Abstract
Metaplastic carcinoma is an uncommon subtype of breast carcinoma which often shows a pathologic incomplete response to neoadjuvant chemotherapy. We report a metaplastic breast carcinoma with squamous differentiation showing pathologic complete response following neoadjuvant chemotherapy. The tumor bed showed keratinous debris without any viable residual tumor. Post-treatment, the acellular keratin persisted as a mass on physical exam and ultrasonography, suggesting an incomplete clinical response and resulting in clinical and pathologic discordance. This report expands the spectrum of histological findings in breast carcinoma with clinical and pathologic discordance in the neoadjuvant setting.
Introduction
Metaplastic breast carcinoma (MBC) is a rare heterogeneous group of diseases that comprises around 0.2%–1% of all breast cancers. 1 It is a unique entity that is characterized by the differentiation of the neoplastic epithelium into squamous and/or mesenchymal elements, resulting into several histologic subtypes.1,2 These tumors typically occur as triple negative breast cancers and behave aggressively due to their poor response to chemoradiotherapy.2,3 Compared to triple negative ductal or lobular carcinoma, metaplastic breast carcinoma was shown to have worse clinical outcomes and lower pathological complete response rates to neoadjuvant chemotherapy. 4
Due to its rare incidence and lack of well-defined treatment guidelines, there are few reports in literature describing excellent response to chemotherapy. 5 We report a MBC showing pathologic complete response (pCR) with a persistent mass-forming acellular keratin that led to discordant clinical, imaging, and pathologic findings following neoadjuvant chemotherapy.
Case Report
A 54-year-old woman presented with a 1-month history of a palpable left breast mass. Bilateral mammogram revealed a 2.0 cm round high-density mass in the left breast at 2 o’clock, middle depth. Ultrasound showed a heterogeneously solid, slightly irregularly marginated 2.0 cm mass, 8.0 cm away from the nipple (Figure 1A). The patient underwent core needle biopsy and was diagnosed with invasive mammary carcinoma of the left breast, Nottingham histologic grade 3. The tumor was a high-grade carcinoma with squamous differentiation (Figure 1B & C). The sample was composed of keratinous debris and reactive fibroblastic stroma (50%) and sheets and rounded nests of cohesive epithelioid polygonal cells with vesicular pleomorphic nuclei. Squamous differentiation was supported by rare intercellular bridges and eosinophilic cytoplasm. Immunohistochemical staining for p63 showed diffuse, intense nuclear staining in tumor cells, supporting a diagnosis of metaplastic carcinoma. The invasive carcinoma was estrogen receptor (ER) negative, progesterone receptor (PR) positive (11%–25%, weak), and HER-2 negative (score 0). The patient was treated as a triple negative carcinoma and subsequently underwent four cycles of dose dense adriamycin and cyclophosphamide followed by eight cycles of paclitaxel and carboplatin. Post-therapy ultrasonography showed a reduction in the mass size from 2.0 × 2.0 × 1.8 cm to 1.4 × 0.9 × 0.8 cm, suggesting incomplete response. A localized partial mastectomy with sentinel node sampling was performed. On gross examination, the breast specimen measured 5.5 × 4.0 × 6.2 cm. No discrete lesion was identified near the biopsy site/clip. Microscopic examination revealed benign breast tissue; no residual invasive or in situ carcinoma was identified. Prior biopsy site changes were observed. The fibrous tumor bed showed abundant keratinous debris, accompanied by foamy histiocytes, vague granulomatous response, and multinucleated giant cells (Figure 1 D & E). The tumor bed was comprised of reactive fibroblasts and cholesterol clefts. A morphological comparison was performed with MBC in the pre-therapy CNB. No viable atypical epithelioid or spindle cells were identified. The entire tumor bed was submitted for microscopic examination. The keratin (AE1/AE3) immunohistochemical staining highlighted the keratin debris (Figure 1F). The KRT IHC was negative in the reactive fibroblasts in the tumor bed. Three sentinel lymph nodes were negative for carcinoma and revealed no treatment effects. The final histological diagnosis was pathologic complete response, with no residual invasive or in situ carcinoma. The acellular keratinous debris and associated fibrous tumor bed represented the mass lesion that was presumed to be the residual “tumor” in post-therapy radiology scans.
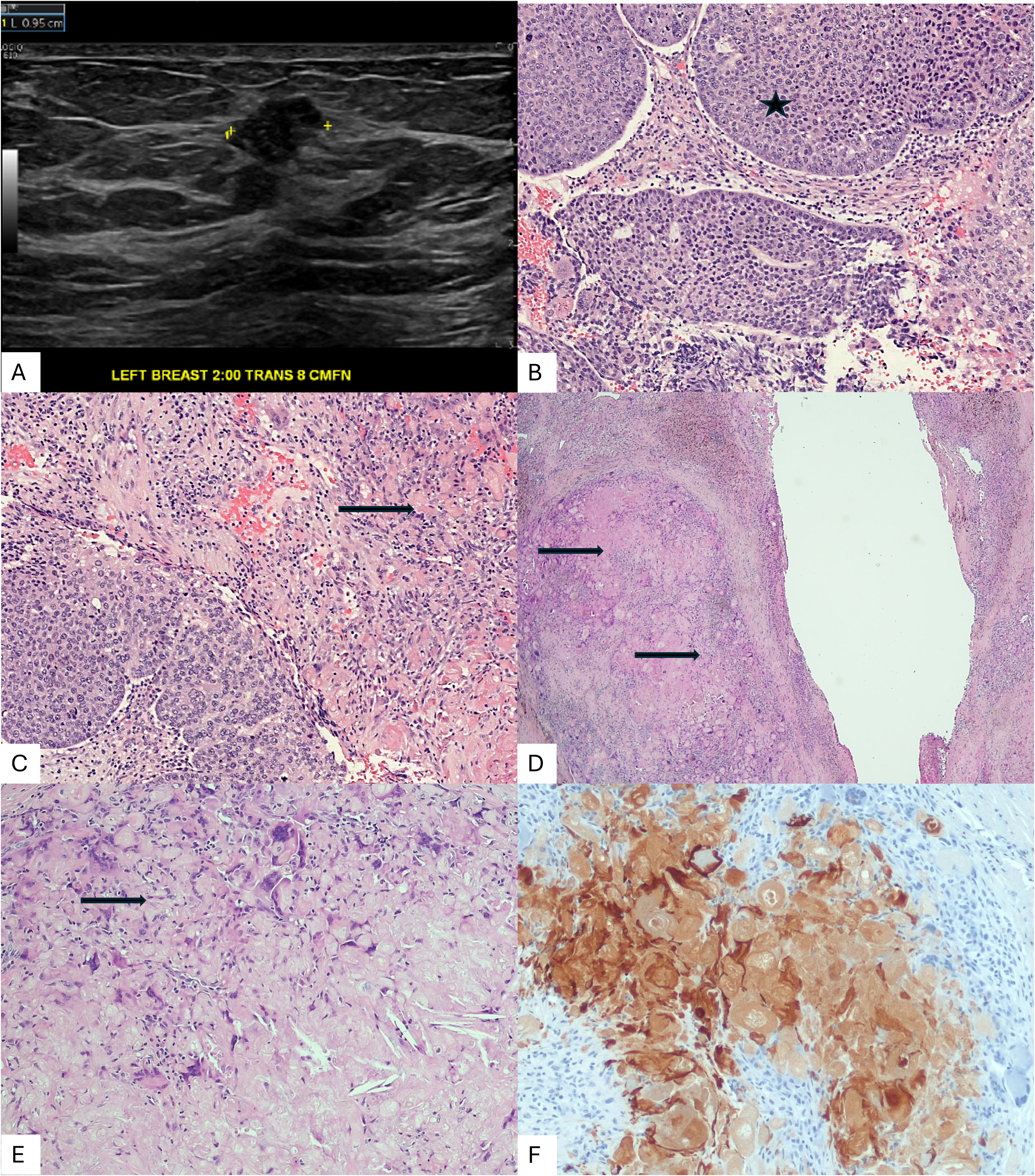
Discussion
MBC is resistant to conventional chemotherapy and has consistently exhibited lower pCR rates. 4 The reported response rates in MBC range from 6.3% to 31.7%,2,3,6 while some studies report a lack of complete response in MBC. 4 The poor response to conventional chemotherapy is said to be caused by the EMT phenomenon, which upregulates cancer-initiating cells or cancer stem cells that are known to be chemoresistant. 4 The same phenomenon also accounts for the high risk of metastasis in MBC. 4
Histological components may determine treatment response, reflecting the morphologic and biological heterogeneity among subtypes. 6 Min Han et al reported that the matrix-producing subtype was associated with pCR, although there was no difference in overall survival among subtypes. 6 Other studies have reported favorable outcomes in MBC showing <40% squamous component.3,7 Our patient achieved pCR following combined paclitaxel and carboplatin NAC. A similar outcome was reported by Noro et al in a patient of metaplastic squamous cell carcinoma subjected to weekly paclitaxel therapy. 8 As per WHO, a pure metaplastic carcinoma is diagnosed when > 90% of tumor shows histological evidence of squamous and or mesenchymal differentiation. In pre-therapy core needle biopsy sample, histologically, the carcinoma was composed of a mixed high-grade ductal carcinoma and squamous cell carcinoma. Since post therapy, there was no residual carcinoma, it is difficult to estimate pre-therapy proportion of squamous component and thus pin down the exact histological classification of this tumor as pure MBC or a mixed MBC with squamous and high-grade ductal carcinoma. A diffuse p63 positivity in pre-therapy high grade carcinoma, suggests squamous differentiation, however, an outcome of pCR suggests smaller overt squamous component/differentiation.
Post-therapy tumor bed comprised of acellular keratin debris, which is akin to the tumor's “tombstone”. The presentation of the keratinous debris and associated granulomatous reaction as a persistent mass-forming lesion, which was confirmed on radiological examination, led to a diagnosis of incomplete clinical response (ycT1c). Keratin is derived from anucleate ghost squamous cells, which in turn elicit a T cell-mediated granulomatous inflammatory response composed of multinucleated giant cells, lymphocytes, plasma cells, and histiocytes. Keratin granulomas without viable tumor cells persisting after systemic treatment have been described in a report of endometrioid carcinoma with squamous differentiation in the uterus. 9 An analogous occurrence has been reported in mucinous breast carcinoma. Didonato et al reported a mucinous breast carcinoma treated with neoadjuvant chemotherapy, which showed pCR in both the treated breast and axillary lymph nodes; only acellular mucin pools remained, which presented as a persistent mass-forming lesion that was also clinically and radiologically interpreted as an incomplete response. 10 It appears that keratin derived from breast carcinoma cells, especially in a setting of squamous differentiation, can form acellular proteinaceous debris, similar to mucin, which may persist at tumor bed site as a mass-forming lesion, and mimic residual tumor. Post-therapy residual mucin or keratin at the tumor bed location is a pitfall, which can masquerade as residual tumor/incomplete pathologic response.
In conclusion, this report highlights the crucial role of pathologic evaluation in accurately assessing the response to neoadjuvant chemotherapy. Acellular material at the tumor bed site, like keratinous debris and acellular mucin pools, may present as residual “tumor-like” masses on physical examination and imaging. MBC can show pCR; further studies evaluating the correlation of therapy response with histological components of MBC are needed.
Footnotes
Ethical Approval
WIH IRB, Institution does not require IRB approval for a case report
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
