Abstract
Mycobacterial spindle cell pseudotumor is a rare manifestation of atypical mycobacterial infection. The process consists of a tumor-like proliferation of bland, spindle-shaped histiocytes that contain acid-fast-positive mycobacteria. Most patients are immunocompromised, and the lymph nodes are the most commonly involved site. The presentation of this process as an isolated aggressive osseous lesion is rare and conjures up a wide differential diagnosis, including neoplastic conditions. Accurate diagnosis of this entity is essential for proper management. We report a 79-year-old man with hypogammaglobulinemia who presented with an aggressively appearing lytic tibial lesion after sustaining a fall. Biopsy of the lesion revealed a mycobacterium-laden spindle cell pseudotumor without granuloma formation. The mycobacteria were visible on acid-fast Ziehl-Neelsen (AFB) stain and modified acid-fast (Fite) stain as well as PAS and GMS stains. PCR of the microbiology culture identified Mycobacterium kansasii. Our report demonstrates that, although rare, mycobacterial spindle cell pseudotumor is a diagnostic consideration when encountering a lytic bone lesion in immunocompromised patients composed of cytologically benign, spindled histiocytes without characteristic caseating granuloma formation. Histochemical stains, including PAS, GMS, AFP, or Fit, are imperative to identify intracellular organisms.
Introduction
Mycobacterial infection typically incites a granulomatous reaction through a cell-mediated delayed-type hypersensitivity reaction.1,2 Rarely, mycobacterial infection may manifest as a mycobacterial spindle cell pseudotumor (MSCP), a mass-forming lesion composed of spindle-shaped histiocytes that contain acid-fast-positive mycobacteria, without caseating granulomas. 3 Most patients were immunocompromised, including those with acquired immunodeficiency from human immunodeficiency virus infection,4–6 stem cell or solid organ transplant,7,8 or patients on immunosuppressants for autoimmune/inflammatory disorders.9,10 Lymph node involvement accounted for nearly half of the MSCPs, followed by involvement of the skin, soft tissue, abdomen, and lungs.3,11 Presentation as an isolated osseous lesion is rare. 12 The most commonly encountered causative species is Mycobacterium avium complex,3,11 while Mycobacterium kansasii is rarely reported as the pathogen in MSCP. 13 In patients with late-stage AIDS, M. kansasii infection typically affects the lungs with thoracic lymph node involvement, predominantly in the form of granulomatous inflammation. 14 Herein, we report a patient with hypogammaglobulinemia who presented with an aggressively appearing tibial lesion that showed MSCP on biopsy and was identified as M. kansasii infection on mycobacterial PCR.
Case Report
The 79-year-old male patient had had persistent pain in his anterior left knee after a fall 1 month before. The past medical history was significant for neurocognitive decline, hypertension, insulin-dependent type 2 diabetes mellitus, a diabetic right foot ulcer, peripheral artery disease, bioprosthetic aortic valves, low-grade B-cell lymphoma off treatment for 7 years, and hypogammaglobulinemia on regular intravenous immunoglobulin. He denied fever, chills, diaphoresis, fatigue, and night sweats. On physical examination, he had tenderness below the patellar tendon along the proximal tibia with an intact skin surface and no evidence of erythema, warmth, or bruising. He had unrestricted range of motion of his left knee without joint laxity or signs and symptoms of neurovascular injury. The erythrocyte sedimentation rate was 50 mm/h (reference: 0-20 mm/h) and C-reactive protein 10.2 mg/dL (reference: 0.0-1.0 mg/dL), but his complete blood count was within normal limits.
Both the plain radiograph and computed tomography (CT) showed a permeative, ill-defined lesion extending from the proximal tibial metaphysis into the diaphysis, with endosteal cortical scalloping and periosteal reaction. Fat infiltration was also noted on CT, although a soft tissue mass was not present (Figure 1A). Magnetic resonance imaging revealed a geographic marrow-replacing lesion with endosteal scalloping (Figure 1B). Chest CT showed scattered punctate calcified granulomas and subpleural millimeter pulmonary nodules. He did not have any abdominal mass lesions on imaging. With both osteomyelitis and neoplasm as the differential diagnoses, the patient underwent a biopsy of the tibial lesion.
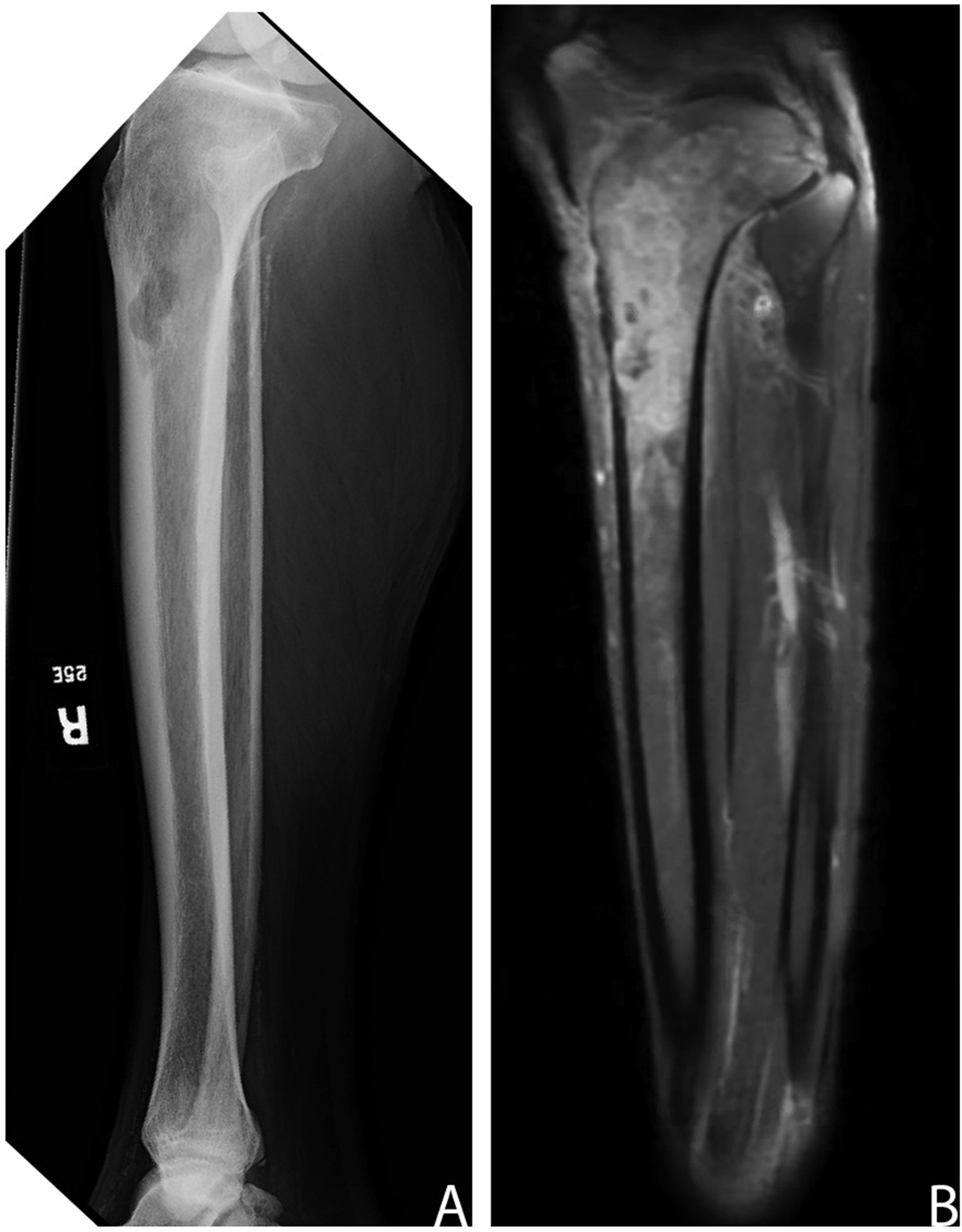
(A). Plain radiograph shows an ill-defined intramedullary lytic lesion in the tibia, involving the meta-diaphysis. Cortical scalloping is present at the medial aspect. (B). T1 fat-saturated MRI image shows a marrow-replacing lesion in the meta-diaphysis. Calcification of the tibia vessels is also evident.
Histological examination of the tibial biopsy revealed a cytologically bland spindle cell proliferation replacing the marrow without granulomas (Figure 2A). The spindle cells were arranged in a vaguely storiform to fascicular growth pattern with chronic inflammatory cells dispersed throughout (Figure 2B). The lamellar bony trabeculae surrounded by spindle cells were necrotic, with some areas showing “creeping substitution” by benign woven bone and scattered small clusters of neutrophils (Figure 2C). On high power examination, the monotonous spindle cells had low nuclear-to-cytoplasmic ratios with abundant pale to granular cytoplasm. There was no nuclear overlap among the spindled histiocytes. The round to oval, normochromatic nuclei were similar in size to those of adjacent lymphocytes and possessed thin nuclear membranes and finely dispersed chromatin. A distinct chromocenter was noted in scattered lesional cells (Figure 2D). The spindle cells were strong and diffusely immunoreactive for histiocyte marker CD163 (not shown). Abundant intracytoplasmic filamentous bacteria were highlighted on AFB (Ziehl-Neelsen), GMS, and PAS histochemical stains, and weakly demonstrated with Fite (modified acid-fast) stain (Figure 3). No microorganism was seen on tissue Gram stain (not shown). Concurrent atypical mycobacteria culture of the aspirate identified M. kansasii on GeneXpert MTB-RIF PCR. There was no growth on tissue and fungal cultures. Overall diagnostic features were those of an MSCP. The patient passed away due to an accident before the initiation of antimicrobial treatment.
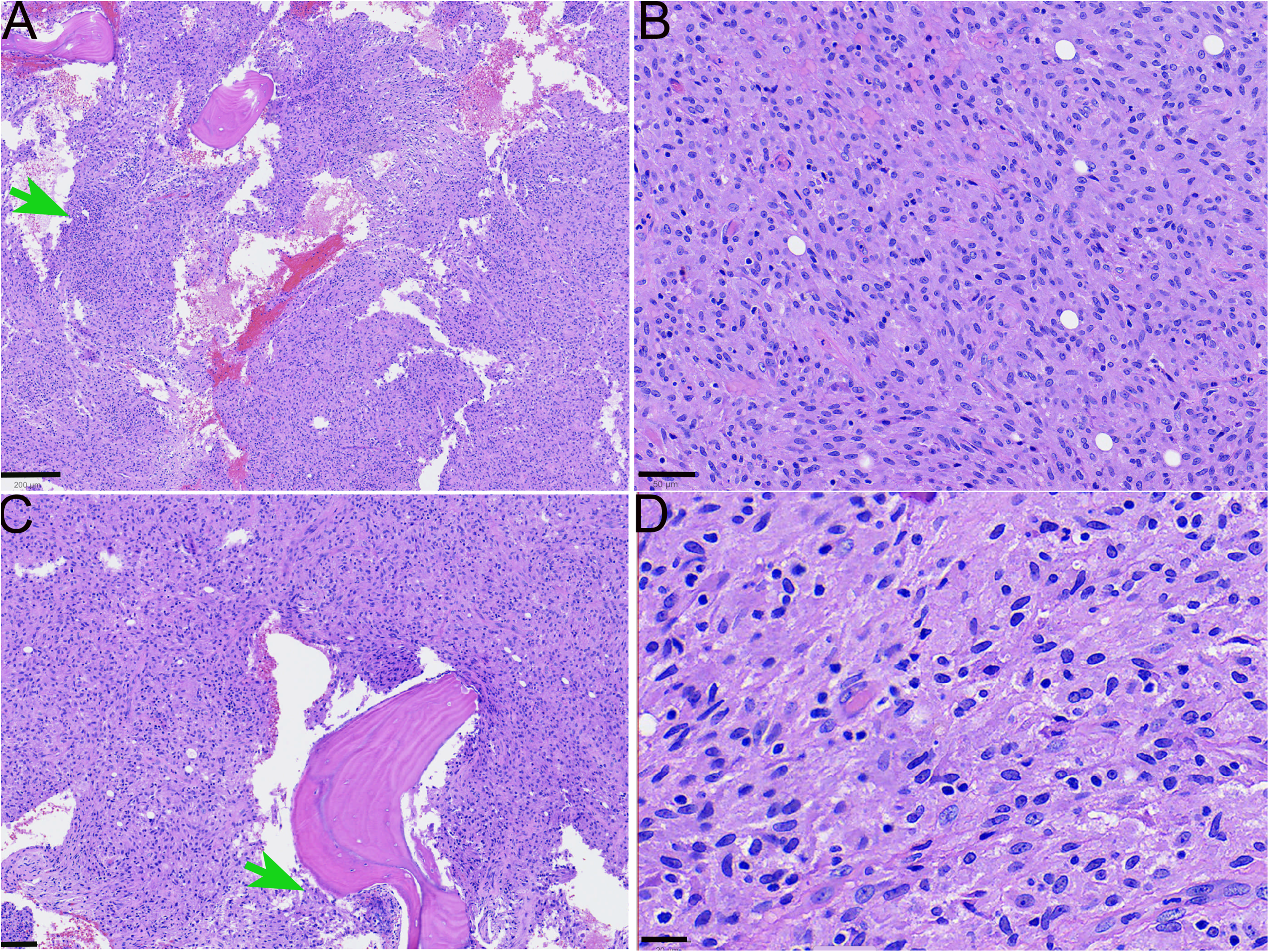
Microscopic findings in the osseous MSCP. (A). Low power view of the biopsy shows marrow replacement by a cellular spindle cell proliferation with spotty inflammation (arrow). No distinct granulomas are appreciated. (H&E, Scale bar: 200 micrometers). (B). Medium power demonstrates a monotonous spindle cell proliferation in vaguely fascicular to storiform patterns. There are scattered chronic inflammatory cells. (H&E, Scale bar: 50 micrometers). (C). Necrotic bone is seen off center to the right, and “creeping substitution” by woven bone is present surfacing necrotic bone to the left of the reversal line. The arrow points to a small neutrophilic microabscess. (H&E, Scale bar: 100 micrometers). (D). High power shows the spindle cells have abundant pale cytoplasm. The small nuclei have smooth nuclear contours and are euchromatic. (H&E, Scale bar: 20 micrometers).
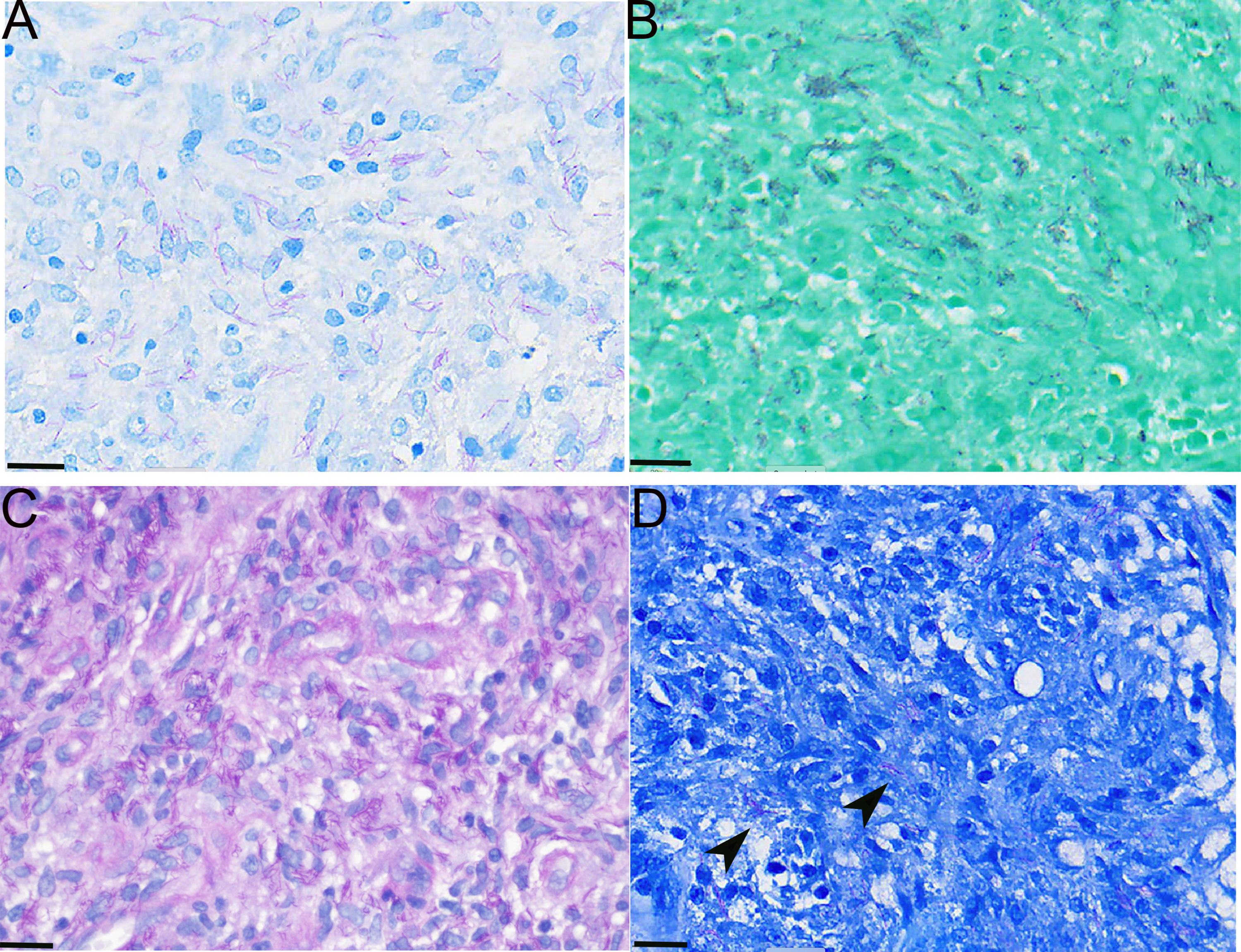
Mycobacterium kansasii is detectable on AFB (A), GMS (B), PAS (C), and Fite (D) stains (scale bars for all: 20 micrometers).
Discussion
Mycobacterial infection typically elicits a delayed, cell-mediated (Type IV) hypersensitivity reaction characterized by granuloma formation.1,2,15 A histiocytic reaction resulting in an MSCP without well-defined granuloma formation is rare and predominantly observed in immunocompromised patients who fail to mount a hypersensitivity response.3,11 MSCP can pose diagnostic challenges due to its clinicopathological resemblance to spindle cell mesenchymal tumors.
In our patient with osseous MSCP, the radiologic differential diagnoses were neoplasm and osteomyelitis. For the first consideration, an aggressive lytic lesion in the meta-diaphysis of a long tubular bone without any radiologically detectable matrix in an elderly male patient would suggest a myeloma, metastatic carcinoma (most commonly prostate and kidney), or an aggressive mesenchymal tumor. The first 2 can be easily excluded on routine light microscopy. The spindled histiocytes exhibited bland cytomorphologic features, which militate against a sarcoma diagnosis. However, other potential mesenchymal mimickers of MSCP include benign fibrous histiocytoma of the bone, desmoplastic fibroma of bone, and angiomatous fibrous histiocytoma. A classic giant cell tumor of bone is not a differential diagnosis for MSCP, due to its unique radiologic findings and classic morphology, characterized by an even distribution of osteoclast-like giant cells among the mononuclear tumor cells. When extensive fibrohistiocytic change obscures the cytoarchitecture, previously known as a benign fibrous histiocytoma, 16 giant cell tumor of bone may mimic MSCP histologically. In this situation, finding the classic giant cell tumor area in a well-defined, eccentric, and expansile lytic lesion in the epimetaphysis can aid the diagnosis. About 95% of the giant cell tumors of bone harbor pathogenic H3-3A gene mutations, which can be detected by sequencing or mutation-specific antibodies in areas with mononuclear tumor cells. Desmoplastic fibroma of bone, similar to its morphologic counterpart in the soft tissue, desmoid fibromatosis, is characterized by sweeping fascicles of fibroblasts in a background of dense fibrosis. 17 An inflammatory infiltrate is not a typical feature. Cytologically, the fibroblasts in desmoplastic fibroma of bone lack the abundant pale to granular cytoplasm and exhibit a more elongated nucleus than the spindled histiocytes of MSCP. The fibroblasts in desmoplastic fibroma of bone do not react with CD163. Angiomatoid fibrous histiocytoma, primarily a soft tissue tumor, rarely occurs in the bone. 18 Like MSCP, it typically exhibits a dense lymphoplasmacytic infiltrate. As its name implies, angiomatoid fibrous histiocytoma is composed of lesional cells with a histiocytoid appearance, but the vesicular nuclei are generally larger than the intermixed lymphocytes, and nuclear overlap is more common. Angiomatoid fibrous histiocytoma tumor cells are characteristically arranged in a syncytial pattern, and concentric whirling and storiform patterns are common. Lastly, their eosinophilic cytoplasm lacks the granularity seen in the spindled histiocytes of MSCP.19,20 Notably, both desmoplastic fibroma of bone and angiomatoid fibrous histiocytoma occur more commonly in young adults.17,20 As a translocation-associated neoplasm, detecting EWSR1/FUS fusion with CREB1 or ATF1 can help confirm the diagnosis of angiomatoid fibrous histiocytoma. Because the intracellular mycobacteria of an MSCP may cross-react with desmin and actin, a smooth muscle neoplasm may enter the differential diagnosis. However, the spindled histiocytes of MSCP lack rigid, intersecting fascicular growth, do not possess brightly eosinophilic cytoplasm, and the aforementioned immunomarkers do not highlight a fibrillar cytoplasm as they do in smooth muscle lesions. 4
With the combination of spindled histiocytes and lymphoplasmacytic inflammation, histiocytosis could be another microscopic differential diagnosis, and none of them possesses intracytoplasmic microorganisms. Osseous Rosai-Dorfman disease often presents as an intramedullary lytic lesion with a sclerotic border, and large epithelioid histiocytes with emperipolesis should be at least focally present. 21 Erdheim-Chester disease typically involves the long bones bilaterally and symmetrically, showing cortical osteosclerosis in the diaphysis and metaphysis on plain X-rays. 22 Langerhans cell histiocytosis is extremely rare in older individuals, but it can present as an aggressive lytic lesion. 23 Grooved nuclei are a distinct feature of Langerhans cell histiocytosis. Histiocytosis is a clonal neoplastic disorder characterized by recurrent somatic alterations in the mitogen-activated protein kinase-extracellular signal-regulated kinase (ERK) pathway. The strong expression of cyclin D1 and p-ERK in neoplastic histiocytes serves as a strong surrogate to distinguish them from the reactive histiocytic infiltrates, as seen in MSCP. 24
Clinically, the patient had an elevation of inflammatory markers, which would be supportive of an acute osteomyelitis. The intact skin excluded a contiguous osteomyelitis stemming from an overlying ulcer, and the absence of leukocytosis or fever would be unusual for a hematogenously derived osteomyelitis. Neutrophilic infiltration and necrotic bone were present in our patient's biopsy, and neutrophilic inflammation was observed in 50% of patients in a recent multiinstitutional study of MSCP. 11 Neutrophils and dead bone are key histologic features for the diagnosis of acute osteomyelitis, 25 for which the most common pathogens include Staphylococcus, Streptococcus, Pseudomonas, and Enterobacter. However, the neutrophilic inflammation presented as scattered, minute clusters of cells in an otherwise spindle cell predominant MSCP and was not intimately associated with degenerating shards of bone. In our MSCP patient, scattered chronic inflammatory cells, including plasma cells, were observed percolating throughout the lesion. In the absence of a granuloma, a chronic osteomyelitis might enter the differential diagnosis. Compared to nonmycobacterial chronic osteomyelitis, our osseous MSCP lacked marrow fibrosis. Rather, MSCP contains vaguely storiform to fascicular spindled histiocytes with clear to granular cytoplasm resembling a neoplasm. 11 The spindled histiocytes are different in cytomorphology from the fibroblasts and myofibroblasts of nonmycobacterial chronic osteomyelitis, and thickened remodeling bone would be more common than necrotic bone in the latter entity. 25 Nonetheless, a careful examination of the whole biopsy and evaluation of each cellular component can help avoid missing an MSCP that has a minor component of acute or chronic active osteomyelitis.
Even though typically used as histochemical stains for fungal microorganisms, PAS and GMS stain phagocytosed atypical mycobacteria in paraffin sections.26,27 The microorganisms detected on both stains in our patient had a curvilinear to filamentous morphology, lacked branching structures, and were significantly thinner than typical hyphae. Mycobacteria are generally 0.2 to 0.6 micrometers in diameter, whereas hyphae are 2 to 8 micrometers. Therefore, detecting the microorganisms on PAS and/or GMS does not equate to a fungal infection, and careful evaluation of the size and morphology can help avoid a wrong conclusion. The long, curvilinear structures seen on the AFB stain may also make Nocardia a diagnostic consideration. Similar to atypical mycobacteria, Nocardia primarily affects immunocompromised patients, 27 and they have a branching, filamentous morphology on AFB and GMS. Unlike atypical mycobacteria, Nocardia is typically PAS-negative and Gram-positive.
In conclusion, we report a mycobacterial spindle cell pseudotumor of the bone without typical granulomatous inflammation. MSCP is a diagnostic consideration when confronted with an immunosuppressed individual with a solitary lytic bone lesion. Routine histochemical stains for fungi can detect intracellular mycobacteria, and considering the differences in size and morphology of the microorganisms can help establish the correct diagnosis.
Footnotes
Ethical Approval
Case reports and reviews are exempt from formal ethics as per our institutional review policies.
Informed Consent (to Participate and for Publication)
Not applicable. This article received Yale IRB exemption and thus did not require informed consent beyond checking patient opt out status for research through the Joint Data Analytics Team.
Author Contributions
HW wrote the manuscript; DL and WL critically reviewed the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
Not applicable; this article did not contain any clinical trials.
