. Statistical analysis and generalizability were constrained by our small sample size, making comparison to similar studies difficult. Information about participant disease stage (Gleason score, prostate-specific antigen levels, and time on active surveillance) and comorbidities that may account for chronic inflammation were not collected, which may have influenced biomarker levels. Although the FitBit devices were used to gauge adherence to the exercise intervention, the trackers did not have heart rate monitoring capabilities; thus, information on intensity and self-reported physical activity was not collected during the study. This information may have allowed further analysis by physical activity level to determine if exercise outside the study may have impacted the findings.
Despite these limitations, the clinically significant increase in functional fitness and decreased inflammatory biomarkers suggests that a home-based exercise program may be effective in reducing inflammatory cytokines and increasing functional capacity in men with PCa. Additionally, these biomarker changes add to the growing body of knowledge of physiological mechanisms underlying observed outcomes in exercise oncology. Future studies with statistically powered samples would be needed to confirm the non-statistically significant trends observed in the exercise group for inflammatory biomarker and 6MWT.
Conclusion
The results of this study suggest that men with prostate cancer on active surveillance can potentially benefit from a home-based exercise program. Our data suggest that a home-based exercise program can modulate inflammatory cytokines associated with tumor progression while also positively impacting physical function and quality of life. Though not significant, the findings of our study may have clinical implications. Future studies are needed to confirm these findings in a powered sample.
Footnotes
Acknowledgments
The authors would like to thank the men who volunteered their time and effort for this study. Without willing volunteers like the participants in the study, we would never be able to understand the clinical impact of exercise. The authors would also like to acknowledge the Bioanalytics and Single-Cell (BASiC) Core at UT Health San Antonio’s Greehy Children’s Cancer Research Institute in support of the multiplex analysis performed for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by an internal research grant from the School of Nursing Advisory Council at the University of Texas Health Science Center and a grant from the United States Department of Education (P031S150048). The National Cancer Institute designated UT Health San Antonio Mays Cancer Center also supported this work (P30CA54174OD).
Ethical Approval
Ethical approval was granted by the Institutional Review Board at the University of Texas Health Science Center at San Antonio (UT Health San Antonio), the Audie L. Murphy Memorial Veterans Administration Hospital, and the Mays Cancer Center at UT Health San Antonio (#20160604HU) Participants were informed about rights and informed consent was obtained prior to enrollment and given a written copy of the consent form. The study was registered at ![]() under NCT03397030.
under NCT03397030.
