Abstract
In recent years, the incidence of cancer has been increasing worldwide. Conventional cancer treatments include surgery, chemotherapy, and radiation, which mostly kill tumor cells at the expense of normal and immune cells. Although immunotherapy is an accurate, rapid, efficient tumor immune treatment, it causes serious adverse reactions, such as cytokine release syndrome (CRS) and neurotoxicity. Therefore, there is an urgent need to develop an effective and nontoxic procedure for immunotherapy. The clinical combination of phototherapy and immunoadjuvant therapy can induce immunogenic cell death and enhance antigen presentation synergy. It also causes a systemic antitumor immune response to manage residual tumors and distant metastases. Photoimmunotherapy (PIT) is a tumor treatment combining phototherapy with immunotherapy based on injecting a conjugate photosensitizer (IR700) and a monoclonal antibody (mAb) to target an expressed antigen on the tumor surface. This combination can enhance the immune response ability, thus having a good effect on the treatment of residual tumor and metastatic cancer. In this review, we summarize the recent progress in photoimmunotherapy, including photoimmunoconjugate (PIC), the activation mechanism of immunogenic cell death (ICD), the combination therapy model, opportunities and prospects. Specifically, we aim to provide a promising clinical therapy for solid tumor clinical transformation.
Keywords
Introduction
In recent years, the incidence of cancer has increased worldwide, and conventional treatments, including surgery, chemotherapy, and radiation, are very likely to kill tumor cells at the expense of normal and immune cells.1-3 Immunotherapy is a new strategy for tumor therapy, including the use of T cell-activated cytokines, immune checkpoint blockade (ICB) or programmed cell death-1 (PD-1), programmed cell death ligand-1 (PD-L1), and chimeric antigen receptor T cell/NK cell (CAR-T/CAR-NK) cell therapy.4-6 These strategies for immunotherapy do not destroy cancer cells directly. It is entirely dependent on activating the immune system to selectively control tumor growth.1,7 Currently, immunosuppressants include PD-1/PD-L1 inhibitors and cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) inhibitors. Some solid tumors, such as lung cancer, melanoma, kidney cancer, bladder cancer, head, and neck cancer, and Hodgkin’s lymphoma are treated with these drugs in a better way. CAR-T cells have achieved a good effect in treating clinical blood cancer, which is very promising. Clinical data show that this therapy is only effective in some patients and has limited efficacy in solid tumors, with serious side effects such as cytokine storms in some cases. 7 Therefore, researchers who study clinical solid tumor therapy have sought new methods that can kill tumor cells and reduce the immune damage of normal tissues.
Photoimmunotherapy (PIT) is a therapy to treat diseases by linking specific antibodies with photosensitizers to form photoimmunoconjugates (PICs).2,8 PIT not only has the advantages of traditional PDT but also has the precision targeting ability of antibodies.
9
This therapy is known to have great potential in the prevention, treatment, and diagnosis of tumors. This combination of tumor photodynamic therapy and immunotherapy can enhance the immune response, thus having a good effect on the treatment of metastatic cancer. Phototherapy-induced cell apoptosis,10,11 autophagy,12,13 or necrosis
14
can result in excellent tumor antigen sources. Immunotherapy combined with phototherapy utilizes tumor antigens to trigger an antitumor immune response. Thus, PIT can treat both residual tumors and metastases (Figure 1). The first human study (NCT03769506) of PIT used cetuximab-IR700 targeting epidermal growth factor receptor (EGFR) to treat inoperable head and neck cancer.
15
This drug was approved by the U.S. Food and Drug Administration (FDA) for fast-track global Phase III clinical studies in 2018.8,16 On 25 September 2020, Japan’s Ministry of Health, Labor, and Welfare approved a photoimmunotherapy drug to treat head and neck malignancies, the first such drug to be approved globally. The BioBlade laser system was approved on Sept. 2 for use in combination with Akalux, a new cancer drug for patients with head and neck cancers that cannot be surgically removed. A monoclonal antibody is administered by intravenous injection of a target to the surface of the tumor. MAb is an antibody-photoabsorber 700DX (IR700) conjugate (APC) for localized near-infrared imaging at the tumor site. NIR light selectively induces rapid tumor cell death, thus achieving highly targeted tumor therapy with minimal normal tissue damage. IR700 is a water-soluble optical dye. Free IR700 separated from APC can be excreted directly from urine with good biosafety. Unlike other traditional therapies, PIT does not impair the host antitumor immune response but activates the specific antitumor immune response. It induces immunogenic cell death (ICD) and rapidly releases tumor-specific antigen and membrane injury danger signals, attracting dendritic cells. DCs migrate to the tumor site and present tumor-specific antigens, activate tumor-specific T cells to induce their proliferation, and mediate tumor cell death. Therefore, PIT can overcome the problems of uneven or inadequate delivery existing in traditional antibody therapy.
17
In addition, PIT is not limited by the frequency of treatment, and repeated PIT is also an effective strategy to solve the insufficiency of antibody therapy.
17
Given the unique advantages and great potential of PIT in the clinical treatment of tumors, this paper reviewed photoimmunoconjugates (PICs), immunogenic cell death activation mechanisms, combination therapy modes, and prospects. The framework of this review is shown in Figure 2. This review aims to provide a reference for its clinical transformation research and applications. Schematic diagrams of photoimmunotherapy for treating residual tumors and metastases. Note. DAMPs, danger-associated molecular patterns; PDT, photodynamic therapy; PTT, photothermal therapy. Schematic framework of the review design.

Photoimmunoconjugate
Phthalocyanine dye (IR700) IR700 is a completely synthetic dye with many advantages. Phthalocyanine compounds have a strong absorption capacity in the NIR region and are promising photosensitizers that can be used for fluorescence imaging and PDT tumor therapy.
18
Hisataka Kobayashi’s team,19,20 in their search for a new near-infrared (NIR) fluorescent group, discovered a dye called IRDye700DX,
21
a water-soluble silico Rn phthalocyanine derivative that, when coupled with antibodies, was highly lethal only to target cells irradiated in near-infrared, with almost no off-target toxicity. IRDye700DX (IR700), a photoactivated substance that binds antibodies, was activated by 690-nm near-infrared light to induce cell killing. NIR penetrates tissue several centimeters without causing damage to DNA or normal cells.
15
Since photoactivation chemicals primarily bind to tumor cells that overexpress specific antigens, photoactivation selectively kills cancer cells without damaging adjacent normal cells, including tumor-infiltrating immune cells.22,23 Figure 3 shows NIR PIT for cancer based on a monoclonal antibody conjugated to a photoimmunoconjugate, such as IR700 or IR800. Schematic of Near-infrared photoimmunotherapy for cancer based on a monoclonal antibody conjugated to a photoimmunoconjugate (IR700, IR800, etc.). EGFR, epidermal growth factor receptor; HER2, human epidermal growth factor receptor 2.
List of Photoimmunotherapy For Cancer at ClinicalTrials.gov.
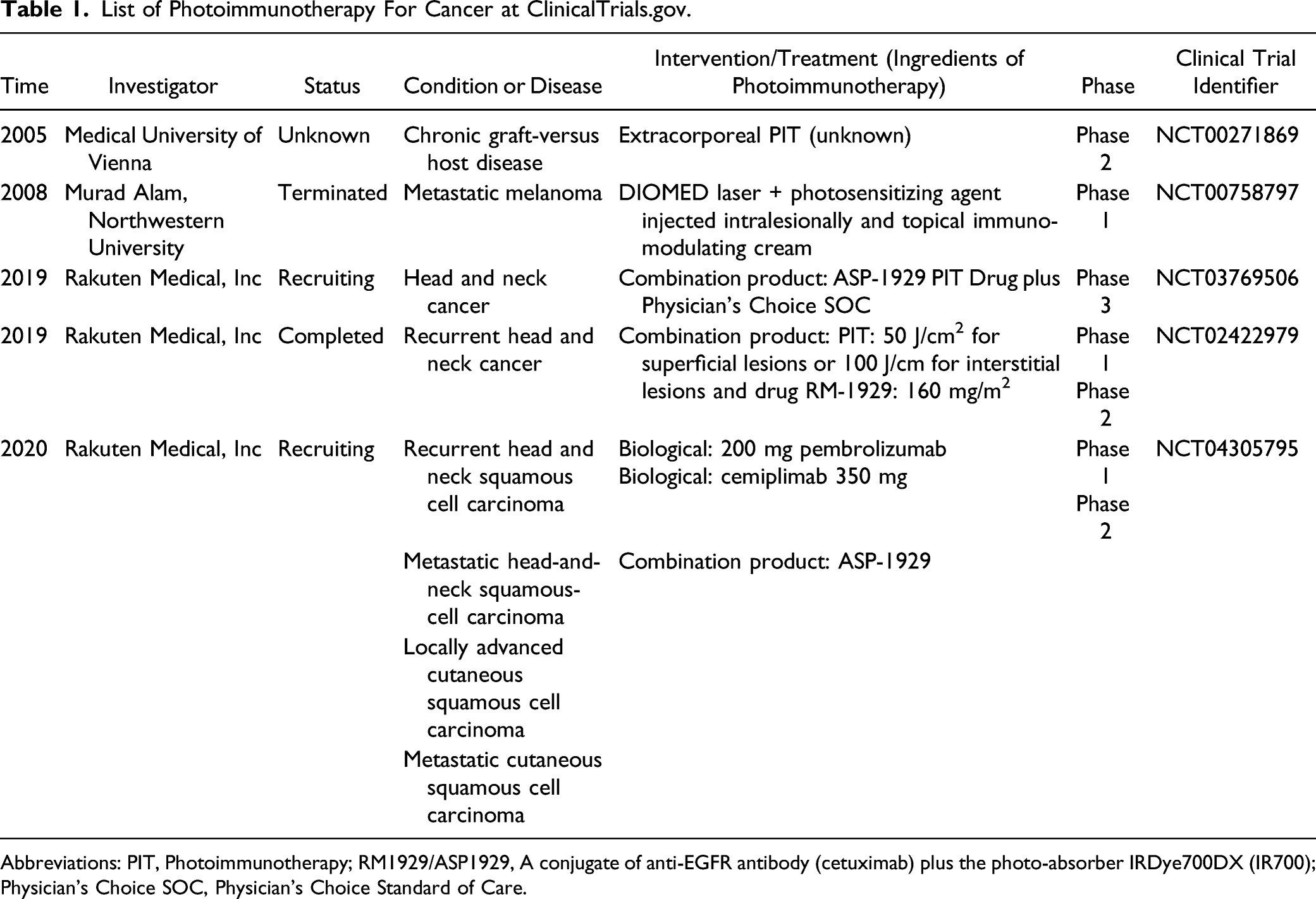
Abbreviations: PIT, Photoimmunotherapy; RM1929/ASP1929, A conjugate of anti-EGFR antibody (cetuximab) plus the photo-absorber IRDye700DX (IR700); Physician’s Choice SOC, Physician’s Choice Standard of Care.
ATAC4 cells with high expression of human epidermal growth factor receptor (EGFR) and low expression of interleukin-2 receptor-alpha (CD25). The results showed that the Pan-Bas-IR700 combination improved the antitumor effect compared to either Pan-IR700 or Bas-IR700 alone.19,24 IR700 was cross-linked with nanobody (NB) antibodies D12 and R2 and bispecific antibody 7D12-9G8, and the results showed that these conjugates could specifically induce EGFR-overexpressing cells to die at low concentrations, and diheterosite conjugates had more obvious phototoxicity. 22 A photosensitizer, IR-700, conjugated to either cetuximab (cet-IR700) or panitumumab (pan-IR700), was evaluated using EGFR-expressing A431 and MDAMB468-luc cells in 2D- and 3D-culture. Although cet-IR700 and pan-IR700 showed identical in vitro characteristics, pan-IR700 showed better therapeutic tumor responses than cet-IR700 in in vivo mouse models because they prolonged retention of the conjugate in the circulation, which is advantageous for tumor responses to PIT. 25 Nagava et al 26 investigated the microdistribution of APCs in a variety of NIR-PIT-treated tumors. Either cetuximab (cet) or trastuzumab (tra) conjugated with IR700 (cet-tra-IR700) was administered, as appropriate, to each mouse model of tumor. Cell killing was primarily on the surface after NIR-PIT, and APCs administered immediately penetrated deeper into tissue to achieve an improved killing effect. 26 Warram et al 23 used activated NHS ester cross-linking to make IR700 and C225 study the biological distribution of cystostomy monkeys and evaluated its efficacy in treating recurrent head tumors. Mesothelins are overexpressed in various malignancies, and recently humanized antibodies against mesothelin (hYP218) have been found, showing high affinity. Nagaya et al 27 examined avelumab-IR700 as a novel cancer treatment candidate. Finally, the conjugate specifically binds to H441 cells expressing PD-L1. Treatment with near-infrared immunotherapy (NIR-PIT) of hematoxylin and eosin (H&E)-stained H441 tumors showed diffuse necrosis, and microhemorrhage significantly inhibited tumor growth but did not cause skin necrosis or toxicity and significantly prolonged survival. From a biological and medical perspective, humanized antibodies are used as delivery vectors for targeted drugs in clinical applications due to their highest binding specificity and maximum in vivo target delivery volume. Hydrophilic phthalocyanine was used as an activated cytotoxic “nano explosive” reagent because of its large absorption of near-infrared light at 700 nm and strong cytotoxicity only when combined with cell membranes. NIR uses 700-nm light as an initiator to activate cytotoxicity from a physical point of view. Due to its high energy, NIR has great tissue penetration ability under nontoxic nonionizing photon stimulation. 9 Based on the photosensitizer and its localization, the acute vascular response can be useful in predicting the overall response to PDT. These approaches are particularly important for predominantly vascular-based photosensitizers such as dextran-benzoporphyrin derivative (BPD) 28 and TOOKAD. Thus, it is important to consider blood flow in tissue undergoing PDT, such as laser speckle imaging. 29
Porphyrins. Porphyrins are special compounds that exist widely in nature. Porphyrin photosensitizers have a strong absorption band at 400 nm, which is called the “Soret” band. At 600∼800 nm, there is a band (Q band) that can be used for PDT treatment. Porphyrin-based PDT is able to remain in tumors and produce cytotoxic singlet oxygen when exposed to appropriate wavelengths of light. They are preferentially localized in tumors and have low dark toxicity. In recent years, researchers have applied nanotechnology to the delivery of drugs. Liang et al 30 developed novel photoimmunoconjugated nanoliposomes (PIC-NALs) that selectively bind EGFR-positive cells, which can deliver irinotecan in vitro and be used in collaborative photoimmunochemotherapy to improve treatment efficiency. By coating gold nanoparticles with photosensitizer and monoclonal antibody at the same time, nanoparticle conjugates with high phototoxicity and low dark toxicity were obtained. 31 Adnan et al 32 combined benzoporphyrin derivative mono acid ring A (BPDMA) with cetuximab C225, which selectively interacts in Chinese hamster ovary cells with EGFR overexpression. Then, the results showed that the EGFR-mediated signaling pathway was inhibited. However, as phototherapy drugs, the main disadvantage of benzoporphyrin derivatives is that they are insoluble in water, and the introduction of hydrophilic groups in the molecules to improve water solubility is the key to the curative effect of such compounds. It has strong absorption in the near infrared region (NIR) and can penetrate deeply into biological tissues.
List of Current Represent Preclinical Studies in Photoimmunotherapy.

Abbreviations: PC, Pancreatic carcinoma; CC, Cholangiocarcinoma; IR700, A photo-absorber dye, IR700DX, activated by NIR light; NIR-PIT, Near-infrared photoimmunotherapy.
Activation Mechanism of Immunogenic Cell Death
ICD is a form of cell death that activates an adaptive immune response to the antigens associated with dead cells, while PIT is a therapy that induces and utilizes ICD to trigger an antitumor immune response.
2
ICD is characterized by causing rapid swelling and rupturing the cell membrane of target tumor cells. The mechanism of the antitumor host immune response induced by PIT is shown in Figure 4.1,2 Biological mechanism by Near-infrared photoimmunotherapy-induced immunogenic cell death enhances the host antitumor immune response. Notes. CRT, Calreticulin; HSP, heat shock proteins; ATP, adenosine triphosphate; HMGB1, high mobility group box; DAMPs, damage-associated molecular patterns; TLR4, Toll-like receptor 4.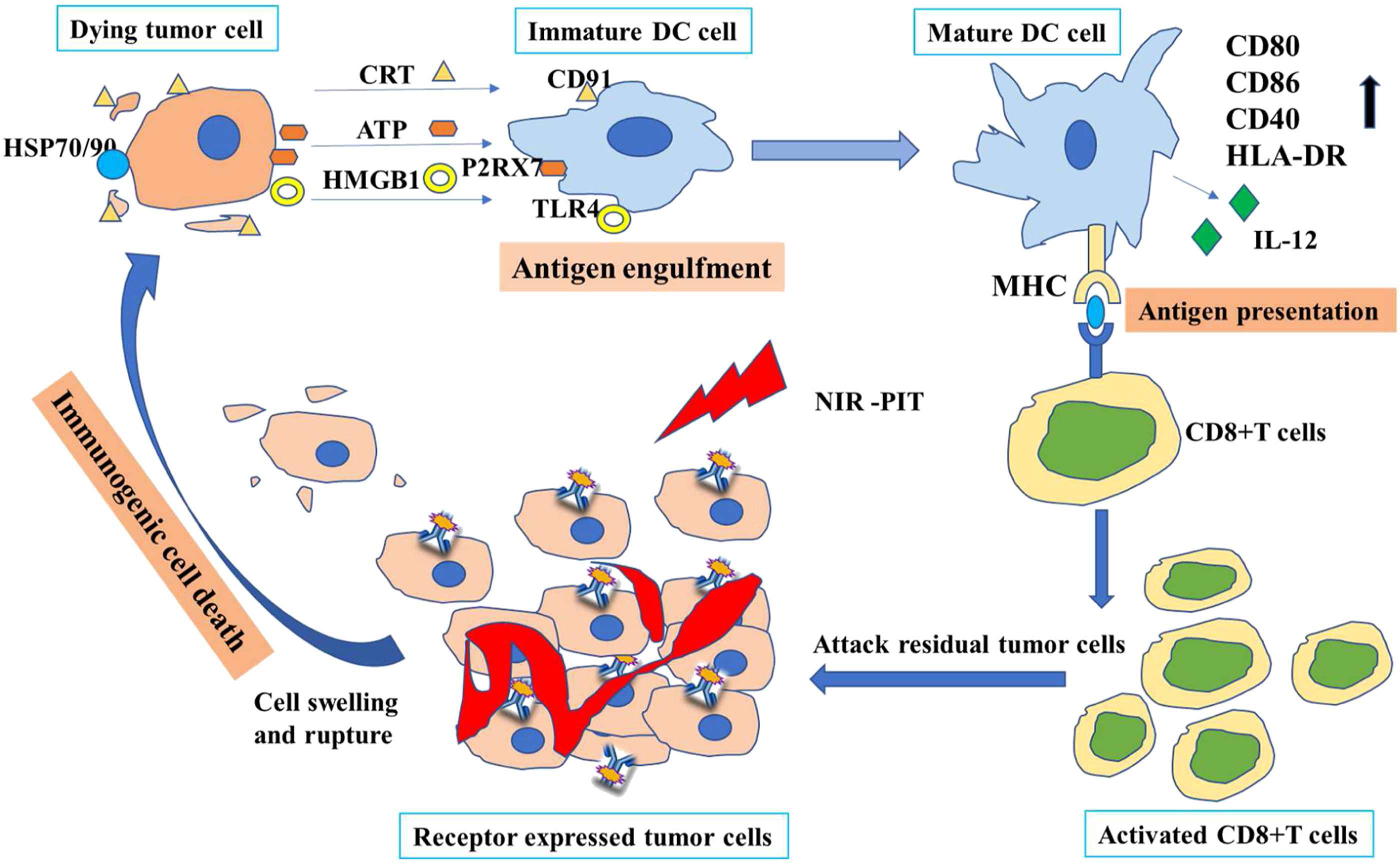
ICD relies on expressing and releasing specific damage-associated molecular patterns (DAMPs) on the cell surface. These DAMPs include calreticulin (CRT) and heat shock proteins (HSP) 70 and HSP 90, adenosine triphosphate (ATP), and high mobility group box 1 (HMGB1).3,4 CRT, HSP70, and HSP90 exposed to the surface of dying cells cooperated with the extracellular release of ATP and HMGB1 to provide immunogenic signals to the host immune system. This plays an important role in establishing antitumor immunity and immune memory. 38 One of the most important markers of ICD is the transfer of CRT from the endoplasmic reticulum to the outside of the cell membrane. CRT on the surface of the cell membrane binds to the CD91 receptor on the surface of innate immune cells to enhance the immune response. Phototherapy can promote the expression of HSP on the surface of tumor cells. 39 For example, HSP70 is an endogenous danger signal of the immune system, and innate or adaptive immune cells can destroy it by recognizing the expression of HSP70 on the surface of tumor cells. Extracellular HSP70 can promote the maturation of antigen-presenting cells and form an HSP-peptide complex to encourage the cross-presentation of HSP-coupled peptide antigens in DCs, thus further triggering the activation and differentiation of antigen-specific helper T cells and cytotoxic T cells. 40 HMGB1 is a marker of advanced ICD. The intracellular release of HMGB1 can be recognized by immune cells expressing pattern recognition receptors such as Toll-like receptor 4 (TLR4), thus initiating immune responses. Extracellular ATP is a dangerous signal used by eukaryotes to detect cell damage. 10 ATP released during ICD binds to the purinergic receptor P2RX7 41 of target cells to generate immune stimulation through activation of inflammasomes.
Ogawa et al 42 co-incubated immature DCs with tumor cells killed by PIT and found that DC maturity markers CD80, CD86, and human leukocyte antigen-DR. The expression of HLA-DR on the surface of DCs was significantly higher than that on immature DCs, suggesting that tumor cells killed by PIT could cause DC maturation. Subsequently, a tumor-associated antigen (TAA) was presented to CD8+ T cells by mature DCs, and CD8+ T cells attacked the remaining tumor cells by recognizing TAA. After implementing tumor cell-targeted PIT, the newly induced CD8+ T cells can react with more tumor antigens and proliferate in the tumor bed compared with the CD8+ T cells before PIT. 43 Due to the activation and proliferation of CD8+ T cells, the antitumor immune response was greatly enhanced after PIT. Ogawa et al. also measured the expression of biological markers of ICD on the surface of A431 cells. Flow cytometry data showed that the expression of CRT, HSP70, and HSP90 increased on the surface of dying cells after PIT. Additionally, the determination of extracellular concentrations of HMGB1 and ATP confirmed that PIT could induce the rapid release of HMGB1 and ATP from tumor cells. PIT-induced ICD results in the rapid release of tumor-specific antigens (such as DAMPs) by dying tumor cells, which can be captured by antigen-presenting cells, especially DCs, thereby promoting the maturation of immature DCs in the tumor cell microenvironment. The mature state of DCs determines their ability to phagocytose, process, and present antigens from dying cells to successfully induce antitumor immunity. 22 Unlike conventional tumor immunotherapy, which mainly activates existing CD8+ T cells in the tumor bed and other parts of the body, PIT locally enhances the host immune response and does not produce side effects similar to those of autoimmune diseases.8,44 In addition, APCs using Treg-targeted or MDSC can selectively clear immunosuppressive cells from the tumor bed, further enhancing the host immune response.8,45
Combination Therapy
Although PIT can induce ICD of target tumor cells and stimulate the host immune response, most of it fails to induce a lasting antitumor response in syngeneic tumor mouse models due to adaptive immune resistance. 43 In addition, intratumor heterogeneity and incomplete PIT conditions (such as low expression of tumor surface receptors, an insufficient dose of APC, and insufficient intensity of NIR light) often lead to poor antitumor effects or tumor recurrence.9,46 In these cases, a combination of chemotherapy and immunotherapy will help improve PIT outcomes. Table 2 showed represent preclinical studies in photoimmunotherapy. Most studied involved in IR700 photoimmunoconjugate, in addition their combined therapy achieved improved outcome in survival and tumor growth inhibition.
Near-Infrared Photoimmunotherapy
Due to abnormal morphology and function of tumor neovascularization, that is, poor arrangement of endothelial cells, lack of smooth muscle layer, impaired angiotensin ii receptor function, and insufficient lymphatic drainage within the tumor, macromolecule drugs and nanoparticles will be retained in the tumor site once they enter. This phenomenon is called the enhanced permeability and retention (EPR) effect. Utilizing the EPR effect, targeted nanocarriers such as liposomes and nanoparticles are expected to deliver more drugs to tumors. However, the literature has revealed that only approximately .7% of nanomaterials reach solid tumors after systematic administration. Although the traditional EPR effect can increase the delivery of nanomedicine to the site of the tumor, it is far from achieving a tumor therapeutic effect. After intravenous administration of an antibody-photon absorber conjugate (APC), the antibody conjugate can bind to tumor cells in the perivascular space to the maximum extent. 47 Exposure to NIR light induces toxic effects on these cells, resulting in sudden perivascular cell necrosis and loss of vascular integrity. Blood vessels dilate due to decreased interstitial hydraulic pressure, which leads to a sharp increase in permeability, resulting in rapid infiltration of nanoparticles into the perivascular space and eventual deposition in the entire tumor bed. 48 This phenomenon can be observed by fluorescently labeled nanoparticle imaging. Hanaoka et al 49 used IR700-YP7 bound to A431/G1 cells and induced rapid target-specific necrotic cell death by near-infrared light exposure in vitro. IR700-YP7 accumulated in A431/G1 tumors. As a result, tumor growth was inhibited by PIT compared with the nontreated control. Sano et al 48 took nude mice with A431 tumors as a model and administered panitumumab-IR700 (PAN-IR700) intravenously for 1 day; only the right tumor was treated with light. Pan-IR800 was injected at 1 hour, 6 hour, and 24 hour, and its distribution in tumors was observed. The results showed that pan-IR800 injected 1 hour after PIT had the most distribution in light-side tumors, and the uptake of Pan-IR800 in light-side tumors after background correction was 21.5 times higher than that of the control tumors when the light dose was 50 J/cm2. 48
However, PAN-IR800 injection 24 hour after PIT showed no increase in permeability, which may be due to the restoration of tumor interstitial hydraulic pressure caused by the tumor tissue repair around blood vessels. Therefore, the optimal time of administration to achieve the maximum concentration of PAN-IR800 in the tumor was immediately after PIT, and increased uptake was observed within 6 hour after PIT, but leakage gradually decreased from then to 24 hour. The EPR effect can only moderately increase the tumor transmission of nanomaterials, while the PIT-mediated effect can cause a 24-fold increase in the distribution of nanomaterials in treated tumors compared with control tumors. 17 In addition, the enhanced effect caused by PIT has no special requirements on the properties of nanomaterials. It not only promoted untargeted nanoparticles such as PEG-doped Qdot800 (approximately 50 nm), superparamagnetic iron oxide nanoparticles (approximately 200 nm), an ultrasmall paramagnetic iron oxide contrast agent (average particle size 20 nm), gadolinium-labeled polyethylene diamine dendrimers (average particle size 10 nm). After tumor transmission, 48 other antibodies or APCs can be promoted to the tumor bed. 50 The tumor-bearing mouse model showed that multiple illuminations after a single dose of APC could significantly inhibit tumor growth. 50 This was due to the enhanced effect induced by initial illumination so that APC existing in blood could enter larger tumor areas and deep tumor tissues that were difficult to reach before PIT, and its distribution was more uniform. 51
Photoimmunotherapy Combined with Chemotherapy
Solid tumors composed of phenotypic and functionally heterogeneous malignant cells can interfere with tumor treatment, especially the effectiveness of targeted molecular therapies, including PIT, and ultimately lead to tumor recurrence. 50 Combined chemotherapy is a new strategy to consolidate the PIT treatment effect of solid tumors. Ito et al 52 combined HER2-targeting PIT with 5-fluorouracil in the treatment of gastric cancer. Compared with single PIT, the combination therapy was enhanced in vivo and in vitro against gastric cancer with HER2 expression, resulting in inhibition of tumor growth in vivo, and tumor cell death induced by PIT included necrotizing membrane injury and cell apoptosis induced by 5-fluorouracil and trastuzumab. Drug carriers such as liposomes and nanoparticles can improve the delivery of chemotherapy drugs to tumors. Antitumor nano agents approved by the FDA include Doxil and Abraxane. The combination of PIT and commercial antitumor nanomaterials can not only improve the tumor delivery and enhance the antitumor effect of nanomaterials through the PIT-induced enhanced effect but also provide a basis for their clinical application and transformation. Sano et al 53 constructed a mixed tumor model with EGFR-positive A431 cells and EGFR-negative Balb3T3/deRed cells, injecting DaunoXome (DX) liposomes 1 hour after EGFR-targeted PIT. The volume of tumors treated with DX in PIT increased by 5 times. Compared with DX or PIT alone, the tumor volume of the mice treated with PIT and DX was significantly reduced, and the survival time was significantly prolonged. In another study, 49 glypican-3 (GPC3)-targeted PIT combined with Abraxane showed superior efficacy in treating hepatocellular carcinoma. The IR700-anti-GPC3 antibody (IR700-YP7) mediates PIT to significantly increase Abraxane leakage into the tumor bed. Compared with Abraxane, tumor suppression and survival improved significantly in the combined treatment group. 54 In addition, protein-bound low molecular weight antitumor drug systems, antibody-drug conjugates, and photoactivated drug delivery systems can also be used in conjunction with PIT. 49 Regarding combining chemotherapy, Nagaya et al 54 combined PAN-IR700-mediated PIT with NIR-released Cyet-Pan-duo (Cyanine-Caged Duocarmycin conjugated panitumumab). In vivo data showed that injection of Cyet-Pan-duo immediately after NIR light resulted in high tumor accumulation and a high tumor background ratio, and the combination therapy was superior to PIT or NIR release therapy alone. Based on the above studies, PIT combined with chemotherapy can enhance the synergistic effect and increase target drug delivery.
Photoimmunotherapy Combined with Immunotherapy
The tumor immune antibodies listed at home and abroad include CTLA-4 and PD-1/PD-L1 blockers. One study combined PIT targeting CD276 with PD-1/PD-L1 ICB. 35 PD-L1- ICB therapy alone or IRD-αCD276/Fab PIT only partially inhibited the growth of 4T1 tumors, while combination therapy significantly enhanced the delayed tumor growth effect and resulted in a significantly smaller tumor size than monotherapy. Moreover, compared with monotherapy, combination therapy significantly inhibited secondary tumor growth and prevented lung metastasis by recruiting tumor-infiltrating CD8+ T cells. 35
Maruoka et al 55 carried out a study that compared CTLA-4 blockers alone or PIT targeting CD44, and combination therapy significantly prolonged survival and improved complete response rates (44%) in MOC1 tumors. The complete response rate in MOC1-loaded mice was only 8%. In the Mc38-Luc tumor model, although PIT combined with CTLA-4 blocker significantly prolonged the survival rate in mice compared with monotherapy, the tumor complete response rate was lower than PIT combined with PD-1 blocker. In addition, CTLA-4 blockers had little effect on LL/2 tumor models. The addition of a CTLA-4 blocker did not significantly prolong the survival of mice compared with PIT. IL-15 administration after CD44-targeted PIT can further inhibit tumor growth by enhancing the antitumor host immune response. 56 Compared with monotherapy, combination therapy significantly inhibited tumors in all three tumor models, resulting in growth and prolongation of mouse survival. The distribution of T cells varies among tumor types, and the individual microenvironment of each tumor differs, so the most effective immune checkpoint target for use in combination with PIT will vary by cell line-based tumor type.
Opportunities and Prospects
PIT can specifically reduce the number of tumor cells and increase the strong host immune response, which has obvious advantages over traditional tumor therapy. It can be more beneficial than any monotherapy when combined with chemotherapy and other cancer treatments, especially with immune activation. When combined with other therapies, it can not only treat local tumors but also minimize or eliminate systemic metastasis to prevent recurrence and produce immune memory. NIR-PIT is based on the injection of a conjugate of an antibody, which binds a cell surface marker on the cancer, and a photoimmunoconjugate.
Generally, the most widely used photoimmunoconjugate in PIT is IR700, and it is a commonly used light absorber for PIT due to its excellent properties. In recent years, several other light absorbers have also attracted the attention of researchers. Indocyanine green (ICG) is a commonly used contrast agent in the clinical diagnosis of cardiovascular diseases. The anti-tissue factor (TF) antibody 1849-ICG can specifically bind to TF highly expressed BXPC-3 cells, induce rapid death of tumor cells and significantly inhibit tumor growth in vivo after NIR illumination. 28 The development of new targets and strategies has led to improved effects of photoimmunotherapy. In addition to targeting antigens on the surface of tumor cells, PIT can also act on other targets to achieve the purpose of tumor therapy. Nishimura et al 30 examined the effect of anti-VEGF on the vascular endothelial growth factor receptor 2 (VEGFR-2) antibody DC101 combined with IR700 to produce DC101-IR700, which targets tumor blood vessels. NCI-n87 showed a stronger antitumor effect than trastuzumab-IR700 (TRA-IR700), with reduced tumor microvascular density and destruction of neovascularized tumors. Since its mechanism of action is different from conventional PIT, no SUPR effect was observed in DC101-IR700-mediated PIT, but anti-VEGFR-2 antibodies bind to all vascular endothelial cells and limit normal tissue toxicity by controlling the site of NIR illumination. Therefore, PIT can be applied to most solid tumors, including gastric cancer. Fibroblast activation protein (FAP) is overexpressed on the surface of activated fibroblast CAFTECM. Watanabe et al 57 also used PIT to target cancer-associated fibroblasts (CAFs) located in the tumor microenvironment. Coculture with CAFs accelerated the growth rate of TE cells in vivo, and FAP-IR700-mediated PIT significantly inhibited the growth of TE cells in the coculture model to a level only comparable to the growth rate of cancer cells.
Mao et al 58 evaluated the effect of anti-PGP FAB-IR700 (FAB-IR700) targeting Pgp on chemically resistant tumors. It was also observed that the fab-IR700 complex made of recombinant antibody fragment was superior to the monoclonal antibody IR700 (Pab-IR700). Better tissue penetration, shorter time for intratumoral concentration to peak, rapid tumor absorption and systemic clearance allow for illumination in a shorter time interval after the injection of the conjugate, which is conducive to clinical transformation. Wei et al 59 combined a single-chain antibody (A33scFv) targeting glycoprotein A33 antigen (GPA33) with IR700 for the treatment of colorectal cancer. In LS174T tumor-bearing mice, A33SCFV-IR700 was selectively enriched in GPA33-positive tumors, and a single injection of A33SCFV-IR700 combined with light produced excellent therapeutic effects compared with the non-PIT group.
Conventional PIT only targets single tumor-associated antigens (TAAs). The fixed antigenic specificity severely limits its efficacy and applicability. 60 Solid tumors are usually composed of heterogeneous cell populations that may be derived from tumor stem cells, and the expression pattern of TAA and composition of the tumor microenvironment usually change dynamically with the malignant progression of the tumor and the process of radiotherapy and chemotherapy. For PIT to be effective in multiple cancer types and adapt to changes in TAA expression, a large number of antibodies against various TAAS would need to be synthesized, but this is not practical. Shirasu et al 61 used IRdye700dX-conjugated Neutr Avidin (Av IR) and various biotinylated antibodies. BioAb) constructed a generic strategy for targeting tumors. The target cells were labeled with one or more Biolabs, followed by the addition of AvIR, which connected IR700 to the cell surface using the high affinity and specificity of avidin and biotin and then killed the target cells by irradiation with NIR light. Although IR700 is not directly linked to antibodies, it does not affect PIT efficacy. The phototoxicity of AvIR-mediated PIT is highly antigen-specific, and different BioAbs can kill tumor cells, tumor stem cells, CAFs, and tumor vascular endothelial cells. This strategy can simultaneously target not only multiple tumor cells but also multiple tumors supporting and/or immunosuppressive cells and can integrate and effectively control the tumor and its microenvironment, thereby overcoming tumor heterogeneity.
NIR-PIT can target and treat almost any cell surface antigen in vivo and in vitro such as EGFR, 34 PSMA, 62 PD-L1, 63 CD44, 64 CD133 2 and so on. NIR-PIT directed at CD44 and CD133, 2 which are considered effective in breast cancer and glioblastoma, respectively. Additionally, tumor xenograft flank mouse models located in the peritoneally, pleurally, and miliary lung metastasis and spontaneous lung cancer in transgenic mice were also successfully treated with NIR-PIT. The first human phase 1/2 clinical trial of IR-PIT using cetuximab-IR700 (RM1929) targeting EGFR in patients with inoperable recurrent head and neck cancer was completed in 2017 and is currently underway in 3 “fast track” trials conducted by the FDA8,65 in three countries and territories in Asia, the United States/Canada and four countries and regions of the European Union. NIT-PIT’s next step is to take the immune response further. Studies in animals27,34,35 with intact immune systems have shown that NIT-PIT targeting of intratumor immunosuppressive cells, such as regulatory T cells, can further selectively enhance the systemic host immunity of tumor cells, resulting in a significant response in distant metastatic tumors. In addition, NIR-PIT can enhance the targeted delivery of nanomaterials to tumors by severely damaging tumor blood vessels and causing super leakage. Dr Kobayashi’s NIR immunotherapy is currently licensed to Rakuten Medical in SAN Mateo, California, and led the clinical trial. Conditional clinical approval for NIR-PIT in recurrent head and neck squamous cell cancer (HNSCC) patients was granted in Japan as of September 2020. 56 It kills cancer cells and activates an immune response. Cetuximab sarotalocan phase I/IIa (NCT02422979) for recurrent head and neck cancer has been completed, and phase III (Luzera-301) was launched in December 2018. 65 The drug is expected to benefit cancer patients. 66 On January 25, 2019, Takashi Kobayashi and his team confirmed that animal experiments could greatly improve treatment techniques. The combination of near-infrared photoimmunotherapy with immune checkpoint inhibitors, a cancer treatment drug, can significantly improve the therapeutic effect. 8 When the mice were injected with colon cancer immune checkpoint inhibitors, only 10% of cancer could be cured. “Light” immune therapy using near-infrared light destroys cancer cells, and then, checkpoint inhibitors are administered. More than 80% of the mice can be completely cured in this way. These cured mice no longer have the recurrence of the same cancer.36,67 To date, the NIR-PIT cytotoxic mechanism is based on a photoinduced ligand release reaction of IR700 after NIR light exposure, which leads to physical damage to the cell membrane. 20 Additionally, this photoinduced ligand release results in irreversible loss of IR700 fluorescence, indicating that NIR-PIT has been effective. Therefore, fluorescence images of IR700 during NIR-PIT could be used to monitor the process and provide useful feedback to the operator regarding NIR-PIT. Okuyama et al. used a clinically approved camera for indocyanine green to monitor real-time IR700 fluorescence imaging during near-infrared photoimmunotherapy, which achieved precise light dosimetry that allowed us to decide on the optimal exposure. 68
With the development of radiography, several imaging platforms have been used in PIT safety and effect evaluation. 69 Huang et al4-6 used ICG as a photosensitizer in combination with immunotherapy. They found that liposome ICG could enhance A549 tumor cell apoptosis and improve immunotherapy. To investigate this mechanism for further research, they established a FRET imaging platform to observe cell immunotherapy with photosensitizers. 4 Thus, it is a potential novel method to apply imaging in tumor PIT therapy. In other ways, it will broaden the PIT scope of concept from antibody to cell immunotherapy. In addition, Kobayashi and colleagues 70 assessed acute photoimmunotherapy cytotoxic effects by 18F-FDG PET, and the immediate cytotoxic effects induced by photoimmunotherapy were clearly detected by decreased glucose uptake using 18F-FDG PET even before tumor size changes. It is suggested that 18F-FDG be used as the clinical assessment of the photoimmunotherapy effects earlier than tumor size observation methods. 70 In the future, real-time IR700 fluorescence imaging during near-infrared photoimmunotherapy will become mainstream in cancer diagnosis and therapy.
Although PIT is a very impressive method in solid tumor therapy, there are some limitations of the review. First, not all photoimmunoconjugates (PICs) in PIT studies are summarized, as this PIT treatment is a novel therapy that includes many photosensitizers. Second, photoimmunotherapy contains different combinations therapy due to individual study aims, and there is no consensus. We could not cover all aspects. Third, this review content based on Kobayashi’s team’s first proposed concept may have some bias. This PIT strategy has been approved by the FDA in the clinic, and it will be developed more popularly for wide use in clinical trials in the future.
Conclusion
PIT can specifically reduce the number of tumor cells and enhance the host immune response, which has obvious advantages over traditional tumor therapy. When used in combination with chemotherapy and other cancer treatments, it can produce better results than any other monotherapy. When used in combination with immune activation therapy, it can not only treat local tumors but also reduce or eliminate systemic metastasis, prevent their recurrence, and produce immune memory. With the gradual deepening of research and its continuous maturity in combination therapy of cancer, tumor-targeted PIT has great potential to replace chemotherapy and become a widely used first-line cancer therapy, which can provide novel clinical therapeutic transformation for cancer treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Authors' Contributions
Zheng Peng and Shigao Huang conceived and designed the research. Xiaolan Lv reviewed literature, Shigao Huang edited the paper. All authors read the approved final manuscript.
