Abstract
Multiple myeloma (MM) is characterized by bone pain, pathologic fractures, bone destruction, and secondary hypercalcemia, all these conditions impact on health-related quality of life of patients. The objective was to evaluate the global health state and health-related quality of life in a group of patients with MM who attended a tertiary health-care center of the Instituto Mexicano del Seguro Social in Mexico, through the questionnaires designed by European Organization for Research and Treatment of Cancer (EORTC) quality of life group. Exploratory cross-sectional study in patients with MM treated in a Department of Hematology in a High-Specialty Medical Unit was conducted. Patients older than 18 years of age, men and women, were selected, and their informed written consent was obtained. We included all consecutive cases treated from January 2012 to December 2014. Questionnaires EORTC QLQ-C30, EORTC QLQ-MY20, and EORTC IN-PATSAT-32 were used. We studied 37 patients, 19 (51%) men and 18 women. The mean age was 61.9 years. Twenty-two (59.46%) patients presented with clinical stage III. The mean time for diagnosis was 33.11 months. The most used first-line treatment schedule was melphalan/prednisone/thalidomide (15; 40%). The global health median was 66.67, and symptoms showed a median score of 22.22. Treatment side effects score was 16.67; for general satisfaction, the median score was 75. In conclusion, the patients showed an advanced clinical stage and poor prognosis but had scores higher than 50 in functional scales and lower than 50 for symptom scales. The scores for symptom scales were related to age, renal failure, and disease-free survival. Identification of quality of life and satisfaction of care markers allow for early therapeutic intervention and efficiency and enable a change in quality of life and perception of care in Health Services.
Introduction
Multiple myeloma (MM) is a malignant neoplasm due to B lymphocyte-derived plasmatic cells clonal proliferation and immunoglobulin clonal production. 1 Marrow infiltration causes hematopoiesis dysfunction with anemia and other cytopenias. 1 The associated clinical data are bone pain, pathologic fractures, hypercalcemia, and renal failure. 2 Multiple myeloma is the second most common hematologic malignant neoplasm, and it represents 1% of all cancers and 10% of hematologic malignant neoplasms. 3,4 In Western countries, annual age-adjusted incidence is 5.6 cases per 100 000 people. 5 In the United States, 20, 520 new cases were diagnosed in 2011. 6 In Mexico, a report published in 2007 mentions that during 2002, 597 incident cases were diagnosed, 7 and other report published in 2011 mentions that 1068 new cases were detected during 2006 (increment 1.01%), 8 with a mortality of 1 per 100 000 people. 1 An additional work published in 2015 reported 98 consecutive MM cases from May to December 2012. 9
Relevant issues in MM pathogenesis are close relation between plasmatic cells and bone marrow stromal tissue to permit survival, growth, and differentiation, 2 and clinical characteristics influenced by primary and secondary genetic alterations that impact the disease clinical course, clinical response, and prognosis. 3,10 Prognosis is estimated through 2 scales: Durie and Salmon (D&S) 11 and International Staging System (ISS) 12 for MM.
Health-Related Quality of Life in MM
Health-related quality of life has been used as health state synonymous, and its principal objective is to analyze whether a disease or a chronic health condition and its symptoms interfere with the daily life of a patient. 13,14
Initially, in oncologic diseases, some scales were designed to estimate physical function in this group of patients, 15 and afterward, different questionnaires have been developed to measure different patient-reported outcomes (PROs), maybe the most important is the health-related quality of life (HRQoL).
Currently, many clinical trials that analyze overall survival, disease-free survival, tumor activity, treatment efficacy, relapse, or death also analyze HRQoL and other PRO’s such as patient satisfaction as part of the standards and outcomes for the specific clinical trial, considering that many oncologic diseases are chronic conditions in which it is mandatory to preserve functionality and HRQoL in surviving patients. 16 As part of the questionnaires used to evaluate HRQoL, different items study patient perspective with respect to health problems related to quality of life. 17 -19 In patients with cancer in general, and those with malignant hematologic neoplasm, it is difficult to apply an unique questionnaire, and this is the reason to develop disease-specific modules. 19 -23
In MM, the clinical course has been related to chronic pain, pathologic fractures, renal failure, 24 and treatment itself and is related to bothering procedures as part of the follow-up such as intravenous injection needs, repeated bone marrow biopsy, secondary effects of treatment like myelosuppression, mucositis, thrombosis, neuropathy, jaw osteonecrosis, prolonged hospital stay, and repeated transfusions 25 -28 that potentially impact HRQoL.
The objective of this work was to evaluate the global health state and HRQoL in a group of patients with MM, 29,30 attended in a tertiary health-care center of the Instituto Mexicano del Seguro Social (IMSS) in Mexico, through the questionnaires designed by European Organization for Research and Treatment of Cancer (EORTC) quality-of-life group.
Materials and Methods
Study Design: Exploratory Cross-Sectional Study
Research setting
The research was conducted at Tertiary health care center (Unidad Médica de Alta Especialidad Hospital de Especialidades, Centro Médico Nacional de Occidente. Instituto Mexicano del Seguro Social) at Guadalajara, México. This is a reference hospital for secondary health-care facilities in Western Mexico. All patients were selected from Hematology Clinical Department.
Patients
Patients were selected with MM diagnosis, older than 18 years of age, either gender, and providing a signed institutional informed consent form. We included all consecutive cases treated from January 2012 to December 2014.
Data collection
The information was obtained through 2 ways: (1) from patient’s clinical charts, including comorbid conditions and complemented directly by (2) applying the following questionnaires. EORTC QLQ-C30: In 1980, The European Organization for Research and Treatment of Cancer began the research program to develop an integrative scope based on specific modules per each malignant disease to evaluate HRQoL in patients enrolled as cases in clinical trials. This action derived in the design of a core questionnaire to measure HRQoL in patients with cancer: EORTC QLQ-C30.
31,32
This is a generic questionnaire and must be complemented with specific malignant disease modules.
32
EORTC QLQ-MY20: This questionnaire was developed to study HRQoL MM, and it is constituted by 20 items to evaluate symptoms, treatment side effects, and impact on daily life. The module must be used with EORTC QLQ-C30.
16
The module has been validated and applied in different populations, including Mexican patients.
9,33,34
EORTC IN PATSAT32: The EORTC quality-of-life group designed the questionnaire IN PATSAT32 to evaluate patient perception about quality of health care in hospitals for patients with cancer. It is constituted by 32 items and arranged in 11 multi-item scales; they evaluate the quality of health care provided by the physicians, nurses, hospital environment, and administrative aspects of the hospital. 35 The psychometric properties of the Spanish version EORTC IN-PATSAT32 have been proven in 80 different tumors. 36 The current treatment objective in MM is to achieve disease control and enhance survival; in this stage, the issues related to HRQoL are important; 37 lately, HRQoL has been considered an important aspect influencing patient outcome in cancer treatment clinical assays. A better understanding about HRQoL could help to give a better health care to this kind of patients. 38
All the questionnaires described earlier 16,17,31 -36 were used and all of them validated in the Mexican–Spanish language and based on the Likert-type scale. The mean time to answer the questions is about 15 minutes. The questions have shown to be easy to understand, and in previous international reports, patients had no sensation of discomfort with the question related to their symptoms or troubles related to their disease.
The way to obtain the score of the items is the following: in the case of multi-item scales (1) a raw score (RS) for the specific scale through the formula: (I1+I2+I3…In)/n; (2) range R (maximum value for RS − minimum value for RS); and (3) after that a linear transformation is performed to get a 0 to 100 score, using the following formulas: (1) functional scales: S=(1 − ((RS − 1)/range)) × 100; (2) symptoms scales: S = ((RS − 1)/range) × 100; and (3) global health scale: S = ((RS − 1)/range) × 100.
The standardized score of 0 to 100 is the same for the 3 questionnaires, and the score reflects the levels of patient affectation or satisfaction for each scale.
Sample size
This is an exploratory study that included consecutive outpatients during the mentioned period. We calculated the power for the study once that data collection was completed.
Statistical Analysis
The results were described as mean and (standard deviation) and median and (interquartile range) for the HRQoL scales because these data show a nonparametrical distribution, and proportions are described as percentages. Comparisons between groups were evaluated by Student t test for independent samples or Mann-Whitney U test, according to data distribution. Proportions were compared with χ2 or Fisher exact test. Obtaining a P value <.05 was considered significant. Reliability and internal consistency for multi-item scales were calculated through Cronbach’s α. The association between variables was estimated through Pearson coefficient correlation (rP), and a P value <.05 was considered significant. We performed a logistic regression analysis to identify variables associated with HRQoL. Data were analyzed with the statistical SPSS v21.0 (SPSS, Chicago, Illinois) software. This study has a risk lower than the minimum according to the Reglamento de la Ley General de Salud en Materia de Investigación en Salud in Mexico, and it complies with the guidelines of the Declaration of Helsinki adopted by the 18th WMA General Assembly, Helsinki, Finland.
The project was evaluated by the Comité Local de Investigación y Ética en Investigación en Salud 1301 from the Hospital de Especialidades Centro Médico Nacional de Occidente, with the approval number R-2015-1301-24. Data of each patient were managed confidentially and anonymously; data were analyzed and identified by the social security number. Patients signed the institutional informed consent form.
Results
The study comprised 37 patients, with a median age of 62 years (interquartile interval: 32 to 98). Of these, 18 (48.6%) were women. The most frequent clinical stage was Durie and Salmon III (59.46%) and ISS (54%). The mean follow-up time was 35.11 months. Other comorbid conditions were found such as arterial hypertension (29.72%), diabetes mellitus type 2, chronic renal failure (13.5%), and previous episode of thrombosis in 3 patients (8.1%); additional features are included in Table 1. Lytic images were found in 86.5% and plasmacytoma in 46% (lumbar, 35%; cranium 29%; and ribs and femur, 12%).
Sociodemographic and Clinical Features of 37 Mexican Patients With MM Attending IMSS Facilities in the Western Region of Mexico.

Abbreviations: IMSS, Instituto Mexicano del Seguro Social; MM, Multiple Myeloma.
a According to clinical stage proposed by Durie and Salmon.
Table 2 presents the biomarkers related to prognosis and tumor activity. We found 32 study patients with ECOG (the Eastern Cooperative Oncology Group ECOG, part of the ECOG-ACRIN Cancer Research Group, and published in 1982) performance status 2, and 5% with ECOG 3; the remaining patients had ECOG 1.
Laboratory Features of 37 Patients With Multiple Myeloma Attending IMSS Facilities in the Western Region of Mexico.

When considering the frequency of treatments, the most used schedule as first-line treatment was melphalan/prednisone/ thalidomide (MPT) in 15 (40%) patients and thalidomide /prednisone in 13 (35%) patients. The most used schedule as second-line treatment was MPT in 9 (24%) patients, and liposomal doxorubicin/dexamethasone (DLD) in 7 (19%) patients. The third- and fourth-line treatment included treatment with proteasome inhibitor in 10 (32%) cases.
Regarding the relationship between treatment and HRQoL, we found a significant correlation in global health status/quality of life and second-line treatment (P = .042; see Table 3). The correlation between clinical stage, ECOG, prognostic index, lytic images, and current response status were also analyzed. Progressive disease was shown by 14 (38%) patients, while 6 (16%) patients showed complete clinical response (Table 1).
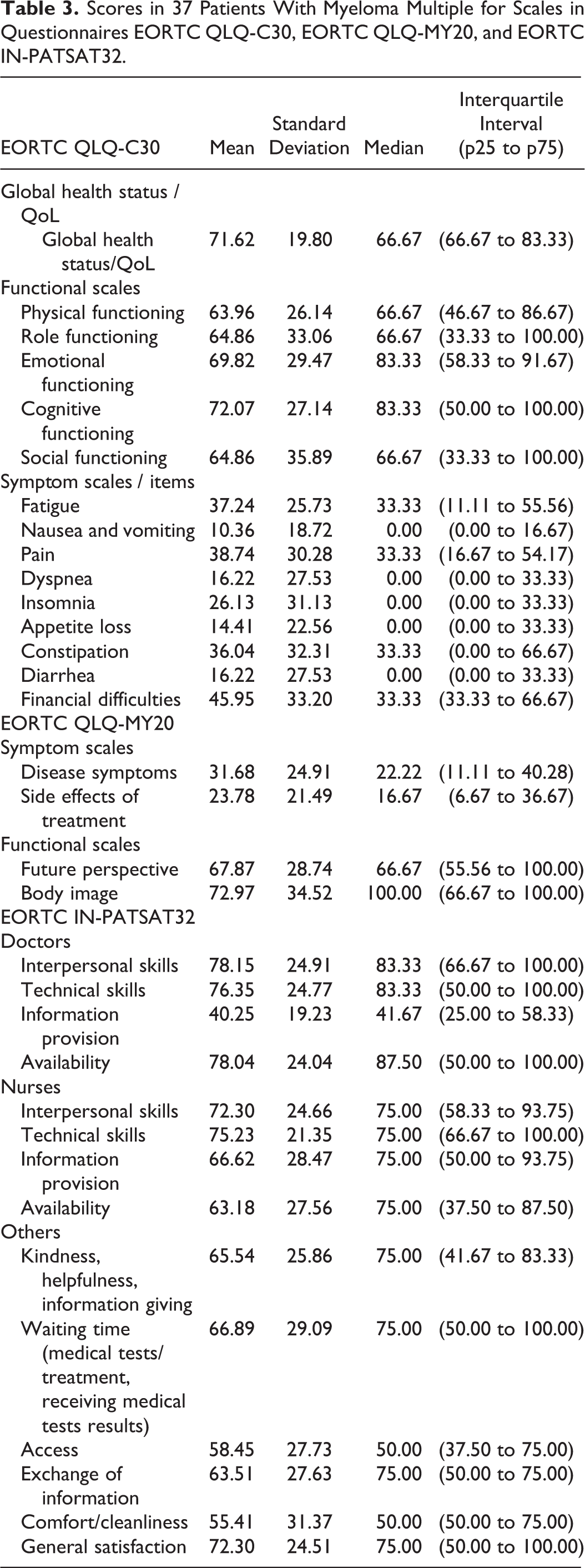
Scores in 37 Patients With Myeloma Multiple for Scales in Questionnaires EORTC QLQ-C30, EORTC QLQ-MY20, and EORTC IN-PATSAT32.
The mean time of thalidomide treatment was 15.37 months and most used doses were 50 mg/day orally, and it was longer in patients with sensory neuropathy grade II.
Table 3 describes the results obtained in the MM-specific module (EORTC QLQ-MY20), and the scores for functional scales and symptom scales. The questionnaire EORTC IN-PATSAT32 is shown in the same table.
Functional scales in questionnaire EORTC QLQ-C30 show some correlations with clinical variables: global health state/quality of life was related to serum calcium (P = .002); emotional function was associated with immunoglobulin G and albumin and cognitive function. In the symptom scales, we observed some association with clinical values (see Table 4).
Variables Association in 37 Patients With Myeloma Multiple With Scales From Questionnaires EORTC QLQ-C30, EORTC QLQ-MY20, and EORTC IN-PATSAT32.

The scales in the questionnaire EORTC QLQ-MY20 show relationship between functional and symptom scales. In the EORTC IN-PATSAT32, satisfaction with medical care showed association with plasmatic cells percentage and albumin (P = .04). General satisfaction was associated to κ serum level and plasmatic cells percentage (P = .04 and .03, respectively, Table 4).
Clinical stage ISS, ECOG, lytic images, and global health status/QoL did not show association with general satisfaction. The first-line treatment with therapeutic schedules, melphalan/prednisone/thalidomide or thalidomide/dexamethasone, were related to higher scores in general satisfaction (P = .03).
Discussion
Results revealed that patients with MM showed high scores in HRQoL scales, approximately 11 points higher than those observed in reference studies. In these studies, the highest difference was observed in fatigue and pain scales. In our sample, the score was under 50 points (0-100 scales by linear transformation), which represents a low level of symptoms. 28 The observation is probably related to the HRQoL expected by this group of Mexican patients with MM.
A first report from a research group from Mexico validates the EORTC QLQ-C30 and EORTC QLQ-MY20 instruments in their Mexican–Spanish versions, 9 and other reports from Latin American research groups 7,13 describe the use of the EORTC QLQ-C30 instrument in patients with hematologic neoplasm; but until now, the current data are the only report from the Western region of Mexico.
Multiple myeloma, as a chronic disease, is concurrent with complications and treatment failure, and both conditions affect HRQoL and is correlated with disease-free survival and overall survival. 39
In our data, we found renal failure in 13.5% of patients at the time of diagnosis and in the literature 20% to 25% is reported. 40 Renal failure is related significantly to high scores in fatigue, pain, and insomnia scales and reflects a poor functional status as a complication derived from MM. The biomarkers are related to higher tumor charge; most patients are in clinical stage III, and the renal damage could be reversed. But there higher rates of early mortality have been observed in patients with persistent renal damage 40
Hypercalcemia was significantly associated with global health status/QoL. In other reports, hypercalcemia has been related to fatigue, nausea, and vomiting as well as cognitive functioning. In our study, we did not observe the cited associations.
Hypercalcemia complicates the treatment because it is related to dehydration and prerenal azotemia 41 Our patients are treated through hyperhydration and dexamethasone; we limit use of bisphosphonates and other treatments such as the use of calcitonin as well as plasmapheresis, hemodialysis, and RANK antibodies (receptor activator and nuclear factor kb). 41
Known poor prognosis factors such as clinical stage, ISS, ECOG and lytic images were not associated with HRQoL or patient satisfaction with health care. Most of our study patients showed an advanced clinical stage. The patients with good clinical response showed a better HRQoL and diminished symptoms. The overall survival was not associated with global health status/QoL or general satisfaction, coinciding with results described by Cömert et al. After the first year of diagnosis, no significant differences exist in toxicity between schedules, symptoms, or scores for HRQoL. 39
We observed a correlation among global health status/QoL, general satisfaction, and treatment schedules. The second-line treated patients had better scores, and this condition could be explained because the patients were strictly observed regarding their disease progression.
The patients treated with MPT or thalidomide/dexamethasone showed higher scores in general satisfaction, probably due to a treatment with low toxicity and ambulatory administration. The third- and fourth-line treatments did not show significant correlation.
The immune modulators used (IMiDs) such as thalidomide or second-generation IMiDs are related to higher HRQoL scores. 42 In our sample, thalidomide use was not related to high scores in HRQoL, and its use was limited to first-line schedules. With a second or third relapse, thalidomide is counterindicated because it can exhibit higher toxicity rates, mainly peripheral neurotoxicity. Weisel et al 43 report that the use of second-generation IMiDs + low doses of dexamethasone in a phase III study, improved symptoms, and decreased disease progression in a group of patients in who lenalidomide and bortezomib had failed. They propose that a maintenance period second-generation IMiDs + low doses of dexamethasone could prolong overall survival and result in clinical benefits in patients with relapsed or refractory and advanced stage of the disease 43
In patients treated with proteasome inhibitors, we did not observe association with higher HRQoL scores, and this situation probably is associated with this kind of treatment used as a third- or fourth-line treatment. Some reports with proteasome inhibitors being used as first-line treatment showed high scores in HRQoL and less adverse effects.
Lee et al 44 compared melphalan/prednisone versus melphalan/prednisone/ bortezomib, and the best results in HRQoL were observed in the patient group treated with bortezomib. Delforge et at 31 reported better results with low doses of bortezomib and less side effects.
We found a mean hemoglobin of 10.88 g/dL, and this variable has been related to higher HRQoL scores, better exercise capacity, less fatigue, and a sensation of wellness. 45 In the evaluation related to patient satisfaction, we found higher scores in the scales related to willingness and kindness, helpfulness, and information provided by other hospital personnel and technical skills, whereas for information provided by the physician the scores were lower, this situation is probably related to the time spent during the medical appointment in a public health institution, which does not allow explaining different disease issues to patients and their families. Rood et al 46 reported that the need for information in patients with hematologic malignant neoplasm is more evident in younger patients and in patients affected by comorbid diseases and in those patients who perceive his or her HRQoL as bad.
Other authors, like Wagner et al, 47 reported that the information provided by the physician is an important factor associated with HRQoL, together with multidisciplinary support in palliative care, pain treatment, orthopedic procedures, and rehabilitation. Supportive treatments could help to maximize treatment benefits and enhance the patients’ well-being in all disease stages. 47
In the evaluation related to patient satisfaction, we found high scores in the scales. Identification of markers that are related to the quality of life in patients with MM, such as pain, fatigue, anemia, and renal failure, is pivotal, as it indicates the need to be persistent in the relevant therapeutic intervention aimed at modifying these markers to improve quality of life. A good quality of life is related to overall survival.
Identifying the level of satisfaction of care in these patients that correlate with markers of tumor burden as a percentage of plasma cells, serum level of light chains, allows us to anticipate the efficacy and opportunity of treatment, and early intervention will improve the perception of care in health services.
Limitations of the Study
This study with an exploratory design includes a small sample, and because of this feature, the results have very limited generalizability.
Conclusion
Most patients in our study had an advanced clinical stage and poor prognosis, but they showed high scores in functional and symptom scales. Markers related to poor prognosis, such as clinical stage, ISS, and ECOG, are not related to poor HRQoL or patient satisfaction. The highest scores in symptom scales are related to aging, renal damage, and disease-free survival.
Footnotes
Authors’ Note
The project was evaluated by the Comité Local de Investigación y Ética en Investigación en Salud 1301 from the Hospital de Especialidades Centro Médico Nacional de Occidente, with the approval number R-2015-1301-24. Data of each patient were managed confidentially and anonymously; data were analyzed and identified by the social security number. Patients signed the institutional informed consent form.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
