Abstract
Background:
Despite substantial increase in total ankle arthroplasty (TAA) nationwide, there are few studies comparing flat-cut vs chamfer-cut talar systems in TAA with regard to radiographic aseptic loosening rates of the implant.
Methods:
This retrospective study included 189 Salto-Talaris TAA and 132 INBONE II primary TAA with a minimum 1-year follow-up. Patient characteristics were obtained including gender, age at surgery, body mass index (BMI), smoking status, primary diagnosis, surgical time, and the presence of diabetes. Radiographic evidence for aseptic loosening was assessed. Statistical analysis was performed for comparison in outcomes between Salto-Talaris and INBONE II.
Results:
The mean age of the study population was 63.5 ± 9.8 years at surgery. Mean follow-up was 4.9 ± 3.0 years. Radiographic aseptic loosening of the tibial implant showed no significant difference between the 2 groups: Salto-Talaris, 18%, and INBONE II, 18.9% (P = .829). Aseptic loosening of the talar implant also showed no significant difference between the 2 groups: Salto-Talaris, 1.6%, and INBONE II, 1.5% (P = .959). No variables, including the implant type, were found to contribute to the aseptic loosening rate of either the tibia or talus.
Conclusion:
In our cohort, we observed no difference in radiographic implant aseptic loosening between Salto-Talaris and INBONE II systems.
Level of Evidence:
Level IV, retrospective case series study.
Introduction
Total ankle arthroplasty (TAA) is growing in popularity given favorable outcomes with newer systems and enhanced understanding of foot and ankle biomechanics. In the correctly selected patient, outcomes can be comparable or superior to ankle arthrodesis, long considered the gold standard treatment for end-stage ankle osteoarthritis.6,19 A recent study of the National Inpatient Sample database in the United States reported a 5-fold increase in TAAs performed in 2017 when compared to 2005. 20 Accordingly, failed TAA is expected to grow in incidence over the next few decades.16,20
Aseptic loosening of the TAA implant is one of the more common complications encountered with the procedure and remains a consistent cause for revision TAA. 12 Thus, design teams seeking to manufacture implants with improved survival rates rely on well-executed biomechanical and clinical research to iterate on existing designs. For example, a study using finite elementary analysis suggested that the potential for micromotion at the interface between the implant and bone was greater with use of the INBONE II as opposed to the INFINITY. 23 Such micromotion may lead to aseptic loosening of TAA. Although biomechanical studies can be helpful, there are few clinical studies focusing on aseptic loosening rates of specific TAA implants providing direct comparison between manufacturers.
The INBONE II (Stryker; Kalamazoo, MI) and Salto-Talaris (Smith & Nephew; Memphis, TN) TAA are popular systems. Both represent third-generation TAA implant designs with fixed-bearing components. Recently, several studies have been published regarding midterm outcomes of these systems.3,4,13,22 Each of these TAA designs features distinct elements. The INBONE II also has a modular tibial stem, in contrast to the monoblock stem used for the Salto-Talaris. Further, the talar implant of the INBONE II is symmetric with regard to laterality whereas the Salto-Talaris talar implant has a laterally specific design. The 2 implants have differently shaped joint surfaces, and thus different kinematics and different stresses on the bone-implant interface. The INBONE II employs a flat-cut design for the talus, whereas the Salto-Talaris uses a chamfer-cut design.
To date, there has been no clinical evidence providing a direct comparison between implants regarding radiographic aseptic loosening rates, irrespective of the need for subsequent revision. 9 Moreover, there are few comparison studies between 2 fixed-bearing TAA designs in large-volume, single-center samples.7,17 Rajan et al 17 compared these 2 systems and reported similar longevity. However, these authors did not report asymptomatic loosening cases, which can portend failure and may warrant revision procedures such as grafting or component exchange. Therefore, the purpose of this study was to compare the aseptic loosening rate between the INBONE II and the Salto-Talaris systems. Our hypothesis was that the aseptic loosening rates of the Salto-Talaris would be lower than those of the INBONE II.
Methods
Study Design
Institutional review board approval was obtained prior to conducting the study. A large institutional TAA database was queried for patients undergoing primary TAA from 2003 to 2019. A total of 1312 patients were identified during that period. With regard to implant selection, several TAA implant systems were used throughout the study period, including the INBONE I, INBONE II, STAR, Salto-Talaris, Infinity, and the Vantage TAA. Implant choice is based on surgeon preference and suitability to various patient-specific factors. For example, in cases of more severe coronal or sagittal plane deformity, the INBONE II is preferentially used. Of the implants used at our institution, the INBONE I is not currently available on the market, the Infinity was performed in only a very small number of patients, and the Vantage is a newer, fourth-generation implant with novel design elements. For these reasons, we chose the INBONE II and the Salto-Talaris as comparison material to evaluate the impact of the talar-cut system on aseptic loosening rates. Moreover, both the INBONE II and the Salto-Talaris are popular third-generation models with existing series presenting favorable mid- to long-term clinical outcomes.
Of these, 255 patients underwent TAA with the Salto-Talaris system and 250 patients with the INBONE II system (Wright Medical/Stryker, Kalamazoo, MI) underwent primary TAA. Of these, we excluded the following: (1) all revision cases and cases revised at follow-up for the causes such as infection, periprosthetic fracture other than aseptic loosening or subsidence; (2) cases without appropriate medical images available; (3) cases with <1 year of follow-up; (4) cases where patients also underwent arthrodesis at the subtalar or talonavicular joints; and (5) cases where the patient underwent bilateral TAA. 15 All cases were performed following the specifications of manufacturer-provided technical guides, and no use of patient-specific instrumentation for the INBONE II was included. After exclusion criteria were applied, 189 Salto-Talaris and 132 INBONE II implants were included in this study. All procedures were performed in a single institution by one of 4 fellowship-trained surgeons with significant experience in TAA.
Data Variables
Manual chart review was performed for collection of data variables. Information on patient demographics and characteristics were obtained including patient gender, age at primary TAA, body mass index (BMI), smoking status, primary diagnosis including the presence of inflammatory arthritis, and the presence of diabetes. Total surgical time for the TAA was acquired.
Radiographic Outcomes
Final follow-up appointments involved the use of ankle anteroposterior and lateral weightbearing radiographs to assess the state of the components of the TAA implant. Immediate postoperative and final radiographic images were used to measure the alignment changes of the implants with regard to the tibia and talus. Alignment changes of implants were assessed by a specialty-trained foot and ankle surgeon using calibration function in the PACS. Tibial subsidence was defined as more than 2 mm of implant displacement compared with previous radiographs, and talar subsidence was defined as more than 5 mm of implant displacement compared with previous radiographs in accordance with previous literature. 8 The decision was made to include subsidence of implants in the category of aseptic loosening given semantic overlap between these terms. Thus, the same parameters used to determine subsidence can be interchangeably applied to diagnose implant aseptic loosening. In line with this reasoning, aseptic loosening was also diagnosed in the following circumstances: (1) when there was a change in either the coronal or sagittal distal tibial component angle of more than 2 degrees or the presence of a >2-mm-thick radiolucent line that spanned >5 mm in length at the bone-implant interface was observed, or (2) when there was a change in the talar component flexion angle of >5 degrees. 1
Statistical Analysis
Descriptive statistics were performed to report the data variables collected. Continuous data were reported as means (SDs) or medians (interquartile ranges), and categorical variables were reported as counts (percentages). The Student t test was used to compare characteristics according to the type of implant. χ2 test was used for analysis of categorical variables. Aseptic loosening of implants was analyzed using logistic regression analysis and χ2 test. In logistic regression analysis, dependent variable was set as 1 when there was aseptic loosening of tibial or talar implant and was otherwise set as 0. Statistical significance was defined as a P value <.05. All statistical analyses were performed using SPSS, version 22 (IBM, Armonk, NY).
Results
Study Population Characteristic
The characteristics of this study population are listed in Table 1. The mean age of the study population was 63.5 ± 9.8 years at surgery. The patients undergoing Salto-Talaris TAA surgery (86 male and 103 female) were 63.6 ± 10.0 years of age and those undergoing INBONE II TAA (96 male and 36 female) were 63.5 ± 9.4 years of age (Table 1). There was no significant difference in age between the cohorts. The study patients had a mean BMI of 29.9 ± 5.5. The mean operative time was 2.3 ± 0.7 hours. The cohort included 173 nonsmokers (55.3%), 130 former smokers (41.5%) and 10 active smokers (3.2%). Mean follow-up was 4.9 ± 3.0 years. Primary diagnosis for TAA included posttraumatic osteoarthritis (53.9%), idiopathic osteoarthritis (38.9%), rheumatoid arthritis (2.8%), and others (3.4%).
Characteristics of the Study Population.
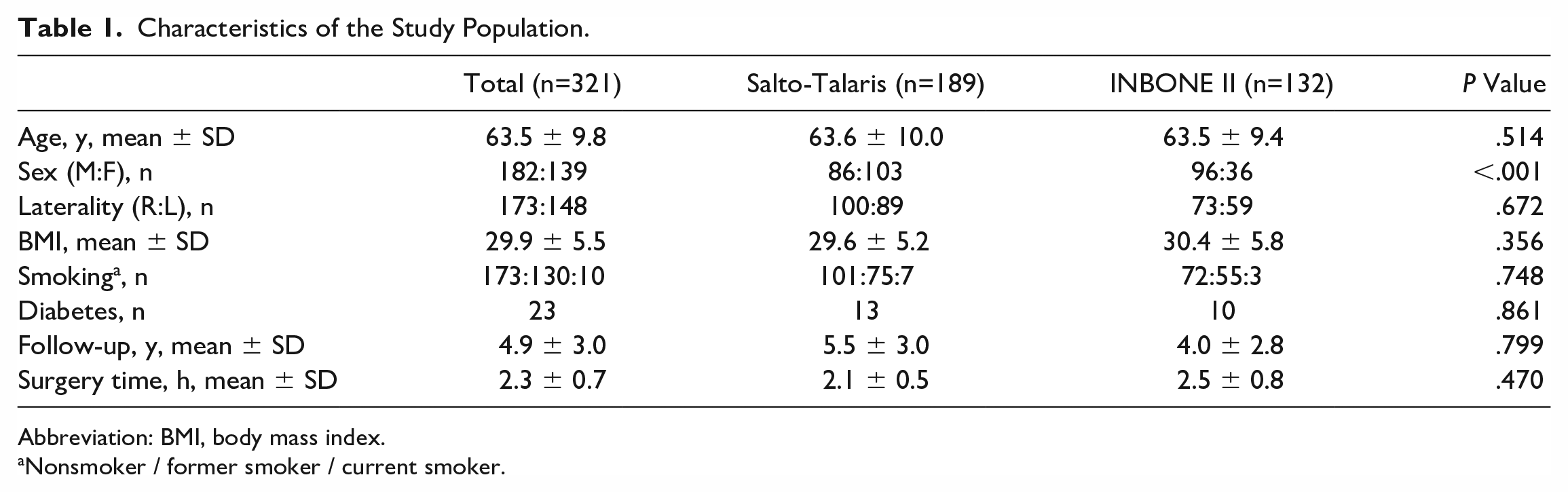
Abbreviation: BMI, body mass index.
Nonsmoker / former smoker / current smoker.
Radiographic Outcomes
There was no difference in tibial implant loosening between the 2 groups: Salto-Talaris, 18%, and INBONE II, 18.9% (P = .829) (Figures 1 and 2). Likewise, aseptic loosening of the talar implant showed no significant difference between the 2 groups: Salto-Talaris, 1.6%, and INBONE II, 1.5% (P = .959). There was no variable included in the regression analysis (including the implant type) associated with the aseptic loosening rate of the tibia or talus implant (Table 2). Information regarding patients who experienced talar component aseptic loosening or osteolysis is outlined in Table 3.
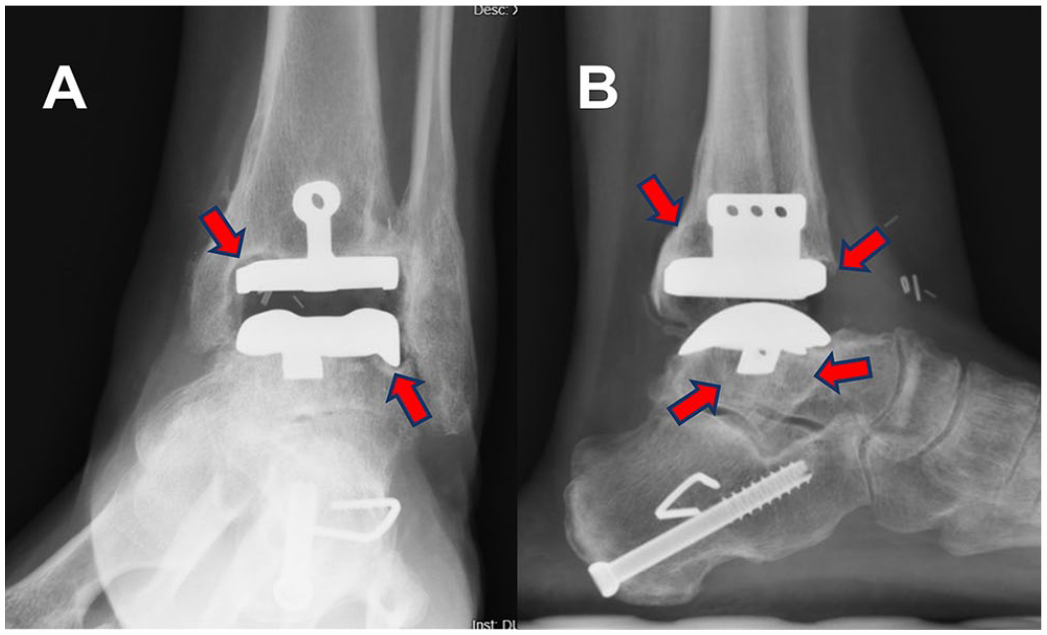
(A) Anteroposterior (AP) and (B) lateral radiograph of a Salto-Talaris Talar implant, demonstrating both tibial and talar peri-implant lucency, consistent with the diagnosis of aseptic loosening. Notably, substantial cystic formation is present beneath the talar component, appreciable on the anterior and posterior quadrants below the implant on the lateral image and on the lateral quadrant below on the implant on the AP image.
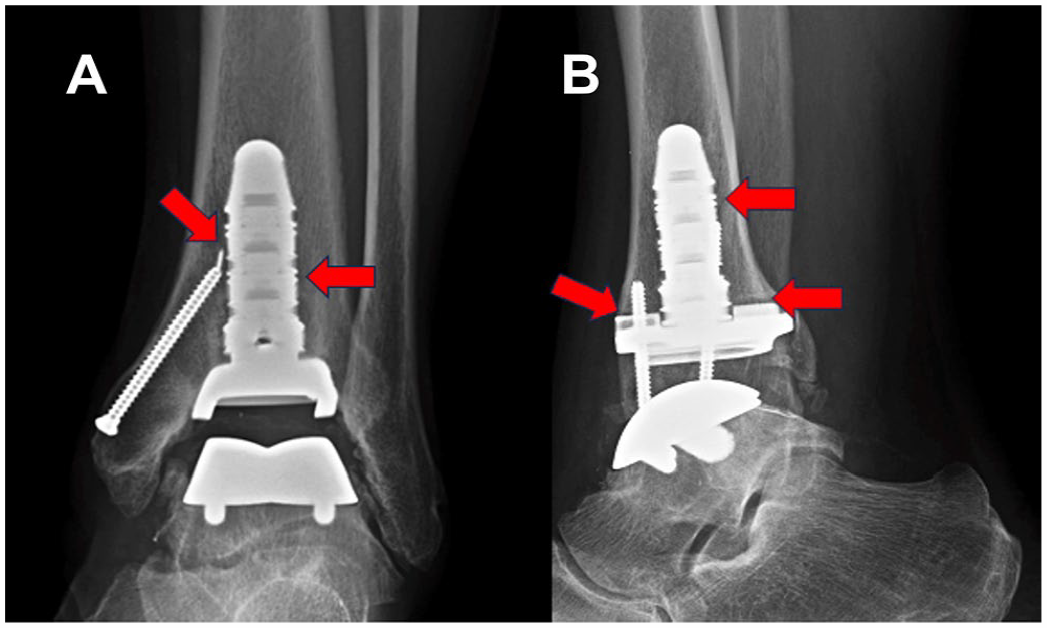
(A) Anteroposterior and (B) lateral radiograph of a INBONE II implant with primarily tibial-sided periimplant lucency indicating aseptic loosening. The red arrows in the image above mark sites of peri-implant lucency. [See online article for color figure.]
Logistic Regression Analysis for Loosening or Subsidence of the Implant.
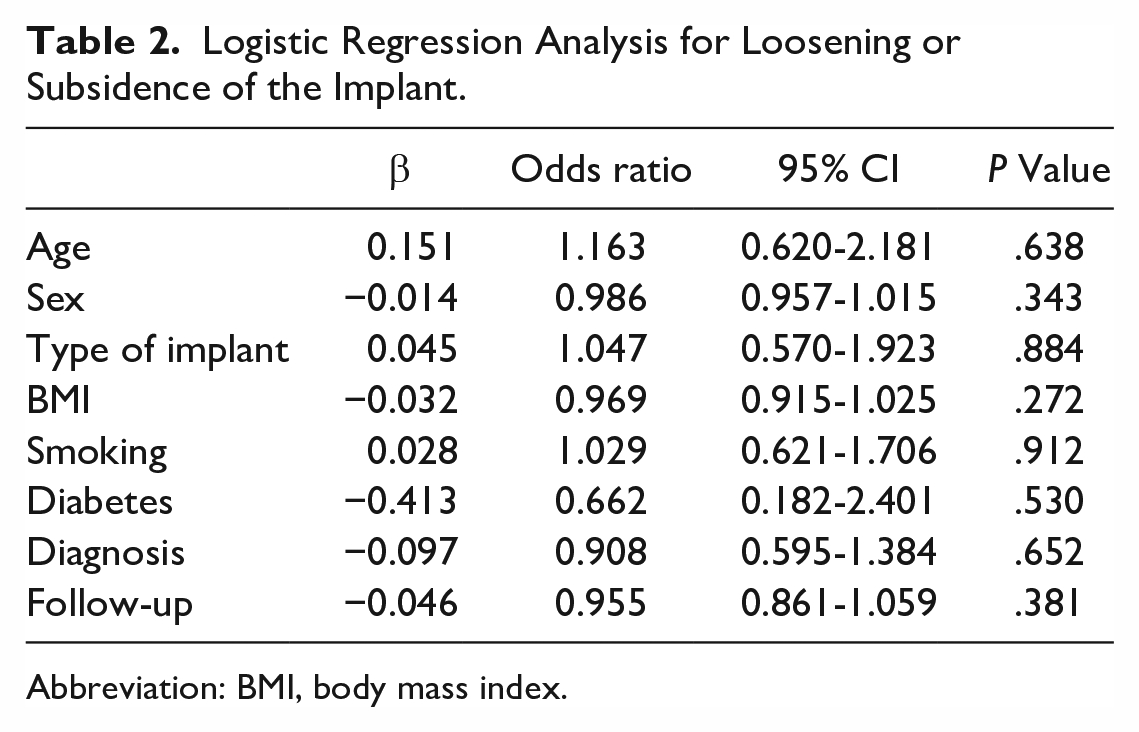
Abbreviation: BMI, body mass index.
Five Patients Who Experienced Talar Component Aseptic Loosening or Osteolysis.
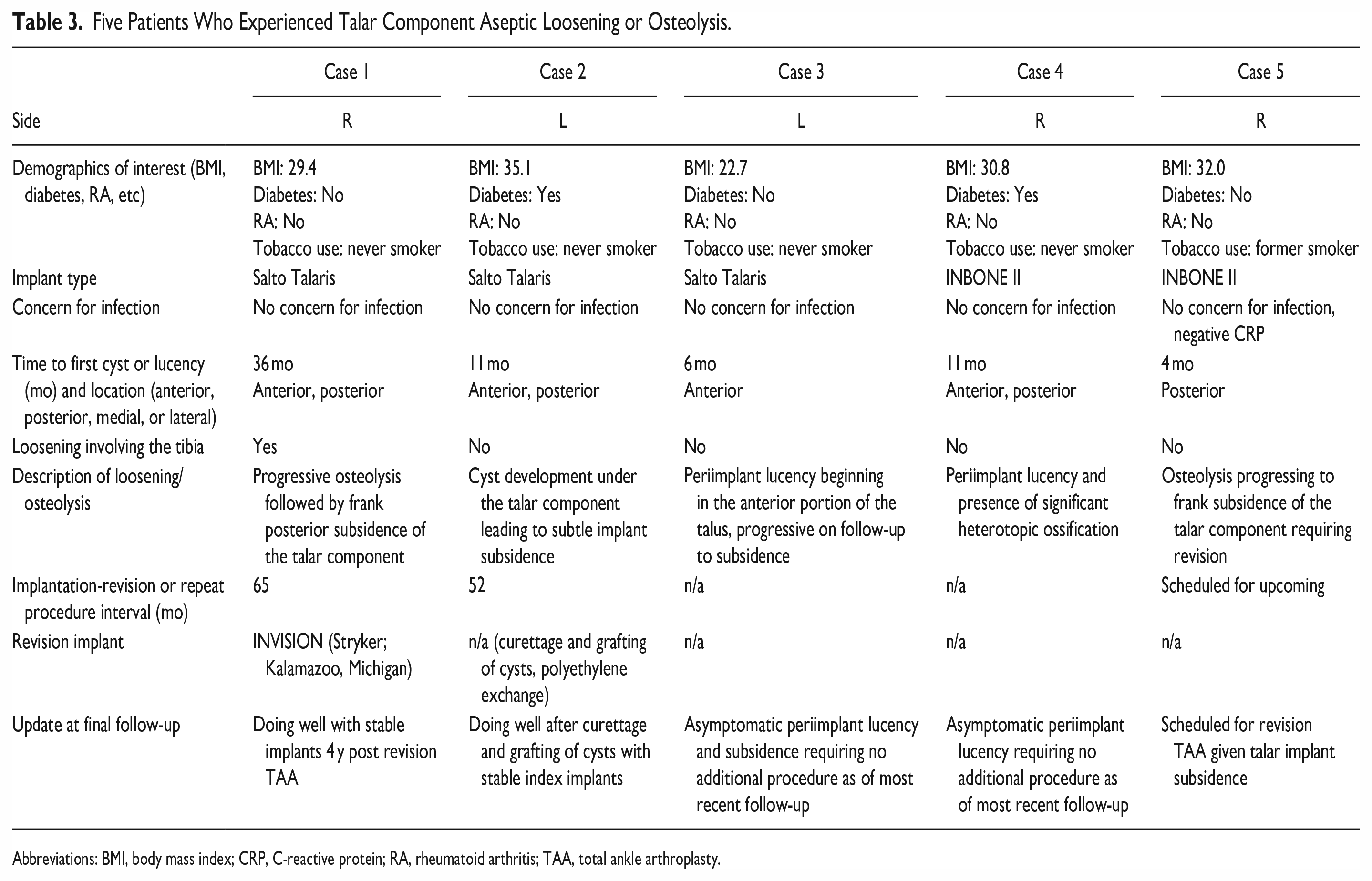
Abbreviations: BMI, body mass index; CRP, C-reactive protein; RA, rheumatoid arthritis; TAA, total ankle arthroplasty.
Discussion
Our study found no significant difference between the Salto-Talaris and INBONE II TAA in midterm radiographic aseptic loosening rates. Moreover, all independent factors included in our analysis did not impact the aseptic loosening rates in our sample. Prior to initiation of the study, we hypothesized that the talar aseptic loosening rate of the INBONE II would be higher than that of Salto-Talaris because of several factors. First, the shearing force resulting from flexion and extension movement through the ankle joint may be larger in the flat-cut interface between the talar implant and bone. Second, there is a larger amount of bone resection in the INBONE II flat-cut system when compared to the Salto-Talaris chamfer-cut system. In contradiction to our working hypothesis, our results revealed no differences between the 2 implants in the patient groups studied.
Component loosening is one of the most common complications in TAA, and talar loosening is known to be more frequent than tibial loosening. 12 The previous literature is not congruent with our results, where aseptic loosening of the tibial implant was more common than that of the talar implant. However, the vast majority of cases of tibial aseptic loosening in our study group was asymptomatic and did not progress to subsidence, whereas 4 of the 5 cases of talar aseptic loosening progressed to subsidence. Thus, our results support the notion that catastrophic implant failure is more common on the side of the talus than the tibia in TAA.
In many cases of failed TAA, there is considerable bone loss, making revision TAA more difficult, particularly with regard to the talus because of the unique anatomy of the bone. 12 The etiology of implant aseptic loosening is still unclear. Most of the recent design systems for TAA employ chamfer-cut talar preparation in the belief that a flat-cut talar component may be more likely to subside, despite minimal clinical evidence in support of this concept. Thus, our research team sought to directly compare a flat-cut talar system to a chamfer-cut system.
The quantity of bone resection is larger in flat-cut talar systems, and this has been raised as a potential concern with use of implants employing this design feature. 5 In addition to the flat talar cut, the INBONE II requires creating a central 6-mm hole in the talus for implantation of the tibial modular stemmed component. The clinical impact of these elements has been the subject of controversy. Lewis et al 11 found that all cases of failure in TAA combined with subtalar fusion used the INBONE model, theorizing that the central hole may contribute to these findings. Moreover, several authors have proposed the potential for development of avascular necrosis of the talus following TAA with a central talar hole. 21 However, our results imply that such concerns may not be supported by clinical evidence. The talar preparation for the Salto-Talaris also violates the talar dome integrity through the use of a hollow drill. Thus, determining optimal talar preparation methodologies proves extremely difficult, given the relatively minor differences between implant systems and the small incidence of talar loosening in our sample.
There are few studies comparing the INBONE II and Salto Talaris. Rajan et al 17 reported in a retrospective series that both implants had similar longevity, but the INBONE II resulted in greater clinical improvement and fewer reoperations than the Salto-Talaris. However, these authors did not report the rate of radiographic aseptic loosening, potentially failing to account for patients who may ultimately require future revision TAA if progression of subsidence occurs.
Although few studies compare flat-cut to chamfer-cut talar systems, there are several reports regarding the loosening of implants in TAA. Kihara et al 10 demonstrate that loosening of the talar implant correlates with periprosthetic osteolysis after TAA. Moreover, there are concerns regarding tibial component aseptic loosening with certain fourth-generation TAA systems within the first postoperative year.2,14,18 In contrast, Miner et al 14 demonstrated in their hybrid implantation series that there was no significantly higher loosening rate with use of that particular system, although the study was limited by the short-term nature of clinical follow-up.
In the present series, we directly compare a talar flat-cut system to a chamfer-cut system with regard to loosening rates. There were few cases with talar loosening in both groups, and no significant difference of loosening rates between the cohorts. The flat cut system has several advantages over chamfer-cut systems in various aspects, including convenience, ease of use, and reducing the surgical time required for the multiple chamfer cuts. Additionally, flat-cut systems allow for easier talar preparation during revision TAA procedures.
Limitations
Our study has several limitations. First, this is a retrospective study of prospectively collected data. Implants were chosen based on surgeon preference, and so no randomization protocol was implemented. Moreover, there are many differences between the 2 implants investigated in this study, including the talar-cut methodology and preparations, but also notably differences in the bearing surface size, shape, and constraints resulting in different kinematics and stresses at the bone implant interface. Thus, it may be difficult to define which particular implant-specific factor may contribute to implant loosening. Second, we are aware that high radiographic aseptic loosening rates do not necessarily equate with high revision rates or poor clinical outcomes such as pain and functional impairment. Indeed, there is a substantial portion of patients who remain asymptomatic even in cases of implant loosening or subsidence. Third, our ability to visualize areas of lack of bony ingrowth or lucency depended on the exact radiographic view, and the shape of the talar implant. We may have underestimated the extent of problems. Fourth, we excluded cases with arthrodesis of the subtalar or talonavicular joints because of the effect of adjacent joint arthrodesis as a confounder on aseptic loosening of the implant. However, arthritis or stiffness of either the subtalar or talonavicular joint may have affected the loosening rates, and we did not include radiographic or clinical evidence of degenerative change to either of these joints in our regression analysis. Lastly, we report only midterm outcomes, and aseptic loosening rates were therefore not evaluated in the long term.
In conclusion, the rates of radiographic aseptic loosening of the tibial and talar implant showed no significant difference between the Salto-Talaris and the INBONE II system. Moreover, there was no independent risk factor identified to explain these findings.
Supplemental Material
sj-pdf-1-fai-10.1177_10711007231209763 – Supplemental material for Comparison of Radiographic Talar Loosening Rates Between Salto-Talaris and INBONE II
Supplemental material, sj-pdf-1-fai-10.1177_10711007231209763 for Comparison of Radiographic Talar Loosening Rates Between Salto-Talaris and INBONE II by Albert Thomas Anastasio, Samuel B. Adams, James K. DeOrio, Mark E. Easley, James A. Nunley and Dong-Oh Lee in Foot & Ankle International
Footnotes
Acknowledgements
The authors gratefully acknowledge the contributions of Kevin Wu to this article.
Ethical Approval
Ethical approval for this study was obtained from Duke University Hospital Institutional Review Board (IRB number: Pro00111414).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
