Abstract
Background
Prostate cancer is the commonest cancer among men worldwide and serum prostate specific antigen (PSA) has remained the most commonly applied screening test for the disease till date. Current PSA test results guidelines in our population are informed by reference intervals derived from studies from Caucasians and other racial groups. With scanty data on PSA reference values from our local population, this study evaluated the serum PSA levels of apparently healthy Nigerian male subjects in whom prostate cancer and urinary tract infection have been excluded.
Method
This study had participants aged 40 to 70 years, with no lower urinary tract symptoms or other symptoms suggestive of prostate disease recruited from the male staff population of the University of Abuja and University of Abuja Teaching Hospital and the adjoining local community. They were physically examined, had prostate ultrasonography, urine analysis, and blood sample collected for PSA testing. Data collected was analyzed using Statistical Package for Social Science (SPSS) version 24.
Result
Of a total of 210 men who participated in the study, 191 eventually met the inclusion criteria. The average age was 52.9 years, ninety seven percent of them had heard of prostate cancer before now. The mean total PSA was 1.46 ng/mL (SD +/−1.55), while the reference interval was .23–5.60 ng/mL. The average prostate size was 41.8 mL (SD+/-20.11), and there was a positive correlation between the PSA and the prostate size (.418) as well as the age of the subjects (.446). There was no significant difference in the mean PSA value for those with or without family history of prostate cancer (P=.979).
Conclusion
The reference range of PSA in Nigeria is higher than in other races, hence utilizing a local value in decision making would help to reduce unnecessary invasive procedures.
Introduction
Prostate cancer (CaP) is the most commonly diagnosed malignancy affecting men worldwide 1 and is certainly the commonest cancer among Nigerian men, accounting for 29.8% of all male cancers. 1 Prostate cancer burden is postulated to grow to 1.7 million new cases and 499 000 new deaths by year 2030. 2 Several studies have reported an increasing high incidence and mortality of this malignancy in blacks than in other races. The differences were observed to be a result of the differences in early detection of the disease in US men than in men of African ancestry (MAA) as well as the possibility that African-specific risk profiles, disease aggressiveness, and health care access and treatment strategies are different in Africa compared with the situation in Europe and North America.3,4
Serum Prostate specific antigen (PSA) has remained the standard biochemical screening test for CaP till date.5-7 In Nigeria, the currently and widely accepted PSA reference interval is 0–4 ng/ml. 8 This reference interval value was informed by reference intervals derived from studies amongst Caucasians and other racial groups, 9 considering that there is scanty data on the subject from our local population8,10,11 and notwithstanding the well-documented evidence of the influence of race and ethnicity on PSA levels.12-14
One of the hallmarks of the utility of a tumor marker as a screening test is the use of its reference interval values as a cutoff point for making clinical treatment decisions. Such reference interval values when not determined in the environment of use carry the potential risk of false positivity or negativity with consequences for over-diagnosis, under-diagnosis, and overtreatment of prostatic carcinoma. Elevated serum PSA is not specific for prostatic carcinoma as it can be seen in benign conditions such as urinary tract infections, benign prostatic enlargement (BPE), acute and chronic urinary retention, and prostatic trauma from trucut diagnostic biopsy or vigorous digital rectal examination. Beyond these probable confounding factors for elevated serum PSA is the necessity to improve the utility of PSA as a screening test in terms of positive and negative predictive values by utilizing ethno and age-specific reference interval values.
Researchers15-17 have shown that majority of cases of CaP in Nigeria and other low- and middle-income countries are diagnosed among symptomatic men at advanced stages with attendant high morbidity and mortality.
Prostate cancer is more likely to develop in older men. The average age of men at diagnosis is about 66. The risk of developing the cancer increases gradually from age 40. Six out of 10 cases are diagnosed in men who are 65 or older and it is rare in men under 40. 18
This pilot study to determine a PSA reference interval in our environment using the Clinical and Laboratory Standards Institute (CLSI) and International Federation of Clinical Chemistry’s (IFCC) guidelines for establishing reference intervals19,20 evaluated the serum PSA levels of healthy Nigerian male subjects aged 40 to 70 years in whom prostate cancer has been excluded by digital rectal examination and prostate-focused ultrasound, and in whom urinary tract infections (UTI) had been objectively excluded by laboratory assessment of urine.
Methodology
This collaborative multi-disciplinary (team of investigators comprised two Urologists, a Chemical Pathologist, a Medical Microbiologist, a Radiologist, and a Public Health practitioner) and descriptive cross-sectional study had participants aged 40 to 70 years with no LUT symptoms nor symptoms suggestive of prostate disease recruited from the male staff population of the University of Abuja and University of Abuja Teaching Hospital and the adjoining local community. The study had earlier been publicized to the members of the respective communities using posters, flyers, and various social media platforms. Two prostate health lectures were delivered at community forums to a well-attended audience (some of whom were eventual study participants) before the actual recruitment for the study commenced.
In line with the CLSI and IFCC guidelines, a minimum total sample size of 120 subjects is required for reference interval determination using the non-parametric method.19,20
The study which was to include a physical examination (including a DRE), measurements for body mass index (BMI), urinalysis, a prostate-focused abdomino-pelvic ultrasound scan, and phlebotomy for PSA analysis were to be carried out at no cost to volunteering participants. This was made possible following the support of the study in part by a grant from the Nigeria Tertiary Education Fund (TETFUND) 2014–2016 (Merged) Batch 3 Research Projects (RP) disbursement in year 2020.
Those who indicated interest in participating in the study sent their biodata to a dedicated phone number for the study and were subsequently scheduled for a study appointment.
At study appointment, participants consented verbally to the study and had to complete a self-administered questionnaire. Urine was then collected for urinalysis and blood drawn for PSA. Next, a physical examination including digital rectal examination (DRE) and lastly a KUB +P ultrasound scan were performed. Those who had features suggestive of a urinary tract infection (ie, positive for nitrites and leukocyte esterase activity) and those with features suggestive of CaP (ie, an abnormal DRE findings and or hypoechoic nodule(s) on ultrasound) were excluded from the study. However, the serum of those participants excluded from the study was still analyzed for PSA and the affected participants informed about their respective result, but the PSA results were not included in the final study data analysis. The patients were serially recruited over a period of 3 weeks until the sample size of 208 was reached.
The PSA was analyzed using chemiluminescence immunoassay-Access Hybritech PSA (Beckman Access-UniCelDxl 800). This Hybritech-calibrated assay is a two-site immunoenzymatic “sandwich” assay with a limit of detection of .008 ng/mL and a lowest reportable PSA result of 0.1 ng/mL
The prostate scans were performed for patients with a full urinary bladder; determined as patient having a strong urge to micturate after they had consumed water. With patient in supine position on the couch and abdomen expose, a 3–5 MHz curvilinear probe was placed in the lower abdomen after coupling gel was applied. The prostate was identified posterior to the urinary bladder and its volume was automatically generated by the machine after the transverse (width), craniocaudal (length), and anteroposterior (height) dimensions of the prostate were measured.
All scans were performed by the study team Radiologist to prevent inter-observer assessment variability.
At the conclusion of PSA analysis, all participating subjects were given a copy of their PSA results. Additionally, every participant with either an abnormal DRE or abnormal prostate ultrasound findings or a raised PSA were referred to the study team Urologists for further consultation and evaluation. The reporting of this study conforms to strobe guidelines. 21
Data Collection
Data collected was entered into Statistical Package for the Social Sciences (SPSS) version 24. These included relevant demographic data, digital rectal examination (DRE) findings, prostate ultrasound findings, results of urinalysis, and total PSA levels in ng/ml. Descriptive statistics such as frequencies, percentages, mean, and standard deviation were used to summarize the qualitative and quantitative variables. The PSA reference interval was determined using CLSI and IFCC guidelines for establishing reference intervals.
The t-test statistic was used to test for mean difference between the quantitative variables, while the correlation analysis was used to state the relationship between the variables. Confidence interval of 95% was used while a P value <.05 was considered as statistically significant.
Ethical approval for the study was obtained from the University of Abuja Teaching Hospital Health Research Ethics Committee (UATH/HREC/PR/2020/003/008).
Results
Of the 208 apparently healthy men who participated in the study, six had abnormal findings on DRE suggestive of a prostate malignancy. One of the six subjects with abnormal DRE findings had a PSA of 7.4 ng/mL, while the PSA of the remaining 5 was greater than 10 ng/mL. Furthermore, only two of the six subjects with abnormal DRE findings (33.3%) were found to also have abnormal prostate ultrasound findings. Interestingly, these two subjects with abnormal DRE and abnormal prostate ultrasound findings had PSA values of 3.9 ng/mL and 13.67 ng/mL, respectively.
Nine of the 208 study subjects (4.3%) had abnormal urinalysis. Of this number, two had PSA values of 5.77 ng/mL and 9.54 ng/mL, respectively, while the remaining 7 subjects had PSA values greater than 10 ng/mL. All these participants, 6 with abnormal DRE, 2 with abnormal prostate ultrasound findings, and 9 with abnormal urinalysis, were excluded from the final analysis of the study data.
Frequency of age groups.

The mean total PSA value among participants was 1.46 ng/mL while the minimum and maximum values recorded were .15 ng/mL and 9.93 ng/mL, respectively. Approximately 91% of the study subjects had PSA values less than 4 ng/mL while about 70% had PSA values less than the mean value (Figure 1). The distribution of PSA (ng/ml) values.
The PSA reference interval as determined by non-parametric method was .23–5.60 ng/mL (95% CI) in our study
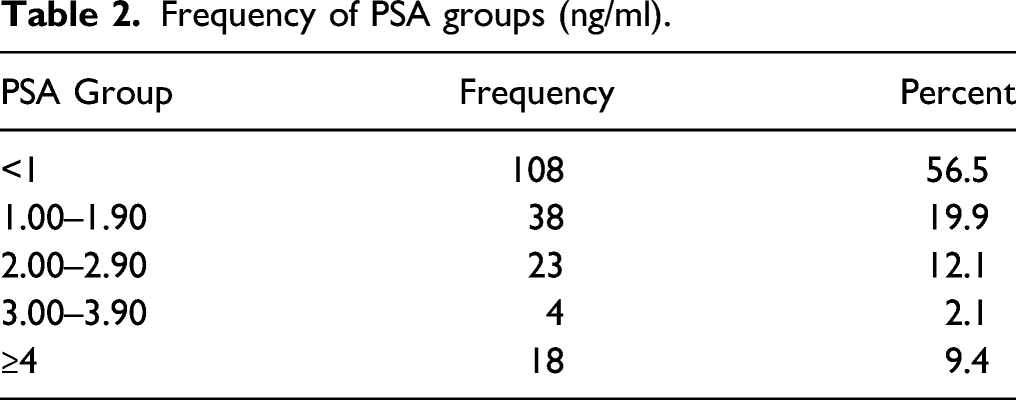
Frequency of PSA groups (ng/ml).
Age groups (years) and PSA values (ng/ml).

Age group (years) and prostate size (mls).

There was no significant difference in the mean PSA value for those with and without family history of prostate cancer (P = .979).
Discussion
The importance of PSA in the early diagnosis of CaP cannot be overemphasized, as its introduction has increased the early detection of CaP, decreased the incidence of high-grade diseases, as well as reduced CaP-specific mortality. 22 Whilst there is no PSA value at which CaP is completely eliminated, it is important to have a useful and consistent reference interval that would guide proper clinical treatment decision making and avoid the unnecessary anxiety and invasive procedure associated with a diagnosis of CaP.
Existing guidelines and reference interval on PSA in our environment are currently predicated on reference intervals from studies amongst Caucasians and other racial groups.9,12 This study to our knowledge is one of the few that has worked on total serum PSA reference range in our environment, while excluding subjects with possible urinary tract infection and possible occult malignancy. The other available studies in their methodology did not screen for urinary tract infection in their subjects nor excluded possible prostate malignance using prostate ultrasonography.8,10,11
In our current study, we made deliberate attempts to exclude as much as possible those participants that had factors that could affect their PSA values by performing a DRE and focused prostate ultrasound to exclude prostate cancer, urinalysis to exclude urinary tract infection, and we ensured that the blood sample for PSA analysis was collected before DRE was performed. We are not unmindful however that some subjects with UTIs and indolent prostate cancer may still be missed as DRE and ultrasound findings in patient with T1 prostate tumors may be normal, and patients with prostatitis may also have normal urinalysis test results. However, it is instructive that approximately 4% of those who had been adjudged healthy and did not report symptoms of UTI were found to have UTI on urinalysis. This further highlights the importance of objective exclusion of UTI in assessment of PSA even in apparently healthy individuals especially in the setting of population-wide evaluations to PSA reference intervals.
Average PSA value amongst our participants was 1.46 ng/mL. This value is at variance with 1.84 ng/mL by Ikuerowo et al, 10 2.21 ng/mL by Eboreime and Atoe, 11 and much lower than the average of 2.9 ng/mL by Amadi and Odum. 8 Perhaps the fact that these studies did not screen for UTI or attempt to screen clinically for CaP using ultrasound may account for the variations seen in them relative to our current finding.
Interestingly, 56.5% of our study participants had serum PSA values less than 1 ng/mL, 52 of the 67 participants (77.6%) aged 40 to 49 years were predominant in this category. Only 9.4% of the subjects had their PSA values equal or greater than 4 ng/mL; this value approximates closely to 8% of the population studied by Ikuerowo et al. 10
The average PSA for the different age groups 40–49, 50–59, and >60, which were .83 ng/mL, 1.47 ng/mL, and 2.6 ng/mL respectively, were lower than those obtained in other local studies.10,11 The latter may still not be unconnected to the screening processes we adopted to exclude factors such as UTI. Our finding that the average prostate size increases per decade was not unusual as was already documented in several other studies from Nigeria, part of Africa, and the Western world. Furthermore, we found a positive correlation between PSA and age in our study.8,10,11,23,24
From our study, the reference interval of PSA is .23–5.60 ng/mL and this is similar to 0–5.22 ng/mL obtained by Ikuerowo et al. 10 Studies like those by Imai et al 25 amongst Japanese subjects and by Bakir et al from a Syrian population 26 found the reference intervals to be 0–3.7 ng/mL and 0–4.3 ng/mL, respectively, while a much lower value of 2.6 ng/mL was proposed as the threshold for biopsy among US men by Punglia et al. 27= There is therefore evidence to believe that PSA reference values from other races are much lower than what obtains in MAA.
The upper limit of 5.22 ng/mL in this study is certainly higher than the traditional 4 ng/mL currently used as a cutoff point for clinical treatment decisions for suspected CaP; hence, adopting this value as the threshold for prostate biopsy in our population should significantly reduce the number of unnecessary invasive prostate biopsies performed to diagnose CaP.
The prostate size is of paramount importance when interpreting PSA results that are in the grey zone (PSA values between 4 and 10), in patients with normal DRE findings. 28 In our study, the prostate size ranged between 9.97 mls and 115.24 mls, with an average of 41.8 mls. The average prostate size for the age groups 40–49 years, 50–59, years and 60 and above were 35.01 mls, 42.48 mls, and 52.24 mls, respectively. This trend of progressively increasing prostate size per decade was similar to the finding by Okuja et al. 23 This may account in part for the progressively increasing total PSA per decade, as the quantity of PSA elaborated by prostate tends to increase with prostate size. Yet again, our study confirms the association between prostate volume and PSA though with a weak positive correlation similar to Okuja et al and Elijah et al in their respective studies.23,29
Some limitations of our study are, first, that subjects who had PSA values higher than 4 ng/mL and no clinical signs of CaP did not have their serum-free PSA evaluated in order to assess the percentage-free PSA (percent-free PSA).
Secondly, we were only able to generate information on comparative age-group mean PSA values and not age-specific reference intervals due to the sample size utilized. To undertake the latter, based on CLSI guidelines, 20 would have required 120 subjects for each age category.
Lastly, prostate biopsy was not carried out on the participants because of its invasiveness and arguably most of the participants would not have consented to it; therefore, it is possible that a few of the participants included in the final study analysis may harbor clinically undetectable CaP. We find some justification all the same because we only set out to investigate only “apparently healthy Nigerian males”!
Conclusion
The findings in this study corroborate the long held suspicion of variations in PSA values among different ethno-racial groups. The reference interval of PSA in Nigerian men obtained from this study and as deduced from other local studies is higher than those obtained in Caucasian populations, and so utilizing this reference value in clinical decision making in our patients offers the potential of minimizing the need for unnecessary invasive prostate biopsy procedures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Tertiary Education Trust Fund (TETFund) under the auspices of its Institution-Based Research Grant id TETFUND/DESS/UNI/ABUJA/RG/2016.
Ethical Approval
Ethical approval for the study was obtained from the University of Abuja Teaching Hospital Health Research Ethics Committee (FCT/UATH/HREC/1085).
Informed Consent
Verbal informed consent was obtained from the participants for their anonymized information to be published in this article.
