Abstract
Background
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has overwhelmed the capacity of healthcare systems worldwide. Cancer patients, in particular, are vulnerable and oncology departments drastically needed to modify their care systems and established new priorities. We evaluated the impact of SARS-CoV-2 on the activity of a single cancer center.
Methods
We performed a retrospective analysis of (i) volumes of oncological activities (2020 vs 2019), (ii) patients’ perception rate of the preventive measures, (iii) patients’ SARS-CoV-2 infections, clinical signs thereof, and (iv) new diagnoses made during the SARS-CoV-2 pandemic.
Results
As compared with a similar time frame in 2019, the overall activity in total numbers of outpatient chemotherapy administrations and specialist visits was not statistically different (P = .961 and P = .252), while inpatient admissions decreased for both medical oncology and thoracic oncology (18% (P = .0018) and 44% (P < .0001), respectively). Cancer diagnosis plummeted (−34%), but no stage shift could be demonstrated.
Acceptance and adoption of hygienic measures was high, as measured by a targeted questionnaire (>85%). However, only 46.2% of responding patients regarded telemedicine, although widely deployed, as an efficient surrogate to a consultation.
Thirty-three patients developed SARS-CoV-2, 27 were hospitalized, and 11 died within this time frame. These infected patients were younger, current smokers, and suffered more comorbidities.
Conclusions
This retrospective cohort analysis adds to the evidence that continuation of active cancer therapy and specialist visits is feasible and safe with the implementation of telemedicine. These data further confirm the impact of SARS-CoV-2 on cancer care management, cancer diagnosis, and impact of infection on cancer patients.
Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a ribonucleic acid coronavirus, similar to SARS-CoV-2 1 and Middle East respiratory syndrome coronavirus. 2 The genome of the virus demonstrates 96% of concordance with the genome of a bat coronavirus suggesting its potential origin.3,4
SARS-CoV-2 was first identified in humans in December 2019 and presented as a severe viral pneumonia. 5 As SARS-CoV-2 spread rapidly, this virus challenged the world’s finite health resources and the traditional healthcare delivery system, forcing immediate change in health management that is likely to last for a prolonged time.
Traditionally, cancer patients especially were regarded as at higher risk for SARS-CoV-2 transmission and disease because of the underlying disease, but also due to comorbid conditions and treatment-related immune dysfunction. Early data from China seemed to confirm the higher infection rates, 6 but these were later contradicted7,8 and ultimately refined as the United Kingdom—researchers demonstrated a differential susceptibility relative to tumor type. 9 Similar to patients without cancer, the severity or fatality rate in cancer patients is confounded by factors such as age, gender, cardiovascular morbidities, obesity, diabetes, and even tumor type.10-13 Still, when cancer patients suffer severe SARS-CoV-2, the in-hospital mortality rate is higher compared to non-oncology patients, particularly in patients with metastatic disease and patients with hematological cancers.14,15
The SARS-CoV-2 forced oncology professionals to navigate—mostly centralized—cancer care with shortages of resources and the possibility of a potential higher transmission exposure. The scientific community responded with adjusted guidelines in order to provide the safe and efficient care to all; all of which are based on broad consensus and expert peer review.16-20
In order to present a real-world experience, we report in this single-center cohort analysis the changes implemented during the first peak of the SARS-CoV-2 pandemic and the evolution in oncologic care activities and diagnoses. In addition, we evaluated the patients’ perception of the safety measures and finally describe the SARS-CoV-2 prevalence and comorbidity in a cohort of cancer patients treated at a tertiary care hospital in Belgium.
Material and Methods
Study Design, Data Sources, and Collection
We report a retrospective analysis of the clinical activity at the Antwerp University Hospital Cancer Center (MOCA) from March 17th to August 28th (week 8 until 34), and compared these data to the data of the same period in 2019.
We evaluated the effect of SARS-CoV-2 preventive measures on volumes of oncological activity by tracking the (1) number of consultations, the (2) number of outpatient treatments, (3) hospital admissions (pre-planned and not), and finally (4) the number of new cancer diagnoses presented to the multidisciplinary oncology boards. (Supplementary methods).
All cancer patients with a laboratory confirmed SARS-CoV-2 infection; both out- and inpatients at the Antwerp University Hospital were identified by reviewing medical charts and by screening for patients with positive nasopharyngeal polymerase chain reaction test from March 17th to August 28th. The clinical data such as patient demographics, comorbidities (including history of cancer), and clinical presentation were retrieved from electronic medical records after anonymization.
The clinical data were compared to a population-based national dataset obtained by the Belgian public health institute (Sciensano) and published in literature. This dataset concerns 13594 confirmed SARS-CoV-2 hospitalized patients (1187 with a solid cancer), and were retrieved by Sciensano, on May 24, 2020. 15
Patient-Reported Perception of Organizational Measures
To assess patients’ perception of risks, their fears, and the acceptance of protective/organizational measures, anonymous questionnaires (Supplementary methods), approved by the Antwerp University Hospital psycho-oncology service, were administered at triage to all patients accessing our outpatient facilities over a 28-day period; questionnaires were returned on a voluntary basis.
Statistical Analysis
Qualitative variables were presented with the observed numbers and percentages. For oncological activity volumes, 27 weekly average and total numbers +/− standard deviations were reported for weeks 8 until 35 of 2019 and 2020. Variations and differences in average activity volumes between the two periods were compared. Normality per group was tested with a Kolmogorov–Smirnov test and if normality was acceptable per group an independent samples t-test was used, if not a Mann–Whitney test. The Levene’s test evaluated if equal variances could be assumed, if not a Welch t-test was used.
The answers to relevant questionnaire items are reported as percentages. When evaluating cancer diagnosis made during a similar period in 2019 as in 2020, numbers were described for 4 solid tumor types and per type the distribution of the stages between 2019 and 2020 was compared using a chi-square test or Fisher’s exact test as appropriate.
The clinical characteristics of SARS-CoV-2-infected patients were compared to a national dataset (15) using a chi-square test for the categorical variables and a Fisher’s exact test if more than 20% of the expected values were lower than 5. The corresponding odds ratio with 95% confidence interval was also calculated. For the age as continuous variable, the Welch t-test was used. The statistical analysis was done using SPSS version 21, and non-parametric confidence interval for odds ratio was computed in R-3.5.2 for Windows.
Results
SARS-CoV-2-Related Events and Organizational Protective Measures
Cancer-specific protective measures provided by the Belgian authoroties, the European Society of Medical Oncology (ESMO) and adapted by the Antwerp University Hospital.

Overall Volumes of Oncology Activities
Between February 17th and August 28th, 2020, we evaluated the volumes of clinical oncological activity and compared them to the activities from a similar period in 2019. During these first months of the SARS-CoV-2 pandemic, local and regional public health authorities prioritized urgent help. Since cancer care was defined as urgent care, all ongoing systemic therapies were continued and new treatments were initiated following careful evaluation of necessity (Supplementary data file).
Changes in volumes of oncology activities during week 8 until 34 2020 as compared with the same period in 2019. Average weekly numbers (calculated over the entire period) ± standard deviation (SD) of inpatient admissions, outpatient admissions and consultations. Overall volumes refer to all disciplines at the Antwerp University Hospital. Oncology refers to Cancer care for solid tumors other than Thoracic Oncology. Gynecological oncology refers to surgical interventions / treatment for gynecologic malignancies.
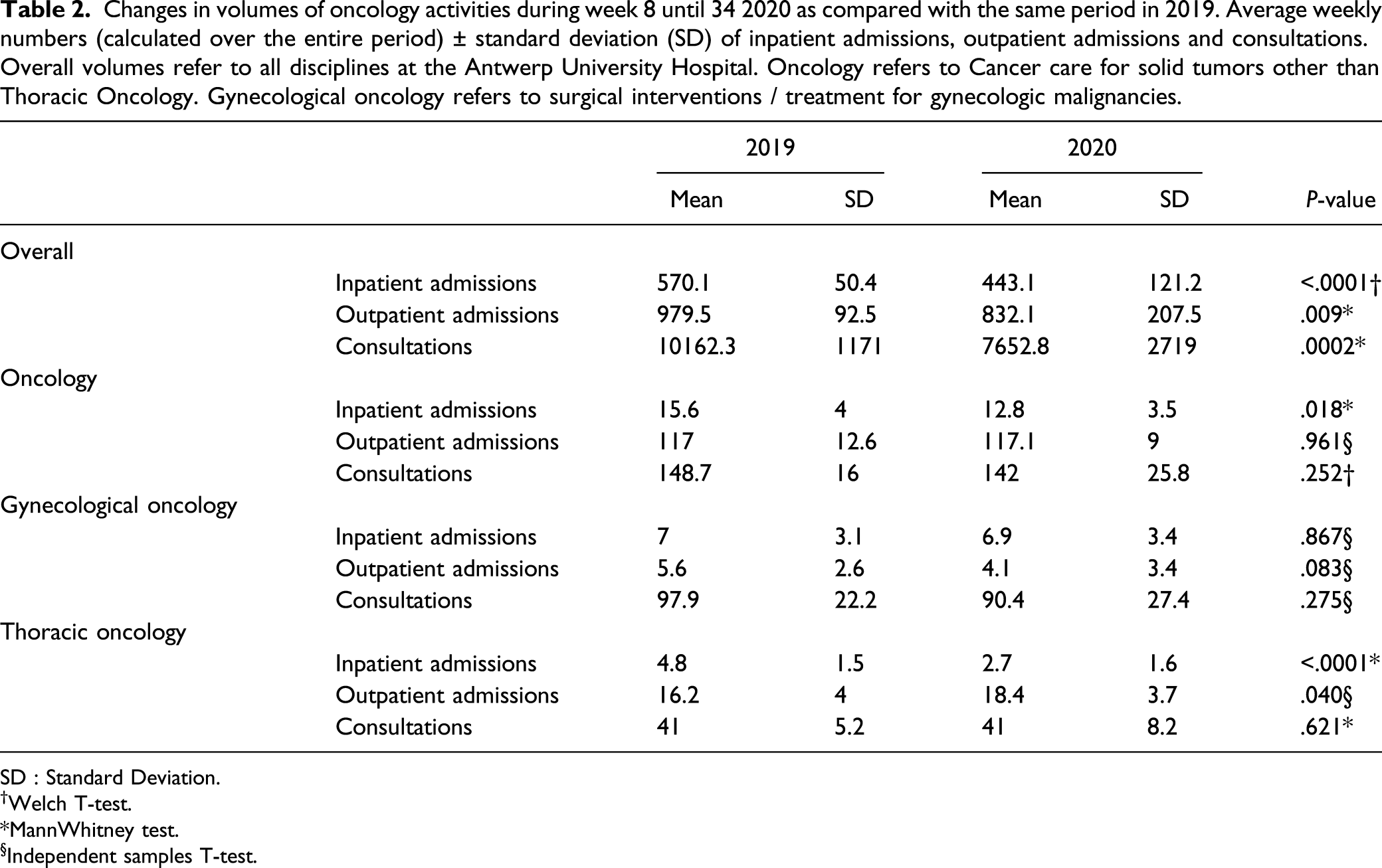
SD : Standard Deviation.†Welch T‐test. *MannWhitney test.§Independent samples T‐test.
In-hospital consultations with oncology specialists did not demonstrate statistical meaningful differences between 2019 and 2020 (oncology: mean = 148.7 vs mean = 142, respectively; P = .252).
Diagnoses
We retrospectively evaluated all new cancer diagnoses made at the Antwerp University Hospital Cancer Center during the period from February to July 2019 and 2020, for which a registration to the National Cancer Registry was made. The overall incidence of solid tumor plummeted with 34%. (818 and 1243 incidences in 2019 and 2020, respectively). In order to evaluate stage migration because of delayed presentation, we evaluated 4 major solid tumor types: breast, colorectal, lung, and prostate cancer. All of which were profoundly less frequently diagnosed in 2020 (136, 43, 106, and 48) compared to 2019 (210, 59, 120, and 86). There was no statistical difference when evaluating the 2019 and 2020 cohort per tumor type and stage (e.g., breast (P = .995), CRC (P = .152), lung (P = .474), and prostate (P = .535)).
Questionnaire on the Knowledge About and Acceptance of Risk-Mitigating Protective Measures
We sent out a questionnaire in a cohort of outpatients (n = 119); the questions regarded organization and implementation of the coronavirus disease 2019 (COVID-19) protective measures at the hospital and oncology clinic; the acceptance thereof by cancer patients; and anxiety concerning infection risk, regardless of these measures. The demographics of the 119 responders can be reviewed in supplementary data file.
A majority of patients were knowledgeable about the protective measures (88%), most declared to have received this information at the entrance of the hospital (billboards) or subsequently at the triage gate following the entrance of the hospital (69 and 7.6%).
Apprehension for waiting rooms or other hospital facilities was little and patients felt safe 73.1% and 79.8%, respectively. Contrary and perhaps as a consequence, patients were somewhat reluctant to regard e-health/ phone-based consultations as a valid surrogate (46.2% thought this to be an alternative and 34.5% only quite so).
We registered a broad appreciation of the risk-mitigating strategies (68.9% of respondents high and 25.2% quite high); however, slight differences according to specific measures were noted. Wearing a face mask was well accepted by 79.0% (n = 94/119) of patients and isolation within a “domestic cluster” (= a cluster of all people residing under the same roof) or self-quarantine (in case of high-risk contacts) demonstrated feasible for 63.9% (n = 76/119) and 60.5% (n = 72/119); maintaining or restricting to a small social cluster proved less manageable and was appreciated by merely 49.6% (n = 59/119).
46.2% (n = 55/119) of cancer patients claimed to be knowledgeable about the relative interaction of comorbidities and SARS-CoV-2, and 38.7% (n = 46/119) of patients answered that they were afraid (or quite afraid) to receiving chemotherapy because of a higher risk of transmission with the SARS-CoV-2 virus (Figure 1). Distribution of answers to a voluntary questionnaire in an outpatient cohort (numbers indicat percentages of total responders, n = 119 patients). The top two pie charts refer to communication (a) and knowledge (b) of protective measures to prevent SARS-CoV-2 infection, and the relative risk that cancer patients have. The bottom pie chartst indicate the fears of patients regarding visiting an oncology ward (c) or receiving cancer therapy (d) SARS-CoV-2: Severe acute respiratory syndrome Coronavirus-2.
SARS-CoV-2 and Clinical Presentation in Cancer Patients
On August 11th, we retrospectively identified 37 in-and out-hospital Cancer patients with a laboratory-confirmed diagnosis of SARS-CoV-2 infection, at the Antwerp University Hospital. Clinical data of 33 of these patients were evaluated; we excluded 4 because no previous contact was retrieved and no clinical data were evaluable in the electronic patient file.
Patients baseline characteristics and clinical presentation when infected with SARS-CoV-2. SARS-CoV-2 infected patients with solid cancer (diagnosed at the Antwerp University hospital) are compared with patients without cancer (as presented by Sciensano (15)). P value for the univariate comparison between the two groups (patients with solid cancer vs without cancer). OR for age (as a categorical variable) was adjusted for gender; OR for gender was adjusted for age (as a continuous variable); all other ORs were adjusted for age (as a continuous variable) and gender. Systemic symptoms cluster: presence of fever/chills reported by the patient and/or fatigue; respiratory symptoms cluster: presence of cough, sore throat, rhinorrhoea, anosmia and/or shortness of breath; gastrointestinal symptoms cluster: presence of diarrhoea and/or nausea/vomiting; neurological symptoms cluster: presence of headache and/or irritability/mental confusion. Respiratory signs cluster: presence of pharyngeal exudate, dyspnoea/tachypnoea, abnormal pulmonary auscultation and/or abnormal lung imaging; neurological signs cluster: presence of coma and/or convulsions.

†Welch T‐test. *Fisher's exact test. §Chisquare test.
We made no analysis on SARS-CoV-2-related outcome due to the small number of SARS-CoV-2-positive cancer patients.
The retrospective cohort of 33 patients were compared to the clinical data from a population-based national dataset obtained by the Belgian public health institute (Sciensano) and published in literature. 15
P values were used for the univariate comparison between the two groups (patients with solid cancer vs without cancer). We used a chi-square§ test for the categorical variables and a Fisher’s exact* test if more than 20% of the expected values were lower than 5.
OR: odds ratio: The corresponding odds ratio with 95% confidence interval was also calculated. For age, as continuous variable, the Welch t-test† was used.
Discussion
We report on the continued care provided in a single tertiary cancer center, during the first surge of the SARS-CoV-2 pandemic. As the SARS-CoV-2 pandemic spread globally, it outpaced health systems everywhere. Competing with healthcare logistics and man power, SARS-CoV-2 challenged the cancer care continuum as we knew it.
Our dataset adds to the real-life experience and demonstrates that continued care proved feasible and safe. In order to maintain active treatment, the hospital implemented structural changes to safeguard social distance and shorten the time spent at the oncology out-clinic. Telehealth was introduced with some mixed results: 1) A modified e-tool offering not only toxicity registration and management but also screened for SARS-CoV-2-related symptoms was well accepted. 22 However, we encountered reluctance to tele-consultations, which only 46.2% of patients regarded as a worthy surrogate to a face-to-face consultation. This probably resulted in the near similar number of specialist consultations, regardless of SARS-CoV-2 pandemic or not.
Telemedicine has been heralded as the silver lining in the cloud of the SARS-CoV-2 pandemic. 23 Before it can be integrated into longitudinal cancer care, still some challenges remain. These concern technological issues to allow all patients (and care providers) to navigate telemedicine, 24 but also operational issues such as the deployment of the support staff in the clinics, that will engage differently with their patients. And finally, policy makers will need to address payment parity in order to fulfill the full potential of telemedicine.23,25,26
As in other cancer centers, we too saw a robust decline in diagnoses and appreciate the future excess in cancer morbidity (in case of stage shift and delayed treatment) and cost due to COVID-19-related disruption of care pathways.27-30
Fear, uncertainty, and even mistrust toward health systems and hospitals may perhaps explain this backlog in cancer diagnoses. Adequate and timely information, an effective doctor/care team–patient relationship, and prompt psychological support are critical to overcome new physical or social barriers.31,32 This can be somewhat illustrated by the results of the patient questionnaire of patients’ reported perception of organizational measures. Even though 21.9% of patients claimed to be afraid of an interaction between cancer treatment and SARS-CoV-2 infection, over 90% felt safe (or quite safe) at the cancer care outpatient unit. We postulate that this may be explained by the support of the care team, the information provided about, and acceptance of hygienic measures (94.1% clear and reasonably clear).
In our final dataset, we demonstrated a low rate of detectable SARS-CoV-2 infections in cancer patients and compared their clinical characteristics to a national dataset by Sciensano, which included only hospitalized patients. 15 Although this comparison is flawed, our analysis was retrospective and limited in sample size of both in- and outpatients, we believe that presenting these real-life data illustrate the fact that cancer patients received faster testing and screening in absence of or with mild symptoms, that they suffered the same symptoms, when infected with SARS-CoV-2, but may have demonstrated an overlap with chronic underlying afflictions. Finally, that they presented earlier, as assumed by the lack of symptoms at presentation.
This cohort analysis has notable limitations. We reported several datasets and retrospective cohort analyses retrieved from single University Hospital site (also SARS-CoV-2 reference center); therefore, results may not be generalizable to different settings. It is also important to note that the patient questionnaire was not randomized but on a voluntary basis; the patient population may therefore be sociodemographically homogeneous (additional data).
Still, this report illustrates a real-life experience during the first surge of the COVID-19 pandemic in a single tertiary cancer center.
The agility and flexibility of all healthcare professionals to continue care in difficult circumstances is to be applauded. However, the experiences during this crisis will need to guide future cancer care provision.
As the SARS-CoV-2 health crisis lingers on, we must address the possible ramifications of delaying or cancelling possible curative interventions and the societal costs that will ensue. 30 Although we successfully introduced telemedicine in active cancer care and toxicity management, we need to learn from past experiences to prepare the opportunity to offer innovative state-of-the-art cancer care to patients and their caregivers from diagnosis onward. As the benefits of collecting and addressing patient-reported outcomes remotely in order to gain overall survival and reduce unplanned healthcare utilization are well known,33,34 offering a broader set of (web-based or electronic) features to a multitude of stakeholders can allow telemedicine to fulfill its role in a more longitudinal care path.
Conclusion
COVID-19 has impacted the clinical cancer management in many ways; and the full extent on care delivery remains for now largely unknown. This retrospective cohort analysis adds to the evidence that continuation of active cancer therapy and specialist visits is feasible and safe with the implementation of telemedicine.
Supplemental Material
sj-pdf-1-ccx-10.1177_10732748211045275 – Supplemental Material for What Makes a Military Professional? Evaluating Norm Socialization in West Point Cadets
Supplemental Material, sj-pdf-1-ccx-10.1177_10732748211045275 for Meeting the Challenges in Cancer Care Management During the SARS-Cov-2 Pandemic: A Retrospective Analysis by Marika Rasschaert, Pieterjan Vanclooster, Laura Depauw, Tim Mertens, Ella Roelant, Elke Coenen, Sebastien Anguille, Annelies Janssens, Peter Van Dam and Marc Peeters in Cancer Control
Footnotes
Acknowledgments
We thank the study participants and the many physicians and nurses who reviewed, responded and interacted to symptom reports of our patients.
We also thank all medical staff members involved in the patient centered care offered in the outpatient facilities during trying times.
Author Contributions
M.R. and PVD: Conceptualization. M.R. Data Curation, formal analysis, writing original draft, Writing review & editing. P.V. and L.D.P. Data Curation, Writing review & editing. T.M. and E.C. Resources, data curation. E.R. Validation, Methodology Formal analysis, Writing review & editing. S.A., A.J., P.V.D., M.P. Writing review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All methods were carried out in accordance with relevant guidelines and regulations. Patient registries and epidemiological data were captured in accordance to the General Data Protection Regulation (GDPR) (EU) 22016/679 [21] on the protection of natural persons with regard to the processing of personal data and on the free movement of such data.
Informed Consent
Informed consent was waived and the study protocol was approved by the medical ethics committee of the Antwerp University Hospital. (EC UZA 20/39/506; EDGE:001380). Belgian Registration number: B.3002020000180.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are not publicly available due to privacy issues but are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
