Abstract
The assessment of medically unexplained symptoms and “somatic symptom disorders” in older adults is challenging due to somatic multimorbidity, which threatens the validity of somatization questionnaires. In a systematic review study, the Patient Health Questionnaire–15 (PHQ-15) and the somatization subscale of the Symptom Checklist 90-item version (SCL-90 SOM) are recommended out of 40 questionnaires for usage in large-scale studies. While both scales measure physical symptoms which in younger persons often refer to unexplained symptoms, in older persons, these symptoms may originate from somatic diseases. Using empirical data, we show that PHQ-15 and SCL-90 SOM among older patients correlate with proxies of somatization as with somatic disease burden. Updating the previous systematic review, revealed six additional questionnaires. Cross-validation studies are needed as none of 46 identified scales met the criteria of suitability for an older population. Nonetheless, specific recommendations can be made for studying older persons, namely the SCL-90 SOM and PHQ-15 for population-based studies, the Freiburg Complaint List and somatization subscale of the Brief Symptom Inventory 53-item version for studies in primary care, and finally the Schedule for Evaluating Persistent Symptoms and Somatic Symptom Experiences Questionnaire for monitoring treatment studies.
Keywords
Introduction
Somatization is the tendency to experience and communicate somatic distress in response to psychosocial stress and to seek medical help for it (Lipowski, 1988). The severity of somatization, however, is difficult to measure. In younger people, it is often assessed by a physical symptom count, especially by counting symptoms that often remain medically unexplained in clinical practice, like fatigue or dizziness. Medically unexplained physical symptoms (MUS) might thus be the result of a process of somatization and are a core criterion of somatoform disorders in the Diagnostic and Statistical Manual of Mental Disorders, Fourth edition, text revised (DSM-IV-TR).
With the introduction of the DSM-5, the DSM-IV-TR section of somatoform disorders has been replaced by the new section of somatic symptom and related disorders (American Psychiatric Association, 2013). The most important change was the focus on so-called positive criteria for establishing a diagnosis, that is, the prominence of a physical symptom associated with significant distress and impairment. The major diagnosis in this section, somatic symptom disorder, emphasizes diagnosis made on the basis of positive symptoms and signs, that is, distressing somatic symptoms plus abnormal thoughts, feelings, and behaviors in response to these symptoms. This contrasts with somatoform disorders in the DSM-IV-TR that poses the absence of a medical explanation for the somatic symptoms a key feature. Although somatic symptom disorders might be more useful for diagnosis in both primary and specialized (somatic) health care, the criteria for these disorders do not necessarily apply to all patients burdened by MUS. MUS, irrespective of the DSM classification rules, have consistently been associated with a lower quality of life, psychological distress, and increased medical consumption (Hanssen, Lucassen, Hilderink, Naarding, & Oude Voshaar, 2016; Weiss, Kleinstuber, & Rief, 2016).
A systematic review has identified 40 scales to assess self-report somatic symptoms (Zijlema et al., 2013). The Patient Health Questionnaire–15 (PHQ-15) and the somatization subscale of the Symptom Checklist 90-item version (SCL-90 SOM) were considered the best options to be used in large-scale population–based studies, based on several criteria among which type of symptoms, time frame, response scale, psychometric characteristics, and patient burden (Zijlema et al., 2013). Since older persons often suffer from physical symptoms due to one or more chronic somatic diseases (multimorbidity; Fortin, Stewart, Poitras, Almirall, & Maddocks, 2012), somatic symptom questionnaires may easily overestimate the severity of somatization in an older sample. Nonetheless, accumulating data emerge that somatization, MUS, and somatoform disorders are highly relevant in older persons, posing a significant burden on health-related quality of life, increased level of health care usage and potentially iatrogenic damage (Hanssen et al., 2016). Nonetheless, actual prevalence rates are somewhat lower among persons older than 65 years compared with younger persons (Hilderink, Collard, Rosmalen, & Oude Voshaar, 2013). In a systematic review of six cohort studies including both younger and older people, prevalence rates for DSM-IV defined somatoform disorders ranged from 1.5% through 13.0% (median 5.4%) among people aged 65 years and older, and from 10.7% through 26.8% (median 15.3%) in younger people (Hilderink et al., 2013). Whether these lower prevalence rates in later life are simply artefacts due to falsely attributing physical symptoms to (comorbid) chronic somatic diseases, or reflect real differences due to better coping with chronic illnesses and/or less disease benefits in later life have to be established.
The main objective of the present article is to explore which assessment instrument is optimal for assessing somatization in older adults. In our research program on MUS in later life, we have applied both the PHQ-15 as well as the SCL-90 SOM among older patients with MUS and medically explained symptoms. Therefore, we first test empirically how the PHQ-15 and the SCL-90 SOM relate to other measures of somatization, namely the presence of a somatoform disorder determined by a semistructured psychiatric interview (Lecrubier et al., 1997), and health anxiety measured with the Whitely Index (Pilowsky, 1967), as well as to medically explained somatic disease burden. Since these results were rather disappointing, we decided to update and extend the previously conducted systematic review of Zijlema et al. (2013) to examine which of the assessments of somatic symptoms might be most relevant for assessing somatization in older persons.
Method
Empirical Tests of the PHQ-15 and SCL-90 SOM
For the empirical tests, we used our data set of a pilot study on MUS in older patients as well as a data set on a larger case control study. Both samples have been described elsewhere in detail (see Hanssen et al., 2016; Hilderink et al., 2009), but can be summarized as follows.
The pilot study was a consecutive case series of 37 older patients referred to our outpatient mental health clinic for a diagnostic procedure and treatment for MUS (Hilderink et al., 2009). In this pilot study, we administered the SCL-90 as well as the PHQ-15 (Kocalevent, Hinz, & Brähler, 2013), whereby the item on menstrual cycle was omitted (being not relevant in later life).
The case control study, acronym OPUS study (Older Persons with medically Unexplained Symptoms study) included 118 older persons suffering from MUS (cases) and 154 older patients suffering from medically explained symptoms for which they frequently attended their general practitioner (Hanssen et al., 2016). In this study, the Brief Symptom Inventory, 53 items (Derogatis & Melisaratos, 1983), an abbreviated version of the SCL-90 with similar psychometric properties, has been administered as a measure for psychopathological distress (Derogatis, 1975).
In both studies, the same two indicators of somatization were administered. First, the Mini International Neuropsychiatric Interview (Lecrubier et al., 1997), a semistructured psychiatric interview administered by an old age psychiatrist, to diagnose psychiatric morbidity according to DSM-IV-TR criteria. Therefore, the presence of a somatoform disorder is the first indicator of somatization. Second, health anxiety was in both studies assessed with the Whitely Index (Pilowsky, 1967) and used as the second indicator of somatization in the present study. The somatic disease burden was assessed differently in both studies. In the pilot study, the Cumulative Illness Rating Scale for Geriatrics (CIRS-G; Miller et al., 1992) was administered by a geriatrician after a full geriatric assessment. In the OPUS study, the self-report version of the Charlson Index was applied (Charlson, Pompei, Ales, & MacKenzie, 1987; Katz, Chang, Sngha, Fossel, & Bates, 1996).
As a measure of construct validity, Pearson correlations coefficient of both the SCL-90/BSI-53 SOM subscale as well as the PHQ-15 sum score were calculated with both proxies of somatization (Whitely Index, somatoform disorder) expecting a high correlation, and with both measures of somatic disease burden (CIRS-G, Charlson Index) expecting a low correlation. The Pearson r correlation can be interpreted as no or minimal (0.00 < r < 0.30), low (0.30 < r < 0.50), moderate (0.50 < r < 0.70), high (0.70 < r < 0.90), or extremely high (0.90 < r < 1.00) correlation.
Update and Extension of the Systematic Review by Zijlema et al. (2013)
The systematic review by Zijlema et al. (2013) has been conducted to systematically search and judge all self-report questionnaires for common somatic symptoms, generally used to assess somatic symptom burden and/or level of somatization. After an update of the literature search, in this article all questionnaires will be evaluated on their suitability for use in an older population (see Instrument Evaluation section).
Search Strategy
Since the literature search of the previous systematic review was conducted until October 2012, we repeated exactly the same literature search in the databases Medline, EMBASE, and PsycINFO from October 2012 until October 1, 2016, to select additional questionnaires. The search term contained a combination of somatoform disorder or synonyms and questionnaire or synonyms and symptoms. For Medline, the following search term was used: (“somatoform disorders/classification” [MeSH Major Topic] OR “somatoform disorders/diagnosis” [MeSH Major Topic] OR “somatoform disorders/epidemiology” [MeSH Major Topic] OR “functional somatic symptoms” [Title/Abstract]) AND (questionnaire [Title/Abstract] OR screen* [Title/Abstract] OR “self-report” [Title/Abstract] OR “index” [Title/Abstract]) AND symptoms. For EMBASE and PsycINFO, comparable search terms were used. The search was conducted without language restrictions.
Screening and Selection Procedures
The first two authors independently screened the retrieved articles. The articles were included if they described the development, evaluation, or review of self-report somatization questionnaires. The questionnaires selected had to include symptoms from more than one symptom cluster; not just symptoms of the gastrointestinal tract or cardiopulmonary system. When the symptom questionnaire was a subscale derived from a larger questionnaire, the symptom subscale had to have been separately validated and used. There were no criteria for the target population of the questionnaire.
Discrepancies between the two researchers were resolved by consensus. Full articles were then obtained for all potentially eligible studies. Based on the full text, articles that still fulfilled the inclusion criteria were included in the review.
Data Extraction
Name of questionnaire, number of items, domains assessed, answering scale, time frame, and target population were extracted for every questionnaire. Table 2 shows an overview of the questionnaires before and after October 2012.
Instrument Evaluation
The evaluation on the suitability of the identified questionnaires for an older population included (a) the number of items not applicable in older adults (e.g., items on menstrual cycle) and the number of somatic symptoms included that in older persons usually reflect somatic disease burden, (b) the presence of standardized scores (normative data) for older persons, and (c) finally whether the instrument has been applied in an older sample previously.
Although each somatic symptom or sign can be due to somatic disease, some symptoms can be assumed typically for old age and/or common geriatric syndromes. To assess the most common geriatric syndromes, we used the Brief Assessment Tool (BAT), a geriatric assessment tool specifically developed for general practitioners (Senn & Monod, 2015). The BAT aims to identify the following geriatric syndromes: cognitive impairment, mood disorder, gait and balance impairment/falls, visual impairment, hearing impairment, urinary incontinence, malnutrition/loss of weight, and osteoporosis. The first and fourth author, being a clinical psychologist and old age psychiatrist, respectively, assessed which items of each somatization scale corresponded with a geriatric symptom or syndrome as defined by the BAT. In case of disagreement, the last author, an old age psychiatrist, made a decision.
For the third criterion, systematic literature searches were additionally conducted in Medline, EMBASE, and PsycINFO for each questionnaire separately. This was done by search strings combining the name of the questionnaire with the words indicative for a research sample consisting of older persons defined as an age older than 60 years, for example, “older,” “aged,” or “elderly” and in Medline also the MeSH-terms “aged” and “aged, 80 years and over.”
Results
Empirical Tests of the PHQ-15 and SCL-90 SOM
Pearson correlation coefficients of the PHQ-15 sum score and the SCL-90 SOM with the proxies for somatization as well as somatic disease burden are presented in Table 1. Collectively, these results showed that the presence of a somatoform disorder, as proxy for somatization, was neither significantly associated with the sum score of the SCL-90/BSI-53 SOM, nor with the PHQ-15 sum score. Furthermore, the second proxy for somatization, health anxiety, was moderately associated with both measures of somatization (SCL-90/BSI-53 SOM and PHQ-15) as well as moderately associated with the measures of somatic disease burden (CIRS-G and Charlson Index).
Construct Validity of the PHQ Sum Score and SCL-90/BSI-53 Somatization Scale as Measures of Somatization.
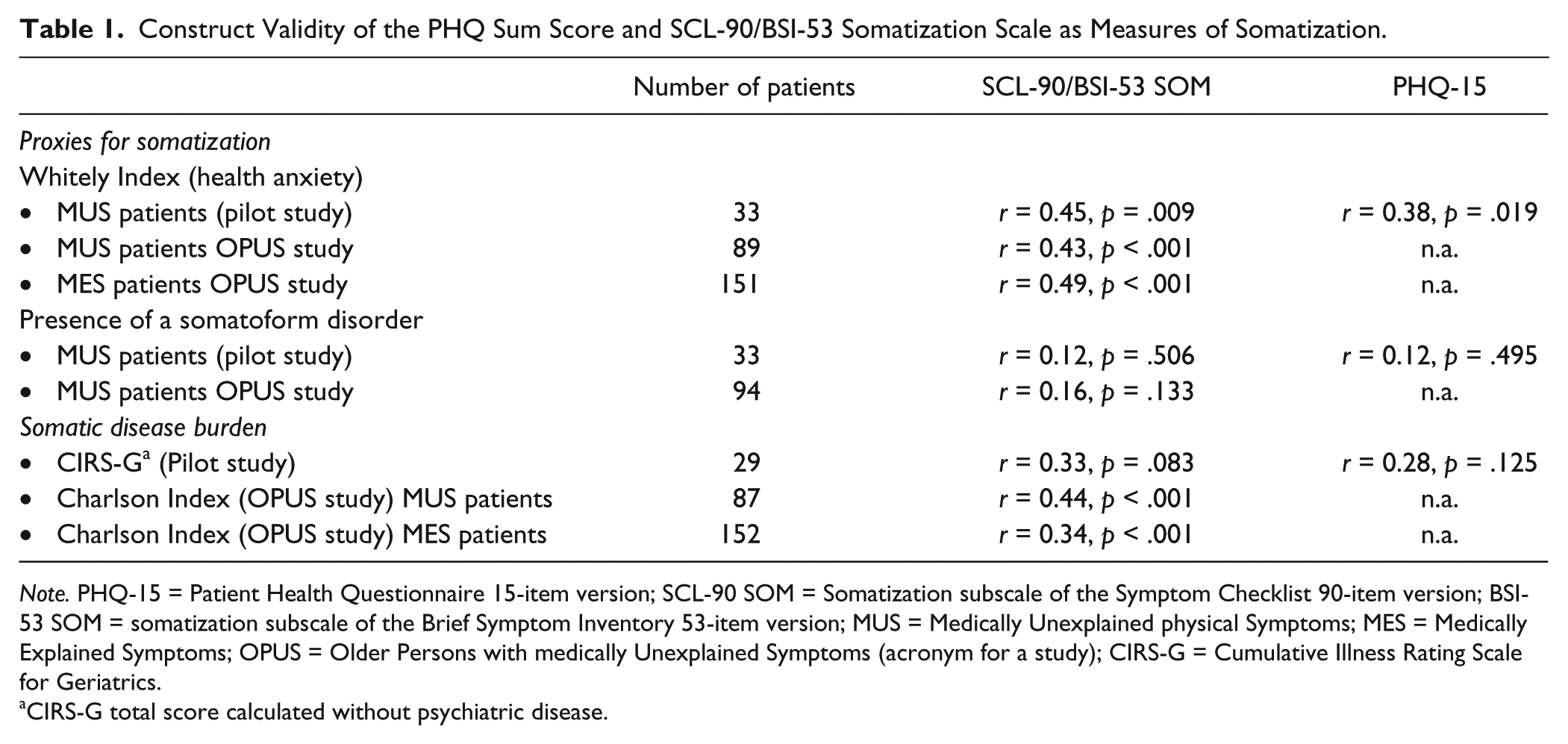
Note. PHQ-15 = Patient Health Questionnaire 15-item version; SCL-90 SOM = Somatization subscale of the Symptom Checklist 90-item version; BSI-53 SOM = somatization subscale of the Brief Symptom Inventory 53-item version; MUS = Medically Unexplained physical Symptoms; MES = Medically Explained Symptoms; OPUS = Older Persons with medically Unexplained Symptoms (acronym for a study); CIRS-G = Cumulative Illness Rating Scale for Geriatrics.
CIRS-G total score calculated without psychiatric disease.
Update and Extension of the Systematic Review
The literature research of Zijlema et al. (2013) until October 2012 had resulted in 40 symptom questionnaires. In contrast to Zijlema et al. (2013), however, we considered the two abbreviated versions of the somatization scale of the SCL-90 (SCL-90 SOM), that is, the BSI-18 SOM (six items) and the BSI-53 SOM (seven items), as separate questionnaires. Therefore, 41 symptom questionnaires were available based on Zijlema et al. (2013). The extended literature search from October 2012 until October 1, 2016, retrieved a total of 631 hits (Medline, n = 187, EMBASE, n = 157, PsycINFO, n = 287), including 75 duplicates. We excluded 436 studies identified on title and abstract. After full text screening, we excluded 114 studies because they were not about somatization (n = 34), were not a self-report questionnaire (n = 3), or were about questionnaires already included (n = 77). A total of six articles were additionally included in the review, describing five new questionnaires. This resulted in a final number of 46 questionnaires for the present review (Table 2).
Overview of the 46 Somatization Questionnaires and Their Properties.
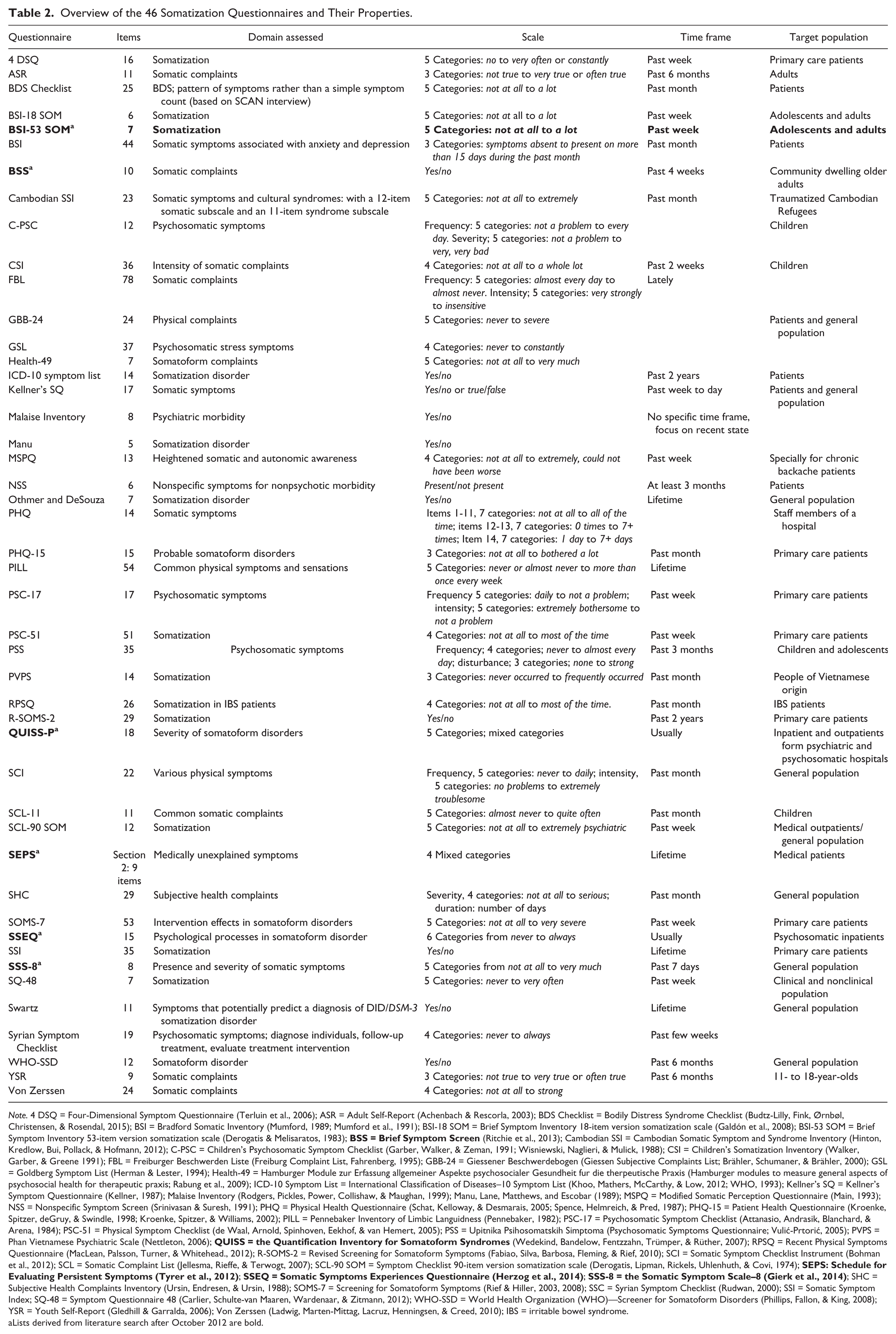
Note. 4 DSQ = Four-Dimensional Symptom Questionnaire (Terluin et al., 2006); ASR = Adult Self-Report (Achenbach & Rescorla, 2003); BDS Checklist = Bodily Distress Syndrome Checklist (Budtz-Lilly, Fink, Ørnbøl, Christensen, & Rosendal, 2015); BSI = Bradford Somatic Inventory (Mumford, 1989; Mumford et al., 1991); BSI-18 SOM = Brief Symptom Inventory 18-item version somatization scale (Galdón et al., 2008); BSI-53 SOM = Brief Symptom Inventory 53-item version somatization scale (Derogatis & Melisaratos, 1983);
Lists derived from literature search after October 2012 are bold.
Evaluation of Questionnaires
Table 3 shows all questionnaires identified. While 8 out of 46 (17%) questionnaires have normative data for older persons, only the Brief Symptom Screen (BSS) was specifically validated in an older sample. The additional questionnaire-specific searches in PubMed, EMBASE, and PsycINFO revealed that only 20 out of 46 (43%) questionnaires were ever used in an older population.
The 46 Somatic Screening Lists and Their Properties for the Old Aged.
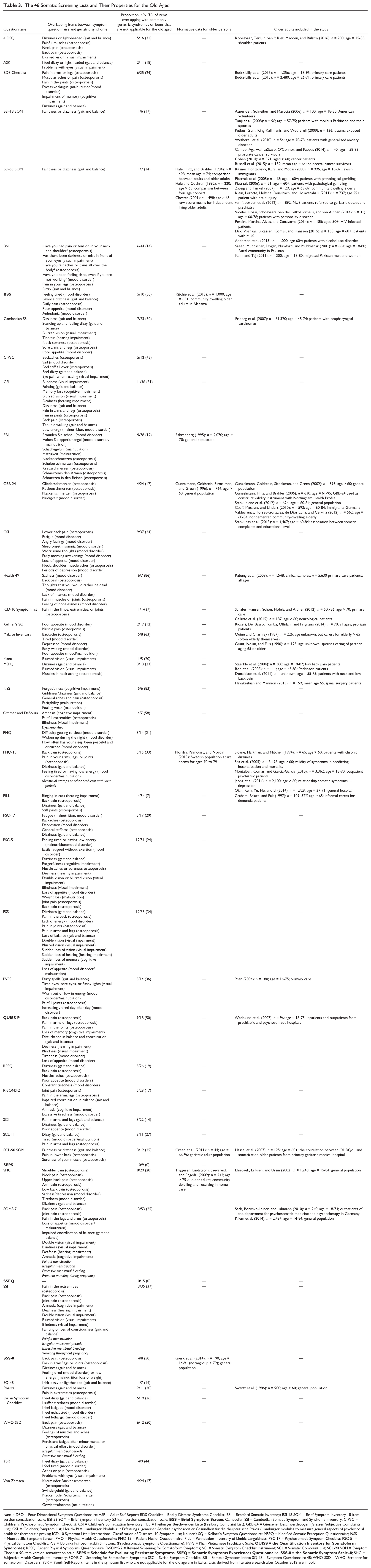
Note. 4 DSQ = Four-Dimensional Symptom Questionnaire; ASR = Adult Self-Report; BDS Checklist = Bodily Distress Syndrome Checklist; BSI = Bradford Somatic Inventory; BSI-18 SOM = Brief Symptom Inventory 18-item version somatization scale; BSI-53 SOM = Brief Symptom Inventory 53-item version somatization scale;
The identification of items overlapping with common geriatric syndromes revealed that all items identified by the first rater (first author) were also identified by the second rater (fourth author). Discrepancies could be clustered in three groups. The first group of items included fatigue, tiredness, loss of energy, and feeling weak; the second group of items loss of appetite and weight loss; and finally the third group (an individual item) was insomnia (sleep onset). The third rater (last author) concluded that these symptoms could all be classified as somatic symptoms of depression and therefore overlap with depression. Taken this decision into account, the median proportion of items overlapping with common geriatric syndromes, plus the number of items not applicable for old age, was 25%. The variability between questionnaire was large, with a range from 0% for the Schedule for Evaluating Persistent Symptoms (SEPS) through as high as 87% for the Health-49. For the PHQ-15, SCL-90 SOM, and SCL-53 SOM, these percentages were 33%, 25%, and 14%, respectively.
Discussion
The PHQ-15 and the SCL-90/BSI-53 SOM both had low correlations with the severity of health anxiety among MUS patients, while neither the PHQ-15 nor the SCL-90/BSI-53 SOM correlated with the presence of a somatoform disorder according to DSM-IV-TR criteria. These findings did not match with our expectations of a moderate to high correlation between both symptom scales and both proxies of somatization. Even more important to note, the PHQ-15 and the SCL-90/BSI-53 SOM were both correlated with measures of somatic disease burden with an effect size comparable to their association with health anxiety. Although the findings with respect to the PHQ-15 need replication in a larger sample, collectively, these findings indicate that both scales do not perform well as indices of somatization in older persons.
An update of the literature search conducted by Zijlema et al. (2013) resulted in five additional self-report somatic screenings lists, namely the BSS (Ritchie et al., 2013), the Quantification Inventory for Somatoform Syndromes (Wedekind et al., 2007), the SEPS (Tyrer et al., 2012), the Somatic Symptoms Experiences Questionnaire (SSEQ; Herzog et al., 2014), and the Somatic Symptom Scale–8 (Gierk et al., 2014).
Less than half (20 out of 46, 43%) of these 46 scales have been administered in studies with exclusively or a substantial number of older adults. Of these studies, the BSI-18 SOM (Galdón et al., 2008), the BSI-53 SOM (Derogatis & Melisaratos, 1983), the Giessener Beschwerdebogen (Brähler et al., 2000), the Modified Somatic Perception Questionnaire (Main, 1983), and the PHQ-15-item version (PHQ-15; Kocalevent et al., 2013) have been applied in more than two studies (see Table 3). As pointed out below, these questionnaires, however, do not have the most optimal characteristics for an older population.
Of the 46 questionnaires, only the BSS has been validated for older adults (Ritchie et al., 2013). Nonetheless, the aim of the BSS was to evaluate overall symptom load in older adult populations in order to estimate illness burden and distress, so not necessarily somatization. Since especially symptoms related to common chronic conditions are included, it may more or less result in a symptom count relevant for the A-criterion of somatic symptom disorder in DSM-5 (American Psychiatric Association, 2013). As 50% of the items overlap with common geriatric syndromes, the BSS is not a good (severity) indicator of possible MUS or somatization in later life.
For eight questionnaires (8 out of 46, 17%), normative data for older persons are available, that is, the BSI-53 SOM, BSS, Freiburg Complaint List (FBL; Fahrenberg, 1995), Giessener Beschwerdebogen, PHQ-15, SCL-90 SOM, Subjective Health Complaints Inventory (Ursin, Endresen, & Ursin, 1988), and the Somatic Symptom Scale–8. Therefore, future results based on any of these scales can be interpreted in the context of scores derived from another geriatric population.
With respect to overlap with common geriatric syndromes, we cannot give a cutoff value above which a specific list should be discouraged to use in geriatric population. The findings of Objective 1 suggests that even a low proportion of overlap may already be too much (i.e., 14%, 25%, and 33% for the BSI-53 SOM, SCL-90 SOM, and PHQ-15, respectively). Of the eight scales with normative data of a geriatric population, only one scale, the FBL has less than 14% of their items overlapping with symptoms of common geriatric syndromes. Since all questionnaires that use symptom counts to measure the level of somatization, we advise to only use these questionnaires in older population when adjustment for the common geriatric syndromes is possible.
Our review identified two scales without any overlap with common geriatric syndromes, that is, the SEPS and the SSEQ. Both scales focus on subjective experiences related to somatic symptoms instead of the symptoms themselves. Unfortunately, none of these scales have normative data for older age groups.
Previously, the PHQ-15 and SCL-90 SOM have been identified as most suitable self-report somatic symptom questionnaires to be used in large-scale studies, because they have been extensively validated, are relatively short, easy to use, and of little burden to participants (Zijlema et al., 2013). Our findings, however, show that the PHQ-15 and the SCL-90 SOM considerably overlap with common geriatric symptoms and probably overestimate the level of somatization in older persons. Recently, somatic symptom count (based on the PHQ-15, added with 10 items on specific neurological symptoms and 5 on mental state) hardly differentiated between patients with (n = 1,144) and without (n = 2,637) MUS referred to an outpatient neurology clinic (Carson, Stone, Hansen, Duncan, & Cavanagh, 2015). Therefore, these questionnaires should only be used when adjustment for MES or common geriatric syndromes is possible.
Some methodological comments, however, need to be made. First of all, many recommendations have been given to increase validity and reliability when designing self-report questionnaires for older adults. Examples of these recommendations include the avoidance of reverse-scored items, a short questionnaire with preferably short, easy to understand items (to avoid fatigue of the participant), a dichotomized response scale (e.g., yes/no), and short reference period (time window) to avoid recall bias (e.g., Green, Goldstein, Sirockman, & Green, 1993; Johnco, Knight, Tadic, & Wuthrich, 2015; Pachana et al., 2007; Yesavage et al., 1983). To our knowledge, none of these recommendations have been empirically tested in order to show that adapting a questionnaire will indeed increase the validity of reliability when applied in older samples. Moreover, many studies, even among frail elderly, generally use several scales together without (reporting) any problems (e.g., Collard, Comijs, Naarding, & Oude Voshaar, 2014; Hanssen et al., 2016). Also, in clinical practice, older patients often have difficulty in making a dichotomous, often black or white choice. And finally, a short reference period seems less applicable for the often, chronic somatic symptoms related to somatization. Nonetheless, although the scientific merits of these criteria have to be established, they may be relevant when choosing a list for a specific study (e.g., as short as possible being a secondary outcome measure). Therefore, these characteristics have been summarized in Table 2.
Second, we focused on self-report questionnaires being most relevant to apply in research studies, acknowledging limited resources being at odds with sample size needs. Moreover, self-report questionnaires are also relevant in later life to get unbiased information about the complaints by minimizing the influence of formal and informal caregivers, often involved with older adults with physical complaints. On the other hand, older persons are more inclined than their younger counterparts to give socially desirable answers (e.g., Saeed et al., 2001).
Third, the validity of all questionnaires can be questioned as no gold standard exists for the measurement of explained and unexplained physical symptoms (especially not in later life). As the agreement between geriatricians whether a physical symptom is explained, partly explained or fully explained is quite high (Benraad et al., 2013), this could be considered as gold standard in future cross-validation studies in old-age samples.
To conclude, our review does not allow to give a simple advice which scale is most optimal to administer in studies in old age. This implies that the field is served by more studies cross-validating these questionnaires in an older population. Depending on the exact research question, however, specific choices can be made. In case overestimation of somatization should be excluded, the SEPS or SSEQ can be used (although both questionnaires are not validated in an older sample yet). These questionnaires, however, do not simply count symptoms, but rely on subjective experiences about physical symptoms. When symptom counts are needed as indication for possible somatization, the FBL emerges as the most optimal questionnaire when simply based on our criteria of the presence of normative data and a low percentage of overlap with common geriatric syndromes. Nonetheless, to our knowledge this questionnaire is only available in the German language and has as much as 78 items. For pragmatic reasons, therefore, one has to rely on less optimal alternatives, which are all provided by the present review. We present our recommendations for most optimal choices below, given the research objective.

Adjustment for the common geriatric syndromes is necessary to make age-related comparisons.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
