Abstract
Background:
Specialized treatment of plantar fasciitis that can reduce inflammation and promote healing may be a possible alternative prior to surgical intervention. We report the results of a randomized clinical trial examining the efficacy of micronized dehydrated human amniotic/chorionic membrane (mDHACM) injection as a treatment for chronic refractory plantar fasciitis.
Methods:
An institutional review board–approved, prospective, randomized, single-center clinical trial was performed. Forty-five patients were randomized to receive injection of 2 cc 0.5% Marcaine plain, then either 1.25 cc saline (controls), 0.5 cc mDHACM, or 1.25 cc mDHACM. Follow-up visits occurred over 8 weeks to measure function, pain, and functional health and well-being.
Results:
Significant improvement in plantar fasciitis symptoms was observed in patients receiving 0.5 cc or 1.25 cc mDHACM versus controls within 1 week of treatment and throughout the study period. At 1 week, American Orthopaedic Foot and Ankle Society (AOFAS) Hindfoot scores increased by a mean of 2.2 ± 17.4 points for controls versus 38.7 ± 11.4 points for those receiving 0.5 cc mDHACM (P < .001) and 33.7 ± 14.0 points for those receiving 1.25 cc mDHACM (P < .001). By week 8 AOFAS Hindfoot scores increased by a mean of 12.9 ± 16.9 points for controls versus 51.6 ± 10.1 and 53.3 ± 9.4 for those receiving 0.5 cc and 1.25 cc mDHACM, respectively (both P < .001). No significant difference in treatment response was observed in patients receiving 0.5 cc versus 1.25 cc mDHACM.
Conclusion:
In patients with refractory plantar fasciitis, mDHACM is a viable treatment option. Larger studies are needed to confirm our findings.
Level of Evidence:
Level I, prospective randomized study.
Keywords
Plantar fasciitis is one of the most common orthopedic complaints relating to the foot, affecting more than 1 million persons per year. 9 Plantar fasciitis is a degenerative syndrome of the plantar fascia. The condition may be caused by repeated trauma or overuse creating micro-tears in the plantar fascia.5,20 Although plantar fasciitis is characterized by classic signs of inflammation including pain, swelling, and loss of function, it has been suggested that some presentations of plantar fasciitis may be a noninflammatory, degenerative process which could be more appropriately termed plantar fasciosis. 13
Plantar fasciitis is diagnosed based on patient reported symptoms, history, and physical examination. In cases recalcitrant to conservative treatment additional diagnostic modalities such as ultrasound or magnetic resonance imaging may be used to rule out other pathologies and confirm the diagnosis of plantar fasciitis.
Clinical guidelines for the treatment of plantar fasciitis vary from conservative, nonoperative tier 1 treatments, such as oral analgesics, rest, stretching exercises, orthotics, cryotherapy, weight loss, and corticosteroid injections, to more advanced tier 2 treatments such as night splints or immobilization. 23 Chronic cases may advance to tier 3 treatments such as extracorporeal shock wave therapy, or plantar fasciotomy.9,23 Local injection of platelet-rich plasma and botulinum toxin type A have also been used as a treatment for plantar fasciitis.6,7,19 No single treatment is guaranteed to alleviate the heel pain.
Although nonoperative management leads to resolution of symptoms in approximately 90% of patients, this condition is challenging and frustrating for both patient and clinician as several months to even years of treatment is often required before symptoms abate.5,19,20 During this time the plantar fascia may be undergoing a degenerative process related to repetitive micro-tearing and inflammation. 24 The pain from chronic inflammation and injury to the connective tissue becomes more and more resistant to nonoperative management over time. 23 Treatment for plantar fasciitis imparts an estimated annual burden of $192 to $376 million to the United States health care system. 24 A treatment that reduces inflammation and heals soft-tissue damage, allowing for rapid return to pain free activities of daily living is desirable.
Human amniotic membrane has been used in a variety of clinical applications for over 100 years.1,3,4,11,16,21 In vivo and in vitro studies have shown that the biochemical properties of amniotic membrane help to reduce inflammation and enhance soft tissue healing.15,16 In its natural form, human amniotic membrane has also been shown to have antibacterial and pain reduction properties. 16 Repair is mediated through the growth factors contained in the membrane tissue. These growth factors include EGF, TGF-β, FGF which are known to stimulate epithelial cell migration and proliferation, and PDGF A and B, which stimulate many metabolic processes, including general protein and collagen synthesis, collagenase activity, and chemotaxis of fibroblasts and of smooth muscle cells.14,16
Use of fresh human amniotic membrane in the clinical setting is precluded by a number of issues, including the risk of disease transmission. The PURION process is a method of cleaning, sterilizing, and drying human amniotic/chorionic membrane obtained from screened and tested donors. 8 This proprietary process has allowed for the widespread use of an allograft material that can be stored at ambient temperature for up to 5 years. Recently, the possibility of further refining the dehydrated human amniotic/chorionic membrane with a micronization process to produce a powder has been realized. Dispersion of the powder into suspension with sterile 0.9% saline solution has led to new uses. The ability to inject dehydrated human amniotic membrane allows treatment of soft tissue injuries beyond surface wounds alone. The purpose of the present study was to examine the feasibility and effectiveness of using micronized dehydrated human amniotic/chorionic membrane (mDHACM) in suspension in 0.9% saline solution as an injectable treatment for refractory plantar fasciitis.
Methods
We performed a prospective randomized clinical trial comparing improvement of plantar fasciitis symptoms in patients receiving standard of care treatment alone versus standard of care with the injection of a mDHACM allograft (AmnioFix Injectable, MiMedx Group Inc, Marietta, GA). The study was conducted at a single center in southwest Virginia under the direction of a senior clinician (C.M.Z) with continuous enrollment of all eligible patients who wished to participate. Patients read and signed an institutional review board (IRB)-approved informed consent form prior to any study involvement. The study was reviewed and approved by Western IRB and preregistered in ClinicalTrials.gov (NCT01659827). The study population comprised patients with plantar fasciitis of 8 weeks to 1 year in duration who had not responded to traditional therapies. A newspaper advertisement was placed to recruit study subjects with heel pain. Eligible for enrollment were those patients whose symptoms were recalcitrant to nonoperative management with at least 3 of the following 5 treatments: rest, ice, compression, and elevation (RICE); corticosteroid injection; stretching exercises; nonsteroidal oral antiinflammatory agents; and orthotics. Other inclusion criteria included a minimum age of 18 years, an understanding and willingness to participate in a clinical study, and agreement to comply with weekly visits and follow-up regimen. Patients not eligible for inclusion were those having prior surgery at the site; clinical signs of site infection; evidence of significant neurological disease of the feet; inability to ambulate; current pregnancy, seeking pregnancy, or pregnancy within 6 months; prior radiation at the site; known positive HIV status; treatment with tissue engineered materials in past 30 days; or the presence of the following comorbidities: calcaneal stress fracture; nerve entrapment syndrome; plantar fascia rupture; systemic disorders associated with enthesopathy; Achilles tendinitis; fat pad atrophy; fibromyalgia; or allergy to gentamicin or streptomycin. Diagnosis of plantar fasciitis was confirmed by the primary investigator (C.M.Z.) through history and physical examination.
Patients were randomized into 1 of 3 groups: (1) standard care, plus 2 injections (2 cc of 0.5% Marcaine plain, then 1.25 cc sterile 0.9% saline) (controls), (2) standard care, plus 2 injections (2 cc of 0.5% Marcaine plain, then 0.5 cc of mDHACM injectable) (0.5 cc mDHACM group), or (3) standard care, plus 2 injections (2 cc of 0.5% Marcaine plain, then 1.25 cc of mDHACM injectable) (1.25 cc mDHACM group). The randomization was balanced and permuted in a block of 45 patients with 15 in each group. Although the clinician performing the injection and follow-up examinations (C.M.Z.) was not blinded as to the study group, patients were blinded as to which treatment they received.
Both injections were given in the heel of the affected foot. The mDHACM was reconstituted with sterile 0.9% saline. The patient after sterile prep received an injection of 2 cc of Marcaine to the plantar surface of the foot immediately distal to the medial calcaneal tuberacle with a 25 gauge needle and 3 cc syringe, along the medial origin of the plantar fascia. This was then followed with an injection of 0.9% saline or saline plus mDHACM, with a 25 gauge needle and 3 cc syringe. The needle was placed down to the level of the periosteum of the heel and pulled back less than 5 mm and the study medication or saline control was injected.
Follow-up and Evaluation
Following randomization and treatment according to group assignment, follow-up visits were scheduled to occur weekly for 6 weeks, and a final study visit was scheduled at 8 weeks postinjection. All patients were prescribed Tramadol 50 mg to be taken as needed for any pain or discomfort associated with the injection. Standard of care received by all patients included instructions on daytime use of a CamBoot (Active Offloading Walker, Royce Medical, Inc, Camarillo, CA) and nightly splinting (Darco International Inc, Huntington, WV) for the first 2 weeks postinjection. After 2 weeks patients could return to tennis shoes with an over-the-counter orthotic. By week 4 patients were instructed to resume normal activity as tolerated.
As pain is a subjective measure and validity of measurement tools is often in question, we used 3 different scales to evaluate symptom improvement. The American Orthopaedic Foot and Ankle Society (AOFAS) Hindfoot Scale is a 100-point scale used to assess pain, function, and alignment. 12 The Wong–Baker FACES Pain Rating Scale uses pictures of faces and asks the patient to rate their current pain from 0 (no hurt) to 10 (hurts worst). Both of these scales were used at baseline (preinjection), then again at each study visit. QualityMetric’s SF-36v2 Standard Health Survey was completed at baseline and at the completion of the study at week 8 to measure functional health and well-being from the patient’s point of view during the study period. The Health Survey is 36 questions in length and takes about 5 to 10 minutes to complete. Results were interpreted to provide psychometrically based physical component summary and mental component summary scores. Primary study outcome was reduction of symptoms between baseline and 8 weeks posttreatment.
Data analysis
Two senior orthopedic surgeons (A.P. and J.A.) reviewed and served as validators of the information collected on the case report forms and the subsequent data analysis, which were used to report results and formulate conclusions. Intention-to-treat 10 methods were used to compare data within and across the 3 study groups. The Mann–Whitney test or Kruskal–Wallis test was used to perform a comparison between 2 or more than 2 samples of continuous data, respectively. The chi-square test was used to compare 2 or more samples of binary data. The level of statistical significance was set at P < .05.
Results
Two hundred seventy-four patients were initially screened for eligibility (270 responded to newspaper advertisement and 4 existing patients). Of those screened via telephone, 229 (85%) did not meet inclusion criteria including heel pain of at least 8 weeks duration and having already received a minimum of 3 nonoperative treatments as described in methods. Sixty-eight screened patients were scheduled for an office evaluation, and 49 patients completed the visit. Of the 49 patients seen in the clinic, 45 were consented and enrolled in the study. Forty-five patients were randomized to 1 of 3 study groups described above. Two patients in the control group failed to complete the study, 1 withdrew consent within the first week, and 1 was lost to follow-up after 5 weeks.
In the study sample overall, 64.4% (29/45) were female, 62.2% (28/45) were over 50 years of age, and 77.8% (35/45) were obese. Plantar fasciitis symptoms without intermittent resolution had been present for a mean of 21.8 ± 12.0 weeks. Patient characteristics by treatment group are presented in Table 1. With the numbers available, no significant differences in patient characteristics were observed at study enrollment.
Patient Characteristics.
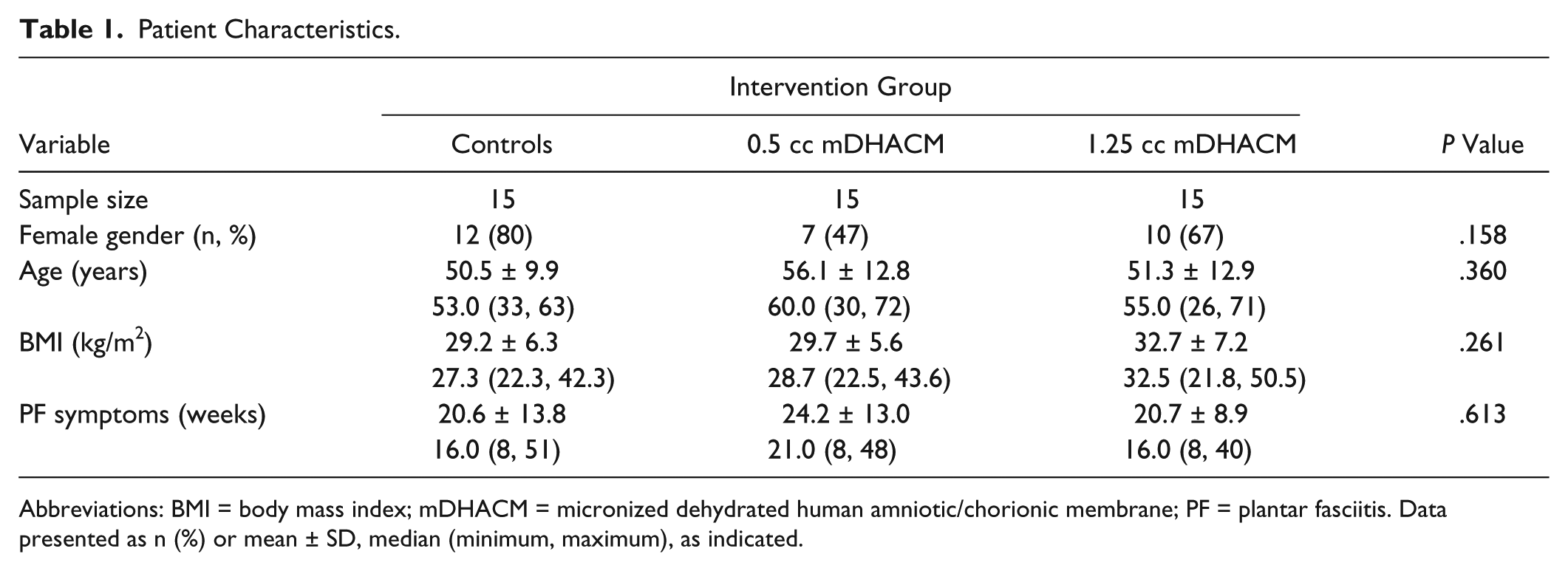
Abbreviations: BMI = body mass index; mDHACM = micronized dehydrated human amniotic/chorionic membrane; PF = plantar fasciitis. Data presented as n (%) or mean ± SD, median (minimum, maximum), as indicated.
AOFAS Hindfoot Score was calculated at baseline, weekly between weeks 1 and 6, and at 8 weeks posttreatment. AOFAS Hindfoot scores are compared in Table 2. Between baseline measurement and week 1, significant improvement was noted in AOFAS Hindfoot score for both mDHACM groups (P < .001 within each mDHACM group), while no improvement was noted within controls (P = .316). At week 1, 7.2% (1/14) of controls, 86.7% (13/15) of patients receiving 0.5 cc mDHACM, and 80.0% (12/15) receiving 1.25 cc mDHACM showed a minimum of a 25-point increase in AOFAS Hindfoot score. Changes in average AOFAS Hindfoot scores throughout the study period are presented in Figure 1. Within each group significantly higher scores were observed between baseline and week 8 (all Ps ≤ .01), although significantly greater improvement was noted in the groups receiving mDHACM versus controls (all Ps < .001). Similar improvement in AOFAS Hindfoot scores were observed for those patients receiving 0.5 cc or 1.25 cc mDHACM at any week.
AOFAS Hindfoot Scores.
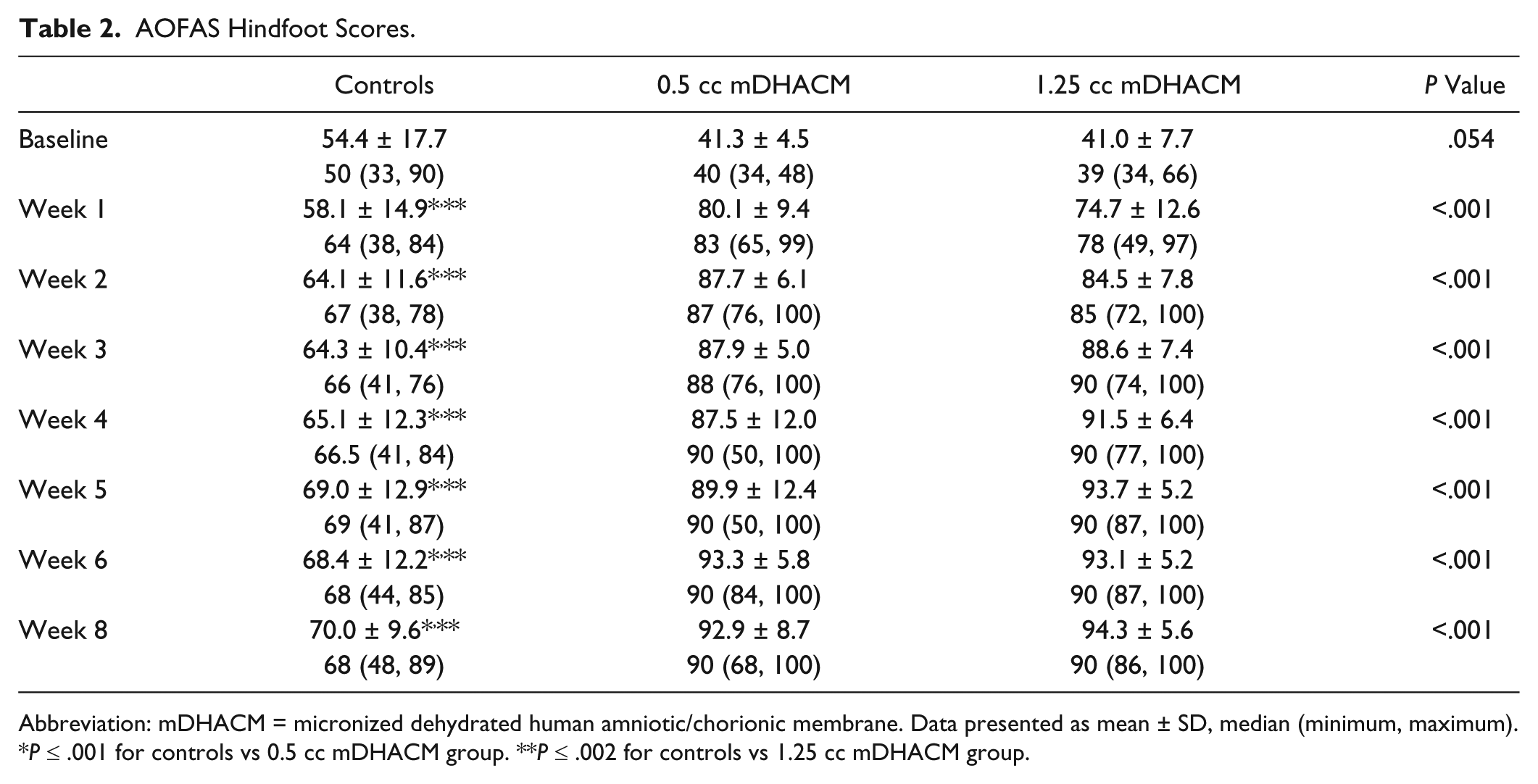
Abbreviation: mDHACM = micronized dehydrated human amniotic/chorionic membrane. Data presented as mean ± SD, median (minimum, maximum).
P ≤ .001 for controls vs 0.5 cc mDHACM group. **P ≤ .002 for controls vs 1.25 cc mDHACM group.
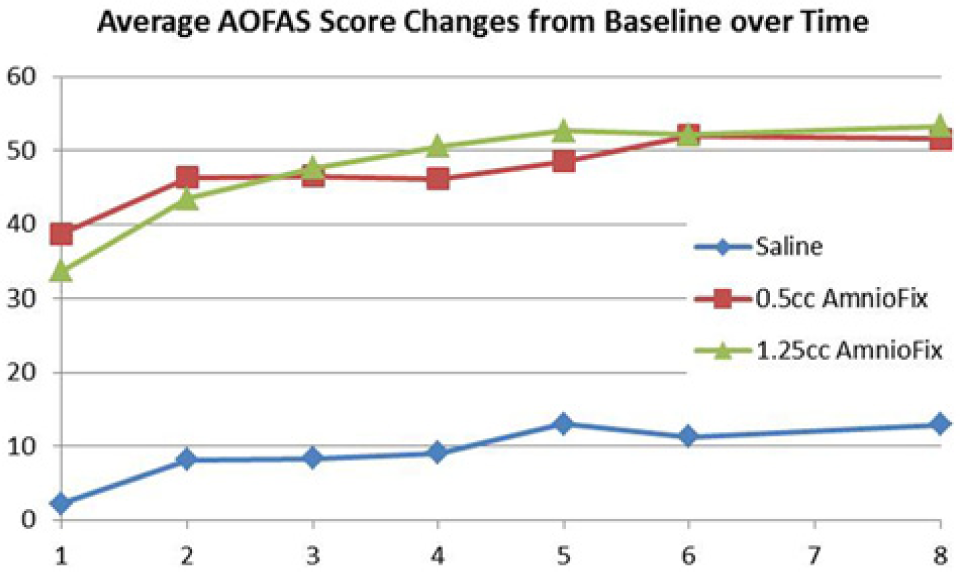
Mean difference in AOFAS Hindfoot score compared to baseline measurement during the study period.
Levels of patient-reported pain according to the Wong–Baker FACES Pain Rating Scale are presented in Table 3. At the time of enrollment, patients in all groups reported having very severe pain according to the scale. Within 1 week of study enrollment, the median reduction in pain was 3 points for controls and 6 points and 5 points for those receiving 0.5 cc and 1.25 cc of mDHACM, respectively (P < .001 controls vs 0.5 cc mDHACM; P = .004 controls vs 1.25 cc mDHACM). Per the FACES scale, controls continued to report moderate to severe pain throughout the 8-week study period, while those receiving mDHACM reported a significant reduction of pain from very severe at baseline to within the mild to moderate range at 1 week and reported continuing reduction in pain over the study period. Overall, at weeks 1 through 8, participants randomized to the mDHACM groups demonstrated statistically significantly lower median pain scores when compared to the controls. The median pain score from the 0.5 cc mDHACM group was 50% that reported by controls at 1 week (P < .001), 40% at 4 weeks (P < .001), and 25% at 8 weeks (P < .001). The median pain score from the 1.25 cc mDHACM group was 50% that reported by controls at 1 week (P = .002), 20% at 4 weeks (P < .001), and 25% at 8 weeks (P < .001). No differences were apparent in pain scores between the mDHACM groups. Mean differences in pain scores from baseline are shown in Figure 2. Patients receiving mDHACM reported significantly greater reductions in pain from baseline reports (all Ps < .001 controls vs 0.5 cc mDHACM, and all Ps < .004 controls vs 1.25 cc mDHACM). Pain reduction from baseline appears similar for the mDHACM groups.
Wong–Baker FACES Pain Scores.
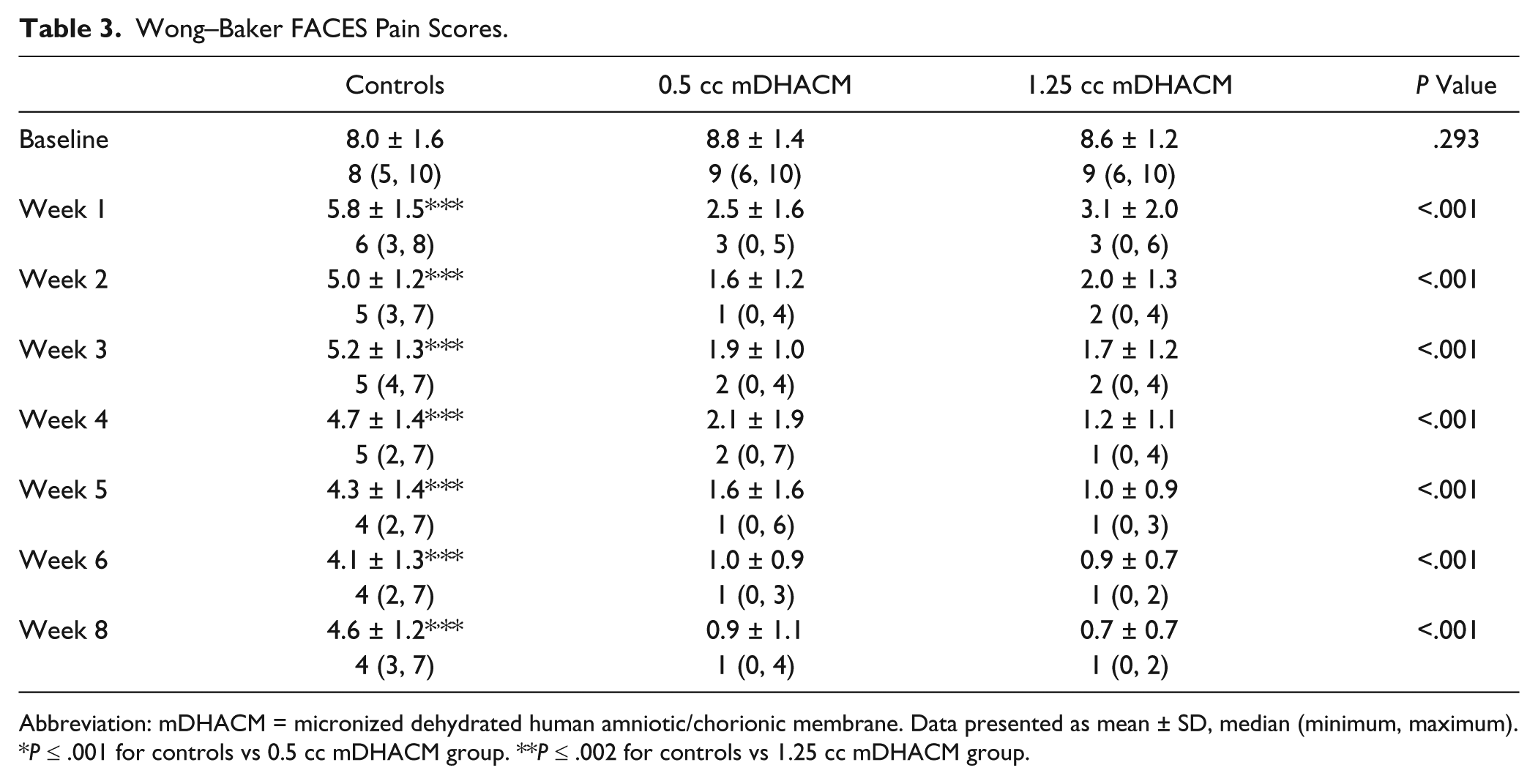
Abbreviation: mDHACM = micronized dehydrated human amniotic/chorionic membrane. Data presented as mean ± SD, median (minimum, maximum).
P ≤ .001 for controls vs 0.5 cc mDHACM group. **P ≤ .002 for controls vs 1.25 cc mDHACM group.
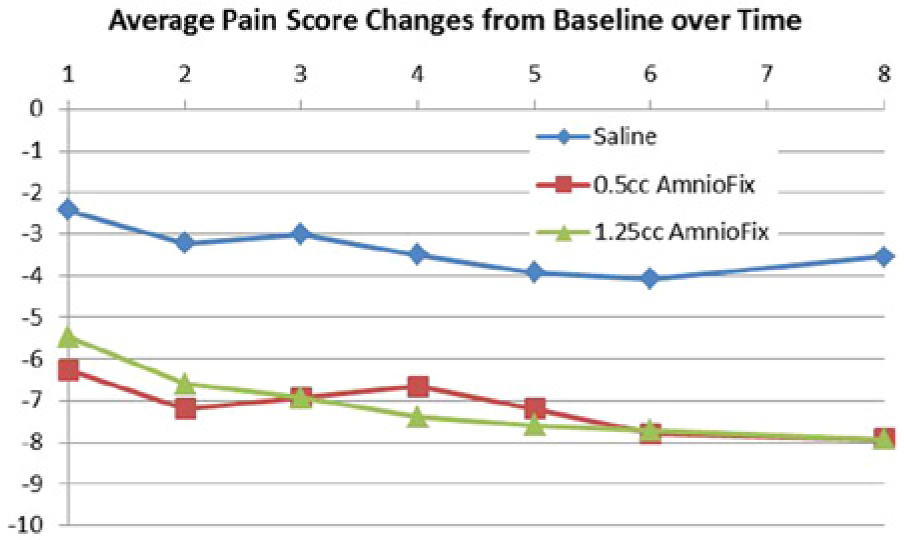
Mean difference in Wong–Baker FACES pain scores from baseline measurement during the study period.
Functional health and well-being were measured using QualityMetric’s SF-36v2 Standard Health Survey at the time of enrollment and completion of the study. Physical component scores are presented in Table 4, and mental component scores are presented in Table 5. No differences were observed in either physical or mental component scores between baseline and study conclusion for controls. In both mDHACM groups significant improvement was observed at study completion for both physical and mental well-being. Mean change in physical and mental scores per group are shown in Figure 3. Patients receiving mDHACM had significantly greater improvement in both physical and mental scores compared to controls (all Ps ≤ .002). The magnitude of difference between baseline and week 8 appeared similar when comparing the mDHACM groups.
Differences in SF36v2 Survey: Physical Component Summary Score From Baseline to Study Completion Within Each Study Group.
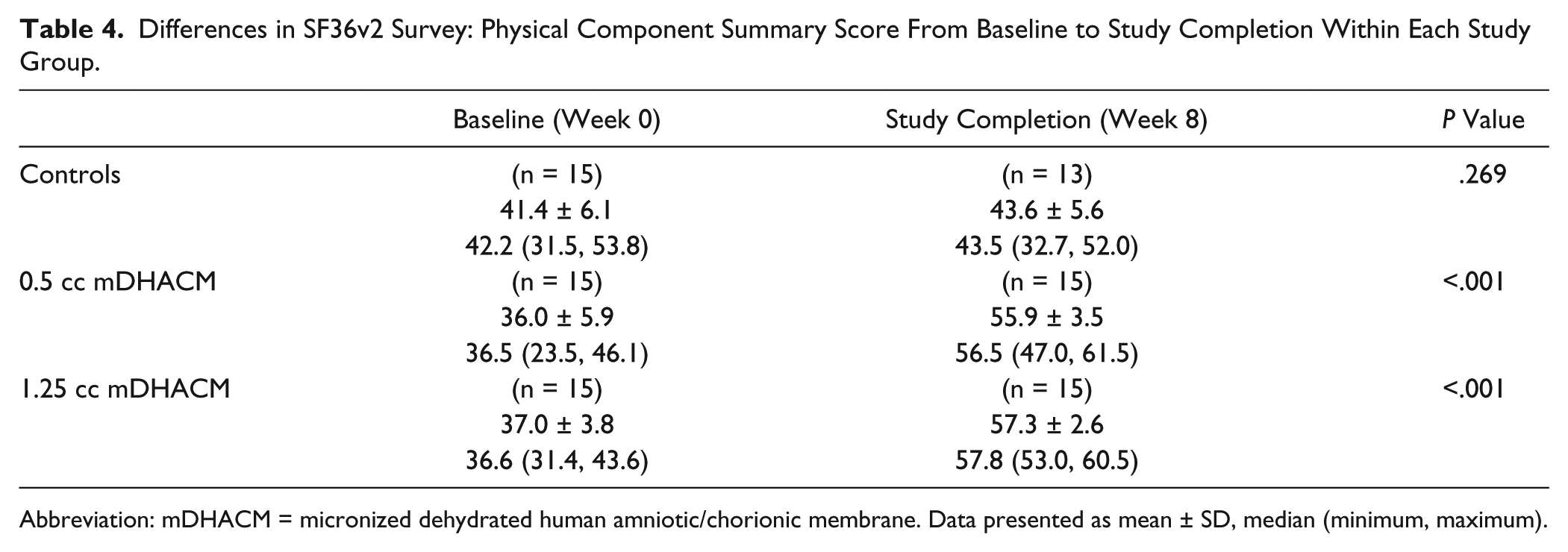
Abbreviation: mDHACM = micronized dehydrated human amniotic/chorionic membrane. Data presented as mean ± SD, median (minimum, maximum).
Differences in SF36v2 Survey: Mental Component Summary Score From Baseline to Study Completion Within Each Study Group.
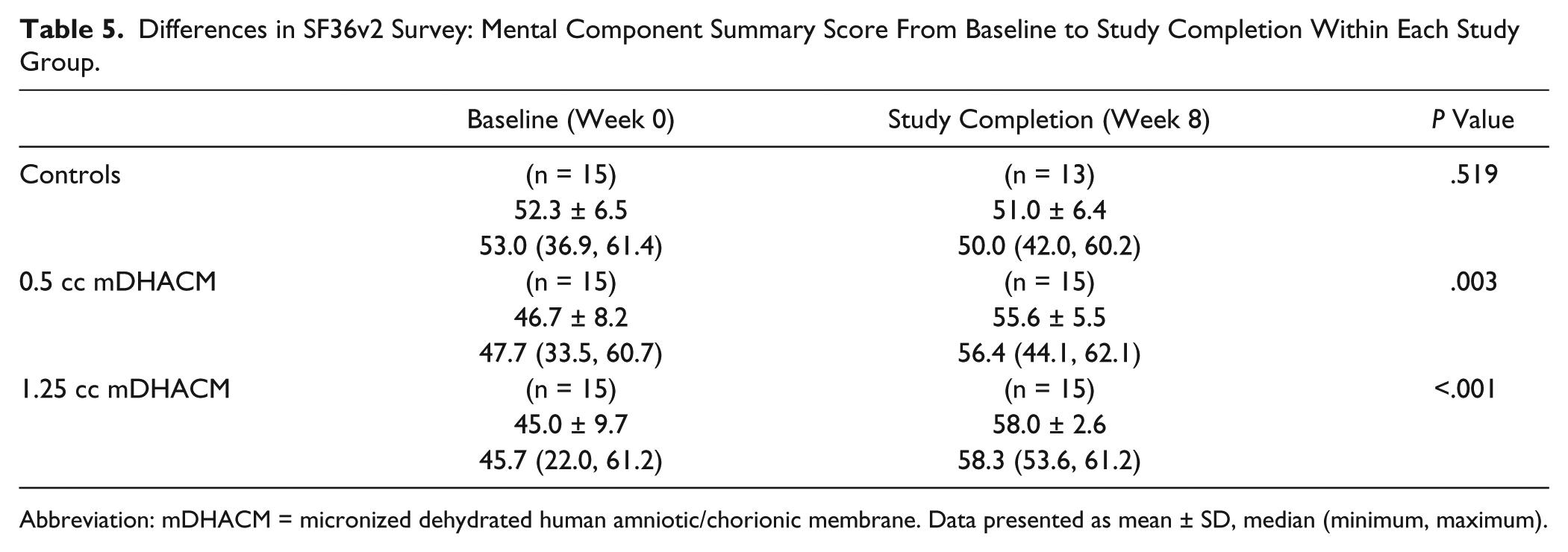
Abbreviation: mDHACM = micronized dehydrated human amniotic/chorionic membrane. Data presented as mean ± SD, median (minimum, maximum).
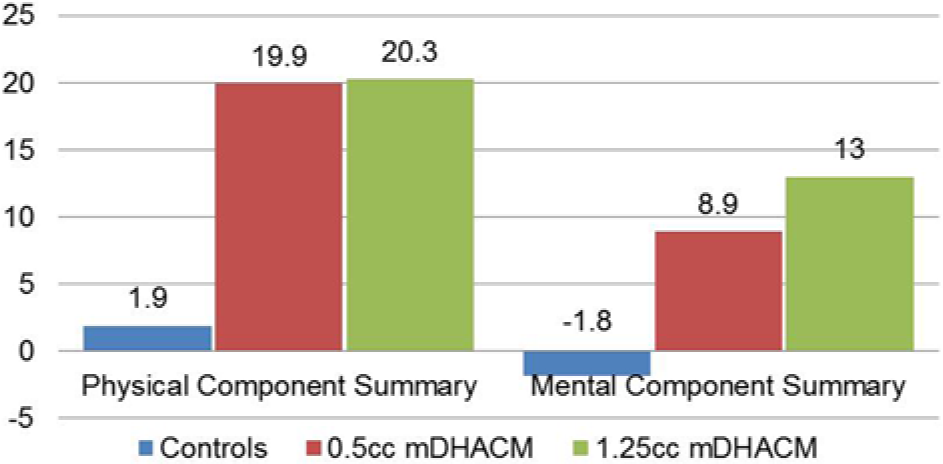
Mean difference in SF36v2 Physical and Mental Scores between baseline and study completion (week 8).
All patients were prescribed tramadol 50 mg to be taken as needed for any pain or discomfort associated with the injection. In the first week following injection 57.1% of controls, 73.3% of those receiving 0.5 cc mDHACM (P = .359 vs controls) and 100% of those receiving 1.25 cc mDHACM (P = .004 vs controls and P = .032 vs 0.5 cc mDHACM group) reported tramadol usage. By the second follow-up visit rates of tramadol use were similar at 50% for controls and 26.7% and 20.0% for 0.5 cc and 1.25 cc mDHACM groups, respectively (all Ps > .05).
Adverse Events
During study enrollment 1 patient that had received 0.5 cc mDHACM was hospitalized for 2 days with severe headache and pain behind the eye. This was not believed to be related to mDHACM and the patient continued in the study. No adverse events related to treatment were observed in any study subjects.
Discussion
This is the first randomized trial on the use of mDHACM for treatment of plantar fasciitis. Patients with chronic/refractory plantar fasciitis receiving a single-dose injection of mDHACM allograft experienced significant improvement in symptoms and increased function within 1 week of injection with continued improvement over the 8-week study period. Patients receiving 0.5 cc injection of mDHACM experienced a mean improvement of AOFAS Hindfoot score of 38.7 points at 1 week, 46.4 points at 2 weeks, and 51.6 points improvement at 8 weeks. Patients receiving 1.25 cc injection of mDHACM had a mean improvement of 33.7 points at week 1, 43.5 points at week 2, and 53.3 points at week 8. Other authors have also used the AOFAS Hindfoot Scale to examine plantar fasciitis treatment outcomes. Elizondo-Rodriguez et al 7 compared outcomes in 19 patients with plantar fasciitis treated with botulinum toxin A and 17 patients treated with intralesional steroids. On average, AOFAS Hindfoot scores increased 39.2 points at 2 weeks and 46.3 points at 8 weeks in patients treated botulinum toxin A. Patients treated with steroid injection had a significantly lower average magnitude of improvement in AOFAS Hindfoot scores at 2 and 8 weeks of 26 and 30 points, respectively.
In patients with chronic refractory plantar fasciitis surgical options may be considered. These options include open release or endoscopic plantar fasciotomy. In a retrospective study of forty-one patients with chronic plantar fasciitis that underwent endoscopic fasciotomy, there was a mean improvement of 37 points in AOFAS Hindfoot scores preoperatively versus postoperatively. 2 In the current study similar reductions in AOFAS Hindfoot scores were observed with mDHACM within 1 week of injection.
Limitations of the present study are those inherent to small sample size. Our findings should be confirmed and expanded with subsequent clinical trials. We were also limited by the validity of the scales used to measure improvement of symptoms, although it strengthens our results in that multiple scales were used. While it is acknowledged that plantar fasciitis is often a condition that may resolve with minimal intervention over time, we observed a greater magnitude of improvement in patients receiving mDHACM versus controls with both the AOFAS Hindfoot Scale as well as the Wong–Baker FACES Pain Rating Scale. While patients receiving treatment were blinded to their group assignment the investigator performing the injection and follow-up was aware of the treatment group, thus may have been biased as to study outcome. As the comparative group was saline we cannot speak to the effectiveness of the mDHACM allograft versus, or as an addition to, other advanced therapies. Additional comparative effectiveness studies are required to address those questions. All patients received an injection of 2 cc of 0.5% Marcaine plain prior to mDHACM or saline (placebo) injection and were instructed on off-loading, night splinting, and orthotics, thus we are unable to comment on how these standard interventions may have impacted study results. Although we did not observe differences in outcome measures between those patients receiving 0.5 cc versus 1.25 cc of mDHACM, the sample size was small, and we do not know if statistical significance would be observed in a larger patient population. Larger studies are required to elucidate if such differences exist.
Plantar fasciitis is a common problem. 18 Although there are many possible treatments, no single treatment is guaranteed to alleviate the heel pain for all patients. When initial nonoperative treatment yields unsatisfactory results, patients are often interested in treatment options other than surgery. Corticosteroid injection has been shown to provide relief from pain, although is associated with a high rate of relapse and may lead to permanent adverse changes within the structure of the fascia. 22 Human amniotic membrane contains growth factors including EGF, TGF-β, and FGF, which are known to stimulate epithelial cell migration and proliferation, and PDGF A and B, which stimulate many metabolic processes, including general protein and collagen synthesis, collagenase activity, and chemotaxis of fibroblasts and of smooth muscle cells. 16 TGF-β has been shown to significantly increase type I collagen production by tendon sheath fibroblasts.16,17 It has been suggested that growth factors work in a synergistic manner to initiate a tendon healing response. 17 These growth factors continue to be present in PURION processed dehydrated amniotic chorionic membrane. 8 The mDHACM (AmnioFix Injectable) is processed from donated human tissue according to the American Association of Tissue Banks (AATB) standards, and is considered a tissue product under section 361 of the Public Health Service Act. AmnioFix is minimally manipulated human amniotic/chorionic membrane intended for homologus use and is regulated as a human tissue by the US Food and Drug Administration. It is neither a medical device nor a drug, therefore does not have to go through a 510(k) premarket approval or new drug application process. As it contains no living cells, a biologics license application is not required. Because of the designation as a tissue, there are no specific on-label and off-label indications for its use.
The results of our clinical trial show that mDHACM allograft injection is an effective treatment for patients with chronic plantar fasciitis and may reduce costs by decreasing the need for repeat office visits or costly surgical interventions. Further studies are needed to better assess the utility of mDHACM within current treatment guidelines for the management of plantar fasciitis.
Footnotes
Editor’s Note
The authors are to be congratulated on evaluating a new treatment for plantar fasciitis which at short-term follow-up seems to be helpful. During the review process, we discussed the short duration of symptoms of some patients, that is, a minimum of 8 weeks, and the short-term follow-up of 8 weeks. For this reason, the study has been termed a feasibility study. The authors are conducting longer-term follow-up now, but due to the novel nature of this treatment it was elected to publish the study to get the information to the readership in a more timely fashion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Andrews has common stock options received for advisory services for the medical board of MiMedx.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by MiMedx Group, Inc, Marietta, GA. This research received no specific grant from any other funding agency in the public, commercial, or not-for-profit sectors.
