Abstract
Circadian misalignment (CM) is a sleep disruption where an individual’s circadian rhythm and actual sleep-wake cycle are unsynchronized. CM is frequently misdiagnosed and difficult to treat but can adversely impact short-term and long-term health outcomes. CM often interacts bidirectionally with chronic conditions affecting cognitive and physical functionality. Continuous physiological monitoring (CPM) of the hormone melatonin would support real-time, patient-driven interventions to address CM. However, patient-driven self-care tasks involve complex information processing and decision-making. Effective CPM use requires situation awareness regarding current circadian health and projected future status, sufficient background knowledge to identify circadian intervention alternatives, and selection of appropriate alternatives given their current state. Melatonin data has characteristics distinct from biomarkers previously subject to CPM, introducing further challenges around data presentation. Task analysis efforts can identify the cognitive processes involved in five different tasks associated with a continuous melatonin monitor to identify necessary informational elements to include in interface design.
Keywords
Introduction
Poor sleep and circadian health are associated with both short-term and long-term health effects, including impaired cognition, emotional regulation, functional performance, and increased risk for chronic illnesses such as cancer, heart disease, diabetes, and mental health disorders (Tubbs & Grandner, 2021). The circadian rhythm is the body’s central “internal clock” and drives several cyclical biological fluctuations (e.g., hormone levels, body temperature) over a roughly 24-hr period. Circadian misalignment is a type of sleep disruption arising from a lack of synchronization between the circadian rhythm and the sleep-wake schedule, leading to insufficient or fragmented sleep. Circadian rhythm disorders and misalignment also interact with existing chronic conditions. Recent research has highlighted bidirectional relationships between circadian misalignment and chronic conditions affecting the cognitive-physical boundary, including chronic pain disorders, mood disorders, traumatic brain injuries and post-concussive syndrome (PCS), metabolic disorders, and others (Boone et al., 2012; Fishbein et al., 2021; Neves et al., 2022; Warfield et al., 2021). This bidirectional relationship increases the risk of ongoing and progressively damaging health impacts to both circadian health and the chronic condition. Despite these important health outcomes, even clinical circadian misalignment can be difficult to detect and effectively treat; the syndrome is commonly misdiagnosed (Flynn-Evans et al., 2017). This can result in care delays and ineffective or even counter-productive treatment regimes.
Patient self-care consists of the cognitive, physical, and emotional tasks a patient performs themselves and is important in diagnosing, treating, and managing chronic conditions (Caldwell et al., 2021), including chronic sleep disruptions. However, for self-care tasks to be triggered, the patient must have adequate situation awareness (SA) for their condition: they must be able to detect health-related signals from their body and environment, make sense of those signals and what they indicate about their current health status, and project that information into the future to understand how it will affect them both in the short- and long-term. The patient must also know what intervention or self-care task alternatives are available to them and decide which is appropriate for their current situation. This represents a complex information processing and decision-making task that can be supported by a clinical care team, formal and informal caregivers, and wearable technologies such as continuous physiological monitoring (CPM) devices.
A continuous melatonin monitoring (CMM) device is one such technology that could support patient self-care to address circadian misalignment. This device is proposed to include a wearable sensor detecting melatonin in interstitial fluid and a patient-facing interface to display collected data and guide self-care tasks. Melatonin is a hormone that is one of several biomarkers that are closely tied to the circadian rhythm (Hilditch & Flynn-Evans, 2021). Melatonin levels generally rise prior to a habitual bedtime, peak a few hours after sleep onset, and decrease through the remainder of the night and upon light exposure after waking. However, the current state of melatonin monitoring utilizes plasma, salivary, or urine samples and relies heavily on the dim light melatonin onset protocol. This protocol is rigid and time-consuming. Further, most health insurance in the United States does not cover such evaluation. This financial barrier limits accessibility for various underserved populations. The results of the current assessment process provide information at a limited temporal grain size and generally for a single onset period; this information cannot be provided in real-time to support short-term circadian intervention (e.g., bright light therapy, exogenous melatonin supplements). CMM could address several of these limitations by increasing flexibility, increasing temporal sensitivity and range, and providing real-time feedback.
CMM offers a unique and non-invasive approach to monitoring the circadian rhythm, both in the short- and long-term. However, melatonin has different properties than biomarkers that have been previously tracked via CPM technologies (e.g., heart rate, blood glucose, blood oxygenation). While many of these previously utilized biomarkers have clear and well-established operating ranges, adequate melatonin levels can vary widely from person to person, and expected melatonin data features, such as the shape of the curve and the peak value, can vary from night to night depending on environmental and behavioral factors. Melatonin levels are highly variable across individuals, more so than biomarkers such as glucose. Thus, a single melatonin value for an individual is not informative or actionable. Melatonin data can be difficult for non-experts to interpret, with erroneous or misguided interpretations or incomplete information about intervention alternatives potentially leading to unsafe self-care interventions (e.g., unnecessary use of exogenous melatonin supplements). Due to these characteristics, the ideal data presentation approach for a CMM device is not yet self-evident.
Methods
A high-level task analysis was performed to identify an initial group of self-care tasks that CMM could enable. This was informed by consultation with an expert sleep researcher and an analysis of continuous glucose monitors (CGMs). CGMs function in a similar manner to the proposed CMM system, measuring interstitial glucose levels and displaying the data pattern on patient mobile devices. Thus, a review of commercially available CGMs (e.g., Dexcom G7, FreeStyle Libre) was conducted, and it focused on types of data displays and what information is being presented. This provided a reasonable analog for the capabilities and limitations of information displays of a CMM interstitial device, especially in terms of data and calculated metrics, though the underlying sensor technologies are distinct. The task analysis focused primarily on prospective user (i.e., patient) goals that would not be accomplishable without the presentation of various features of melatonin data (e.g., melatonin onset and offset time, the melatonin phase curve) to capture tasks specific to the CMM application. Additional goals and tasks that do not require melatonin data or data presentation, such as enhancing sleep or circadian health literacy or utilizing a digital sleep diary, are out of the scope of this analysis. Further, the task analysis focused on identifying “beyond-the-screen” tasks that would trigger or guide self-care interventions; “at-the-screen” levels of analysis – such as entering information – are not within the scope.
Patient self-care, including management of circadian misalignment, requires complex information processing and decision-making tasks. A context-specific information processing model (IP-CMM) was developed to capture the nuances of utilizing a CMM device. This model consisted of two components: the CMM interface system model and the adapted patient information processing model. The CMM interface model is informed by CGM data flows, implementations, and patient care tasks. Information processing and decision-making elements of IP-CMM are based on broader aspects of cognitive situation awareness processes (Caldwell et al., 2021). However, it also incorporates specific skills and competencies critical to health data interpretation, including data and graph literacy and domain-specific health literacy. The interaction between the data flows from the CMM system model and the adapted information processing model emphasizes the ways in which various stages of information processing and decision-making can be supported by interface components.
Finally, the identified tasks were mapped onto the IP-CMM to identify associate cognitive processes with each task. For example, some tasks may aim to simply understand the patient’s current state without necessarily being associated with the full decision-making process. This mapping offers initial insight into the types of information, such as raw data, calculated metrics, machine learning (ML)-augmented data, and supplementary sleep and circadian education content, that is necessary or valuable for successful task completion. In this analysis, it was not assumed that the user is a subject-matter expert in sleep and circadian health, and they may require additional information to accurately interpret the information. This analysis was then furthered by considering the specific information necessary for each task, allowing us to iterate on the IP-CMM.
Results
The task analysis identified a total of five initial data-related goals that a prospective CMM user may want to accomplish using the wearable device. Three tasks were directly identified from the CGM interface analysis; these include detecting and differentiating circadian misalignment from other sleep disorders (T1), detecting and understanding the impact of self-intervention decisions on the circadian rhythm (T2), and planning an intervention for either immediate or delayed implementation (T3). The problem context itself (i.e., supporting patient self-care, especially in individuals with both circadian misalignment and an interactive chronic condition) can inform a fourth task. This involves exploring the relationships between circadian misalignment and other short-term health outcomes like chronic condition symptoms and impaired mood or cognition (T4). The sleep expert consultation re-identified several of the tasks that were already described above and one additional task: predicting when they (i.e., the patient user) will feel tired (T5). Some of these tasks are dependent on the performance of “at-the-screen” tasks such as self-intervention and symptom logging; however, these tasks are not considered here.
The final version of the IP-CMM model is depicted in Figure 1. The CMM system definition depicts the flow and transformation of information. Raw data (C.1) is received from the sensor and subject to feature extraction (C.2), enabling critical melatonin metrics (C.3) to be calculated. According to the sleep expert, metrics of interest include but are not limited to melatonin onset time, peak time, and offset time. Raw data, metrics, and sleep and circadian health information (C.5) could then be further analyzed using a naïve algorithm or ML (C.6), though the effectiveness and accuracy of these methods may vary. This could reasonably provide the output of projected melatonin patterns (C.7) and personalized recommendations for self-care interventions (C.8). These CMM interface data elements support different cognitive processes necessary to complete tasks.
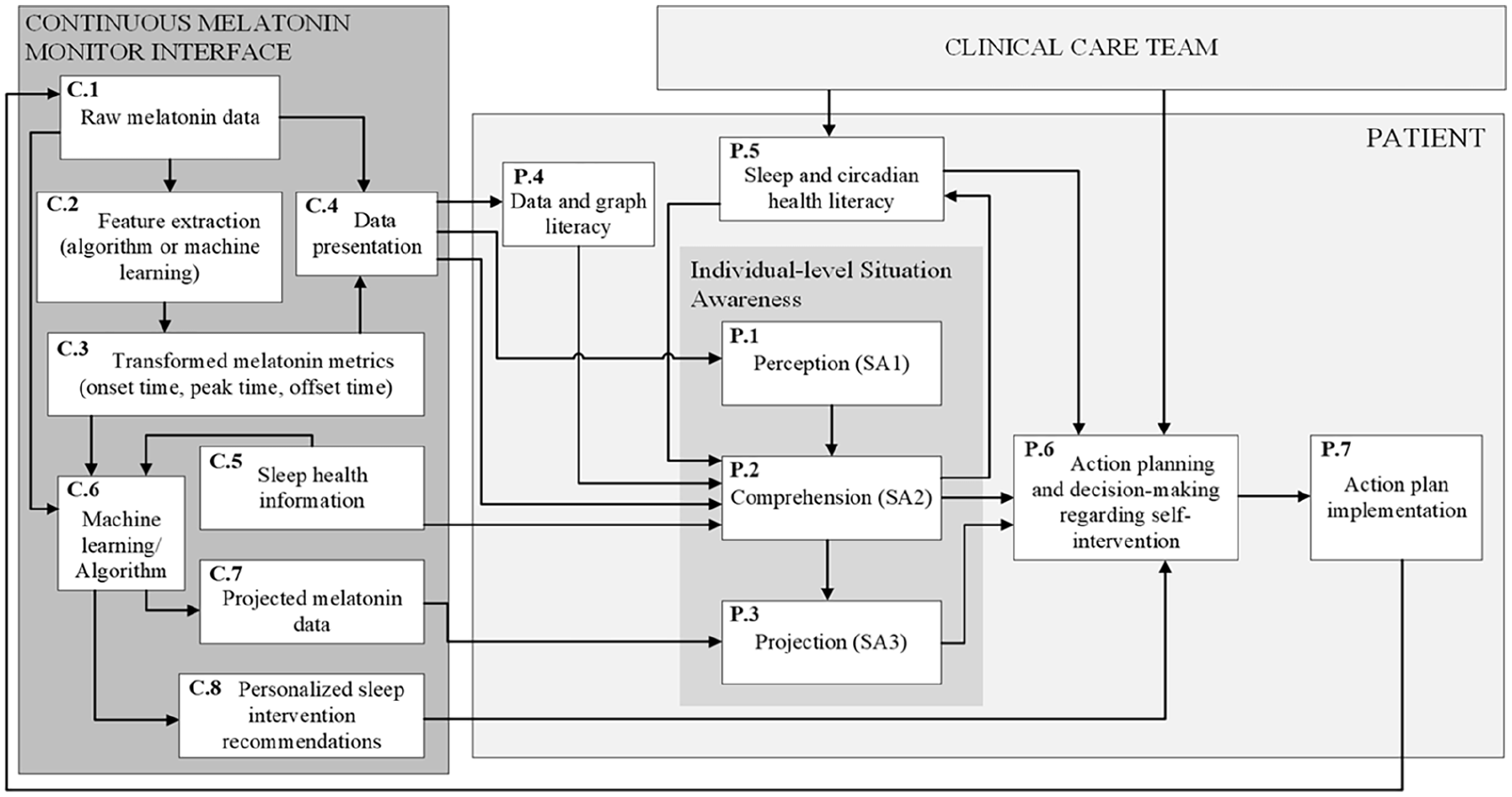
A model of the data and information flows and transformations associated with a continuous melatonin monitoring (CMM) device interface (C.1–C.8) integrated with a context-specific patient-user information processing model (P.1–P.7) that integrates competencies relevant to health data interpretation, such as sleep and circadian health literacy as well as data and graph literacy.
Raw data and calculated metrics are displayed to the user through information presentation methods (C.4). This information is subject to the user’s data and graph literacy (P.4), with lower literacy levels potentially impairing their ability to interpret the information accurately. The presented data influences perception (P.1) by providing signals for the patient to detect. These signals could be enhanced through features like push notifications. The comprehension stage (P.2) is triggered by perception and integrates information from various sources with the goal of the user making sense of their current circadian health status. Critical sources of information include (but are not strictly limited to) the CMM data presentation (C.4), interface-based sleep and circadian health information (C.5), and the patient’s memory, which drives both data and graph literacy as well as sleep health literacy. During the comprehension stage, the user makes sense of their current state. The projection SA level can be triggered by comprehension and involves projecting their current status into the future; this can be informed through the information sources supporting comprehension and could be further augmented by presenting projected melatonin data. Comprehension and projection are critical to decision-making, which involves considering the available alternatives (i.e., circadian health literacy competency retrieved from long-term memory) and selecting the most appropriate alternative based on their understanding of circadian health, their current health status, and clinician recommendations. Following decision-making, the action plan can be implemented; it is critical to note that the self-care task as performed may not be identical to the action plan as originally planned. Self-care intervention implementation provides a feedback loop through its effect on melatonin, which is reflected (subject to a delay) in the raw data. Sleep and circadian health literacy (i.e., the skills, knowledge, and beliefs associated with sleep and circadian health) can be expanded, reinforced, or revised through information provided by clinician teams or indirectly through interaction with educational materials or personal experience.
The tasks require the use of different cognitive processes and can be mapped to the IP-CMM. This process can be used to identify the necessary types of information. Table 1 summarizes this information in relation to the processes depicted and enumerated in Figure 1. Feature extraction (C.2) and application of an algorithm or ML to generate projected melatonin data or personalized recommendations (C.6) are both critical to data transformation processes but do not directly inform task completion. Tasks T1, T2, and T4 emphasize establishing an understanding and do not necessarily include projection into the future, identifying alternative interventions, decision-making, or action plan implementation. These tasks may trigger these processes, but in this case, these tasks would transform into T3. Similarly, while T5 does utilize the cognitive process of projection, it does not include action planning, decision-making between intervention alternatives, or action plan implementation, though it may trigger a T3 task. That said, a decision-making component within the comprehension stage is associated with determining that circadian misalignment is present, for example. All tasks require sufficient data and graphical literacy to interpret the interface’s data and metrics as well as sufficient sleep and circadian health literacy to guide the interpretation and integration of the data, metrics, and qualitative information (i.e., sleep health information, personalized recommendations).
Summary of Tasks, Their Descriptions, and the Cognitive and Continuous Melatonin Monitoring (CMM) Interface Components From Figure 1 That They Map to.
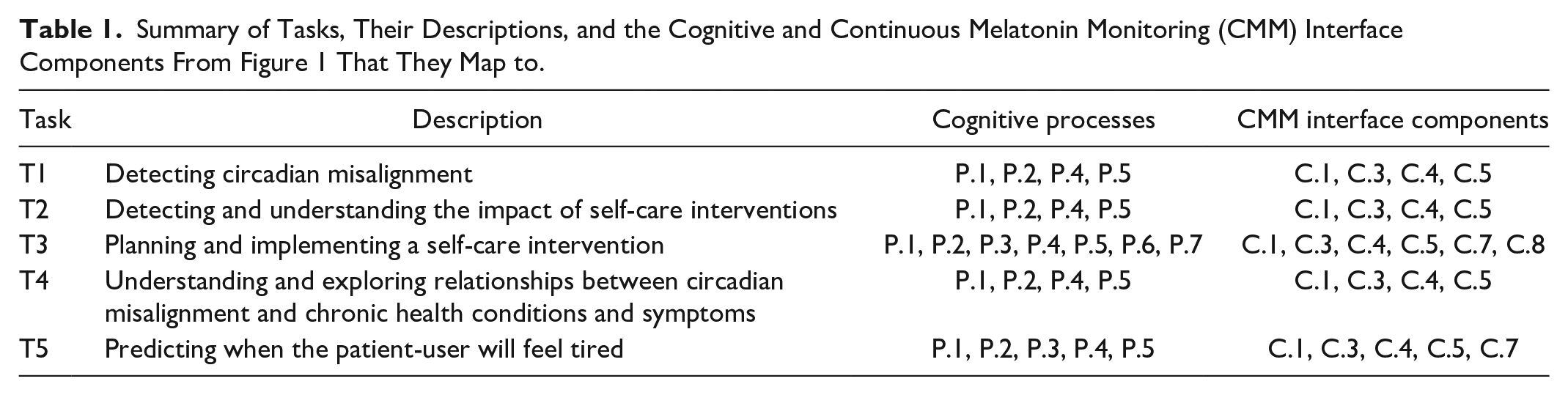
T3 and T5 would benefit from the algorithmic or ML depicted in C.6 in Figure 1. For T3, projected melatonin data could help users more easily determine the impact of taking action (or not), and personalized recommendations could decrease the cognitive load associated with integrating information and selecting an alternative by offering a default option that the user can choose to enact. In T5, projected melatonin data or onset times also decrease mental load and information integration requirements, thus supporting task completion.
Discussion and Conclusions
Systematically relating both user goals and prospective interface components to the human information processing model offers a means of initially identifying features (i.e., data transformations, supplementary information placement) that may be relevant to users even in novel technology contexts. In the CMM context, the display and effective presentation of melatonin data to support the patient’s signal detection and comprehension is critical to all five user tasks. Mapping these data-driven tasks to a context-specific information processing model highlights what processes need support to complete which tasks. For example, the projection level of situation awareness may benefit from support when a patient is identifying when they may feel tired tonight, but the same type of support may not be useful for tasks such as identifying circadian misalignment or understanding how different environmental conditions and behavior affect the circadian rhythm.
Expert analysis and “beyond-the-screen” task analysis can identify critical functional capabilities and user interface design priorities in a novel form of CPM. This can inform the early design process and early interactions with the prospective user base. For example, the tasks identified, described, and mapped to a context-specific information processing model inform a preliminary needs assessment and usability assessment with prospective patient users. The formalized identification of these data-driven tasks and the raw and transformed data necessary to support them have enabled the development of preliminary wireframes. These wireframes include multiple data presentation alternatives, including the inclusion or exclusion of algorithmically or ML-based data transformations, and will be presented to needs assessment participants. These participants will then be asked to complete the different tasks identified above utilizing the example wireframes, which will provide insight into prospective user needs, preferences, sleep and circadian health literacy, and graph and data literacy. This early investigation and design iteration specifically focused on data presentation efforts is critical in the case of CMM because melatonin’s characteristics make it difficult to interpret and different from biomarkers that are more commonly subject to CPM technology and that may be more familiar to the user base.
Ultimately, this analysis represents the first of a multi-step process to develop tools that increase patient empowerment through support of sleep health management. The methodology can be applied early in the design process for a variety of tools but may be especially useful when developing novel technologies and where the prospective user population may have limited exposure to analogous tools or types of data.
Footnotes
Acknowledgements
The authors would like to thank Dr. Michelle Garrison and the Sleep Equity Lab in Purdue University’s Department of Public Health for their ongoing collaboration on this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Partial support for the first author was provided by the Purdue Doctoral Fellowship and John W. Anderson Scholarship.
