Abstract
Introduction
Case series
We describe four patients with
Conclusion
Combination of BRAF/MEK inhibition can be a novel, promising approach as targeted therapy in gliomas with
Introduction
Case #1
A 21-year-old woman was found to have an enhancing mass with a cystic component in the upper cervical cord involving the medulla oblongata on Magnetic Resonance Imaging (MRI) after presenting with a syncopal episode. She underwent biopsy of the lesion at an outside hospital. Pathology showed mild chronic inflammatory disease; however, upon review at Moffitt Cancer Center (MCC), pathology revealed ganglioglioma, World Health Organization (WHO) grade I (Figure 1), Histopathology. Case 1 shows atypical ganglion cells immersed in disorganized glia (A. H&E, original magnification ×200) with positive immunohistochemistry for mutant BRAF V600 E (B. x200). Case 2 has been published in greater detail previously.
29
Case 4 shows mitotically active atypical epithelioid cells (C. H&E x400) which strongly express mutant BRAF V600 E (D. x200). Case 3 not shown. Magnetic Resonance Imaging of the brain of case #1 at initial diagnosis and before and after treatment with BRAF/MEK inhibitors. On 05/2017, initial MRI brain showed interval tumor growth of the ganglioglioma involving brainstem and upper cervical spinal cord as seen with increased enhancing tumor and T2 signal (A, D, G, J.). The enhancing part of the upper largest brainstem lesions measures 1.17 cm on axial view (A.) and 1.06 x 0.83 cm on sagittal view (G.). On 05/2018, MRIs showed disease progression with increase in the size of the lesions, with the enhancing part of the upper brainstem lesion now measuring 1.48 cm in axial view (B.) and 1.42 x 1.43 cm in sagittal view (H.) (B, E, H, K.). BRAF/MEK inhibitors were added. Most current MRIs as of 10/2020 show overall smaller in the size of the lesions, with the enhancing part of the upper brainstem lesion now measuring 1.32 cm in axial view (C.) and 1.10 x 0.80 cm in sagittal view (L.) (C, E, I, L.). A–C and G–L = T1 Post-Gadolinium. D–F and J–L = T2 FLAIR sequences.

Given the superior efficacy of concurrent BRAF/MEKi therapy in other cancer types and gliomas with
Unfortunately, soon after there was radiographic evidence of disease progression and therapy was changed to encorafenib 450 mg PO daily and binimetinib 45 mg PO BID, based upon higher dosing and potentially greater CNS penetrance, while minimizing toxicity. Chloroquine 500 mg PO daily was also added based on early evidence supporting inhibition of autophagy as a potential strategy for overcoming MAPK-pathway resistance.27,28 She had a good response to treatment. After 5 months, the treatment was held due to hyperglycemia. She remained on surveillance with serial scans. Since the treatment was held, she has remained with clinical and radiographic SD ongoing at 29 months after initial start of BRAF/MEKi and 18 months after addition of chloroquine to BRAF/MEKi therapy (Figure 2).
Case #2
A 19-year-old, previously healthy man was found on MRI of the brain to have a large right-sided lesion in the parieto-temporal lobes, with surrounding vasogenic edema and a right-to-left midline shift. He underwent a subtotal surgical resection and was diagnosed with GBM at an outside hospital. He was started on standard treatment with radiation therapy (RT) and concomitant temozolomide (TMZ); however, given tumor recurrence 4 months later, he underwent a second maximal safe gross total resection. Pathology was consistent with a glioma resembling an anaplastic PXA, WHO grade III–IV, non-infiltrating, with a high mitotic index, IDH1 R132H-negative by IHC, negative for EGFRviii and 1p/19q co-deletion, and positive for
Seventeen months following his second resection, an MRI of the brain identified a small increase in the size of his tumor. MRI perfusion scan showed hyperperfusion supporting the diagnosis of recurrent tumor. Based on this, combination therapy with dabrafenib 150 mg PO BID and trametinib 2 mg PO daily was initiated. Imaging remained stable on serial MRIs for 14 months, at which point slight progression was noted on MRI and chloroquine 500 mg PO daily was added to his therapy regimen. 29 The patient remained stable for an additional 21 months with the addition of chloroquine to his BRAF/MEKi regimen. Recently, the patient began to progress (as confirmed on MRI brain with MRI perfusion scans) and underwent resection of his anaplastic PXA, WHO grade III-IV, after approximately 35 months on targeted therapy. This is an update to a case that has been published in greater detail previously. 29
Case #3
A 21-year-old woman presented with complaints of dizziness and severe headaches. Her primary care physician ordered an MRI brain, which showed a left frontal 1.2 cm size lesion. Due to these findings, she was advised to go the emergency department. She was admitted at an outside hospital, where an MRI brain/spectroscopy was performed, confirming the 1.2 cm left frontal rim enhancing cystic lesion with restricted diffusion, abutting the superior margin of the insula. She was evaluated by a neurosurgeon who recommended repeat MRI brain in 1 month. The repeat scans showed an increase in the size of the lesion. A left frontal craniotomy with gross total tumor resection was performed at MCC. Pathology showed an epithelioid GBM, WHO grade IV, negative for
She completed treatment with RT and TMZ followed by maintenance TMZ. After 4 cycles of maintenance TMZ, MRI brain showed two new subcentimeter enhancing nodules around the resection cavity with a mild to moderate increased in vasogenic edema on T2 FLAIR sequence, and no mass effect. These findings were concerning for tumor recurrence. TMZ was switched to BRAF/MEKi with dabrafenib 150 mg PO BID and trametinib 2 mg PO daily. She was not on steroids and given no mass effect or concern for treatment-related changes was not started on steroids or any other treatment for radiation necrosis. Subsequent MRI brain 2 months later showed a reduction in the size of the tumor. She has remained clinically and radiographically stable on this treatment at 16 months after initiation of targeted therapy.
Case #4
A 33-year-old woman with a past medical history of Graves’ disease, presented with a 3-year history of worsening episodes of left eye vision loss. An MRI of the brain showed a cystic right parietal mass with peripheral enhancement measuring 5.5 x 4.4 x 5.5 cm and a nodular enhancing region. She underwent a right parietal craniotomy and pathology confirmed an epithelioid GBM, WHO grade IV (Figure 1), negative for Magnetic Resonance Imaging of the brain of case #4 pre- and post-cystic drainage, and before and after treatment with BRAF/MEK inhibitors. On 01/2016, MRI brain performed pre- (A. and E.) and post-surgical drainage of the cystic component of the lesion and before starting treatment with maintenance temozolomide (B. and F.). On 02/2016, MRI with rapid refilling of the cystic component of the lesion and before starting treatment with BRAF/MEK inhibitors (C. and G.). Most current MRIs as of 10/2020 show stable disease since started on BRAF/MEK inhibitors, without requiring further surgical drainage (D. and H.). A–D = T1 Post-Gadolinium. E–H = T2 FLAIR sequences.
She subsequently progressed after 2 cycles of maintenance TMZ, with MRI brain showing interval increase size of bilobed or 2 adjacent areas of ring enhancement in the right parietal lobe deep to the craniotomy site measuring now 5.0 x 3.3 cm compared to previous MRI measuring 4.2 x 2.7 cm, with persistent surrounding increased T2 FLAIR signal consistent with vasogenic edema, which was consistent with recurrent GBM. TMZ was discontinued and she was started on dabrafenib 150 mg PO BID and trametinib 2 mg PO daily. Dexamethasone was tapered off. She had a subsequent immediate response to therapy with interval decrease in the size of the right frontoparietal epithelioid GBM compared to prior MRI brain a month earlier, consistent with interval response to therapy. The patient remains clinically and radiographically stable at this time, 53 months after initiating treatment with BRAF/MEKi.
Discussion
Primary brain tumors are a heterogeneous group of neoplasms that range from relatively slow-growing (ie, pilocytic astrocytoma) to aggressive and invasive tumors (ie, GBM WHO grade IV) with an overall poor prognosis. 30 The standard of care for GBMs remains limited, which includes maximal safe surgical resection with subsequent RT and concomitant TMZ, followed by 6 cycles of adjuvant TMZ.31,32 Second-line therapy with bevacizumab was shown to improve PFS, but no improvement in overall survival was achieved. 33 Other therapies following the standard first-line of TMZ have provided only modest benefit with much need remaining for improved therapy options. 34
In the setting of primary brain tumors, the VE-basket trial recently demonstrated the feasibility of targeting
Literature review of low-grade and high-grade gliomas treated with combination therapy with BRAF/MEKi or monotherapy with BRAFi.
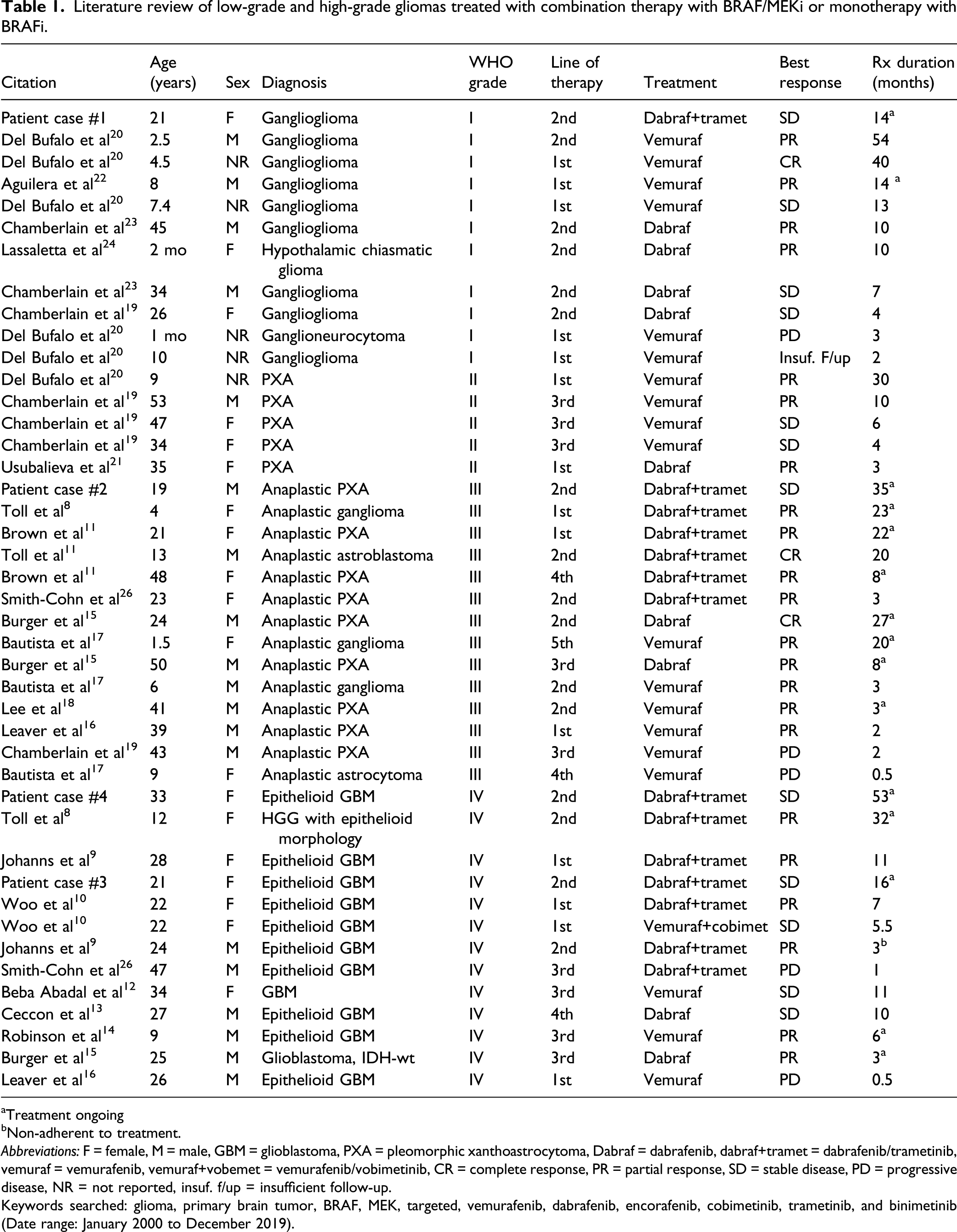
aTreatment ongoing
bNon-adherent to treatment.
Keywords searched: glioma, primary brain tumor, BRAF, MEK, targeted, vemurafenib, dabrafenib, encorafenib, cobimetinib, trametinib, and binimetinib (Date range: January 2000 to December 2019).
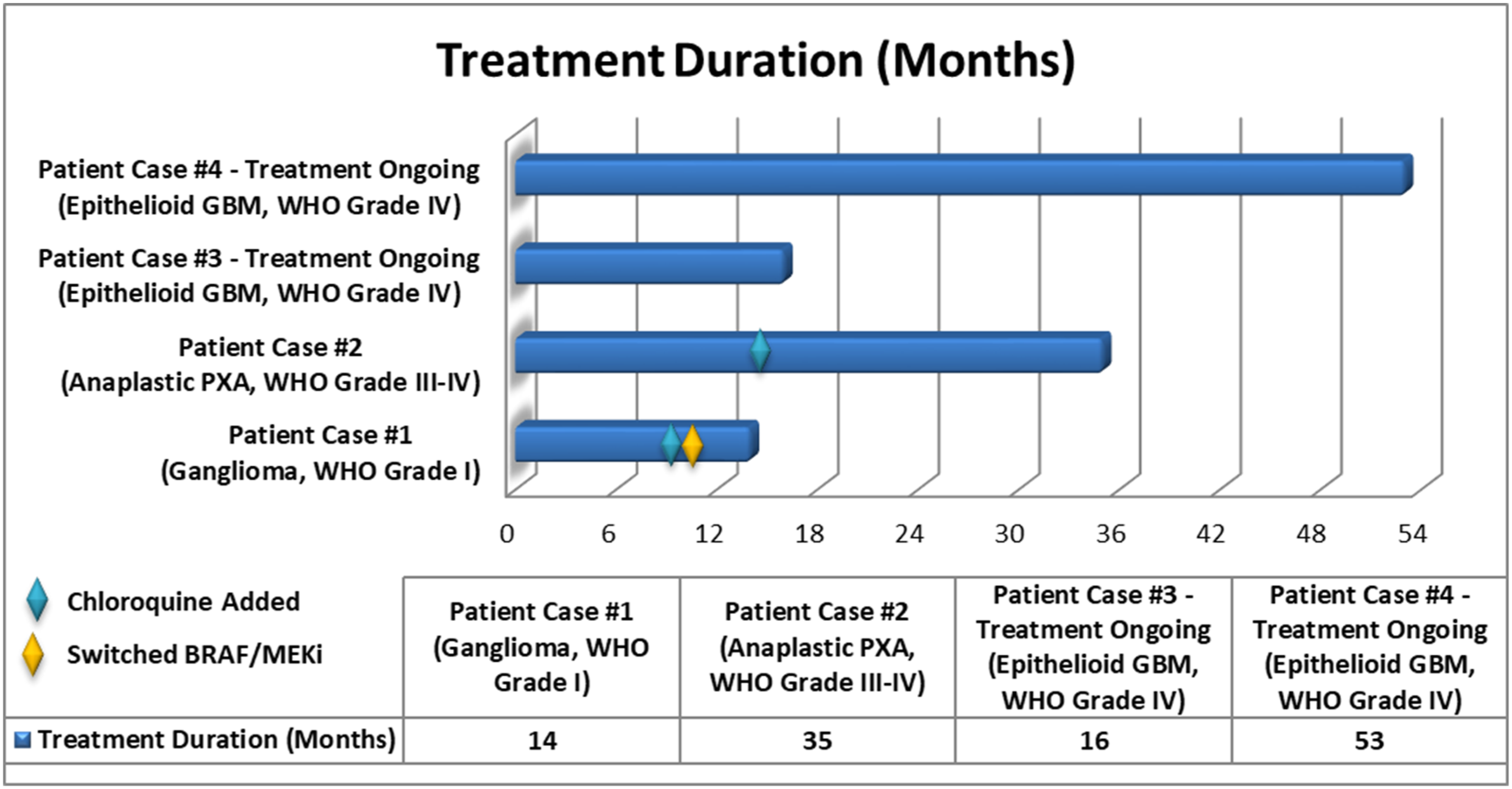
Treatment duration for BRAF V600E-mutant gliomas treated with targeted therapy. Patients were treated with dabrafenib plus trametinib. Patient cases #1 and #2 had chloroquine added to their regimen at approximately 9 months and 14 months, respectively. Patient cases #3 and #4 are ongoing with treatment at 16 months and 53 months, respectively.
The potential for publication-bias remains with described clinical case series of benefit with targeted treatment, but in the setting of rare molecular subsets of cancer they offer important early evidence of clinical activity in rare molecular subsets of cancer. Clinical trials assessing combination dabrafenib/trametinib for both low- and high-grade gliomas are ongoing (NCT02034110) and interim reports have been promising, with an ORR of 62% and 29% in low- and high-grade gliomas, respectively. Durable clinical benefit was also seen in subset of patients with a median treatment duration of 19.5 months (range, 1 to 55 months) and 10.6 months (range, 1 to 43 months) in patients with low- and high-grade gliomas, respectively. 7
Molecular profile of BRAF V600E-mutant GBM cases treated with BRAF/MEKi. Highlighted are the mutations present in the two patients with epithelioid GBM WHO grade IV.

All patients who underwent RT, except case 4, had MRI perfusion performed in addition to the MRI brain to differentiate tumor progression vs treatment-related changes. Case 4 was deemed to have tumor progression based on radiographic appearance, which was further supported by her immediate response clinically and radiographically following treatment with BRAF/MEKi, without the need for dose appropriate glucocorticoids or other therapy for radiation necrosis (ie, bevacizumab). MRI changes observed prior to starting BRAF/MEKi being due to treatment-related changes remain a possibility. Nevertheless, the fact that a patient with epithelioid GBM has remained stable for up to 53 months after starting BRAF/MEKi is noteworthy.
It is worth noting that the ganglioglioma and anaplastic PXA patients had chloroquine added to their regimen of BRAF/MEKi at early signs of progression. Chloroquine was added based upon what was perceived as a relatively low risk of adverse events and promising early clinical data suggesting that autophagy inhibition may help to overcome resistance to targeted treatment with BRAFi.29,42-44
This small case report series has several limitations. These include being unable to generalize to a larger cohort of patients with similar diseases. In addition, it is difficult to determine a cause and effect relationship in these independent, single cases with different histopathological features and WHO grades. Prospective clinical trials are needed to better evaluate the effectiveness of BRAF/MEKi in patients with gliomas. Nevertheless, the positive responses to treatment with BRAF/MEKi seen in these patients can lead to the generation of new hypotheses regarding the pathophysiology of these diseases, and allow the expansion of successful new therapies, when prospective studies are not feasible.
Conclusion
The combination of BRAF/MEK inhibition has the potential to offer clinical benefit in both low-grade and high-grade gliomas that historically have not responded as well to BRAFi monotherapy. Our cases, along with other early reports utilizing dabrafenib/trametinib, highlight the importance of somatic next-generation sequencing, particularly in younger patients. Interim results from clinical trials of dabrafenib/trametinib have been promising thus far, and our case series suggests that durable clinical benefit is possible, even in the setting of glioblastoma, WHO grade IV.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by an NCI Comprehensive Cancer Center support grant (P30 CA076292), Moffitt Pinellas Partners, and the Collins Family Foundation.
Ethical Approval
Our study was approved by the Advarra Ethics Committee (no. Pro00031026, IRB protocol MCC 19859). A waiver of consent was granted for retrospective chart review in accordance with the Declaration of Helsinki and the 21st Century Cures Act.
Informed Consent
All of the patients provided a written informed consent for their information to be published in the current study, except for case #3 who moved out of state and was lost to follow-up. MRI brain and pathology imaging from case #3 were excluded from this study given inability to obtain written informed consent.
Data Availability
All data analyzed during this study are included in this article.
