Abstract
Developmental disorders are diverse, common, and impairing; still, many clinicians lack comprehensive training in their assessment. This review presents thorough guidelines for the evaluation and diagnosis of common communication, sleeping, feeding, and elimination disorders that often onset in the early developmental period and that are commonly encountered in clinical practice. Thorough guidance on the evidence-based assessment of developmental disorders is critical, as they are prevalent, impairing, and commonly comorbid with other psychiatric disorders of childhood. This review is the first of its kind—providing critically needed, step-by-step guidance on the existing evidence-based methodologies and assessment tools available for diagnosis. This review also makes clear the dire need for further development and validation of relevant screening and diagnostic measure and calls for specific attention to the development of specific screening and diagnostic assessment measures for feeding disorders and elimination disorders in particular. Clinicians and researchers alike may find this article useful in guiding diagnostic, treatment, and research procedures.
Keywords
Developmental disorders, including communication, motor, sleeping, feeding, and elimination disorders, are diverse, common, and impairing; still, many clinicians lack comprehensive training in their assessment. This review presents thorough guidelines for the evaluation and diagnosis of common developmental disorders that often onset in the early developmental period and that are commonly encountered in clinical practice. Although all communication, sleep/wake disorders, feeding, motor, and elimination disorders are addressed, it should be noted that guidelines for the evaluation of some important neurodevelopmental conditions—autism spectrum disorder (ASD), attention-deficit/hyperactivity disorder (ADHD), intellectual disability, and specific learning disorders—are independently provided in other articles and will not be discussed here (Eng et al., 2023; Fletcher et al., 2005; Kishore et al., 2019; Ozonoff et al., 2005). In addition, as the scope of this article is limited to disorders that typically onset in the early developmental/early childhood period, assessment guidelines are generally provided only for pediatric populations, although many of the conditions can and do present in or persist into adulthood.
This novel review acts as a comprehensive, clinically useful reference for clinical child psychologists and lifespan psychologists alike and represents a long overdue addition to the evidence-based assessment literature.
Section 1: Communication Disorders
Assessment Overview
Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; American Psychiatric Association [APA], 2013) Communication Disorders include language disorder, speech sound disorder, childhood-onset fluency disorder, and social (pragmatic) communication disorder (SPCD; Figure 1). All are characterized by impairments in sending, receiving, processing, and/or comprehending verbal, nonverbal, or graphic language and therefore may be directly referred to speech and language therapists. Symptom onset for communication disorders typically occurs early in life and can result in lifetime impairment (APA, 2013). Some communication disorders can also be acquired as a result of a neurological disorder or trauma. According to the results of a nationally representative survey of communication disorders among children in the United States, communication disorder prevalence is around 7% to 8% in children aged 3 to 17 years (Black et al., 2015).
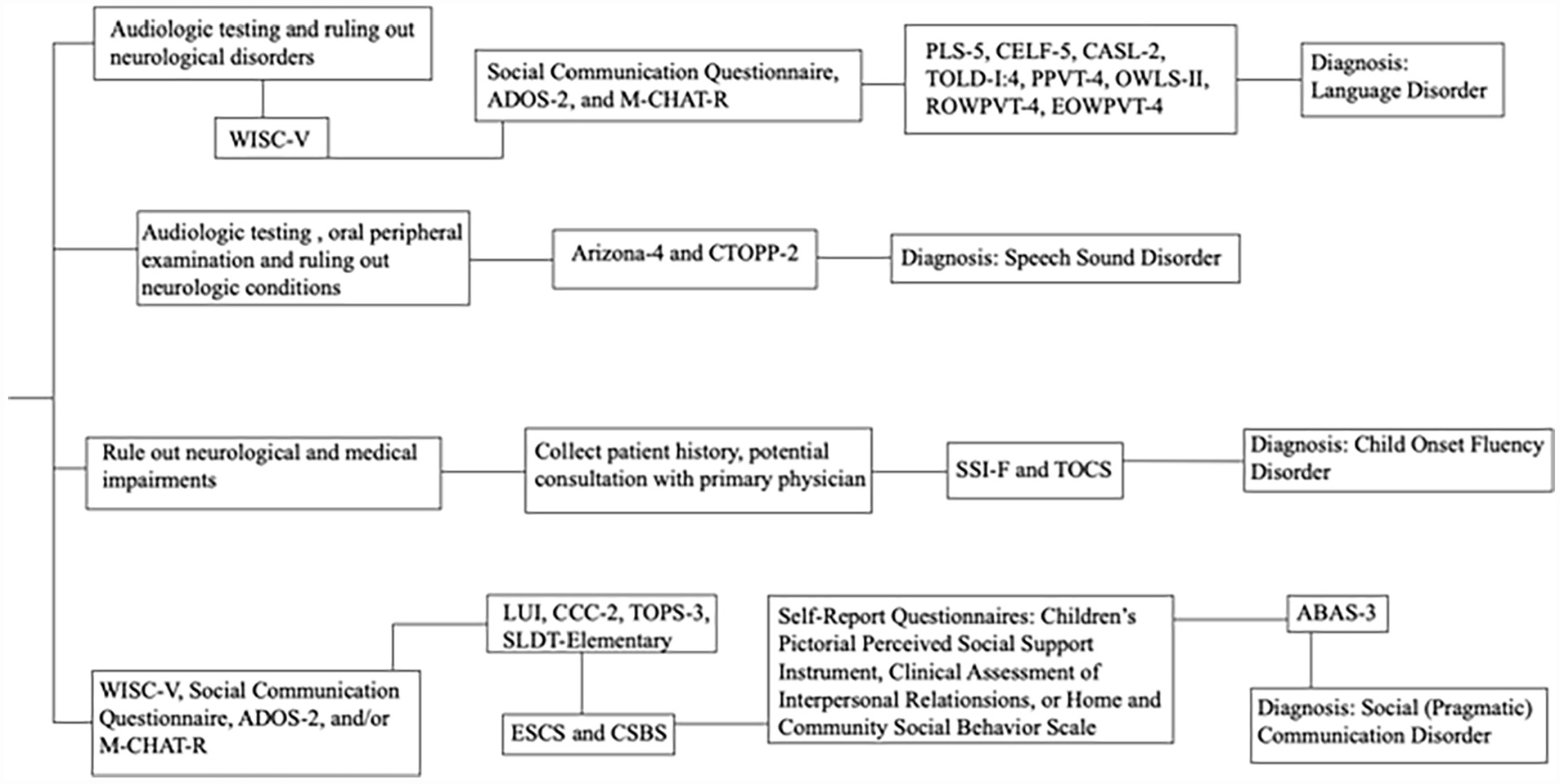
Flowchart of Communication Disorders Assessments.
Language Disorder
Language disorder involves difficulties in the development and use of language that are not considered age appropriate. It involves deficits in verbal and written expression such as comprehension or production of vocabulary, grammar, sentence structure, discourse, sign language, or other forms of language. Difficulties usually arise in language form (phonology, morphology and syntax), content (meaning of words), or function (how language is used by individuals in different contexts), rather than problems in fluency of speech sounds or articulation (APA, 2013). Individuals may struggle with either expressive language (deficits in verbal and written expression) or receptive language (deficits in comprehension), or sometimes both as mixed receptive-expressive language disorder. Differential diagnosis for language disorder must specifically include hearing or sensory impairment, normal variations in language, intellectual disability, ASD, and neurological disorders such as epilepsy, stroke, aphasia, or Landau-Kleffner syndrome (APA, 2013). It is essential that hearing impairment is excluded first as the primary cause of language disorder. A formal audiologic testing is recommended to rule out hearing loss that may be contributing to the language impairment. To rule out neurological disorders, developmental history and symptoms should be evaluated. Consultation with the child’s physician and/or neurologist is advised. Ruling out the presence of intellectual developmental disorder is also crucial, as language delay can be better explained by intellectual disability in some cases. A separate diagnosis is not given for language disorder unless the deficits are in excess of the intellectual limitations of the child. Therefore, administration of a standardized IQ test (e.g., the Weschler Intelligence Scales for Children, 5th edition; WISC-V; Wechsler, 2014) may be warranted. Signs of ASD should also be evaluated. A brief instrument such as the Social Communication Questionnaire (Rutter, Bailey, & Lord, 2003) can be utilized to screen for ASD. In cases where the scores are above the cutoff (>15), a full evaluation is recommended. Primary ASD symptomatology may be evaluated with gold-standard instruments, such as the Autism Diagnostic Observation Schedule (ADOS-2; Kanne et al. 2008; Lord et al., 2012) or Modified Checklist for Autism in Toddlers (M-CHAT-R; Robins, Fein, & Barton, 2009), depending on the age range of the child.
After ruling out specific medical, neurological, and other neurodevelopmental conditions as primary explanations for a child’s symptomatology (Criterion D; APA, 2013), other criteria for language disorder can be considered. Like most developmental disorders, the onset of symptoms must be in the early developmental period to qualify for a language disorder diagnosis (Criterion C; APA, 2013). When a child presents with language difficulties, a thorough case history should be conducted. It should be noted that up until age 4, normal variations in early vocabulary acquisition occur between individuals. These individual differences in language ability tend to stabilize by the age of 4 years at which point they are more predictive of later outcomes (Bornstein et al., 2016; Dale et al., 2003; Norbury et al., 2017; Rice et al., 2008). It is also important to consider regional, social, or cultural/ethnic variations in language before diagnosing with a language disorder. A thorough case history of the child is important to understand if the presenting symptomatology is associated with language disorder or normal variations in the process of learning a language. This is primarily common in bilingual children compared to monolingual children. In the early stages of language acquisition, those learning two languages simultaneously can show some developmental lags relative to children speaking only one language, but this may resolve as they age (Korkman et al., 2012).
The core difficulties such as reduced vocabulary, limited sentence structure, and impairments in discourse (Criterion A; APA, 2013), as well as whether the language abilities are below those expected for age and development (Criterion B; APA, 2013), should then be evaluated. There are several norm-referenced standardized tests that can be used to assess language skills (both receptive and expressive). The Preschool Language Scale–Fifth Edition (PLS-5; Zimmerman et al., 2011) can be used as a screener for a broad range of speech and language skills for children from birth through age 7. In cases where a more comprehensive battery is indicated, the Clinical Evaluation of Language Fundamentals–Fifth Edition (CELF-5; Wiig et al., 2013) is recommended. CELF-5 provides a complete picture of an individual’s language skills through observational and interactive measures evaluating semantics, syntax, morphology, and pragmatics in children aged 5 to 21 years (Wiig et al., 2013). Other alternatives include the Comprehensive Assessment of Spoken Language–Second Edition (CASL-2; Carrow-Woolfolk, 2017), Test of Language Development, Intermediate–Fourth Edition (TOLD-I:4; Hammill & Newcomer, 2008), the Peabody Picture Vocabulary Test–Fourth Edition (PPVT-4; Dunn & Dunn, 2007), the Oral and Written Language Scales–Second Edition (OWLS-II; Carrow-Woolfolk, 2007), the Receptive One-Word Picture Vocabulary Test–Fourth Edition (ROWPVT-4; Martin & Brownell, 2011b), and the Expressive One-Word Picture Vocabulary Test–Fourth Edition (EOWPVT-4; Martin & Brownell, 2011a). All the tests are standardized and norm-referenced (Denman et al., 2017; Rehfeld & Padgett, 2019, Table 8; Community, 2011; Miles et al., 2016, Table 4).
Should deficits emerge through testing, a language disorder diagnosis requires that these deficits should be causing impairments/limitations in other areas of functioning, including but not limited to social functioning and/or academic achievement (Criterion B; APA, 2013). Parent and teacher reports of observation of child’s social interactions and school reports of grades can be useful for evaluation of this criterion.
Speech Sound Disorder
Speech Sound Disorder is characterized by problems with articulation (making sounds) and phonological processes (sound patterns) not typical for a child’s developmental level. Speech sound disorder constitutes about 70% of pediatric speech-language pathology cases and is the most prevalent of childhood communication difficulties (Dodd, 2014). Similar to other communication disorders, onset is typically during early childhood (Criterion C; APA, 2013). Typically developing children start demonstrating overall intelligible speech around the age of 3. While it is not uncommon for developing children to use immature phonological simplification processes such as shortening words and syllables as they are learning to talk, children with speech sound disorder continue to display these deficits past the age of 5 years, when most children can produce words clearly.
Any difficulties that may be congenital or acquired conditions must be ruled out (Criterion D) before diagnosing with speech sound disorder. Differential diagnosis for speech sound disorder should include normal variations in speech, hearing or sensory impairments, structural deficits, dysarthria, and selective mutism. Like language disorder, hearing impairments should be ruled out first. Developmental history should be considered and a thorough audiologic evaluation should be conducted as the first step. Consultation with the child’s physician, and in some cases an audiologist, is recommended. An oral peripheral examination may also be conducted to rule out speech impairments due to structural deficits such as cleft palate or cleft lip. Furthermore, consultations with the child’s physician are recommended to rule out speech impairments attributable to motor disorders, such as cerebral palsy. To exclude neurological conditions such as traumatic brain injury, developmental history should be considered. Anxiety disorders such as selective mutism should be assessed in the child as well. In cases where the child is exhibiting normal speech in familiar and safe settings, such as home or with close friends and people, selective mutism can be ruled out as primary.
After medical, psychological, and neurological impairment have been ruled out as better explanations for a child’s symptomatology (Criterion D; APA, 2013), the other criteria for speech sound disorder may be considered. The presence of difficulty with speech sound production, such as articulation and phonology (Criterion A; APA, 2013), and the role of these disturbances in speech in hindering effective communication (Criterion B; APA, 2013) should be assessed. Currently, only a few psychometrically strong standardized tests exist for the evaluation of speech sound disorder. The Arizona Articulation and Phonology Scale–Fourth Revision (Arizona-4) can be used to assess both articulation and phonology in individuals aged 18 months to 21 years. An alternative is the Comprehensive Test of Phonological Processes (CTOPP-2), which can be used to assess phonological processing for ages 4 to 6 years as well as 7 to 24 years (Wagner et al., 2013). In some cases, a referral to speech/language pathology may be the most appropriate course of action.
Childhood-Onset Fluency Disorder (Stuttering)
Stuttering is characterized by impairment in speech fluency or an interruption in the flow of speaking that is inappropriate for the individual’s age and language skills (APA, 2013). It involves significant problems with normal fluency and flow of speech. Individuals who stutter usually know what they want to say but have difficulty saying it. Symptom onset must occur in the early developmental period (Criterion C) to meet diagnostic criteria for childhood-onset. Age of onset usually ranges from 2 to 7 years, with most affected individuals demonstrating symptomatology by the age of 6 years in about 80% to 90% of cases. While stuttering is common among young children, most children outgrow developmental stuttering. If stuttering develops during or after adolescence, it is diagnosed as adult-onset fluency disorder and is not classified as neurodevelopmental disorder. Furthermore, unlike adult-onset stuttering, which is usually associated with neurological insults (e.g., stroke, tumor, trauma) and a variety of medical conditions, childhood-onset stuttering is not attributable to a speech-motor or sensory deficit, dysfluency associated with neurological insults or another medical condition, and is not better explained by another medical disorder (Criterion D; APA, 2013). Sensory deficits such as hearing impairment should be ruled out. In some cases, stuttering may develop as a side effect of medication, so medication history needs to be carefully evaluated. Some children may exhibit motor movements such as eye blinks, tremors of the face or lips, or jerking of the head, characteristic of Tourette’s disorder. Since Tourette’s disorder is characterized by sudden, repetitive, rapid, and unwanted movements or vocal sounds called tics, it should be easily distinguishable from the repetitive sounds observed in stuttering. Differential diagnosis should be performed through careful collection of developmental history, and in some cases, consultation with the child’s physician and neurologist.
Criterion A (disturbances in normal fluency and time patterning of speech) should be evaluated and distinguished from normal speech dysfluencies that occur in young children, such as whole-word or phrase repetitions. The Stuttering Severity Instrument, 4th edition (SSI-F; Riley, 2009) is recommended for this purpose. SSI-F is a reliable and valid norm-referenced assessment measuring stuttering severity in children at least 2 years of age and in adults (Kahani et al., 2021; Table 1). The SSI-F assesses frequency and duration of stuttering, physical concomitants, and the naturalness of the individual’s speech. An alternative is the Test of Childhood Stuttering (TOCS; Gillam et al., 2009), a standardized assessment which can be used for children aged 4 to 12 years, which evaluates speech disfluency in a variety of speaking contexts.
Psychometric Information of Validated and Reliable Communication Disorders Assessments
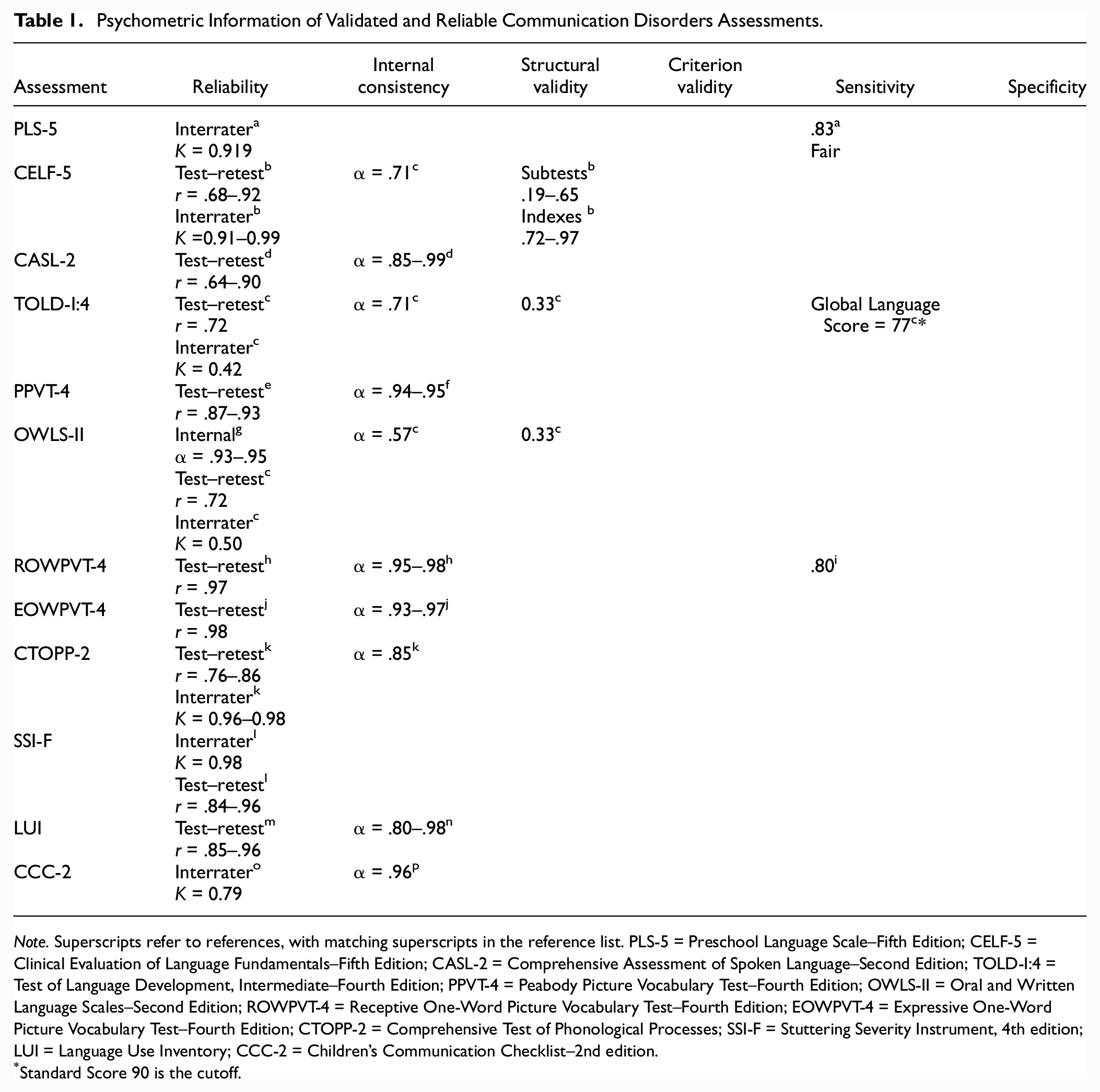
Note. Superscripts refer to references, with matching superscripts in the reference list. PLS-5 = Preschool Language Scale–Fifth Edition; CELF-5 = Clinical Evaluation of Language Fundamentals–Fifth Edition; CASL-2 = Comprehensive Assessment of Spoken Language–Second Edition; TOLD-I:4 = Test of Language Development, Intermediate–Fourth Edition; PPVT-4 = Peabody Picture Vocabulary Test–Fourth Edition; OWLS-II = Oral and Written Language Scales–Second Edition; ROWPVT-4 = Receptive One-Word Picture Vocabulary Test–Fourth Edition; EOWPVT-4 = Expressive One-Word Picture Vocabulary Test–Fourth Edition; CTOPP-2 = Comprehensive Test of Phonological Processes; SSI-F = Stuttering Severity Instrument, 4th edition; LUI = Language Use Inventory; CCC-2 = Children’s Communication Checklist–2nd edition.
Standard Score 90 is the cutoff.
Limitations in effective communicative and social functioning, including anxiety related to speaking (Criterion B; APA, 2013), should also be evaluated. It should be noted that while anxiety is considered a feature of stuttering, it can also worsen dysfluency. If the individual is suffering from anxiety, it is important to determine whether it is related to the dysfluency, so that others form of anxiety, such as social anxiety disorder, can be ruled out or treated concurrently.
Social (Pragmatic) Communication Disorder
SPCD is characterized by persistent difficulties in the use of verbal and nonverbal language for social purposes. This can lead to difficulties in social interaction, language processing, social understanding, speech pragmatics, or any combination of these. SPCD can result in a wide array of problems, including difficulty participating in social settings and developing relationships. SPCD shares symptomatology with ASD; thus, it is essential that primary diagnosis of SPCD makes careful differentiation from ASD, as well as from intellectual disability and other medical/neurological conditions (Criterion E; APA, 2013). To rule out the presence of intellectual disability, administration of a standardized IQ test (e.g., the Weschler Intelligence Scales for Children, 5th edition; WISC-V) may be warranted. If indicated, primary ASD symptomatology may first be evaluated with the Social Communication Questionnaire. In cases where the scores are above the cutoff (>15), a full evaluation is recommended using gold-standard instruments, such as the Autism Diagnostic Observation Schedule (ADOS-2) or Modified Checklist for Autism in Toddlers (M-CHAT-R). ADHD can also cause impairments in areas characteristic of SPCD. Assessment of ADHD should be considered before ruling out SPCD. Similarly, symptoms of social anxiety disorder also resemble the social deficits of SPCD. However, social anxiety disorder can be ruled out by assessing the timing of the onset of symptoms. In cases where social communication skills are not developed appropriately and are not related to anxiety or fear related to social interactions, social anxiety disorder can be excluded.
The onset of symptoms should be in the early developmental period (Criterion D; APA, 2013), which can be determined through the collection of comprehensive personal history from caregivers. Criterion A, persistent difficulties in the social use of verbal and nonverbal communication, can be evaluated using the Language Use Inventory (LUI; O’Neill, 2007) in toddlers (18–47 months of age). The Children’s Communication Checklist–2nd edition (CCC-2; Bishop, 2003) is useful for children aged 4 years and above. Both are norm-referenced instruments that are valid and reliable (Knowledge In Development Inc., n.d.; Wellnitz et al., 2021; Table 1). The CCC-2 is useful in assessing both pragmatic aspects of communication and common co-occurring behaviors. Alternatives include the Test of Problem Solving, 3rd edition (TOPS-3; Bowers et al., 2005), Test of Pragmatic Language, 2nd edition (TOPL-2; Phelps-Terasaki & Phelps-Gunn, 2007), and the Social Language Development Test (SLDT-Elementary; Bowers et al., 2008).
These social communication deficits should also cause significant psychosocial impairment to warrant a diagnosis of SPCD (Criterion B; APA, 2013). This may be seen in the form of ineffective communication with parents, teachers, and peers; low academic achievement; low social participation; and poor social competence. Parent and teacher reports of how the child behaves socially, as well as clinician-rated naturalistic observations, can also be useful to this end. The Early Social Communication Scales (ESCS; Mundy et al., 2003) is a videotaped structured assessment that provides individual differences in nonverbal communication skills of children aged 8 to 30 months. Similarly, the Communication and Symbolic Behavior Scale (CSBS; Wetherby et al., 1990) collects information on communication development through parent interview and direct observation in infants and toddlers (up to 72 months in age). In addition, self-report, parent, and teachers reports of social behavior such as the Clinical Assessment of Interpersonal Relations (CAIR; Bracken, 2006) or Home and Community Social Behavior Scale (HCSBS; Lund & Merrell, 2001) may be used to ascertain child’s social functioning. Evaluating adaptive functioning through the Adaptive Behavior Assessment System–Third Edition (ABAS-3) may also be useful in the assessment of SCPD.
Section 2: Motor Disorders
Assessment Overview
DSM-5 neurodevelopmental motor disorders include developmental coordination disorder, stereotypic movement disorder, and tic disorders (APA, 2013). While many children with motor disorders present to/are referred to occupational or physical therapy clinics directly, clinical psychologists may encounter motor disorders in a variety of clinical settings and should be able to confidently and independently evaluate their symptomatology.
For all DSM-5 motor disorders, it is critically important that both physiological and psychiatric rule-out is performed (Figure 2). Before evaluating candidacy for any primary movement disorder, it is essential that thorough neurological and physical examinations are performed to ensure that motor difficulties are not better explained by disorders such as cerebral palsy, muscular dystrophy, Huntington’s disease, post-viral encephalitis, or by other underlying systemic illnesses or neurological impairments (APA, 2013).
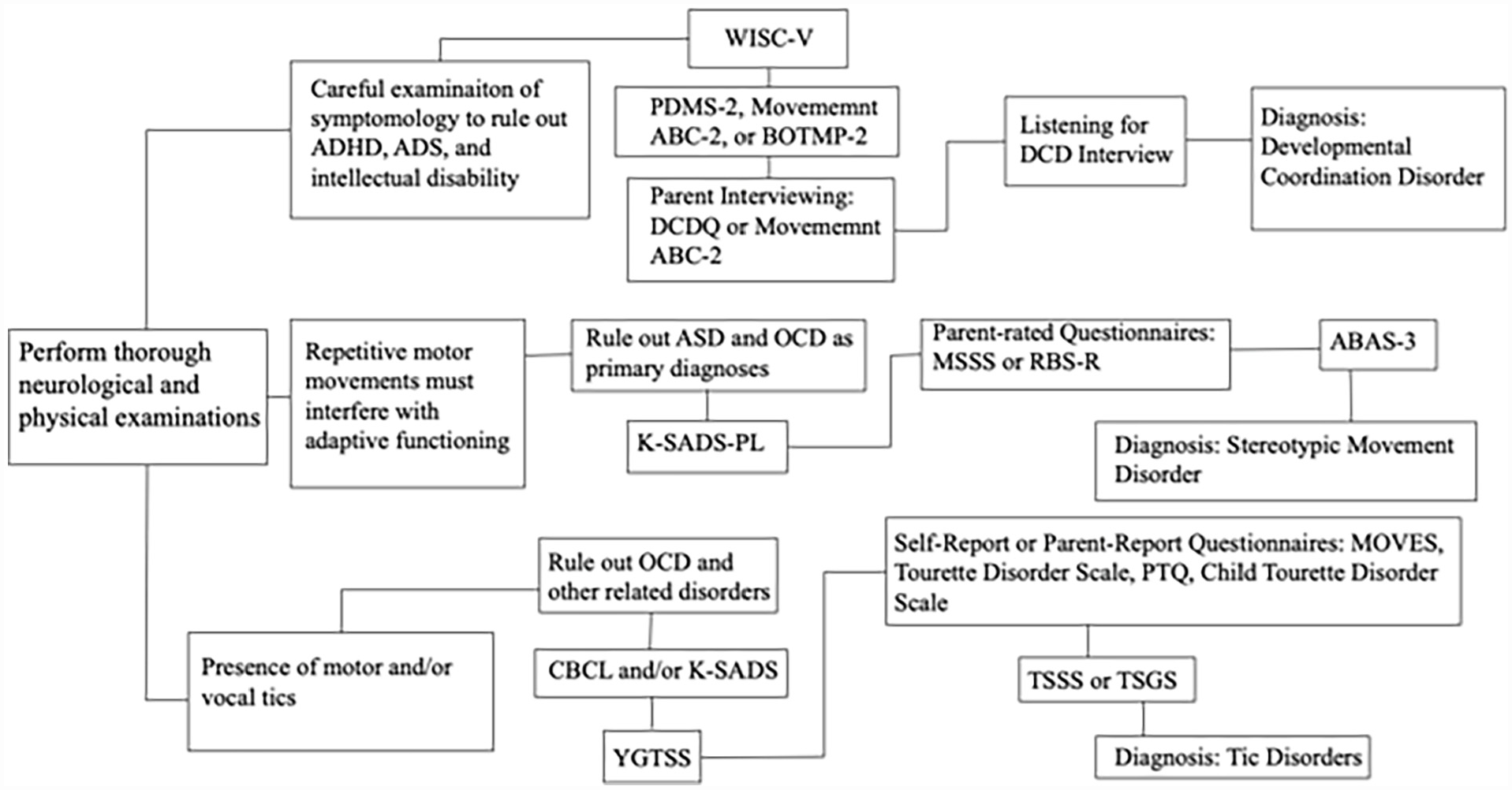
Flowchart of Motor Disorders Assessments.
Developmental Coordination Disorder
Developmental coordination disorder is characterized by deficient and/or delayed acquisition and execution of coordinated motor skills, resulting in motor skill performance that is clumsy, slow, and/or inaccurate. Motor incompetence must be sufficiently interfering that adaptive functioning is impacted. Symptom onset must occur in the early developmental period to meet diagnostic criteria. Developmental coordination disorder occurs in 5% to 6% of children aged 5 to 11 years and has a male:female ratio between 2 and 7:1 (APA, 2013). Differential diagnosis for developmental coordination disorder must include motor impairments due to another medical condition (visual impairment, specific neurological disorders) and joint hypermobility syndrome (APA, 2013; Woods et al., 2007). Although intellectual disability, ADHD, and ASD can and commonly do co-occur with developmental coordination disorder, careful examination of symptomatology is essential to determine the existence of a core motor incompetence (APA, 2013). The presence of primary intellectual disability may require administration of a standardized IQ test (e.g., WISC-V), though school achievement records may be sufficient to rule out intellectual disability in some children (Blank et al., 2012).
After neurological, visual, and intellectual impairment have been ruled out as primary explanations for a child’s symptomatology (criterion D), the other criteria for developmental coordination disorder may be considered. Criterion A (acquisition and execution of coordinated motor skills substantially below chronological age and manifested as motor clumsiness, slowness, and/or inaccuracy) may be evaluated with the Peabody Developmental Motor Scales, 2nd edition (PDMS-2; Folio & Fewell, 2000) in 0- to 5-year-old children, or with the Movement Assessment Battery for Children, 2nd edition (Movement ABC-2; Henderson et al., 2007) in children 3 to 16 years of age. An alternative test for school-aged children is the Bruininks-Oseretsky Test of Motor Proficiency, 2nd edition (BOTMP-2; Bruininks & Bruininks, 2005). All three are norm-referenced, reliable, valid, and sensitive enough to detect fine and gross motor difficulties related to Developmental Coordination Questionnaire (DCDQ; Table 2). They are also useful measures for evaluation of intervention efficacy.
Psychometric Information of Validated and Reliable Motor Disorder Assessments.

Note. Superscripts refer to references, with matching superscripts indicated in the reference list. PDMS-2 = Peabody Developmental Motor Scales, 2nd edition; Movement ABC-2 = Movement Assessment Battery for Children, 2nd edition; BOTMP-2 = Bruininks-Oseretsky Test of Motor Proficiency, 2nd edition; DCDQ = Developmental Coordination Questionnaire.
Interference in adaptive functioning (criterion B) should be evaluated through parent interviewing around the child’s competency in and timeline for performing developmentally appropriate motor tasks, such as tying shoelace, doing buttons, using cutlery, and handwriting (APA, 2013). Parent-rated questionnaires and checklists, such as the DCDQ (ages 5–15 years; Wilson et al., 2009) and/or the Movement ABC-2 checklist (above), are recommended.
Criterion C—onset in the early developmental period—can be evaluated through careful collection of developmental history. The clinician-rated Listening for Developmental Coordination Disorder Interview (Missiuna et al., 2006) can be helpful in evaluation of both criteria B and C. It should be noted that while some children with developmental coordination disorder may have or have had delayed motor milestones, many do not.
Stereotypic Movement Disorder
Stereotypic movement disorder is characterized by repetitive motor movements that seem driven and appear to be purposeless (APA, 2013). These rhythmic movements are commonly of the head, hands, or body. Stereotypic movements can be responsive to efforts to stop them (common among typically developing children) or less responsive (common among children with neurodevelopmental disorders). Some children will attempt to suppress the motor movements through self-restraining behaviors, such as sitting on their hands (APA, 2013). Stereotypic movements can occur multiple times per day or much less frequently and can last from several seconds to several minutes. To meet the criteria for stereotypic movement disorder, these behaviors must interfere with adaptive functioning.
While stereotyped movements are common in young, typically developing children, more complex stereotyped movements occur in only 3% to 4%. In individuals with intellectual disability, stereotypy is much more common (4%–16%; APA, 2013). While stereotypic movements usually begin in the very early developmental period, clinically significant symptoms may not be apparent until 3 years of age.
It is essential that stereotypic movement disorder symptoms be distinguished from the common, simple movements often observed in infancy and early childhood. Differential diagnosis must also rule out ASD. While stereotypic movement disorder can co-occur with ASD, a separate stereotypic movement disorder diagnosis is made only when stereotyped movements are self-injurious or sufficiently interfering that they must be a focus of treatment. Stereotypic movement disorder must additionally be distinguished from obsessive-compulsive disorder and related disorders, tic disorders, and certain medical and neurological conditions (APA, 2013).
Evaluation of stereotypic movement disorder must begin with a thorough medical and neurological examination to rule out underlying causes, as well as a comprehensive clinical psychiatric interview, such as the Kiddie Schedule for Affective Disorders and Schizophrenia (K-SADS-P/L; Kaufman et al., 1997), to rule out psychiatric and neurodevelopmental conditions which may better explain the symptomatology (criterion D). If indicated, stereotyped movements can be evaluated with parent-rated questionnaires, such as the Motor Severity Stereotypy Scale (MSSS; Harris et al., 2015), or in children with ASD, the Repetitive Behavior Scale, Revised (RBS-R; Di Renzo et al., 2017). These questionnaires can aid in evaluation of frequency, intensity, and interference of motor behaviors (criteria A and B) and may be useful measures for evaluation of treatment efficacy. A measure of adaptive functioning (e.g., ABAS-3) may also be useful in evaluating criterion B interference. Criterion C—onset in the early developmental period—should be evaluated through careful collection of developmental history; motor stereotypies should be evidenced in early childhood. If Criteria A to D are met, the stereotypic movement disorder diagnosis must be specified as with or without self-injurious behavior, and if warranted, specified as with a known medical or genetic condition, neurodevelopmental disorder, or environmental factor (APA, 2013). Severity is also coded and is determined by the ease of suppressing the movements and degree of monitoring required to prevent serious injury (APA, 2013).
Tic Disorders
Diagnosis of all tic disorders is based on the presence of motor and/or vocal tics—sudden, rapid, recurrent, and nonrhythmic motor movements or vocalizations (APA, 2013). Tic disorders are diagnostically categorized in a three-fold manner: (a) Tourette’s disorder is the most severe type, appearing in 3% to 8% of school-aged children; (b) persistent motor or vocal tic disorder, with symptomatology less severe than in Tourette’s disorder; and (c) provisional tic disorder, in which symptomatology is less severe than in persistent tic disorder. These diagnoses are hierarchical, such that a prior diagnosis at a high-level category (e.g., Tourette’s) precludes later diagnosis of a low-level category (e.g., persistent tic disorder). Onset of tic behavior is typically observed in kindergarten-aged children (4–6 years of age) and tends to peak in severity between ages 10 and 12 years. While tic disorders can persist into adolescence and adulthood, symptoms are typically diminished, though some adults experience chronic, severe tics (APA, 2013).
Evaluation of tic disorders must begin with thorough medical and neurological examination to rule out medical conditions in which abnormal motor movements may be observed (e.g., chorea, dystonia, substance-induced or paroxysmal dyskinesias, myoclonus; APA, 2013). Rule-out of stereotypic movement disorder is also essential and may be differentiated from tic disorders by its earlier age of onset and by the fixed, rhythmic, and predictable nature of its motor stereotypies. Stereo symptoms are also distinguished from those of tic disorders in their lack of premonitory urge and, in many, their cessation with redirection/distraction. Administration of the MSSS and/or RBS-R may be useful in making this differential. Other psychiatric conditions, such as obsessive-compulsive disorder and related disorders, should be ruled out using screening tools (e.g, the Child Behavior Checklist, and reviewed in other articles), and if necessary, with a clinical interview, such as the K-SADS.
After other disorders have been ruled out (criterion D in all tic categories), and if onset of symptoms occurred before age 18 years (criterion C in all tic categories), criteria A and B may be evaluated. Criterion A differentiates categories through the nature of their tics. Tourette’s disorder requires that multiple motor tics and at least one vocal tic are present, though not necessarily concurrent, at some point since symptom onset. If only one type of tic (i.e., motor or vocal) is present, candidacy for a diagnosis of persistent chronic motor or vocal tic disorder should be evaluated. The nature of the tic(s)’ frequency, complexity, and intensity, as well as the level of distress experienced by the child, can be evaluated with the Yale Global Tic Severity Scale (YGTSS; Leckman et al., 1989), a brief clinical interview. Several useful self-report or parent-report questionnaires are also available for the evaluation of criterion A, including the Motor Tic, Obsessions and Compulsions, Vocal Tic Evaluation Survey (MOVES; Gaffney et al., 1994); the Tourette Disorder Scale (Shytle et al., 2016); the Parent Tic Questionnaire (PTQ; Chang et al., 2009); and the Child Tourette Disorder Impairment Scale (Storch et al., 2007). Clinician-rated scales such as the Tourette Syndrome Severity Scale (TSSS) and Tourette Syndrome Global Scale (TSGS; Storch et al., 2007) can also be useful.
Criterion B distinguishes tic categories by the timeline of its symptomatology. Both Tourette’s disorder and persistent motor or vocal tic disorder require that one or more of the tics have persisted for at least 1 year since symptom onset, though they may wax and wane in frequency. If the tics—motor, vocal, or both—have been present for less than 1 year, provisional tic disorder is diagnosed. Criterion E for both persistent and provisional tic disorder requires, as previously mentioned, that the child has never met the criteria for Tourette’s disorder, and in provisional tic disorder, has never met the criteria for persistent tic disorder (APA, 2013). Criteria B and E should be easily determined during the course of clinical interviewing.
Tic disorder specifiers are only required for persistent tic disorder, in which it should be specified if the tics are motor only or vocal only. An “other specified” or “unspecified” tic disorder diagnosis may be appropriate in children who do not meet the full diagnostic criteria for any of the three tic disorders (APA, 2013).
Section 3: Feeding Disorders
Assessment Overview
DSM-5 feeding disorders include pica, rumination disorder, and avoidant/restrictive food intake disorder (ARFID). Feeding disorders are distinguished from eating disorders in their lack of relation to concerns about weight and/or body shape (APA, 2013). Although eating and feeding disorders can co-occur, differential diagnosis is critical for all feeding disorders (Figure 3), particularly ARFID, in which ARFID symptomatology must be carefully distinguished from that of anorexia nervosa (Seetharaman & Fields, 2020). It should be noted that while a diagnostic assessment measure for DSM-5 feeding and eating disorders exists (i.e., the Eating Disorder Assessment for DSM-5 [EDA-5]; Sysko et al., 2015), its psychometric strengths have only been demonstrated in diagnosis of eating disorders, and it is not a psychometrically sound measure for evaluation of feeding disorders. However, the relatively recent, semi-structured, multi-informant clinical interview, the Pica, ARFID, and Rumination Disorder Interview (PARDI; Bryant-Waugh et al., 2019), has been validated for use in feeding disorder diagnosis (Table 3). The PARDI is available as self-report in youths and adults 14 years of age and older and as parent report in children 4 years of age and older and can be useful in the diagnostic process for all feeding disorders. It is often useful to consult with a nutritionist or pediatrician when evaluating these disorders.
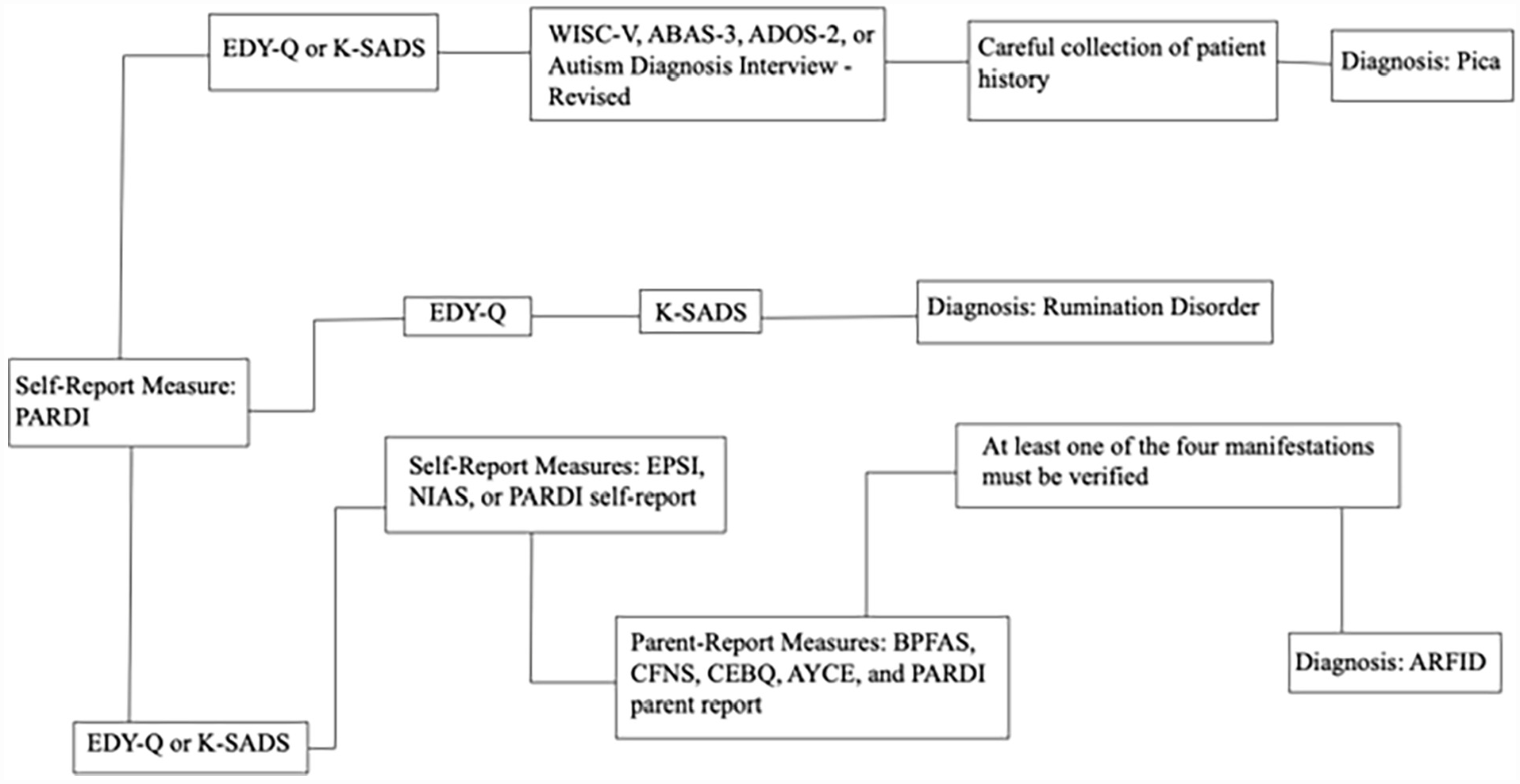
Flowchart of Feeding Disorders Assessments.
Psychometric Information of Validated and Reliable Feeding Disorders Assessments

Note. Superscripts refer to references, with matching superscripts indicated in the reference list. PARDI = Pica, ARFID, and Rumination Disorder Interview.
Pica
Pica is characterized primarily by the consumption of nonnutritive, nonfood substances at a level inappropriate for the child’s developmental level (APA, 2013). Although childhood onset is typical, pica may onset at any point in development, including in adulthood. It should be noted that pica is not typically diagnosed until at least 2 years of age, as mouthing of nonfood items—potentially resulting in ingestion—is common in infancy (APA, 2013). It should also be noted that pica is not diagnosed in pregnant women unless the nonnutritive, nonfood substances consumed pose potential medical risks. Differential diagnosis of pica in youth and in adulthood must rule out anorexia nervosa, in which nonnutritive substances (e.g., tissues) may be consumed in attempts to control appetite (APA, 2013). Candidacy for anorexia nervosa may be evaluated with an eating disorder–specific measure, such as the Eating Disorders in Youth Questionnaire (EDY-Q; Hilbert & van Dyck, 2016), or on a broader clinical interview, such as the K-SADS. Pica should also not be diagnosed if the consumption of nonnutritive substances is related to self-injury, as observed in some neurodevelopmental, mood, and personality disorders. If the eating behavior occurs in the context of another psychiatric condition (e.g., intellectual disability, ASD, schizophrenia), pica is not additionally diagnosed unless the symptomatology is severe enough that specific clinical attention is required (criterion D; APA, 2013). A clinical interview, such as the K-SADS, should typically be administered to rule in or rule out the candidacy of primary or co-occurring psychiatric and neurodevelopmental conditions. Domain-specific testing, such as IQ testing (WISC-5), adaptive function testing (ABAS-3), and autism diagnostics (Autism Diagnostic Observation Schedule, 2nd edition, ADOS-2 [Lord et al., 2012]; Autism Diagnostic Interview, Revised, ADI-R [Rutter, LeCouteur, & Lord, 2003]; Modified Checklist for Autism in Toddlers, M-CHAT-R [Robins, Fein, & Barton, 2009]) may be warranted in more complicated cases. Pica is also not diagnosed if the eating behavior is related to a culturally supported or socially normative practice (criterion C). Information related to this may be gathered during standard collection of demographic information/developmental history or can be specifically queried during other assessment proceedings.
If criteria C and D are met, the nature of the eating behavior may be evaluated. Developmental history must be carefully collected to determine the type of substance(s) consumed and the duration of the eating behavior. Parent or self-report on the substances consumed is essential for verification of their nonnutritive nature and to determine that the eating behavior has occurred for at least 1 month (criterion A). In some instances, referral to a physician for a stool sample may be warranted. If the consumed substances have the potential to cause intestinal blockages and/or toxic side effects (e.g., lead paint, bacteria/parasites from dirt), referral for medical examination is also essential (APA, 2013). Children aged older than 2 years, as previously discussed, is typically sufficient for determination of the impropriety of the eating behavior given developmental level (criterion B). Although limited assessment instruments are available for the evaluation of pica symptoms specifically, the PARDI contains questions that map onto pica’s diagnostic criteria. The PARDI yields both a diagnostic algorithm score and severity score. At this time, the PARDI is the best available clinical instrument for assessment and diagnosis of pica, when combined with medical evaluation, rule-out/comorbidity testing via gold-standard clinical interview and diagnostic instruments (e.g., K-SADS, WISC-V, ADOS-2) when warranted, and careful collection of developmental history.
Pica can be specified as “in remission” if, after formerly meeting full diagnostic criteria, the criteria have not been met for a “sustained” amount of time. DSM-5 does not designate a specific amount of time required to consider the disorder “in remission” and remains an area warranting further research.
Rumination Disorder
Rumination disorder is characterized by the repeated regurgitation of food after feeding or eating, with symptoms present for at least 1 month. Regurgitation must occur in the absence of apparent nausea, involuntary retching, or disgust. Regurgitated food may be ejected from the mouth or swallowed. Regurgitation occurs at least several times per week and commonly occurs daily. While rumination disorder can be diagnosed at any point in development, including in adulthood, infant rumination typically onsets between 3 and 12 months of age. In infancy, rumination can result in serious medical conditions, such as malnutrition, and has the potential to pose a fatal choking hazard (APA, 2013).
Before psychological assessment, medical evaluation is typically warranted—especially in young children—to rule out underlying gastrointestinal or other medical problems and to evaluate any physiological problems that rumination symptoms may be causing. If medical causes are ruled out (criterion B), it is essential that symptoms of rumination disorder be distinguished from those of anorexia nervosa, bulimia nervosa, binge-eating disorder, or ARFID (criterion C). As mentioned, feeding disorders, such as rumination disorder, are characterized by symptomatology not related to weight and/or body shape. To evaluate for the presence of a co-occurring or primary eating disorder, an eating disorder–specific assessment measure may be necessary. The EDY-Q is recommended for typically developing youth. The K-SADS may also be used to evaluate eating disorder symptoms and to either rule out co-occurring disorders or ensure that rumination symptoms do not only occur during the course of the comorbid disorder. As in pica, rumination disorder-specific assessment measures are not available; however, the PARDI can be used both to distinguish rumination from ARFID and to evaluate candidacy for a rumination disorder diagnosis. The PARDI yields a diagnostic algorithm score and severity score for rumination symptoms and is the best available assessment instrument for rumination disorder at this time, when used in combination with careful differential diagnostic proceedings.
Avoidant/Restrictive Food Intake Disorder
ARFID is primarily characterized by avoidance or restriction of food intake that is severe enough to cause clinically significant deficits in nutrition or energy through oral intake of food (APA, 2013). ARFID is distinct from eating disorders, such as anorexia nervosa, in that eating avoidance or restriction is related to lack of interest in food, aversion to sensory characteristics of food, or fear of consequences of eating (e.g., vomiting, choking). ARFID is not related to concerns about weight and body shape.
Initial screening of ARFID symptoms should aim to rule out the primary role of anorexia nervosa or bulimia nervosa symptomatology (criterion C). As in other feeding disorders, the EDY-Q or K-SADS can be used to evaluate eating disorder symptoms. Self-report questionnaire measures such as the Eating Pathology Symptoms Inventory (EPSI; Forbush et al., 2013), Nine-Item ARFID Screen (NIAS; Zickgraf & Ellis, 2018), and PARDI self-report can and should be employed to differentiate symptoms of ARFID and anorexia/bulimia in older youth and adults. This step is essential, as physical and health consequences of both ARFID and anorexia nervosa symptomatology can be similar. Although it is possible to diagnose anorexia nervosa and ARFID concurrently, it is imperative that ARFID symptomatology does not occur exclusively during the course of anorexia. If it is clear that weight and/or body shape are not a concern, the NIAS alone can be used as an initial screening measure for ARFID in adolescents and adults.
Rule-out of underlying medical causes is also essential (criterion D). Many neurological/neuromuscular, structural, congenital disorders and conditions, gastrointestinal diseases, and food allergies/intolerances can produce feeding difficulties and aversions. Physiological dysfunction of oral/esophageal/pharyngeal structures, or other physical symptoms of a medical condition (e.g., nausea), should not be the primary cause of limited food intake (APA, 2013). However, if all diagnostic criteria for ARFID are met, ARFID can be diagnosed concurrently. Ongoing consultation with the child’s physician is often required in the evaluation of avoidant/restrictive feeding symptomatology, both in determining etiological causes of the symptoms and in evaluating other diagnostic criteria (i.e., criterion A; below). After ruling out the primary role of medical or other psychiatric conditions, it should be verified that the eating behavior is not due to a lack of available food or related to cultural practices, such as fasting (criterion B). This should be determined through sensitive clinical interviewing, as it is left to the assessor’s judgment on the PARDI.
In the absence of serious/complicating medical or psychiatric conditions, several screening measures for ARFID in younger children exist. Parent-report measures, such as the Behavioral Pediatrics Feeding Assessment Scale (BPFAS; Crist et al., 1994), Child Food Neophobia Scale (CFNS; Pliner, 1994), Child Eating Behavior Questionnaire (CEBQ; Wardle et al., 2001), About Your Child’s Eating (AYCE; Davies et al., 2007; Hendy et al., 2018), and PARDI parent report, can be used to initially evaluate young children’s eating symptomatology and screen for ARFID symptoms. If screening, broadband clinical interviewing, and/or medical consultation suggest the existence of a primary feeding disturbance associated with inadequate nutrition/energy intake (criterion A), at least one of four manifestations must be verified. The first manifestation is clinically significant weight loss, or in children, failure to meet expected weight trajectories/faltering growth. Judgment about the clinical significance of weight loss/suboptimal growth typically should be rendered by the child’s physician. The second manifestation is significant nutritional deficiency, which also requires physician consultation and commonly involves laboratory testing (e.g., for anemia; vitamin B deficiencies). The third manifestation is dependence on enteral feeding or oral nutritional supplements, in which “dependence” means that supplementary feeding (e.g., nasogastric tube feeding, gastrostomy tube feeding, nutritionally complete oral supplements) is required to maintain adequate nutritional intake. Self/parent report on supplemental feeding may be adequate to verify dependence in some cases but will require physician consultation in others. The final manifestation is significant interference with psychosocial functioning. In children, this is commonly evidenced by difficulty or inability to eat lunch in the cafeteria at school, inability to eat meals at friends’ houses, inability to eat items that people other than their own caregivers have prepared, or difficulty finding things to eat on vacation, sometimes so much so that vacations are impossible. In adolescents/adults, psychosocial interference can present similarly, but often includes interference with relationships. The presence of this manifestation can typically be determined through clinical interviewing and through reference of relevant items on the PARDI.
Important Future Directions for Research
At this time, no pica-specific or rumination disorder-specific screening or questionnaire measures exist. Initial identification of pica and/or rumination disorder candidates through assessment measures is only possible through single-item questions on measures such as the EDY-Q or, in individuals with intellectual disability, the Screening Tool of Feeding Problems (STEP, STEP-child; Matson & Kuhn, 2001; Seiverling et al., 2011). Neither pica nor rumination disorder is included in the K-SADS screen interview. There are also no identified measures recommended for the evaluation of treatment efficacy, although the PARDI may be useful as an outcome measure. As both pica and rumination disorder can be severely impairing, including medically, it is critical that future research efforts be directed toward development of screening and follow-up tools.
Section 4: Sleeping Disorders
Assessment Overview
DSM-5 sleep-wake disorders include insomnia disorder, hypersomnolence disorder, narcolepsy, breathing-related sleep disorders, circadian rhythm sleep-wake disorders, and parasomnias, which include non-rapid eye movement (REM) sleep arousal disorder, nightmare disorder, REM sleep behavior disorder, and restless leg syndrome (RLS; APA, 2013). While many children with sleep disorders present to sleep specialists or pediatricians directly, clinical psychologists may encounter sleep disorders in a variety of settings and should know which disorders they can evaluate and how to independently assess their symptomatology.
For all DSM-5 sleep disorders, it is critically important that both physiological and neurological tests are performed by the child’s pediatrician (Figure 4). To evaluate sleep disorders, it is essential to understand the co-occurring physical and neurological conditions, as these conditions are the rule, not the exception (APA, 2013). These comorbid disorders include neurodegenerative disorders, disorders of the musculoskeletal system, and disorders of the heart and lungs, which not only disturb sleep, but are themselves worsened during sleep (APA, 2013).
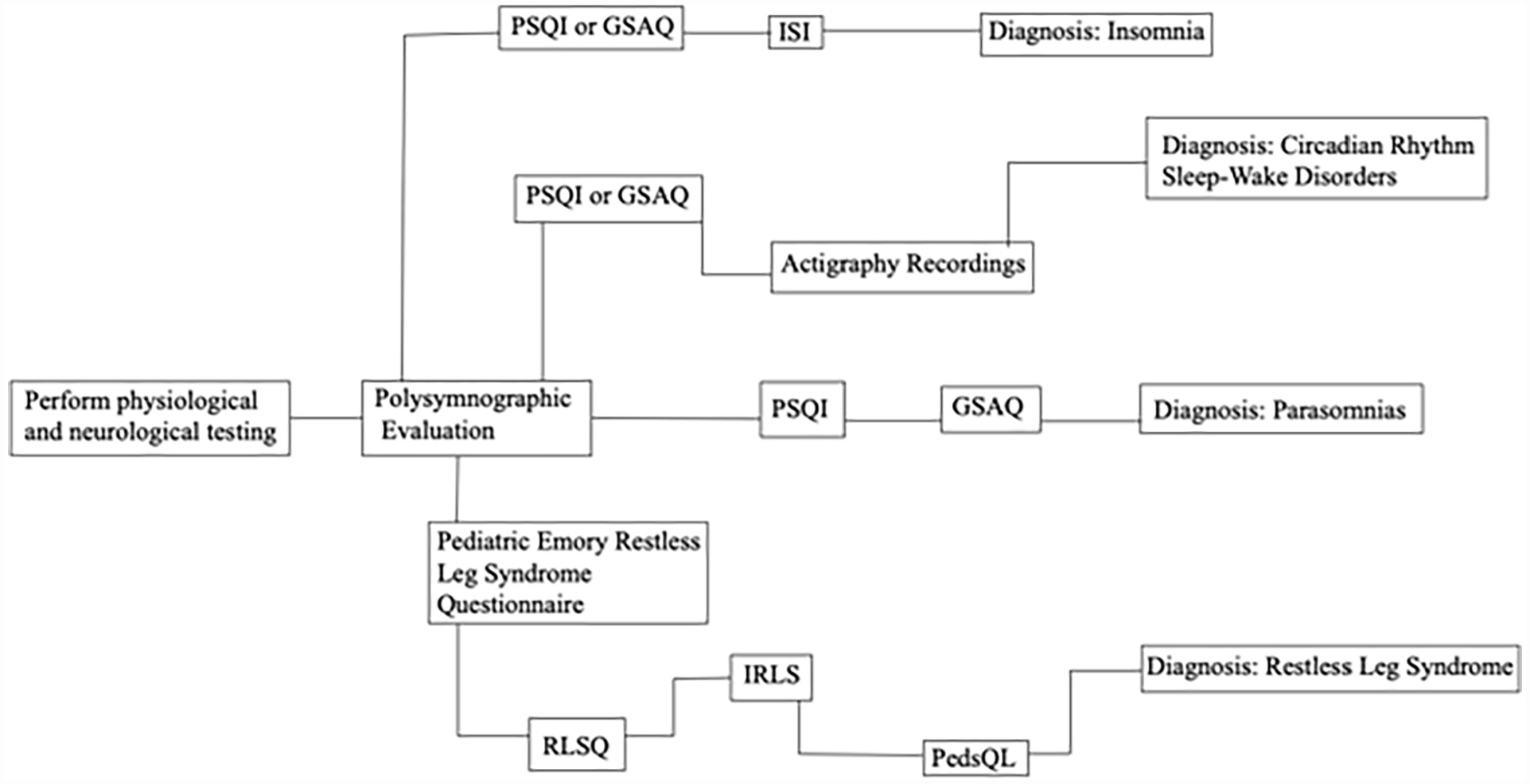
Flowchart of Sleeping Disorders Assessments.
Some specific DSM-5 sleep disorders require polysomnography, a multi-parameter diagnostic tool in sleep research, and must be assessed independently by sleep specialists. Disorders including narcolepsy, obstructive sleep apnea syndrome, REM sleep behavior disorder, non-REM sleep arousal disorders, central sleep apnea, sleep-related hypoventilation, and medication-induced sleep disorder may require polysomnographic evaluation (Yu & Avidan, 2022). Referral to a sleep specialist is also necessary in certain cases of other childhood sleep-wake disorders, including insomnia caused by a child stalling to get in to bed due to leg pain; RLS, which diagnostically requires demonstration periodic limb movements of sleep and elevated arousal indices; and other neurophysiological sleep abnormalities (Yu & Avidan, 2022). A multidisciplinary team including a sleep specialist would be beneficial for identifying potential triggers of parasomnias, but referral is not required (Yu & Avidan, 2022).
Parasomnia
Parasomnias are disorders characterized by abnormal behavioral, experiential, or physiological events occurring in association with sleep, specific sleep stages, or sleep-wake transitions (APA, 2013). As many as 84% of children aged 2 to 6 years experience parasomnias (Yu & Avidan, 2022). The most common parasomnias fall into the categories of non-REM sleep arousal disorders and REM sleep behavior disorders (APA, 2013). Non-REM sleep arousal disorders include sleepwalking; sleep terrors or nightmare disorder; sleep-related eating; and sleep-related sexual behavior. REM sleep behavior disorder includes dream-enactment behaviors that emerge during a loss of REM sleep atonia (APA, 2013). Pediatric parasomnias commonly present as nightmare disorder, a non-REM sleep arousal disorder. Differential diagnosis of nightmare disorder must specifically rule out breathing-related sleep disorders, sleep-related seizures, narcolepsy, panic disorder, and sleep-related dissociative disorders (APA, 2013). In children with parasomnias, elevated scores of depression and anxiety on personality indices are common (APA, 2013).
For non-REM sleep arousal disorders, criterion A (recurrent episodes of incomplete awakening from sleep, accompanied by either sleepwalking or sleep terrors), criterion B (no dream imagery recalled), and criterion C (amnesia for the episodes present) can be evaluated on a global sleep assessment, such as the Pittsburgh Sleep Quality Index (PSQI; Buysse et al., 1989). An alternative global measure of sleep disturbance is the Global Sleep Assessment Questionnaire (GSAQ; Roth et al., 2002). Both the scales are reliable, valid, and sensitive for measuring the type of non-REM sleep arousal disorder (Buysse et al., 1989; Roth et al., 2002; Table 4).
Psychometric Information of Validated and Reliable Sleeping Disorders Assessments

Note. Superscripts refer to references, with matching superscripts indicated in the reference list. PSQI = Pittsburgh Sleep Quality Index; GSAQ = Global Sleep Assessment Questionnaire; ISI = Insomnia Severity Index.
For REM sleep behavior disorder, criterion A (repeated episodes of arousal during sleep associated with vocalization and/or complex motor behaviors), criterion B (behaviors arise during REM sleep; i.e., 90 minutes after sleep onset), criterion C (upon awakening from these episodes, the individual is completely awake), and criterion D (REM sleep without atonia or a history suggestive of REM sleep behavior disorder) can be evaluated using a global sleep assessment like the PSQI or GSAQ, as parasomnias are diagnosed based on clinical sleep history (Klingman et al., 2017). REM sleep behavior disorder may be best assessed by a multidisciplinary team including a sleep specialist to specifically determine the stage of sleep the episodes are occurring in, as well as to determine if atonia is present during REM sleep (APA, 2013).
Insomnia
Another relatively common sleep-wake disorder in children is insomnia. Insomnia is characterized by difficulties falling asleep, staying asleep, or getting good quality of sleep (APA, 2013). Differential diagnosis of insomnia specifically must rule out delayed sleep phase and shift work types of circadian rhythm sleep-wake disorder, RLS, breathing-related sleep disorders, narcolepsy, parasomnias, and substance/medication-induced sleep disorder (APA, 2013). Some of these disorders are best assessed via polysomnography (narcolepsy, breathing-related sleep disorders) by a sleep specialist, and some can be assessed in clinical settings using global sleep assessments like the PSQI and GSAQ (circadian rhythm sleep-wake disorder, parasomnias) (Klingman et al., 2017). Insomnia is a common symptom of many medical conditions, including chronic pain, cancer, diabetes, heart disease, asthma, gastroesophageal reflux disease, overactive thyroid, Parkinson’s disease, and Alzheimer’s disease (Mayo Clinic, n.d.). Insomnia in the presence of these medical conditions can sometimes be better explained by the symptoms of such medical conditions, though can occur comorbidly. Primary insomnia is also commonly comorbid with depressive and anxiety disorders, with the risk relationship appearing bidirectionally (APA, 2013). In these cases, insomnia itself should be assessed and treated as a primary disorder.
After medical and psychological disorders have been ruled out as primary explanations for a child’s symptomatology (criterion F, G, H), the other criteria for insomnia may be considered. Criterion A (predominant complaint of dissatisfaction with sleep quantity or quality) and criterion B (sleep disturbances cause significant distress or impairment) can be evaluated using a global assessment of sleep, like the PSQI or GSAQ (above). Both criteria A and B, along with Criterion C (difficulty occurs at least 3 nights a week), criterion D (difficulty is present for at least 3 months), and criterion E (difficulty occurs despite adequate opportunity for sleep), can be evaluated on the Insomnia Severity Index (ISI; Morin et al., 2011), a specific scale designed to assess the nature, severity, and impact of insomnia. The ISI is a reliable and valid instrument to detect cases of insomnia in children and is also sensitive in monitoring treatment response in children (Morin et al., 2011; Table 4). Along with the questionnaires, children are encouraged to keep a sleep log to track the amount, quality, and frequency of nighttime awakening each night (Yu & Avidan, 2022).
Restless Leg Syndrome
RLS is a sleep-related movement disorder characterized by a desire to move the legs, accompanied by uncomfortable sensations, and occurring primarily during the night (APA, 2013). RLS is seen in about 2% of children (Yu & Avidan, 2022). Along with disturbed sleep, common complaints of RLS children include daytime sleepiness and fatigue. Differential diagnosis of RLS must specifically include leg cramps, positional discomfort, arthritis, myalgia, positional ischemia (numbness), leg edema, peripheral neuropathy, radiculopathy, and habitual foot tapping (APA, 2013). RLS is best assessed via a multidisciplinary team, including a sleep specialist, to demonstrate periodic limb movements of sleep, elevated arousal indices, and other neurophysiological sleep abnormalities, but can be assessed in mental health settings using both global sleep scales and RLS-specific scales (Stubbs & Walters, 2020). Depressive disorders and anxiety disorders are commonly comorbid with RLS (Becker & Novak, 2014). RLS is one of the leading causes of insomnia, with sleep disturbance being one of the primary reasons for seeking assessment (Becker & Novak, 2014). RLS has also been extensively associated with cardiovascular disease, via associations with vascular risk factors such as hypertension, diabetes, and obesity. Sympathetic hyperactivity may be a primary explanation in the increased risk for hypertension and cardiovascular disease among individuals with RLS (Becker & Novak, 2014).
After ruling out specific medical conditions and behavioral conditions (criterion D and E), the other criteria for RLS can be considered. Criterion A (an urge to move the legs, accompanied by uncomfortable sensations in the legs) and criterion B (symptoms occur at least 3 times per week and have persisted for at least 2 months) can be evaluated using the Pediatric Emory Restless Leg Syndrome Questionnaire (Riar et al., 2013), a diagnostic questionnaire based on guidelines for RLS diagnostic criteria; the Restless Leg Syndrome Questionnaire (RLSQ; Evans & Blunden, 2011), a parent-report questionnaire developed for the identification of pediatric RLS; or the International Restless Leg Scale (IRLS; Walters et al., 2003), on which the child rates the severity of RLS symptomatology. All three scales have not yet been validated but have been used in multiple separate studies of pediatric RLS. The IRLS has shown ease of administration in one study and has also been utilized in multiple studies of childhood and adolescent RLS, making it the best-studied scale (Stubbs & Walters, 2020).
Criterion C (symptoms are accompanied by significant distress or impairment) may be assessed using a pediatric quality-of-life questionnaire, such as the Peds Quality of Life Inventory (PedsQL; Silva et al., 2014), a self-report measure that evaluates functioning across multiple environments, including sleep; the PedsOL has been used in multiple childhood and adolescent studies of RLS (Stubbs & Walters, 2020).
Circadian Rhythm Sleep-Wake Disorders
Circadian rhythm sleep-wake disorders are a consequence of misalignment of the circadian rhythm and desired sleep-wake schedule (APA, 2013). The most common circadian rhythm sleep-wake disorder in childhood is delayed sleep-wake phase disorder (Yu & Avidan, 2022). Differential diagnosis of delayed sleep-wake phase disorder must specifically include normal variations in sleep and other sleep disorders. Circadian rhythm sleep-wake disorders are commonly comorbid with depression, personality disorders, and illness anxiety disorder (APA, 2013).
Criterion A (persistent and recurrent pattern of sleep disruption primarily due to an alteration of the circadian system), criterion B (sleep disruption leads to excessive sleepiness or insomnia), and criterion C (sleep disturbances cause clinically significant distress or impairment) may be evaluated using a global sleep scale, such as the PSQI or the GSAQ, discussed prior. Children are also encouraged to keep a sleep log to assess their sleep patterns (Yu & Avidan, 2022). Assessment s may also benefit from actigraphy recordings, which measure sleep parameters and average motor activity via a wristwatch-like device. Actigraphy is more accurate than self-reported sleep duration and may be more useful than sleep diaries in the assessment of pediatric circadian rhythm sleep-wake disorders (Yu & Avidan, 2022).
Section 5: Elimination Disorders
DSM-5 elimination disorders include enuresis, which is repeated urination in inappropriate places; and encopresis, the passage of feces in inappropriate places (APA, 2013). While many children with elimination disorders present to/are referred to pediatricians or urologists directly, clinical psychologists may encounter elimination disorders as primary or secondary diagnoses across clinical settings. It is best practice for a multidisciplinary team including both a pediatrician or urologist and a psychologist to be involved in the diagnosis of elimination disorders (von Gontard, 2013).
For all DSM-5 elimination disorders, it is critically important that a physical and neurological rule-out is performed by a primary care provider (Figure 5). Before a psychologist can evaluate for a primary elimination disorder, it is essential to determine that elimination difficulties are not better explained by organic causes such as a neurogenic bladder, medical conditions that cause polyuria or urgency, urinary tract infections, chronic diarrhea, spina bifida, or anal stenosis (APA, 2013).
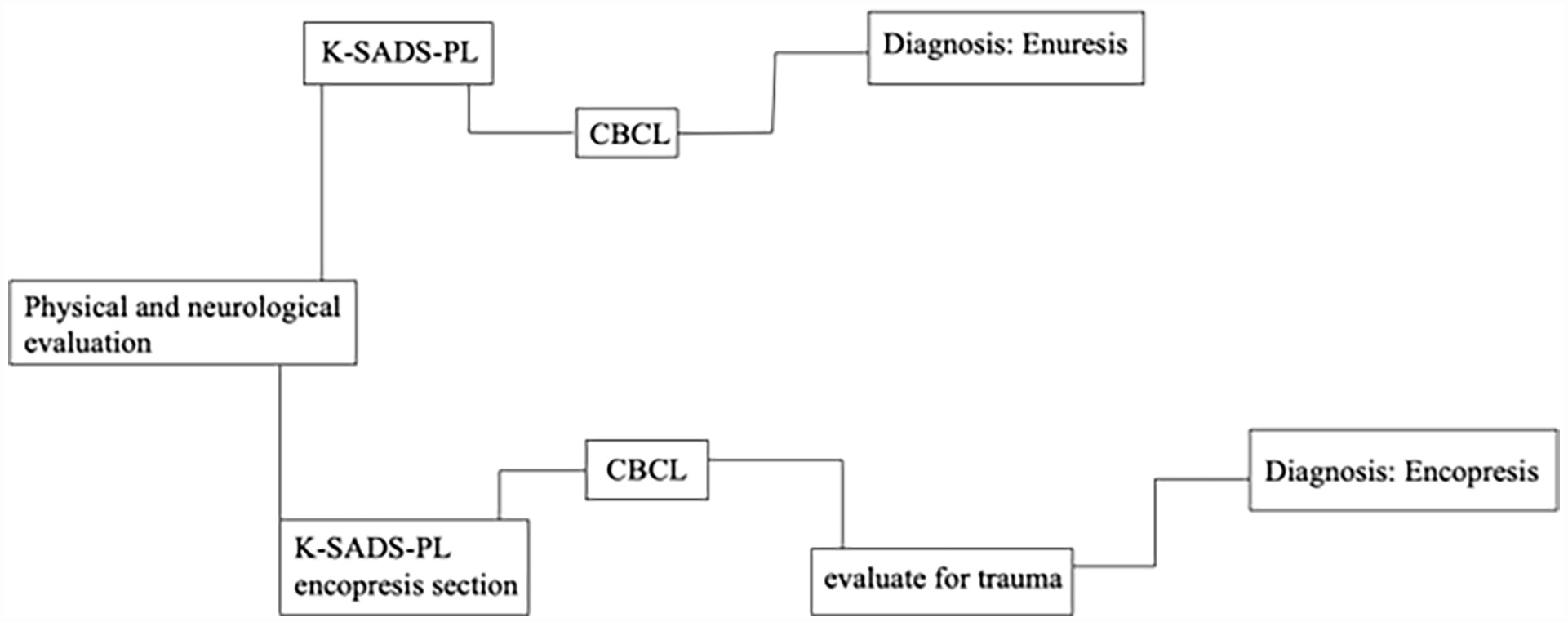
Flowchart of Elimination Disorders Assessments.
Enuresis
Enuresis is the voiding of urine into either the bed or clothes and can be voluntary or involuntary (APA, 2013). Voiding of urine specifically during the day is also known as urinary incontinence, while voiding of urine specifically during nighttime sleep is known as enuresis. Enuresis is significant when inappropriate urination occurs at least twice a week for 3 consecutive months and the child is at least 5 years of age (or the equivalent developmental level). The prevalence of enuresis is 5% to 10% among 5-year-olds and 3% to 5% of 10-year-olds. Differential diagnosis for enuresis must include neurogenic bladder or another medical condition and medication side effects. Children with enuresis commonly present with behavior problems and with the spectrum of developmental delays (APA, 2013).
After ruling out specific medical conditions that could cause elimination problems (criterion D), enuresis can be evaluated by psychologists in a clinical setting. Criterion A (repeated voiding of urine into clothes or bed), criterion B (the behavior occurs at least twice a week for at least 3 months with clinical distress or impairment), and criterion C (chronological age of at least 5 years) can be evaluated using the K-SADS, which queries enuresis symptomatology mapping onto DSM-5 criteria. The K-SADS-PL is reliable, valid, and sensitive for measuring specific subtypes of enuresis (Birmaher et al., 2009; Table 5).
Psychometric Information of Validated and Reliable Elimination Disorders Assessments.
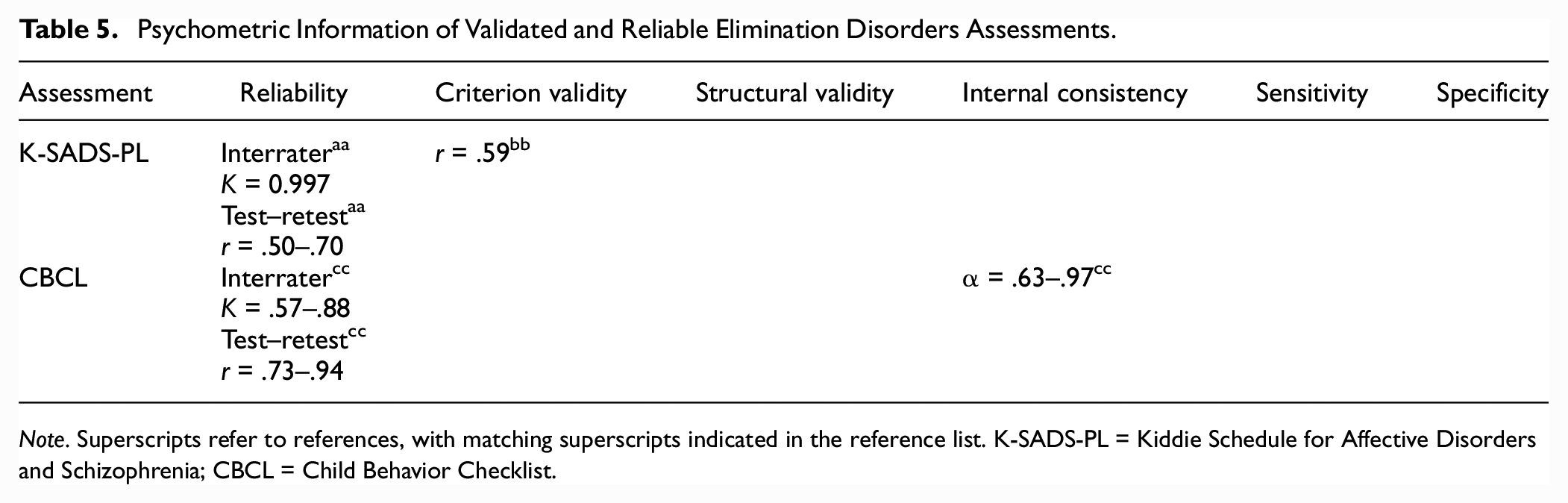
Note. Superscripts refer to references, with matching superscripts indicated in the reference list. K-SADS-PL = Kiddie Schedule for Affective Disorders and Schizophrenia; CBCL = Child Behavior Checklist.
Assessing the child’s psychiatric history and determining the presence of possible comorbidities is important for diagnosis and treatment planning. These criteria may be usefully evaluated using the Child Behavior Checklist (CBCL, Achenbach & Ruffle, 2000) or other broadband measures, available in parent-, teacher-, and self-report, along with a diagnostic interview (e.g., K-SADS). The Achenbach scales are standardized, reliable, and valid and are commonly used as part of characterization of children’s psychiatric profile (Achenbach & Ruffle, 2000; Table 5). When assessing for the presence of other disorders, it is important to check if inappropriate elimination is a response to trauma (acute) or is chronic. Children are also encouraged to keep frequency/volume charts that measure total voided volume over 24 hours, frequency of micturition, mean voided volume, largest single voided volume, and range of voided volume to facilitate documentation of progress over treatment (Siltberg et al., 1997).
Encopresis
Encopresis is the inappropriate passage of feces, either voluntarily or involuntarily. Encopresis is significant when inappropriate passage of feces occurs at least once a month for 3 months and the child is at least 4 years of age (or equivalent developmental level). It is estimated about 1% of 5-year-olds have encopresis. Differential diagnosis for encopresis must include medical conditions like chronic diarrhea, spina bifida, and anal stenosis. Disruptive behavior disorders may co-occur with encopresis, specifically if passage of feces is without constipation and overflow incontinence (APA, 2013).
After medical conditions like chronic diarrhea, spina bifida, and anal stenosis have been ruled out by a child’s primary care provider (criterion D), other assessment for encopresis may be considered. Criterion A (repeated passage of feces into inappropriate places), criterion B (at least one event a month for at least 3 months), and criterion C (chronological age of at least 4 years) can be assessed using the K-SADS-PL encopresis section, which is sensitive for assessing the subtypes of encopresis (Birmaher et al., 2009). Children are also encouraged to keep a food and toileting log that measures amount and type of food consumed, as well as frequency and consistency of fecal passage (von Gontard, 2013).
Comorbidities can be evaluated using a broadband screener and diagnostic interview, as above. When assessing for co-occurring disorders, it is important to determine whether inappropriate elimination is a response to trauma (acute) or is chronic (Achenbach & Ruffle, 2000). Encopresis can be caused by unsuccessful toilet training, negative toilet training practice, stressful family situations, or as a result of traumatic experiences such as sexual or physical molestation. Traumatic experiences can result in more severe, treatment-resistant cases; thus, trauma history is critical for treatment planning.
It is important to note that if a child is suffering from both enuresis and encopresis, fecal incontinence or constipation should be treated first, as urinary incontinence will often terminate once these problems have been addressed. After encopresis, daytime urinary incontinence should be treated, as many children will stop wetting at night once the daytime problems have been treated (von Gontard, 2013).
Conclusion
Thorough guidance on the evidence-based assessment of developmental disorders is critical, as they are prevalent, impairing, and commonly comorbid with other psychiatric disorders of childhood. This review is the first of its kind—providing critically needed, step-by-step guidance on the existing evidence-based methodologies and assessment tools available for diagnosis. This review also makes clear the dire need for further development and validation of relevant screening and diagnostic measure and calls for specific attention to the development of specific screening and diagnostic assessment measures for feeding disorders and elimination disorders in particular. Clinicians and researchers alike may find this article useful in guiding diagnostic, treatment, and research procedures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
