Abstract
Vigilance refers to an individual’s ability to maintain attention over time. Vigilance decrement is particularly concerning in clinical environments where shift work and long working hours are common. This study identifies significant factors and indicators for predicting and monitoring individuals’ vigilance decrement. We enrolled 11 participants and measured their vigilance levels by recording their reaction times while completing the Psychomotor Vigilance Test. Additionally, we measured participants’ physiological responses and collected their sleep deprivation data, demographic information, and self-reported anxiety levels. Using repeated-measures correlation analysis, we found that decreased vigilance levels, indicated by longer reaction times, were associated with higher electrodermal activity (p < .01), lower skin temperature (p < .001), shorter fixation durations (p < .05), and increased saccade frequency (p < .05). Moreover, sleep deprivation significantly affected vigilance decrement (p < .001). Our findings provide the potential to develop a predictive model of vigilance decrements using physiological signals collected from non-intrusive devices, as an alternative to current behavior-based methods.
Keywords
Introduction
Vigilance describes an individual’s capacity to maintain focus over time (Smith et al., 2023; Warm et al., 2008). Vigilance decrement is the gradual decline in performance on tasks requiring sustained attention, typically occurring after extended periods of 5–15 min (van Schie et al., 2021; Warm et al., 2008). This decline manifests as slower response times and reduced accuracy in detecting signals or events (van Schie et al., 2021).
While vigilance decrement is problematic in various high-risk work environments, it is particularly concerning in clinical settings where shift work and long working hours are common (Basner et al., 2017; Fowler & Ellis, 2019). For instance, reports indicate that 84% of medical residents’ sleepiness scores are comparable to, or worse than, those of individuals with diagnosed sleep disorders (Barger et al., 2006; Papp et al., 2004). In clinical environments, monitoring and predicting vigilance decrement is critical due to vigilance decrement’s impact on clinicians’ productivity, patient safety, and the overall quality of healthcare.
Current approaches to detecting vigilance decrement often rely on behavioral responses. Common behavioral responses used to capture individuals’ vigilance decrement include reaction time (Körber et al., 2015; Langner et al., 2010; Mueller et al., 2020), error rates (Mueller et al., 2020), and detection rates (McCarley & Yamani, 2021; Szalma et al., 2006). However, these behavioral measures are limited in their usefulness. For one, they are typically post-hoc and cannot adequately address the need for immediate detection and intervention in clinical settings or other safety-critical environments. In these situations, an error or delayed response can have severe consequences. It is therefore imperative to supplement existing behavioral methods with methods that can provide timely notification of vigilance decrement to operators, allowing them to prevent or mitigate the errors resulting from vigilance decrement.
An alternative approach to identifying vigilance decrement is physiological measurements. Unlike behavioral measures, physiological measurements provide continuous and real-time data. Eye movements, including pupil diameter and saccade metrics, can indicate changes in an individual’s level of vigilance (Bodala et al., 2016; McIntire et al., 2013). Physiological signals related to autonomic nervous system activity, including electrodermal activity (EDA), skin temperature (ST), and heart rate (HR), are known to be associated with vigilance (Al-Shargie et al., 2019; Lara et al., 2018; Posada-Quintero & Bolkhovsky, 2019). Together, these physiological measurements offer a comprehensive view of an individual’s vigilance level and can capture subtle changes that may not be detected by behavioral measures.
Another key limitation of previous studies on vigilance is that most do not simultaneously account for individual differences and contextual factors associated with vigilance decrement. Indeed, research in this area has been fragmented, with separate studies highlighting the impact of sleep quality and deprivation (Weinger & Ancoli-Israel, 2002), demographics (Blatter et al., 2006; Rose et al., 2010), and psychological factors like anxiety (Meeker et al., 2021) on vigilance decrement.
Recognizing the need to develop accurate and reliable methods for monitoring and predicting clinicians’ vigilance decrement, our aims for this study were 2-fold. First, we aimed to identify individuals (including demographics and anxiety scores) and contextual factors (sleep quality and deprivation) related to their vigilance levels. Second, we sought to identify physiological signals that could be used for real-time vigilance decrement detection in clinical settings.
Methods
Participants
We recruited a total of 11 participants, including internal medicine residents and non-clinicians, to participate in this study at the University of Washington. The group consisted of five men and six women, with an average age of 29.91 years (SD = 11.74). We excluded individuals who had visual or hearing impairments, as well as those with photosensitive epilepsy or a history of sleep disorders. All 11 participants had normal or corrected-to-normal eyesight. The study was approved by the institutional review board at the University of Washington.
Apparatus
Experiment Space Setup
The experiment was conducted in the Human and System Lab at the University of Washington. The lighting conditions were controlled to ensure consistency across participants by blocking out natural light and using the same lighting setup. Participants conducted the experiment on a 15-inch Dell laptop with a resolution of 1,920 × 1,080. The laptop was elevated off the desk using a laptop stand to bring it to eye level for participants.
Eye Tracker
We used a Tobii Pro X3-120 eye tracker (Tobii) to collect participants’ eye movement data at a sampling rate of 120 Hz. The eye tracker was mounted below the screen of the laptop. The eye movements were recorded using the Tobii Pro Lab software (Tobii). To help ensure high-quality data, participants were instructed to maintain a consistent distance (approximately 40 cm) from the screen after calibration.
Wristband
We utilized the E4 wristband (Empatica) with sampling rates of 64, 4, and 1 Hz to collect participants’ EDA, ST, and HR data, respectively. To minimize noise from arm movements, participants were asked to wear the wristband on their nondominant hand throughout the experiment.
Psychomotor Vigilance Test (PVT)
PVT is a standardized tool widely used to assess individual vigilance in experimental and clinical settings (Basner & Dinges, 2011). It consists of a simple, monotonous task in which participants are prompted to press the spacebar every time they see a red dot appear on the screen. The red dot appears at random time intervals between 1 and 5 s.
Variables
Sleep Diary
We used a sleep diary to record participants’ sleep duration and sleep quality rating during the 24 hr prior to each visit to the experimental space. The sleep quality score was ranging from 1 (Very poor) to 4 (Very good).
State-Trait Anxiety Inventory (STAI)
The STAI (Spielberger et al., 1971) is a tool widely used to measure two types of anxiety: state anxiety, which is a temporary emotional condition related to current circumstances, and trait anxiety, a more long-term disposition toward experiencing anxiety. We applied a short form of the STAI developed by Zsido et al. (2020), which consists of five statements about state anxiety and five statements about trait anxiety. Participants were asked to rate their agreement with each statement using values ranging from 1 (Not at all) to 4 (Very much so). The general anxiety level was calculated by averaging the scores from all the state and trait anxiety questions.
Behavioral Responses
In this study, participants’ vigilance decrement was measured by their reaction time while completing the PVT. Reaction time was defined as the time interval between the appearance of the red dot on screen and the moment the participant responded by pressing the spacebar. This reaction time was recorded in milliseconds.
Physiological Responses
We collected three types of physiological responses using the E4 wristband: EDA, ST, and HR. Additionally, six variables were extracted from the participants’ eye movement data: left and right pupil diameters, average fixation duration, number of fixations, average saccade duration, and number of saccades. Given the high likelihood of similar patterns between the left and right pupil diameters, we used the average of both pupils’ diameters in our data analysis.
Procedures
We used a within-subject design in this study. Each participant visited the experimental space twice: once with over 6 hr of sleep the previous night (the low sleep-deprived condition) and once with under 5 hr of sleep (the high sleep-deprived condition). These visits could happen in either order. On each visit, we first explained the experimental procedure to the participant. We then guided them in filling out the sleep diary and STAI survey. We also collected their demographic information and caffeine intake on the day of the experiment. After that, we assisted the participant in putting on the E4 wristband and completing the eye tracker calibration. Each participant had a one-minute practice session to familiarize themselves with the PVT. Following the practice session, the participant completed a 15-min version of the PVT. Each visit lasted about 30 min in total. The same procedure was followed for each visit.
Data Preprocessing
Before analyzing the behavioral and physiological data, we performed data preprocessing. We first converted the Unix timestamps from the E4 wristband and eye tracker data to local time. To align these two types of physiological signals, we down-sampled them to 1 Hz by averaging the values per second, as the lowest sampling rate between them was 1 Hz. We calculated the number of fixations and saccades as the total per second. Next, we aggregated the data into 1-min intervals. Afterward, we synchronized the PVT data with the E4 wristband and eye tracker data using the local timestamps. For each participant’s STAI questionnaire responses, we calculated the average score across the ten questions.
Results
Descriptive Statistics
We summarize the mean values and standard deviations for each type of response under the two conditions in Table 1. Before the non-sleep-deprived visits, participants had had an average of 7.508 hr of sleep and an average score of 3.182 for their sleep quality. Before the sleep-deprived visits, they only had had an average of 4.608 hr of sleep and 1.678 for sleep quality.
Descriptive Statistics of Sleep-Deprived and Non-Sleep-Deprived Conditions.
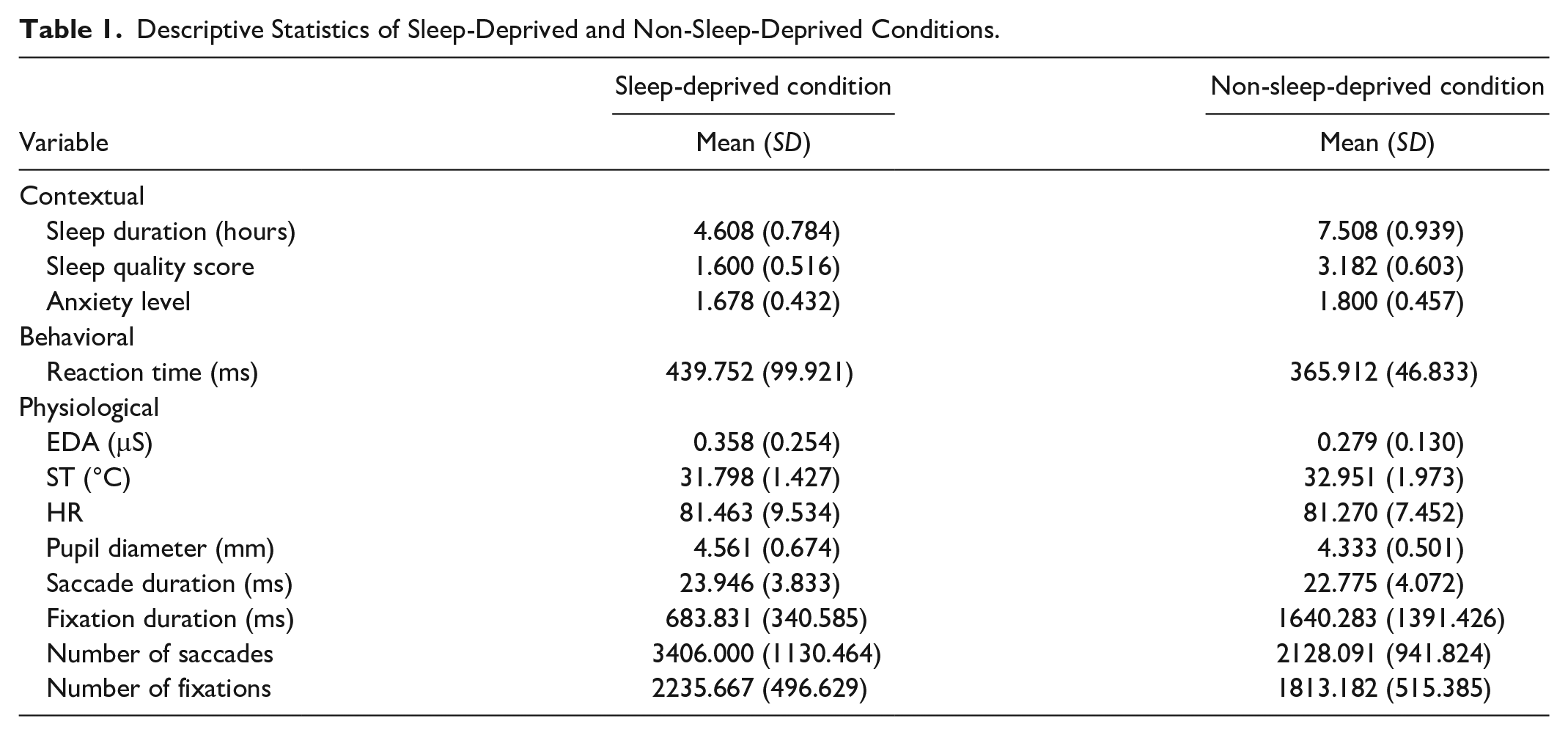
We applied paired Wilcoxon signed-rank tests to compare physiological responses and reaction times between the sleep-deprived and non-sleep-deprived conditions. When participants were sleep-deprived, they had longer reaction times (p < .001, W = 6075), indicating a lower level of vigilance, compared to when they were not sleep-deprived while completing the PVT. They also showed significantly higher EDA (p < .001, W = 5431), lower ST (p < .001, W = 1394), shorter fixation duration (p < .001, W = 1102), and more fixations (p < .001, W = 4540) and saccades (p < .001, W = 4677) under sleep-deprived conditions.
Relationships Among Contextual, Behavioral, and Physiological Variables
We identified a significant negative correlation between sleep duration and sleep quality rating of the night before the PVT and reaction time using repeated-measures correlation, indicating that shorter sleep durations (p < .001,
To identify potential physiological indicators of vigilance levels, we analyzed the relationship between reaction time and physiological response data collected by the E4 wristband and eye tracker. We observed that longer reaction time is correlated with higher EDA (p = .006,
Discussion and Conclusion
In this study, we found that physiological measures, including EDA, ST, fixation duration, fixation frequency, and saccade frequency, can be effective for the real-time monitoring of clinicians’ vigilance levels. These signals can be collected from non-intrusive devices, which are less disruptive to clinicians while they perform their primary tasks (Greenlee et al., 2020). Additionally, we found negative impacts of sleep deprivation on vigilance performance. These findings highlight the importance of integrating sleep-related factors into vigilance level modeling for clinicians.
Our findings will contribute to developing a predictive model of vigilance decrements using physiological signals collected from non-intrusive devices, potentially replacing current behavioral response-based methods. Such a predictive model will be able to alert clinicians when their vigilance levels are likely to decrease, enabling timely interventions to prevent medical errors and accidents. Interventions such as improving shift work strategies and utilizing relaxation and mindfulness techniques (Robbins et al., 2021) could enhance clinicians’ vigilance performance, potentially leading to better patient care and safety.
This pilot study has several limitations that need to be addressed in future research with clinicians. First, the sample size of this pilot study is small, and the demographics of the non-clinician participants do not fully represent those of medical residents. Second, the PVT may not fully capture the nature of prolonged tasks that lead to vigilance decrements in clinical settings. In future studies, we will develop a domain-specific task tailored to medical residents’ daily work and expertise. Third, the time-dependent changes in reaction time suggest the need for dynamic modeling to monitor and predict vigilance decrements over time more accurately. Our future research will explore dynamic modeling techniques, such as dynamic Bayesian networks, to improve the accuracy of vigilance level modeling.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the National Science Foundation under Grant No. 2237661.
