Abstract
Decrements in sensorimotor performance due to vestibular challenges associated with spaceflight are well characterized, and these impairments pose a significant risk to astronaut safety and mission success. However, the impact of fatigue on this impairment is unknown. We hypothesized that the interaction of fatigue and vestibular impairment would lead to greater-than-additive detriments to static and dynamic balance. Thirteen subjects (9 m/4 f) completed a series of sensorimotor tests (static and dynamic tasks, with and without galvanic vestibular stimulation) before and after a fatigue-induction task. Contrary to the hypothesis, the combination of fatigue and stimulation did not lead to increased impairment in sensorimotor task performance. Subjects displayed improved performance under GVS following the fatigue-induction task during eyes-open conditions, but not during eyes-closed or cognitive dual-task conditions, suggesting that the availability of alternate sensory cues as well as an astronaut’s level of mental workload can impact adaptation to vestibular challenges associated with spaceflight.
Introduction
Mental fatigue is a neurobiological state associated with prolonged exposure to mentally taxing activities (Van Cutsem et al., 2022), likely due to persistent global network changes (Lim et al., 2010) and decreased coordination in the brain (Karthikeyan et al., 2022). It is often associated with time-on-task performance decrements (Karthikeyan et al., 2022), feelings of drowsiness and exhaustion, and increased distractibility (Ishii et al., 2014), and it has been previously shown to impact gait, sway, and balance, and to increase the risk of falls in older adults (Brahms et al., 2022; Grobe et al., 2017; Hachard et al., 2020). During spaceflight, astronauts frequently engage in mentally taxing activities that can lead to fatigue, and it is critical to understand how this impacts their performance in order to ensure mission success and astronaut safety.
Decrements in sensorimotor performance due to vestibular challenges associated with spaceflight are well characterized (M. F. Reschke & Clément, 2018; Wood et al., 2011). Following G-transitions (i.e., takeoff and landing), astronauts frequently display motion sickness and impairments to balance, locomotion, and gaze stabilization (M. F. Reschke & Clément, 2018). These impairments pose a significant risk to astronaut safety and mission success, compounded by the critical nature of many tasks that immediately follow G-transitions (e.g., docking a spacecraft to the ISS). Further, with NASA’s plans to return humans to the moon and eventually land humans on Mars, post-flight sensorimotor impairment poses an even greater risk during extraterrestrial landings, with no landing crew present to support the astronauts after their descent. While sensorimotor impairment during and after spaceflight is well characterized, the impact of fatigue on this impairment is unknown.
The brain relies on three major inputs to maintain balance and interpret movement and position: visual cues, proprioceptive feedback, and vestibular inputs (Dixon & Clark, 2020). On Earth, the brain takes into account the constant inputs to the vestibular system from the acceleration due to gravity (Merfeld et al., 1999). Upon entry into a microgravity environment, this input is removed, and the mental model for processing signals from the vestibular system degrades, resulting in disorientation and sensorimotor performance decrements (Shelhamer, 2015). When the vestibular system is impaired, the brain must adapt to rely heavily on vision and proprioceptive feedback for these functions (Sprenger et al., 2017). Over time, the astronaut adapts to these altered inputs, through a central reinterpretation of vestibular otolith afferent signals (Dixon & Clark, 2020), but upon return to earth, these changes become maladaptive and lead to further sensorimotor challenges (Clément et al., 2022).
We hypothesize that this reweighting of sensory inputs and reinterpretation of otolith signals imposes increased mental demands, and therefore, under states of mental fatigue, a person with vestibular impairment will demonstrate greater deficits in balance and sensorimotor function. The goal of this study was to explore this interaction and test this hypothesis using galvanic vestibular stimulation (GVS) (Moore et al., 2006) as an analog for spaceflight-induced vestibular challenges. This analog was recently evaluated with a sample of five crewmembers who subjectively rated it as duplicating 50% to 90% of their early post-landing vestibular disruption (Moudy et al., 2024).
Methods
Participants and Recruitment
A total of 13 participants (9 m/4 f) were recruited from university students and faculty. Mean age of subjects was 24.5 years (SD: 3.97), and subjects reported no history of cardiovascular or neurological disorders and no metal implants. Subjects were instructed to refrain from caffeine consumption for 12 hr prior to the start of the study. Participants provided informed consent with approval from the Texas A&M Institutional Review Board (IRB2022-0933).
Study Protocol
Participants completed a one-day study, which lasted approximately 5 hr (Figure 1). After providing informed consent, subjects were fitted with Galvanic Vestibular Stimulation (GVS) electrodes, placed over the mastoid processes (i.e., bony regions behind the ear). Large 4.5 × 4.5 cm carbon flex electrodes, placed within a tDCS-grade sponge (5 × 7 cm), were used to apply GVS. Saline solution and a small amount of medical-grade gel were applied to the sponges to improve contact with the skin. Each participant completed a sensorimotor test battery based on NASA’s standard measures for sensorimotor assessment (Miller et al., 2018; M. Reschke et al., 2015; M. Reschke et al., 2016), consisting of the tandem walk, the eyes-closed tandem walk, and a balance assessment (Horn et al., 2015; Quijoux et al., 2021). We also introduced a new dual-task tandem walk, to measure performance when cognitive reserves are depleted.
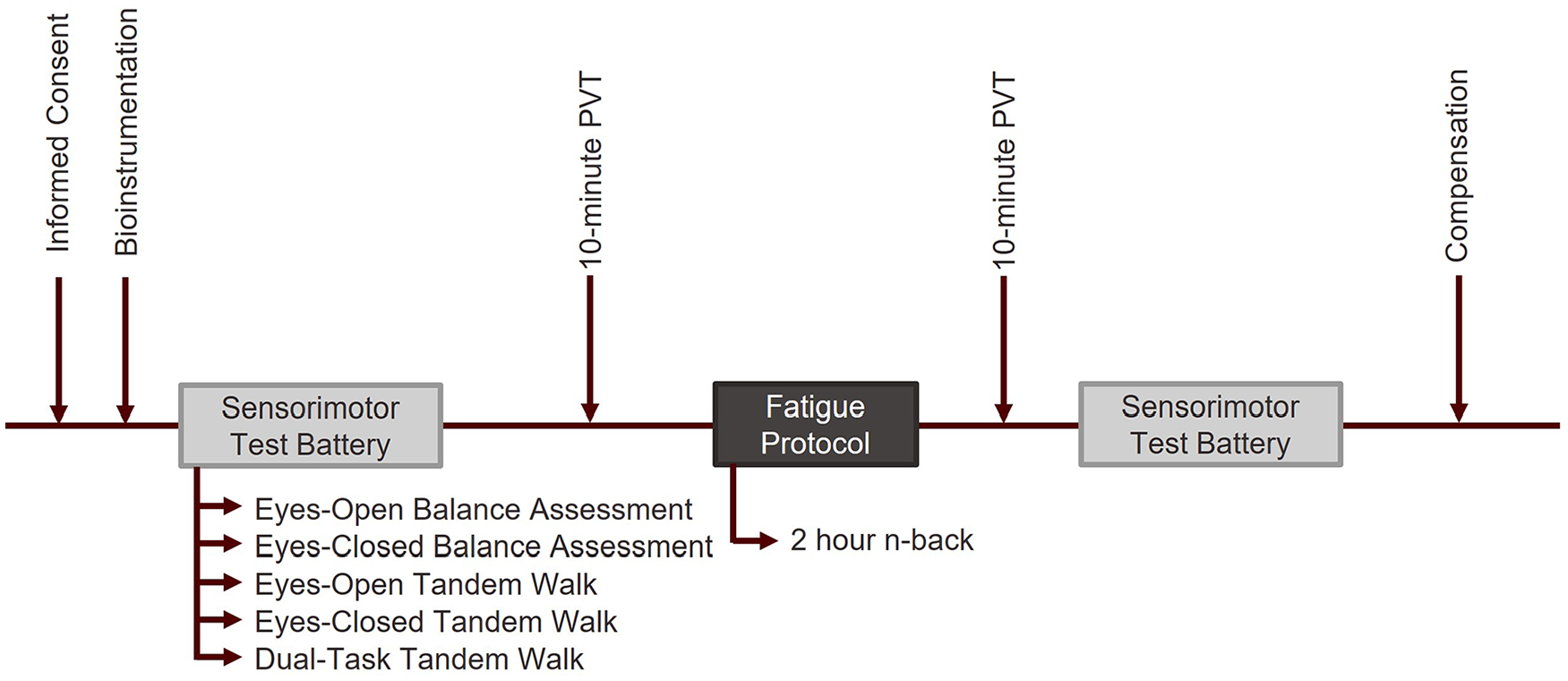
Study protocol.
Subjects completed two trials of each test with active GVS—using a pseudorandom sum-of-sines waveform with a maximum amplitude of 2 mA and frequency near 1 Hz (Moore et al., 2006)—and two trials without GVS. Participants completed a mental fatigue induction protocol, consisting of a 2-hr two-back task—a working memory task previously shown to successfully induce mental fatigue (Karthikeyan et al., 2022). Before and after this task, subjects completed a 10-min Psychomotor Vigilance Test (PVT) (Drummond et al., 2005), as a measure of fatigue. Following fatigue induction, participants repeated the sensorimotor test battery.
Balance data was collected on an AMTI force plate (BP400600, AMTI, USA) at 1,000 Hz. Tandem walk trials consisted of ten steps per trial and were recorded with a digital camera for later analysis of performance. During the dual-task tandem walk, subjects were given a three-digit number and asked to repeatedly subtract seven and state the answer aloud while performing the task.
Measures and Statistical Analysis
All statistical analyses were conducted in R version 4.2.2 (R. Core Team, 2022). PVT and two-back data were used to validate successful fatigue induction. Reaction time and lapses were extracted from the PVT performance data. Three performance measures—sensitivity, specificity, and accuracy—were extracted from the two-back data in 10-min bins (Karthikeyan et al., 2021). The distribution of each measure was tested for normality using the Shapiro-Wilk normality test (Shapiro & Wilk, 1965), and the appropriate parametric or non-parametric test was applied. A paired t-test or paired Mann-Whitney was conducted on each PVT measure. A non-parametric test for repeated measures data was conducted on each two-back performance measure, using the R package nparLD (Noguchi et al., 2012). Post hoc tests were conducted using the mctp.rm function from the R package nparcomp (Konietschke et al., 2015).
Performance measures extracted from the sensorimotor tests were analyzed to examine the impacts of fatigue and GVS. Videos of each trial of tandem walk were scored by a review blinded to the condition of the trial. For each trial, the reviewer quantified how many steps (out of 10) met the requirements of the test (i.e., heel within 2 cm of toe, no side stepping, no wide swinging of leg). Mean sway velocity in the anteroposterior and mediolateral directions, and sway area were extracted from the balance data (Quijoux et al., 2021).
Each measure was tested for normality using the Shapiro-Wilk normality test (Shapiro & Wilk, 1965), and the appropriate parametric or non-parametric analysis was conducted. A two-way repeated measures ANOVA or a two-way non-parametric test for repeated measures data (Noguchi et al., 2012) was conducted for main effects of phase and stimulation condition and for interaction effects. Post hoc tests for ANOVA were conducted using paired t-tests, while post hoc tests for the non-parametric tests were conducted using a paired studentized permutation test (R function npar.t.test.paired) from the R package nparcomp (Konietschke et al., 2015).
Results
Using a Bonferroni-corrected alpha of .025, significant increases in both response time (p < .01) and lapse count (p < .01) were identified from the PVT data. Using a Bonferroni-corrected alpha of .0167, a significant effect of phase was identified for specificity (p < .01), but not for accuracy or sensitivity in the two-back data. Post-hoc analysis found that specificity during the seventh and tenth block of two-back were significantly lower than the first block (Figure 2).
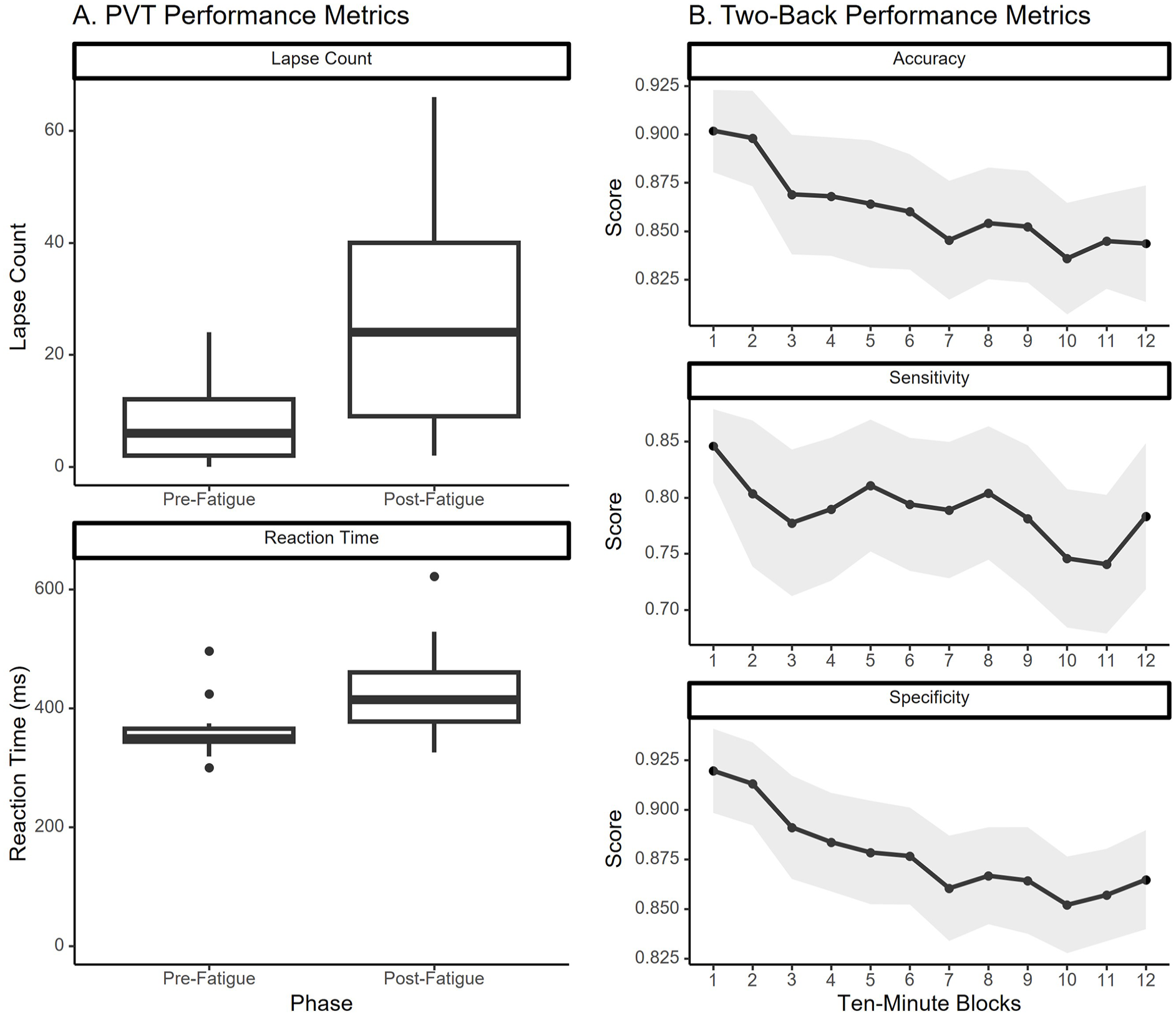
(a) PVT performance (b) Two-back performance (mean with shaded standard error).
Significant results for the balance data (Figure 3) were identified by a Bonferroni-corrected alpha of .0083. A significant main effect of stimulation was identified for mean sway velocity in the AP (p < .001) and ML (p < .001) directions, and for sway area (p < .001) for both the eyes-open and eyes closed conditions. A significant phase × stimulation condition interaction effect was found for mean sway velocity in the AP (p < .001) and ML (p < .001) directions, and for sway area (p < .0083) for the eyes-open conditions only. Post-hoc tests, corrected for multiple comparisons, found that sway velocity in the AP and ML directions decreased under fatigue during the GVS condition but not the no-GVS condition. Additionally, exposure to GVS increased sway area and sway velocity in both the AP and ML directions before fatigue but not after. Significant results for the tandem walk data (Figure 4) were identified by a Bonferroni-corrected alpha of .0167. No main effects of phase (pre/post fatigue) were identified. Subjects completed significantly fewer correct steps while under GVS compared to no GVS (main effect of stimulation) during the eyes open (p < .001), eyes closed (p < .001), and dual task (p < .001) tandem walk tests. There was a significant phase x stimulation condition interaction effect for the eyes open (p < .001) and eyes closed (p < .0167) tests, but not for the dual task test. Post hoc tests, corrected for multiple comparisons, found that during the eyes open task the number of correct steps significantly increased following the fatigue induction task during the GVS condition but not during the no-GVS condition, and that, both before and after fatigue, subjects performed more correct steps when GVS was turned off compared to when it was turned on. Additionally, during the eyes closed tandem walk, the number of correct steps did not change after fatigue for either stimulation condition (GVS, no-GVS), and both before and after fatigue, subjects performed more correct steps when GVS was turned off compared to when it was turned on.
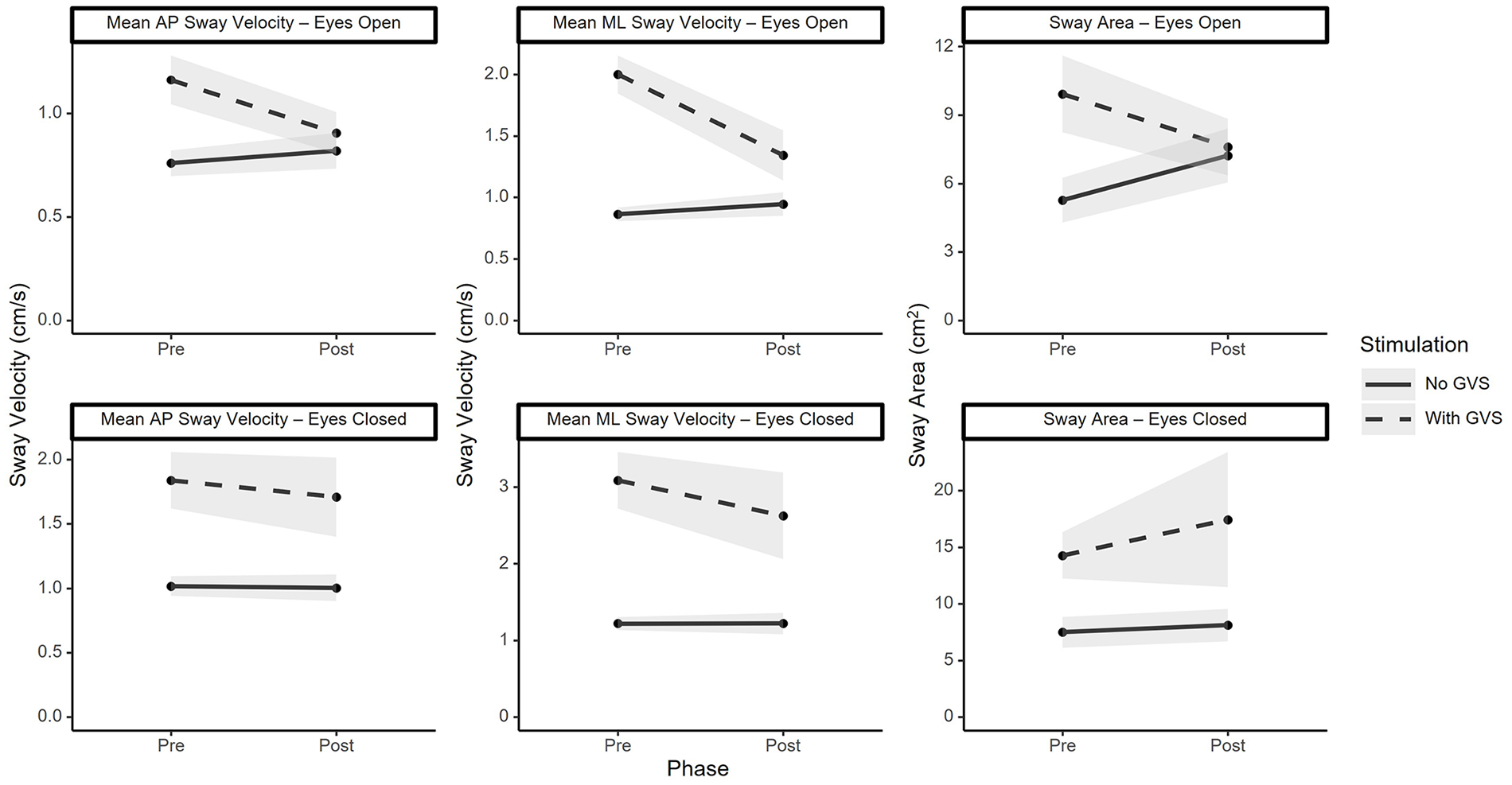
Performance metrics for eyes-open and eyes-closed balance tasks (mean with shaded standard error).
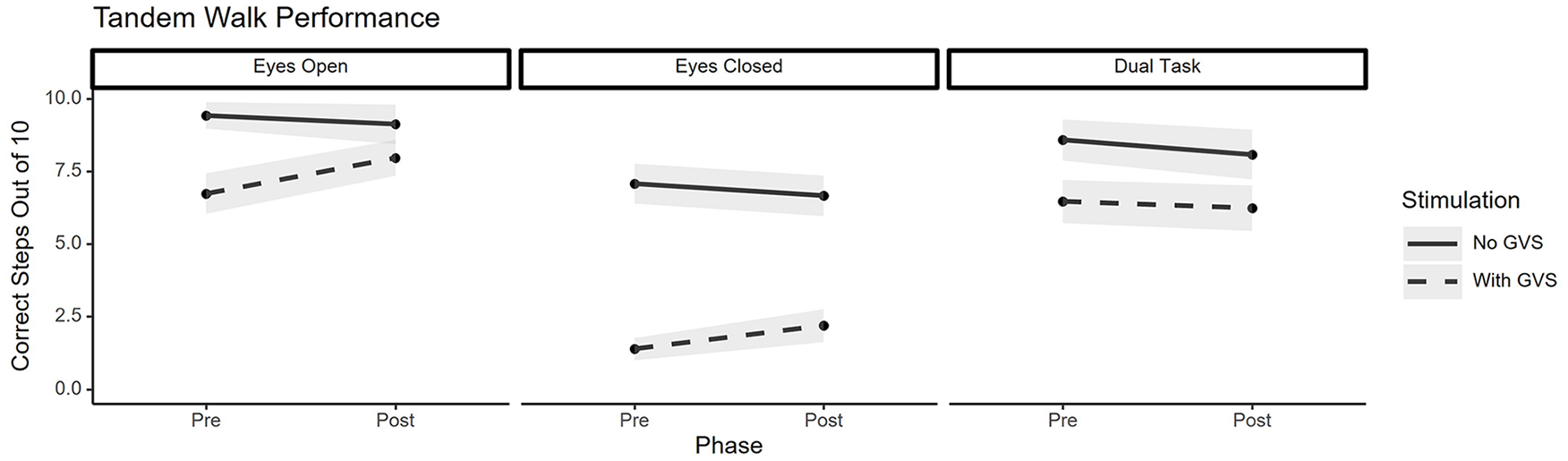
Performance during tandem walk task (mean with shaded standard error).
Discussion
Performance metrics from the PVT and two-back tests were used to validate successful induction of fatigue (Figure 2). Increased reaction time and lapse count on the PVT are consistent with fatigue, and the decrease in specificity during the two-back is especially characteristic of mental fatigue, as response inhibition is known to be impaired by mental fatigue (Guo et al., 2018).
We found that in the absence of GVS neither static nor dynamic balance declined under fatigue. While this does not match previous literature, which has reported balance decrements associated with fatigue (Brahms et al., 2022), prior studies reported relatively small effect sizes, so our study was likely underpowered to detect such differences.
Our hypothesis was that the combined pressures of mental fatigue (i.e., decreased mental efficiency) with the increased mental demands required to maintain balance under uncertain vestibular conditions (induced by GVS) would lead to a greater-than-additive performance decrements. Interestingly, the performance data from the balance and tandem walk tasks did not support this hypothesis. In fact, subjects displayed improved performance under fatigue while exposed to GVS on both the eyes-open balance and eyes-open tandem walk tasks.
A major limitation of this study was a lack of a non-fatigue control group. Prior fatigue studies have included such a group that performed a non-fatiguing task such as watching a movie (Hachard et al., 2020) instead of completing the fatigue induction task. Without this control group, it is difficult to ascertain whether the differences before and after the fatigue task are the result of fatigue or are simply a “time” effect (e.g., learning, adaptation, etc.). It seems unlikely that balance improved because of mental fatigue, although it is possible. It has been previously shown that increased cognitive workload can lead to better performance on highly automated tasks (Engström et al., 2017). It is possible that mental fatigue forced subjects to rely more on automatic postural control mechanisms during the post-fatigue trials, which led to better performance. However, this seems unlikely, as such automatic mechanisms for balance control would rely on the brain’s internal model for interpreting sensory inputs, including vestibular signals. Since vestibular inputs become unreliable under GVS, greater reliance on this internal model without more intentional cognitive control would likely lead to greater instability.
A much likelier explanation is that the improved performance is indicative of adaptation to GVS, and any minor negative effects of fatigue were masked by the much larger positive effect of adaptation. Indeed, effect sizes for balance decrements associated with mental fatigue in prior studies have been small (Brahms et al., 2022). This would suggest that adaptation to vestibular perturbations can occur very rapidly (the study lasted less than 5 hr, and GVS exposure was less than 1 hr of total intermittent exposure throughout the study) for tasks in which alternate sources of sensory data (visual and somatosensory) are available. However, for tasks in which visual input is removed, and the subject must rely more on the perturbed vestibular inputs, adaptation is slower. This finding is consistent with post-flight astronaut performance data. After return to earth, astronauts recover rapidly under eyes open conditions, but this re-adaptation takes longer when visual or somatosensory inputs are perturbed (Clément et al., 2022).
Likely the most important contribution of this study comes from the dual-task tandem walk. Even though both visual and somatosensory inputs were available to the subject (the dual-task tandem walk was conducted with eyes-open for all trials), they did not display adaptation (i.e., improved performance) following the fatigue task (similar to the performance trends on the eyes-closed tasks). This finding suggests that even in environments in which full sensory information is available, adaptation to altered vestibular cues could take longer when under high mental workload in a state of mental fatigue.
Previous studies have reported a rapid initial adaptation, followed by a longer, slower full recovery of sensorimotor function following spaceflight (Clément et al., 2022). This recovery curve can create a sense of false confidence, in which astronauts feel fully recovered and ready to return to duty when visual and somatosensory cues are available, only to discover ongoing decrements when these sensory inputs are disturbed (Jain et al., 2010). Indeed, astronauts have reported suddenly feeling a sense of disorientation or postural instability when walking on the beach at night and while driving at night (i.e., unstable walking surface and/or reduced visual cues) after returning from spaceflight (Jain et al., 2010). This creates major risks for future missions. Astronauts may feel fully adapted while in the good lighting conditions and stable footing of a lunar lander or habitat. However, poor-lighting conditions and unstable, rocky surfaces during an extraplanetary EVA could exacerbate performance decrements if the astronaut has not fully adapted to the altered vestibular inputs.
Our findings suggest that, in addition to situations with limited sensory information, periods of high mental workload while in a state of mental fatigue could also exacerbate otherwise undetectable sensorimotor performance decrements. Future extraplanetary EVAs will likely place high mental demands on crewmembers and both mental and physical fatigue should be expected. If a longer adaptation period is required to maintain sensorimotor performance under these conditions, this must be accounted for when scheduling EVAs after a g-transition and when clearing a crew member for return to service.
Limitations
The lack of a non-fatigued control group made interpretation of the results difficult. Future studies should incorporate such a control to help untangle the effects of fatigue from other confounding factors (i.e., adaptation).
Additionally, due to personnel limitations, the tandem walk data was only scored by a single scorer. Ideally, this would be conducted by three separate scorers and the median score would be used, so the results should be interpreted with caution. However, for consistency in scoring, all tandem walk trials were scored by the same person, who was blinded to the condition of the trial, so we feel confident in the quality of the data.
Finally, a potential limitation of the study was the head strap used to hold the GVS electrodes in place. The material that the strap was made of was partially absorbent, so sweat from the subject and saline used on the GVS electrodes could have partially absorbed into the head strap. This could have influenced the current flow and changed the influence of GVS during the post-fatigue testing session. After identifying this problem, we added plastic insulation to prevent the absorption of saline and tested with one subject, and the results from this subject were consistent with the prior results, so we feel confident that this did not significantly impact the study outcomes. Even so, future studies should be cognizant of this issue when using electrical brain stimulation.
Conclusion
We hypothesized that the interaction of mental fatigue with vestibular challenges would lead to greater-than-additive performance decrements during static and dynamic balance tasks. However, the findings of the study did not support this hypothesis. Instead, subjects displayed rapid adaptation to GVS (<1 hr intermittent exposure) when visual information was available, but not under eyes-closed conditions. Additionally, we found that subjects did not display adaptation when under high mental load in a state of mental fatigue, even when visual cues were present. These findings should be considered when planning future missions and scheduling EVAs, to ensure that astronauts have been given sufficient time to adapt to altered vestibular cues in order to maintain performance and successfully complete the mission.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by a student research grant from the Huffines Institute for Sports Medicine & Human Performance at Texas A&M University.
