Abstract

Mesenchymal chondrosarcoma (MCS) is a rare, high-grade malignant neoplasm that most commonly arises in bone, with approximately 40% of examples occurring in extraskeletal soft tissues. Primary involvement of visceral organs is exceedingly rare. Fewer than 25 primary genitourinary MCSs have been reported in the literature, including tumors arising in the kidney (most commonly), ureter, uterus, ovary, and prostate.1–5 To the best of our knowledge, the primary MCS of the urinary bladder has not been previously reported.
A woman in her late 20s with a 2-year history of chronic pelvic pain presented with acutely worsening hematuria and urinary obstruction. Initial computed tomography (CT) and magnetic resonance imaging (MRI) revealed a 13.5 cm heterogeneous anterior pelvic mass with dense chondroid calcification and aggressive features (Figure 1). The mass invaded through the anterior bladder wall with a bulky tumor both within the bladder lumen and enlarging the retropubic space (Figure 2A). The fact that the bladder was not merely being displaced by the mass supported that the tumor was within the bladder wall. Additional imaging identified a 2.1 cm partially calcified lesion along the left pleural surface and a 1 cm lytic lesion in the left pubic body, both concerning for metastases. Although the large anterior pelvic mass abutted the left pubic body, it did not arise from bone, as there was no osseous destruction or direct contact between the large soft tissue mass and small osseous lesion (Figure 2B).
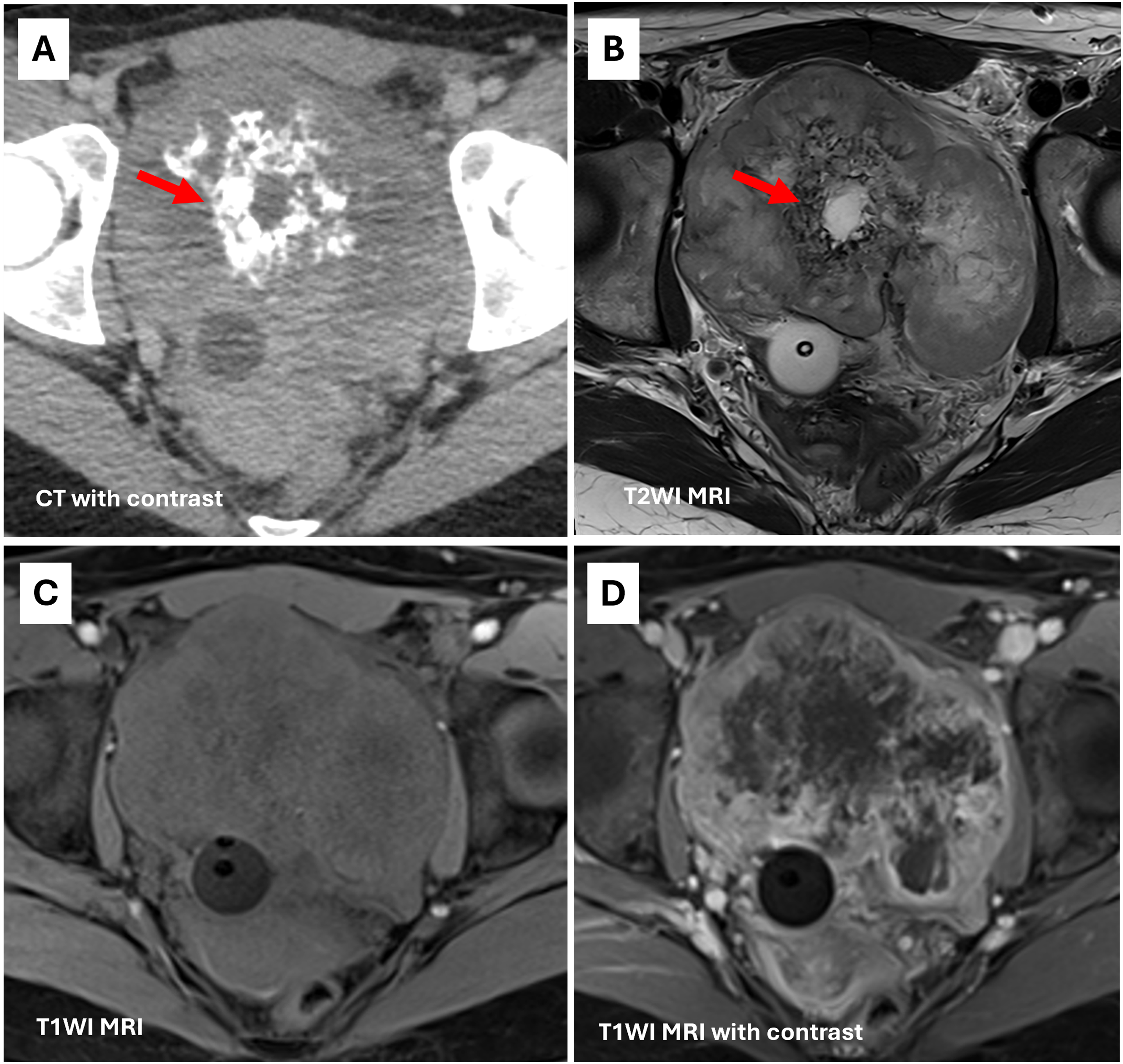
(A, B) Large mass arising from the urinary bladder demonstrates dense chondroid calcifications on CT corresponding with areas of low signal on T2WI MRI (arrow). (C, D) Mass is isointense to muscle on T1WI with heterogeneous enhancement and areas of central necrosis.
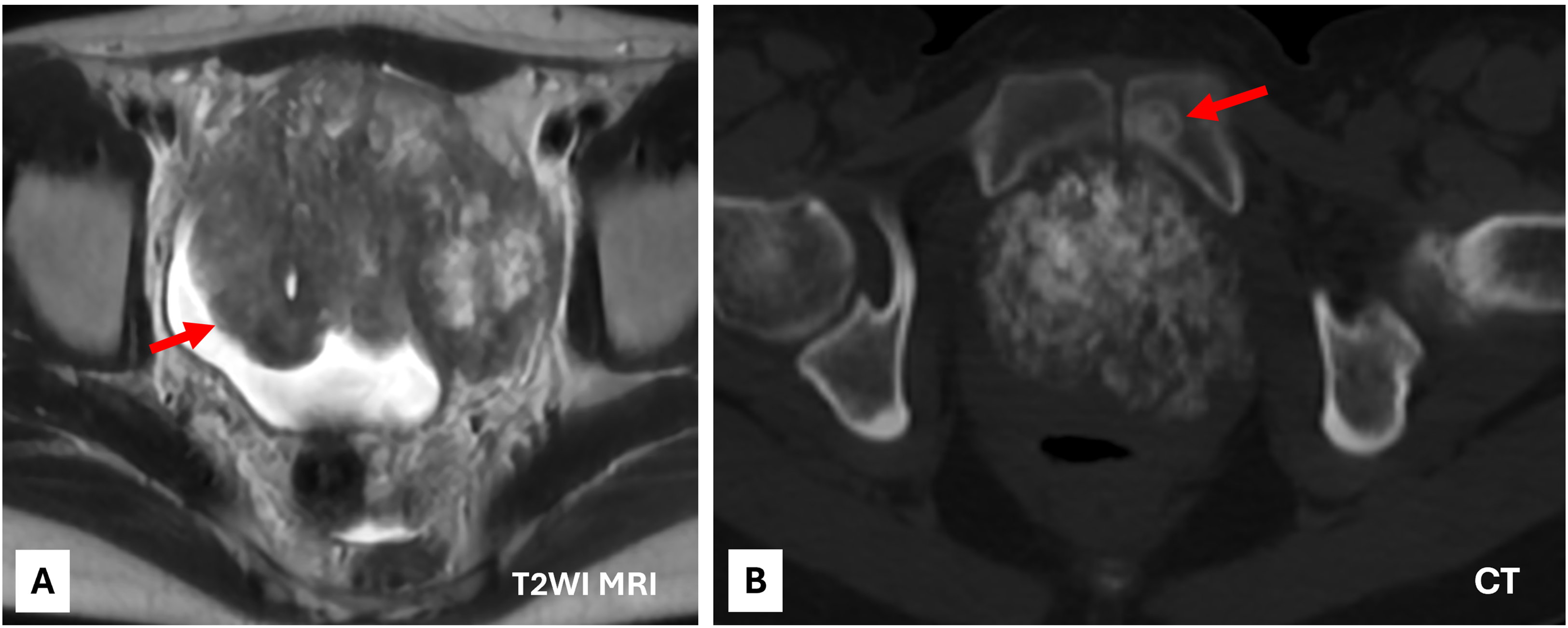
(A) Mass (arrow) along the anterior wall of the bladder with tumor growth into the bladder lumen and filling the retropubic space. Tumor abuts bilateral pelvic sidewalls. (B) Lesion in left pubic body (arrow) is concerning for metastasis.
An ultrasound-guided transabdominal biopsy of the urinary bladder mass revealed sheets of uniform round cells with round nuclei, scant eosinophilic cytoplasm, and brisk mitotic activity (Figure 3A). Frequent apoptotic bodies and multifocal necrosis were present. No definitive matrix or chondroid differentiation was identified. Immunohistochemically, the neoplastic cells were positive for NKX2-2, CD99, and TLE1 (multifocal weak), and negative for desmin, myogenin, CD45, TDT (DNTT), CD20, CD3, SOX10, WT1, S100, keratin AE1/AE3, CAM 5.2, synaptophysin, INSM1, uroplakin 2/3, GATA3, and SS18-SSX (Figure 3B and C). EWSR1 and FUS fluorescence in situ hybridization (FISH) studies were negative for gene rearrangements. Whole transcriptome sequencing (RNA-sequencing) identified a HEY1::NCOA2 fusion involving exon 4 of HEY1 (NM_001040708, chr8:80678886) and exon 13 of NCOA2 (NM_001040708, chr8:71057083), resulting in a predicted in-frame fusion product and confirming the diagnosis of MCS (Figure 3D). Targeted next-generation sequencing panel demonstrated multiple copy number alterations, including gains in 9q, 10q, 11q, 12p/q, and 19p, and losses in 2p, 2q, 3p, 6q, 15q, and 19q. The tumor was microsatellite stable and exhibited a tumor mutational burden of 10.3 mutations per megabase. The patient underwent neoadjuvant chemotherapy [vincristine, doxorubicin, and cyclophosphamide, alternating with ifosfamide and etoposide] prior to planned surgical intervention.
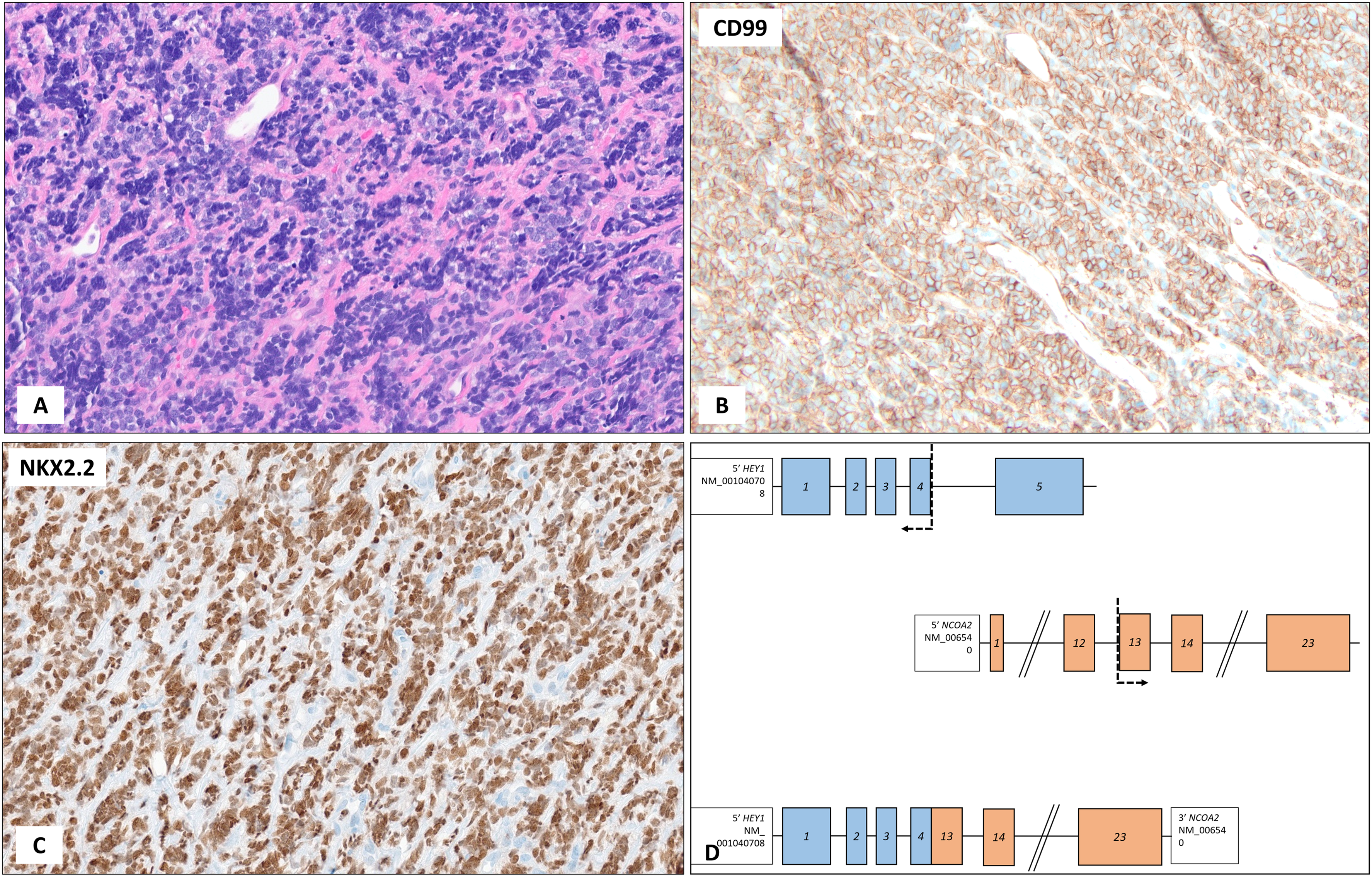
(A) The tumor is composed of small- to medium-sized, poorly differentiated round cells with a high nuclear-to-cytoplasmic ratio. (B, C) The tumor cells are diffusely positive for CD99 and NKX2-2. (D) Model schematic of HEY1::NCOA2 fusion. Whole transcriptome sequencing (RNA-sequencing) identified a fusion involving exon 4 of HEY1 and exon 13 of NCOA2, resulting in a predicted in-frame fusion product.
The morphologic differential diagnosis for a small round blue cell tumor in the urinary bladder is broad and includes lymphoma, small cell neuroendocrine carcinoma, high-grade urothelial carcinoma with small cell features, poorly differentiated solitary fibrous tumor, poorly differentiated synovial sarcoma, and a spectrum of small round cell sarcomas. The latter includes entities such as Ewing sarcoma, alveolar rhabdomyosarcoma, and desmoplastic small round cell tumor.6,7 In this tumor, the absence of epithelial, lymphoid, neuroendocrine, and myogenic marker expression, along with negative immunohistochemical staining for STAT6 and SS18-SSX and the lack of EWSR1 or FUS gene rearrangements by FISH, effectively excluded many of these possibilities. The presence of chondroid calcifications on imaging raised suspicion for an undersampled MCS, which was subsequently confirmed by the identification of a HEY1::NCOA2 gene fusion.
MCS is characterized by a biphasic morphology composed of undifferentiated small round to ovoid cells with scant cytoplasm abruptly juxtaposed with islands of well-differentiated cartilage. A hemangiopericytoma-like vascular pattern is a hallmark feature of the primitive component. In small biopsies, however, the cartilaginous component may be entirely absent, leading to potential misclassification as other round cell neoplasms. This sampling bias underscores the importance of maintaining a high index of suspicion, particularly when imaging shows calcification and histology reveals primitive round cell sarcoma with staghorn vasculature. At the molecular level, the vast majority of MCS harbor a pathognomonic HEY1::NCOA2 fusion resulting from a t(8;8)(q13;q21) translocation, which plays a central role in tumorigenesis by dysregulating transcriptional programs involved in mesenchymal differentiation. 8 An alternative IRF2BP2::CDX1 fusion has been identified in a single example. 9 MCS is an aggressive malignancy characterized by a high risk of local recurrence and distant metastasis, most commonly to the lungs and bones. Prognostic factors affecting survival include tumor size, presence of metastases at diagnosis, completeness of surgical resection, and, to a lesser extent, anatomic site.10,11
In summary, this report expands the known anatomic distribution of MCS to include the urinary bladder and underscores the importance of considering this rare entity in the differential diagnosis of bladder-based small round cell tumors, particularly when imaging reveals calcifications or histologic examination demonstrates a hemangiopericytoma-like vascular pattern.
Footnotes
Ethical Approval
According to the institutional review board policy, individual case reports are exempt from ethics review.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
