Abstract
Affective dysregulation (AD) in children is characterized by persistent irritability and severe temper outbursts. This study developed and evaluated a screening questionnaire for AD in children. The development included the generation of an initial item pool from existing instruments, a Delphi rating of experts, focus groups with experts and parents, and psychometric analyses of clinical and population-based samples. Based on data of a large community-based study, the final screening questionnaire was developed (
Affective dysregulation (AD) is characterized by persistent irritable mood and severe outbursts of temper, which are among the most common and challenging symptoms in child and adolescent psychiatry (Baweja et al., 2016). In the literature, the terms AD and irritability are largely used as synonyms (Döpfner et al., 2019; Giller et al., 2021; Waltereit et al., 2019). Children with AD or irritability are excessively angry and aggressive in response to negative emotional stimuli. AD is often characterized as a transdiagnostic dimension (Döpfner et al., 2019) and is associated with a wide range of internalizing and externalizing mental disorders, including depression, anxiety, attention-deficit hyperactivity disorder (ADHD), oppositional defiant disorder (Roy et al., 2013), and conduct disorder (Axelson et al., 2012; Brotman et al., 2006; Copeland et al., 2013; Roy et al., 2013), which can result in poor diagnostic specificity (Holtmann et al., 2017).
In recent years, there has been a controversial debate about the diagnostic classification of children with AD (Grimmer et al., 2010; Parens & Johnston, 2010). As a consequence of this debate, a new diagnosis for children was introduced in the revision of the
Despite increasing interest among researchers and clinicians, relatively little empirical research has yet been performed on the assessment, etiology, and epidemiology of AD. Depending on the underlying conceptualization of AD, previous epidemiological studies have reported prevalence rates between 0.8% and 9.2% (Brotman et al., 2006; Copeland et al., 2013; Grau et al., 2018; Mayes et al., 2015). Affected children are severely impaired in various life domains, such as school and family life (Copeland et al., 2013; Dougherty et al., 2014). Furthermore, AD in childhood predicts later psychiatric problems such as depression, anxiety, and suicidal ideation (Benarous et al., 2019; Stringaris et al., 2009; Vidal-Ribas et al., 2016) and is a common cause of health services utilization (Dougherty et al., 2016), pointing out a significant public health impact. Overall, these findings underline the importance to identify and treat children with AD at an early stage.
So far, however, knowledge regarding the appropriate assessment of AD in children is still limited. Some standardized questionnaires and structured clinical interviews include items that measure certain aspects of AD, such as irritability (Affective Reactivity Index; Stringaris et al., 2012), anger (PROMIS Anger Scale; Irwin et al., 2012), emotion regulation (Emotion Regulation Checklist; Shields & Cicchetti, 1997), or temper loss (Multidimensional Assessment of Preschool Disruptive Behavior; Wakschlag et al., 2012). Further established rating scales assessing emotion dysregulation include the Emotion Dysregulation Inventory (Mazefsky et al., 2018) and the dysregulation profiles of the Strengths and Difficulties Questionnaire (SDQ-DP; Deutz et al., 2018) and of the Child Behaviour Checklist (CBCL-DP; Geeraerts et al., 2015). However, most existing instruments are not structured around the phenotype of AD, as they do not describe the full spectrum of irritability and temper outbursts or cover additional symptoms that rather capture other disorders (McTate & Leffler, 2017; Waltereit et al., 2019). The development of focused screening and assessment tools to guide diagnosis and treatment for AD should be the focus of future research.
The aim of this study was to develop and evaluate a parent-reported screening questionnaire on AD in 8- to 12-year-old children following a mixed-methods approach. We aimed to investigate the following research questions.
Method
Study and Samples
The research consortium ADOPT (
For developing the preliminary version of the screening questionnaire (i.e., pre-DADYS-Screen), two samples were used.
Sample 1: The clinical pre-DADYS-Screen development sample included data from a clinical pilot study conducted at the outpatient clinic of the School of Child and Adolescent Cognitive Behavior Therapy (AKiP) at the Department of Child and Adolescent Psychiatry, Psychosomatics and Psychotherapy at the University of Cologne, Germany; the clinical sample was recruited during the usual clinical assessment meetings, during which parents were informed about the study and asked to fill out a paper-and-pencil questionnaire. Data were gathered over 4 months (September–December 2017) from parents of
Sample 2: The population-based pre-DADYS-Screen development sample resulted from the longitudinal BELLA study on mental health and well-being in children and adolescents in Germany (Ravens-Sieberer et al., 2015); parent-reported data from the second measurement point of the BELLA study (conducted 2004 to 2007) on 8- to 12-year-olds were used (
For developing and evaluating the final version of the screening questionnaire (i.e., DADYS-Screen), we prospectively recruited a large community sample in the project ADOPT Epidemiology. Data were collected across four German cities (Cologne, Dresden, Mannheim, and Ulm) over 18 months (February 2018 to August 2019). Families with children aged 8 to 12 years were randomly selected from the official registers of the residents’ registration offices. Potential participants were contacted by the ADOPT Epidemiology study team using conventional mail to inform about the study and ask for written informed consent to participate. In addition, the screening questionnaire was sent out to the parents. Parents were reminded once within the study (after 4 weeks). Alternative to the paper-and-pencil questionnaire, participants had the opportunity to fill in the questionnaire online or to answer the questions on the phone. Data collection and management were supported using a secure, web-based application named REDCap (Harris et al., 2009) hosted at the Clinical Trials Center Cologne.
Overall, we contacted
For developing and evaluating the final version of the DADYS-Screen, the following samples from the community-based study ADOPT Epidemiology were used:
Sample 3: The community DADYS-Screen development sample included valid data of the first
Sample 4: The community DADYS-Screen evaluation sample included data of
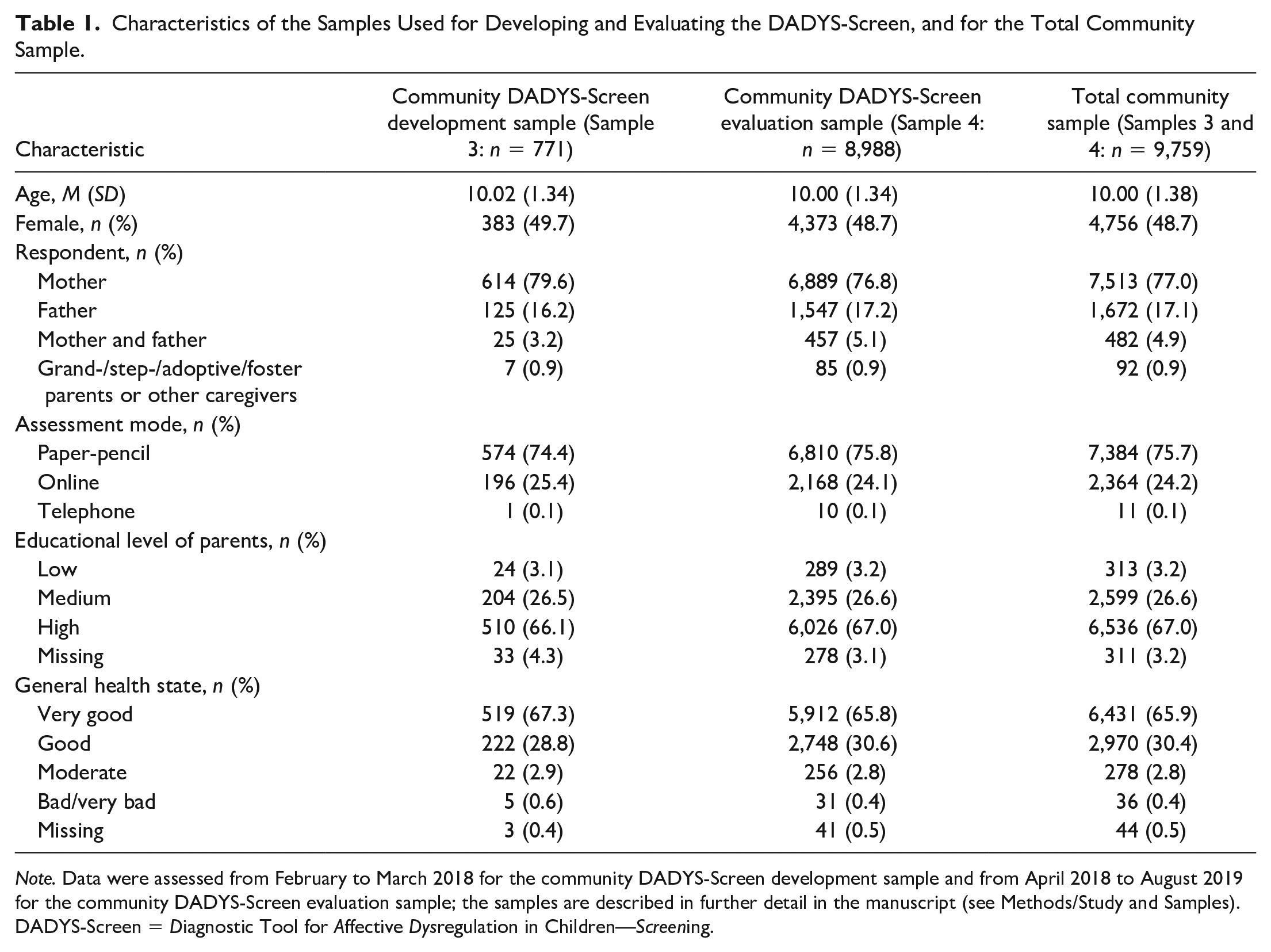
The flowchart on the process of selecting the study participants in the community-based study is presented in Figure 1. In addition, both subsamples (Samples 3 and 4) and the total sample from the community-based study (gathering Samples 3 and 4) are described in Table 1.
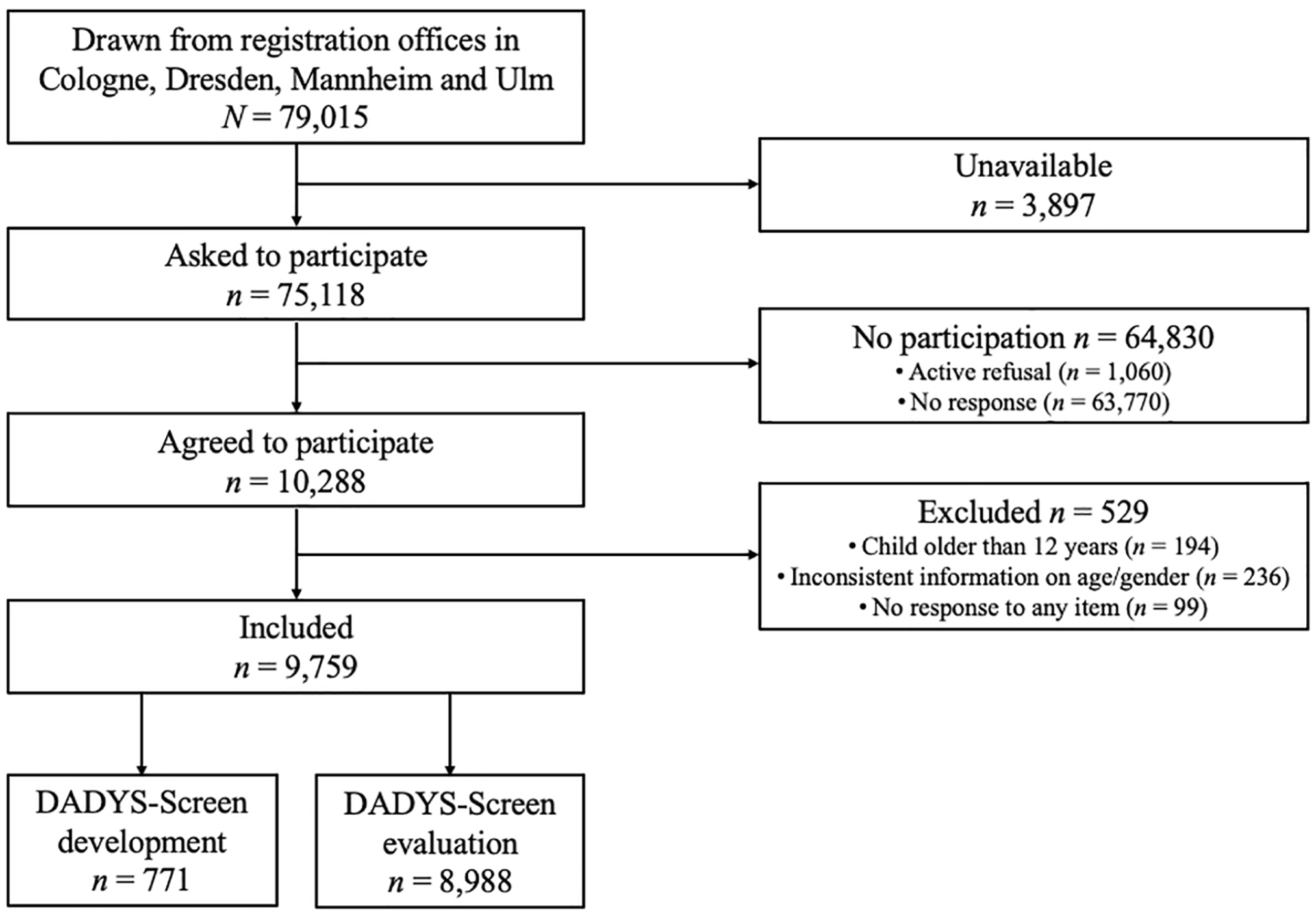
Selection of Study Participants in the Community Sample
Characteristics of the Samples Used for Developing and Evaluating the DADYS-Screen, and for the Total Community Sample.
Measurement
In the community samples of the project ADOPT Epidemiology, the preliminary version of the screening tool with 14 items (i.e., pre-DADYS-Screen) was administered in Sample 3 until March 2018, and the final version with 12 items (i.e., DADYS-Screen) was administered in Sample 4 from April 2018 until August 2019. For assessing data in the community study, the items for pre-DADYS-Screen and for DADYS-Screen were consistently presented with a 4-point response scale, ranging from 0 =
Analyses
Development of DADYS-Screen
The development of the screening tool followed a stepwise process using qualitative and quantitative methods.
Generation of the Initial Item Pool
The initial item pool was created based on existing instruments. Clinical and research experts reviewed well-established, validated, and standardized clinical rating scales and selected items on symptoms and behaviors closely related to AD (such as irritable mood, emotion dysregulation, anger, impulsivity, and reactivity). The following measures were selected: (a) the Affective Reactivity Index (ARI), which is a dimensional measure of irritability and anger that includes six items on behaviors and feelings specific for irritability as well as 1 item on impairment (Stringaris et al., 2012); (b) the Emotion Regulation Checklist (ERC), which is a 24-item scale for assessing the ability of a child to manage and cope with emotions (Shields & Cicchetti, 1997); (c) the PROMIS Anger Scale, which consists of 5 items covering parent-reported irritable and angry mood of the child (Irwin et al., 2012); (d) the Global Index of the Conners’ Rating Scale, which is used to screen for symptoms of ADHD and behavioral problems, and consists of ten items (Conners et al., 2011; Otto et al., 2018); (e) the dysregulation profile of the SDQ, which assesses affective, behavioral, and cognitive aspects of dysregulation (Deutz et al., 2018); and (f) the Disruptive Mood Dysregulation and Irritability Scale from the Symptom-Checklist for Oppositional Defiant Disorder and Conduct Disorder (SCL-ODD), which assesses the symptom criteria of DMDD according to
Qualitative and Quantitative Investigations of the Initial Item Pool to Develop Pre-DADYS-Screen
Qualitative investigations included a Delphi rating and focus groups. The Delphi process is a consensus method frequently used in health research with the aim to determine the extent to which experts agree on a given issue (Jones & Hunter, 1995; Landeta, 2006). In this study, a multidisciplinary group of experts, composed of child and adolescent psychologists, psychotherapists, pediatricians, and researchers, was asked to participate because of their expertise and clinical experience in the field of child mental health (
Focus groups were conducted with (a) clinical experts (
For the quantitative investigation of the initial item pool, Sample 1 (i.e., the clinical pre-DADYS-Screen development sample resulting from the clinical pilot study;
Based on the results of quantitative and qualitative investigations, the initial item pool was reduced following a stepwise procedure. In the first step, we excluded items that did not meet a priori defined criteria. An item was excluded, if (a) only 10% or less of the experts agreed with the inclusion (resulting from the Delphi rating), (b) only 10% or less of the experts rated the item as relevant (resulting from the clinician focus group), (c) the item was difficult to comprehend by parents (resulting from the parent focus groups), (d) the item had a low item difficulty (only relevant for Sample 1;
Empirical Investigation of Pre-DADYS-Screen to Develop DADYS-Screen
The quantitative investigation of pre-DADYS-Screen was conducted based on Sample 3, i.e., that is., the community DADYS-Screen development sample (including data of the first participants of ADOPT Epidemiology;
An item was excluded from pre-DADYS-Screen, if (a) an intercorrelation with another item was very low (
Evaluation of DADYS-Screen
Sample 4 was used to evaluate the final screening tool based on methods from CTT and IRT (i.e., the community DADYS-Screen evaluation sample with data gathered from April 2018 until August 2019;
IBM SPSS Statistics versions 25 to 27 were used for CTT analyses, Mplus version 8.0 for confirmatory factor analyses, and WINMIRA 1.42 and Winsteps 3.67.0 for IRT analyses.
Results
Development of DADYS-Screen
Generation of the Initial Item Pool
Clinical and research experts gathered items on AD from the following measures: ARI (7 items; Stringaris et al., 2012), ERC (24 items; Shields & Cicchetti, 1997), the PROMIS Anger Scale (5 items; Irwin et al., 2012), the Global Index of the Conners’ Rating Scale (10 items; Conners et al., 2011; Otto et al., 2018), SDQ (10 items; Deutz et al., 2018), and the Disruptive Mood Dysregulation and Irritability Scale from SCL-ODD (11 items; DISYPS-III; Döpfner & Görtz-Dorten, 2017). The generated initial item pool included 67 items and is presented in the Supplementary Material (Table S1).
Qualitative
and Quantitative Investigations of The Initial Item Pool to Develop Pre-DADYS-Screen
Using the Delphi rating, a positive consensus was achieved for 17 items (i.e., at least 90 % of the experts agreed that these items should be included in the screening questionnaire). A negative consensus was reached for 7 items, indicating that these items are rather unsuitable to screen children for AD. For the remaining 43 items, no consensus was achieved. Detailed information on the results of the Delphi rating is depicted in Table S2.
In the focus group among clinical experts, 7 items were identified as relevant to screen children for AD by at least 90% of the experts, whereas 8 items were classified as relevant by only 10% or less of the experts (see Table S2). According to the focus groups with parents, the presented items were mostly well understood, some items were rather difficult to answer (especially double-barrelled items [e.g., Items i7 and i25 in Table S2] or those with very long phrasings [e.g., Item i11]).
Quantitative investigations were conducted on the complete initial pool of 67 items using Samples 1 and 2. Sample 1 (i.e., the clinical pre-DADYS-Screen development sample;
Following our stepwise procedure, 30 items were excluded from the initial item pool in the first step because they did not meet the a priori defined qualitative criteria and/or statistical thresholds. In the second step, 23 items were excluded based on a final agreement of experts; excluded items covered the same content as other items but showed less good analyzing results or had limited content validity (e.g., items that were too strongly related to other mental disorders). Overall, this resulted in a remaining set of 14 items that were included in the pre-DADYS-Screen. Results of the conducted analyses and item-specific information on inclusion or reasons for exclusion from pre-DADYS-Screen are shown in Table S2. The pre-DADYS-Screen was subsequently administered in the community sample (Sample 3).
Empirical Investigation of Pre-DADYS-Screen to Develop DADYS-Screen
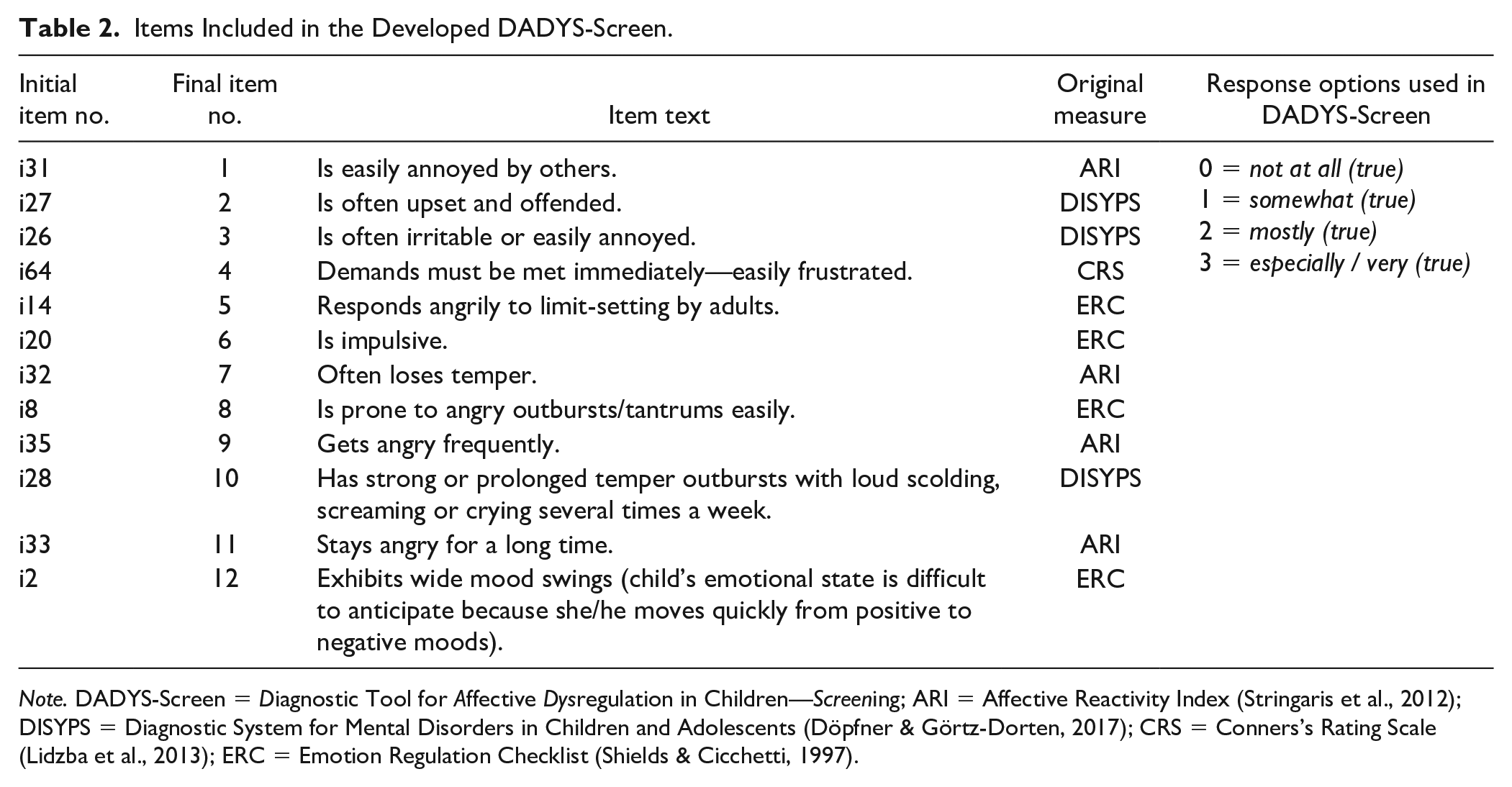
We investigated pre-DADYS-Screen using data of Sample 3 (i.e., the community DADYS-Screen development sample including data gathered until March 2018;
Items Included in the Developed DADYS-Screen.
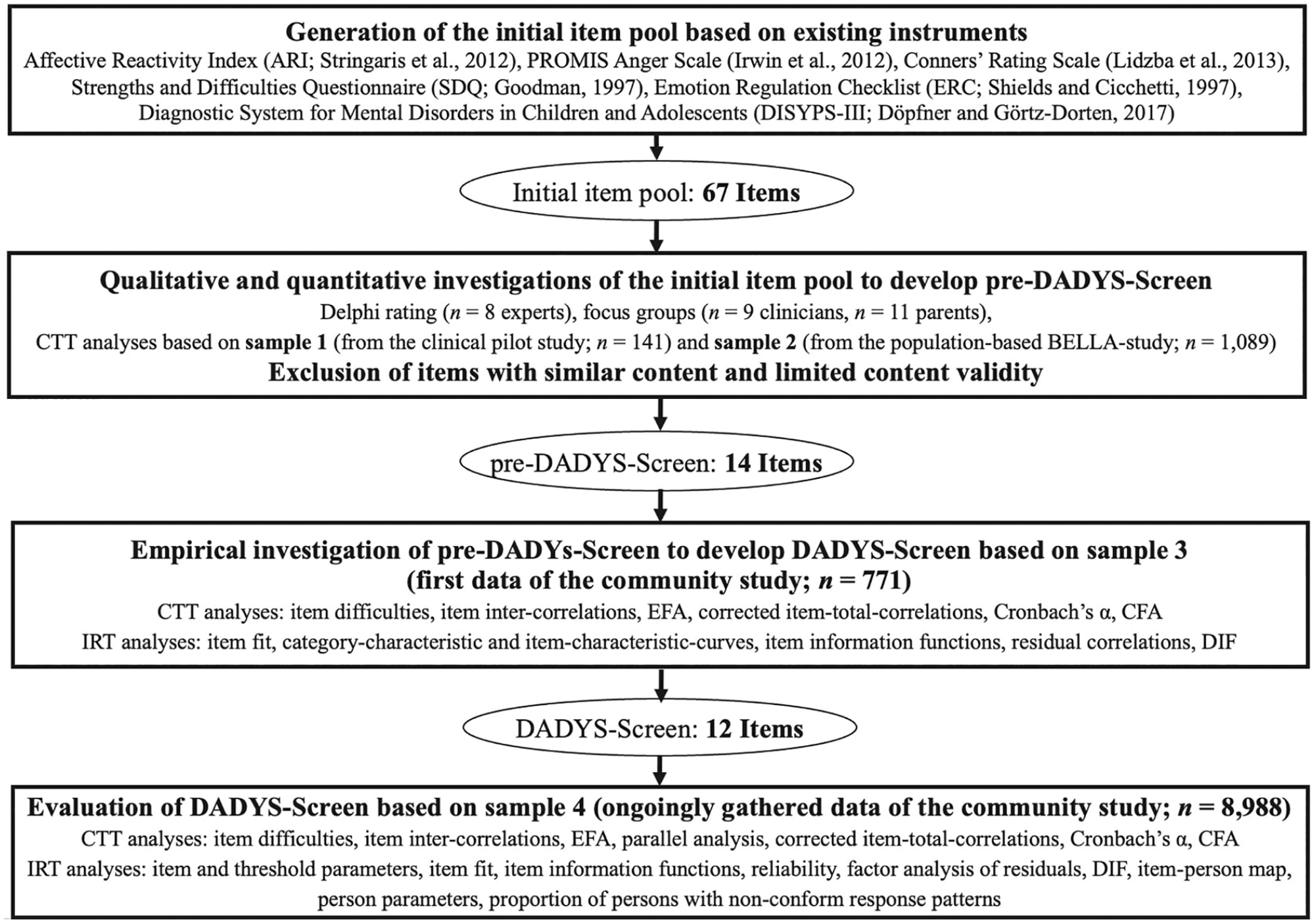
Development and Evaluation Process for the DADYS-Screen
Evaluation of DADYS-Screen
Results Based on Classical Test Theory
Sample 4 was used to evaluate DADYS-Screen (i.e., the community DADYS-Screen evaluation sample including data gathered from April 2018 until August 2019;
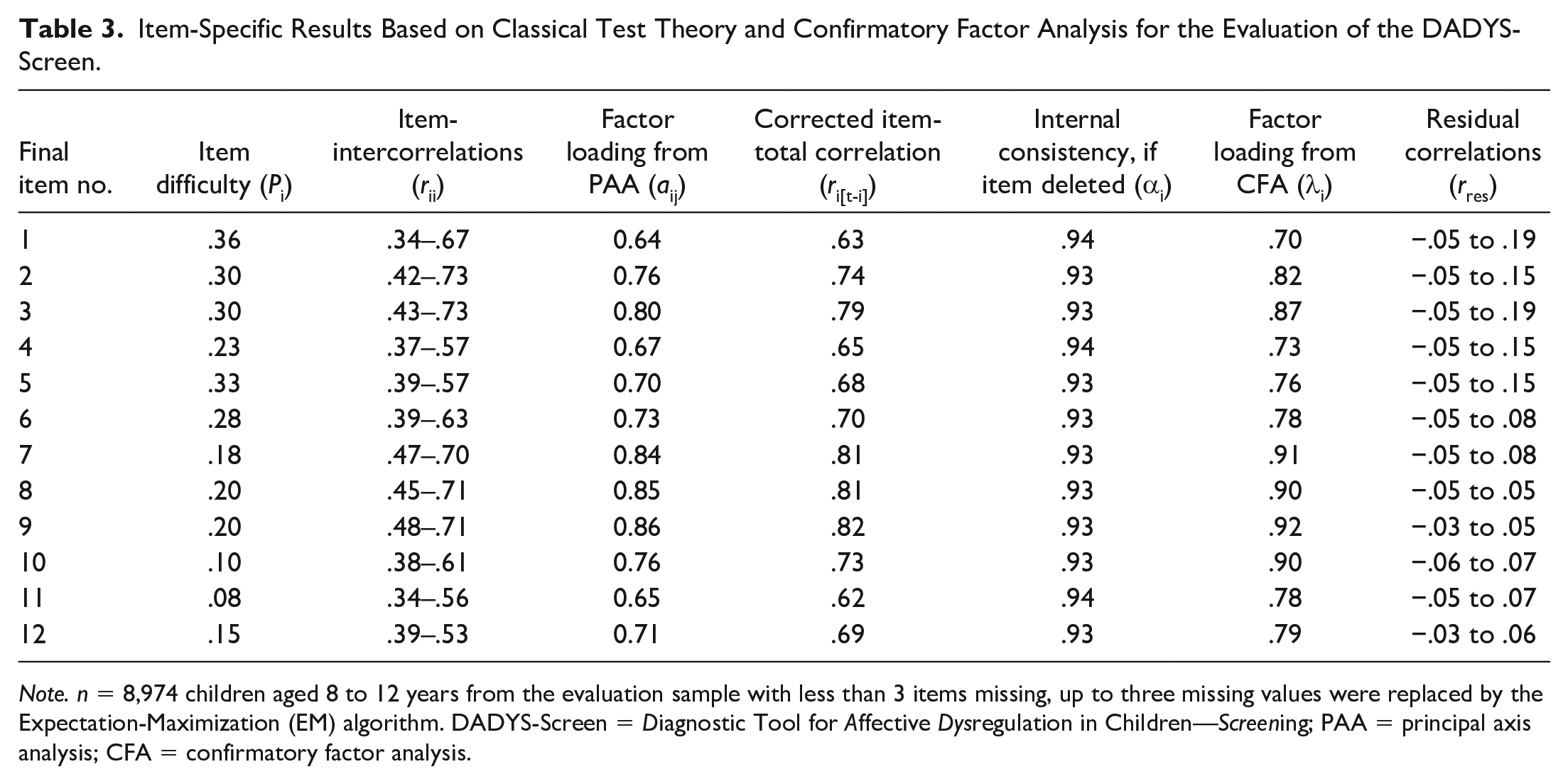
Results are presented in Table 3 and gathered in the following. Item difficulties in our community-based sample (i.e., Sample 4) ranged from .08 to .36 across the 12 items (only presented for descriptive purposes). Correlations indicated moderate to strong associations among items (
Item-Specific Results Based on Classical Test Theory and Confirmatory Factor Analysis for the Evaluation of the DADYS-Screen.
Results Based on Item Response Theory
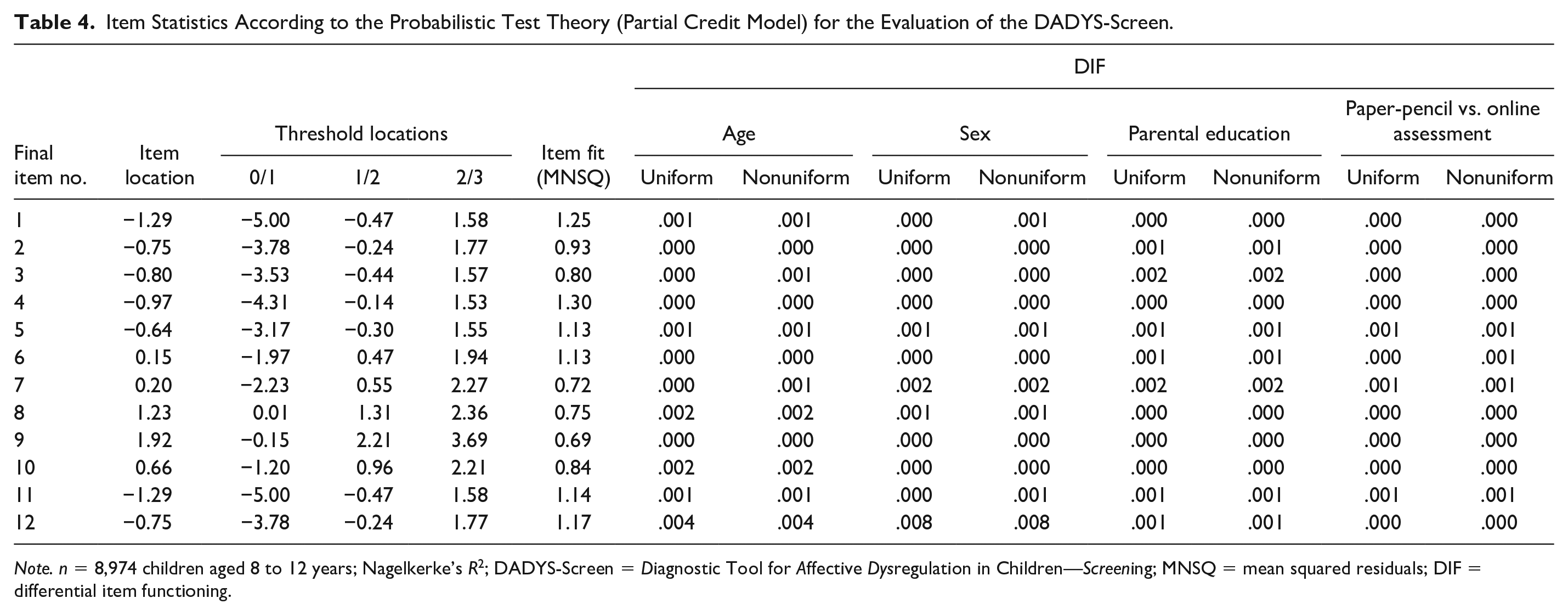
Table 4 shows the item statistics according to the probabilistic test theory following the partial credit model generated for Sample 4 under analyses (
Item Statistics According to the Probabilistic Test Theory (Partial Credit Model) for the Evaluation of the DADYS-Screen.
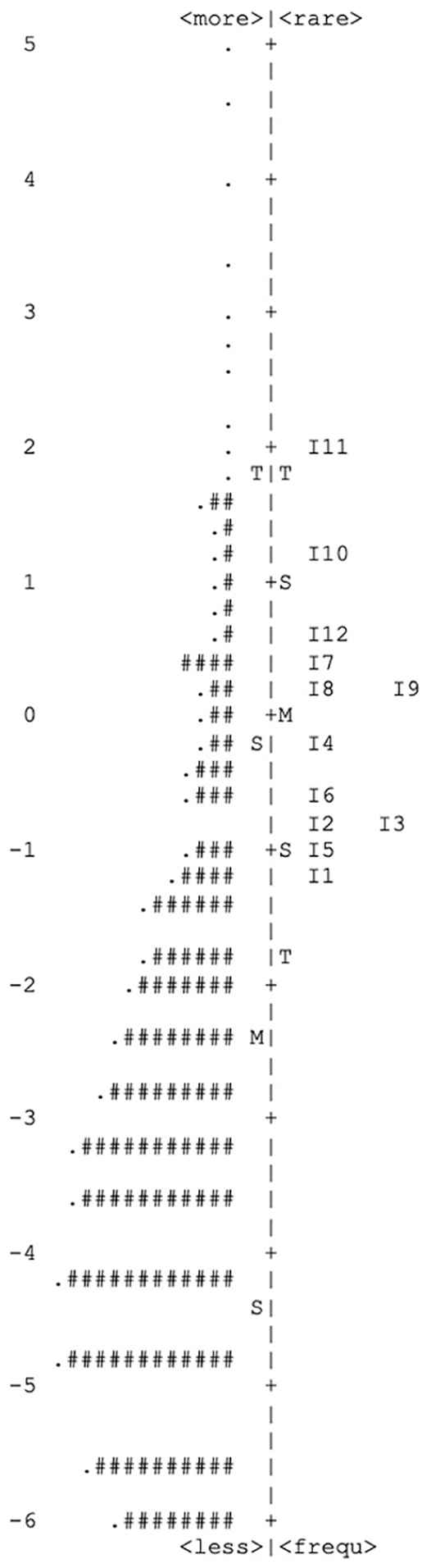
Item-Person Map: Location of Persons and Items on the Latent Trait Affective Dysregulation in a Community Sample of n = 8,974 Children Aged 8 to 12 Years
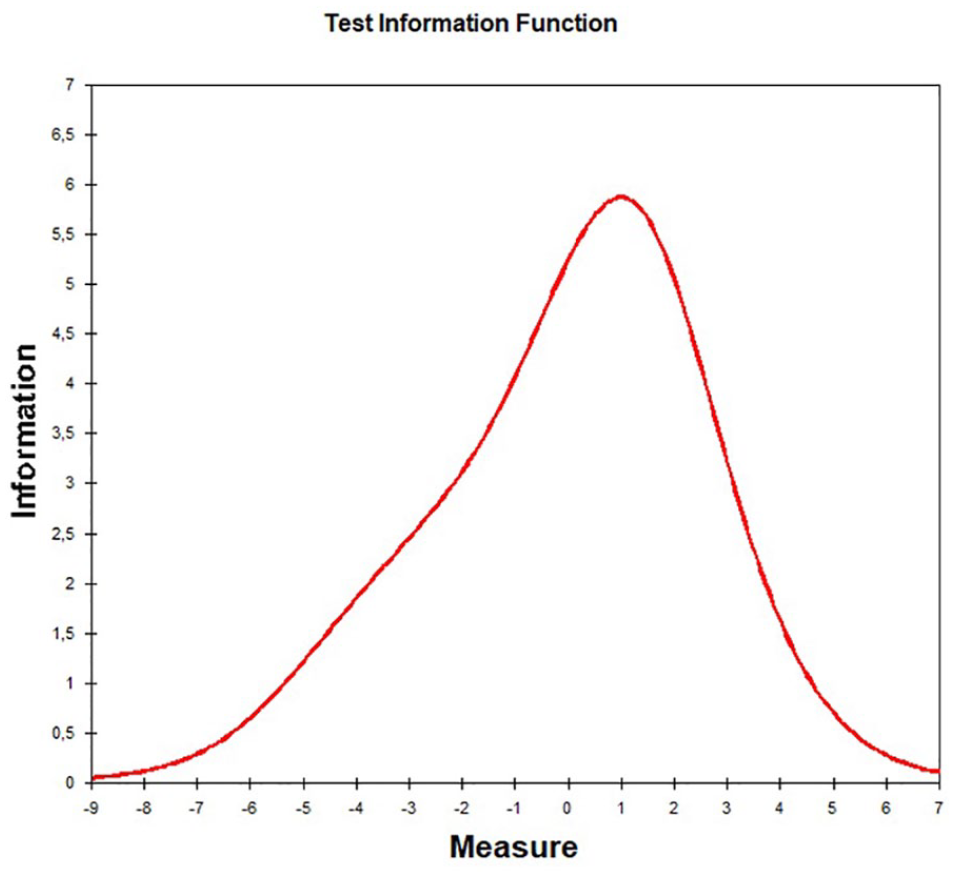
Test Information Function
Discussion
The present study describes the development and evaluation of a screening questionnaire based on parent reports to assess AD in 8- to 12-year-old children. We used qualitative methods like Delphi rating of experts and focus groups with clinicians and parents, and quantitative methods based on CTT and IRT investigating clinical and population-based data. The resulting 12 items of the DADYS-Screen are appropriate and suitable for assessing AD comprehensively due to ratings of clinical experts and comprehensible according to the feedback of parents. The evaluation of the DADYS-Screen demonstrated good feasibility and mainly good psychometric properties according to parameters based on CTT and IRT.
We followed the state of the art in our proceeding using a mixed-methods approach with recommended methods. Furthermore, we chose the items for the initial item pool from existing well-established and validated instruments, such as the Affective Reactivity Index (Stringaris et al., 2012) and the Emotion Regulation Checklist (Shields & Cicchetti, 1997). Moreover, we followed Boateng et al. (2018) and used a deductive method in developing our screening tool. Our item pool covered a broad and comprehensive construct, which was somewhat broader than the target construct of AD and included 5 times as many items as gathered in the final DADYS-Screen (Boateng et al., 2018; Weiner et al., 2012).
In our evaluation of the screening measure, we detected overall good psychometric properties according to the CTT. We found good item-intercorrelations, high factor loadings, good internal consistency, and low residual correlations. Model fit according to confirmatory factor analysis was good in comparison to the baseline model, although the fit was only “mediocre” according to guidelines for interpreting the RMSEA of Brown and Cudeck (1992). We tolerated low item difficulties as our data was gathered in a community sample, but the main target group for the screening tool will be children under risk of AD and clinical samples. The analyses in the probabilistic model on the evaluation of DADYS-Screen confirmed the good measuring characteristics of the scale overall. Although the item difficulties were located rather closely, the category difficulties were sufficiently broadly spread. The slight bimodality of the information functions of Items i14, i27, and i31 (i.e., final item numbers 5, 2 and 1) should be further investigated in future studies. All other characteristics showed a good fit of the Rasch model to the data, also an item bias could not be identified. The item-person map (Figure 3) indicates that the items are somewhat too difficult for a community sample because the location of the item parameters differs somewhat from the location of the person parameters (and reliability is best when both correspond to each other). However, as the target group of the instrument are selected subjects with an elevated symptom level of AD, the screening tool will probably show a better fit in corresponding clinical samples; this will be investigated in future studies.
The parallel analysis indicated a one-factorial solution. However, two-factor models for tonic (irritable mood) and phasic (temper outbursts) irritability are also described in the literature (Klein et al., 2021; Silver et al., 2021). We will consider this in future analyses based on clinical data. We will validate our screening tool based on clinical data collected in the ADOPT consortium. Analyses based on future clinical data will include the determination of item difficulties and the investigation of the factor structure of the screening tool in the clinical population, and we aim to provide a cutoff for our screening measure considering its sensitivity and specificity due to relevant psychiatric diagnoses.
The construct of AD is very closely related to the new diagnosis of DMDD. According to the
Our study has some limitations. First, many different terms are used in the literature to characterize very similar constructs, and a clear consensus definition of what constitutes AD or irritability remains elusive. In line with the literature (Döpfner et al., 2019; Giller et al., 2021; Waltereit et al., 2019), we chose the term AD, which appears to be less established but focuses on a more narrow construct of excessive reactivity to negative emotional stimuli with affective and behavioral components, differentiating it from broader concepts such as emotion dysregulation. Second, considering the developmental context of prepuberty (e.g., hormonal development), our study focused on children aged 8 to 12 years. We did not develop our screening tool for children younger than 8 years or older than 12 years. Future research may wish to investigate the feasibility and validity of our screening tool for use in younger or older children. Third, the validation of the DADYS-Screen and its self-reported version is not provided so far but will add important information for their use, especially in samples under risk of AD. The present study does not provide information on the construct and criterion validity of DADYS-Screen. The questionnaire used for the screening of participants at risk of AD in the community study was kept as short as possible to support the participation of as many willing parents as possible. However, in analyses conducted for another article based on data of Sample 4, a strong correlation was found between the PROMIS Anger Scale (including five items measuring parent-reported irritable and angry mood of the child) and the DADYS-Screen (
Overall, this study provides preliminary evidence for the psychometric performance of the DADYS-Screen and supports its use in identifying children with persistent irritability and severe temper outbursts, although further psychometric investigation using clinical data is needed. DADYS-Screen seems to allow clinicians and researchers a better identification of children with AD and is thus promising to support appropriate diagnosis, treatment, and clinical practice.
Supplemental Material
sj-docx-1-asm-10.1177_10731911221082709 – Supplemental material for The DADYS-Screen: Development and Evaluation of a Screening Tool for Affective Dysregulation in Children
Supplemental material, sj-docx-1-asm-10.1177_10731911221082709 for The DADYS-Screen: Development and Evaluation of a Screening Tool for Affective Dysregulation in Children by Christiane Otto, Anne Kaman, Claus Barkmann, Manfred Döpfner, Anja Görtz-Dorten, Claudia Ginsberg, Sara Zaplana Labarga, Anne-Katrin Treier, Veit Roessner, Charlotte Hanisch, Michael Koelch, Tobias Banaschewski and Ulrike Ravens-Sieberer in Assessment
Footnotes
Acknowledgements
The authors thank all parents who participated in this research for their time and involvement. We are very grateful to all the researchers and students who worked on this project and made it possible. Furthermore, we would like to thank the members of the ADOPT study group. Members of the ADOPT study group are: Pascal Aggensteiner, Tobias Banaschewski, Dorothee Bernheim, Stefanie Bienioschek, Daniel Brandeis, Maurice Breier, Veronika Dobler, Manfred Döpfner, Jörg M. Fegert, Franziska Frenk, Franziska Giller, Claudia Ginsberg, Anja Görtz-Dorten, Monja Groh, Charlotte Hanisch, Stefan Heintz, Martin Hellmich, Sarah Hohmann, Christine Igel, Michaela Junghänel, Anna Kaiser, Anne Kaman, Betül Katmer-Amet, Josepha Katzmann, Michael Kölch, Katrin Koppisch, Kristin Kuhnke, Sabina Millenet, Kristina Mücke, Theresa Nickel, Christiane Otto, Ulrike Ravens-Sieberer, Anne Ritschel, Elisaveta Rodova-Ghasemi, Veit Roessner, Angelina Samaras, Anne Schreiner, Jennifer Schroth, Anne Schüller, Marie Steiner, Marion Steiner, Susanne Steinhauser, Anne-Katrin Treier, Matthias Winkler, Sara Zaplana.
Data Availability
The data and analysis code that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christiane Otto, Anne Kaman, Claus Barkmann, Manfred Döpfner, Anja Görtz-Dorten, Claudia Ginsberg, Sara Zaplana Labarga, Anne-Katrin Treier, Charlotte Hanisch and Ulrike Ravens-Sieberer declare that they have no conflicts of interest.
Veit Roessner has received payment for consulting and writing activities from Lilly, Novartis, and Shire Pharmaceuticals, lecture honoraria from Lilly, Novartis, Shire Pharmaceuticals/Takeda, and Medice Pharma, and support for research from Shire Pharmaceuticals/Takeda and Novartis. He has carried out clinical trials in cooperation with the Novartis, Shire Pharmaceuticals/Takeda, Servier and Otsuka companies; the present work is unrelated to these relationships.
Michael Koelch received funding by German Ministry of Education and Research for several projects. He received royalities from Springer, Hogrefe and Beltz; the present work is unrelated to these relationships.
Tobias Banaschewski served in an advisory or consultancy role for Lundbeck, Medice, Neurim Pharmaceuticals, Oberberg GmbH, Shire, and Infectopharm. He received conference support or speaker’s fee by Lilly, Medice, and Shire. He received royalities from Hogrefe, Kohlhammer, CIP Medien, Oxford University Press; the present work is unrelated to these relationships..
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research consortium on Affective Dysregulation, ADOPT, funded by the German Federal Ministry of Education and Research (FKZ 01GL1741B).
Supplemental Material
Supplemental material for this article is available online.
Methodological Disclosure
We described how we recruited our community-based sample, all data exclusions, and all measures in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
