Abstract
Neuroendocrine differentiation is a common feature observed in epithelial tumors. Both exocrine and neuroendocrine differentiation can be observed in tumors originating from the gastrointestinal system. However, since 1924, these tumors have been described under different names, leading to confusion among clinicians. While such tumorsare generally referred to as mixed adenocarcinoma-neuroendocrine carcinoma in the 4th WHO classification, amphicrine tumors are not currently included in any specific tumor classification. This study aimed to enhance the understanding of the clinicopathological features of amphicrine tumors, thus contributing to their standardized classification. A 52-year-old woman presented to our center with persistent nausea and vomiting. Physical examination revealed no findings except tenderness in the right upper quadrant of the abdomen. Intraoperative consultation revealed a tumor, approximately 8 mm in diameter, in the ampullary region. A tumor showing neuroendocrine differentiation was observed in the hematoxylin and eosin-stained sections. After formalin fixation, tumors with neuroendocrine differentiation were observed in the sections. Trabecular and glandular patterns, and nuclear salt-and-pepper chromatin were observed in the tumor cells. Immunohistochemically, the tumor cells were positive for synaptophysin and chromogranin. Alcian blue staining was positive for cytoplasmic mucin, and diffuse CDX2 expression was observed. As a result, the diagnosis was confirmed as “amphicrine carcinoma.” Based on these findings, this type of tumor should be included as a separate histological type from adenocarcinomas, neuroendocrine carcinomas, and mixed neuroendocrine-nonneuroendocrine neoplasm. This will also support a more standardized approach in terms of the treatment and follow-up protocols for patients with this rare tumor.
Keywords
Introduction
Neuroendocrine differentiation is a common feature observed in epithelial tumors. Both exocrine and neuroendocrine differentiation can be observed in tumors originating from the gastrointestinal system. These tumors were first described in 1924 and have been reported in the literature under different names, such as collision, combined, or amphicrine tumors. 1 However, these definitions have led to confusion among clinicians. To address this, such tumors are referred to as mixed adenocarcinoma-neuroendocrine carcinoma (MANEC) in the 4th edition of the WHO classification. According to this definition, neuroendocrine and adenocarcinoma components are present in the same tumor, and both tumor components must be at least 30%. 2 However, this term has certain limitations. Specifically, it assumes that the nonneuroendocrine component is exclusively adenocarcinoma, and although rare, it does not include entities other than adenocarcinoma, such as squamous cell carcinoma or signet ring cell carcinoma. Therefore, the proposed new nomenclature, mixed neuroendocrine-nonneuroendocrine neoplasm (MiNEN), has been adopted in the 2020 WHO classification. 3 However, MiNEN tumors are seen as collisions or combined tumors in which both components are observed within the same tumor. This nomenclature encompasses tumors with dual differentiation, where both endocrine and nonneuroendocrine components, which are morphologically distinct, coexist within the same lesion.
Amphicrine tumors are rare and exhibit bivalent cell differentiation. These tumor cells simultaneously exhibit mucin and neuroendocrine granules, with no dual components observed in them. Amphicrine tumors were initially included in the intermediate-grade MANEC group, according to earlier WHO classifications. 4 However, the latest WHO classification recommends distinguishing amphicrine tumors from the MiNEN tumor group, citing the importance of more studies on this subject. 5 These tumors have morphological and genetic characteristics different from those of the MiNEN group. 6
Although rare, amphicrine carcinomas can be observed anywhere in the gastrointestinal system and are not currently included in any tumor classification. Therefore, this study aimed to contribute to the existing literature, as it reports a rare example of amphicrine carcinoma, thereby enhancing understanding of its clinicopathological features.
Case Report
A 52-year-old woman presented to our center with persistent nausea and vomiting. Physical examination was unremarkable except for tenderness in the right upper quadrant of the abdomen. Initial biochemical tests revealed the following: Alanine aminotransferase, 54 U/L; aspartate transaminase, 52 U/L; alkaline phosphatase, 550 U/L; gamma-glutamyl glutamine transferase, 616 IU/L; and total bilirubin, 0.98 mg/dL. Magnetic resonance imaging revealed contrast enhancement in the ampullary region. Subsequent magnetic resonance cholangiopancreatography followed by endoscopic retrograde cholangiopancreatography (ERCP) were performed. During ERCP, mucosal protrusions with an adenomatous appearance were observed, and biopsies were obtained and submitted to our department for evaluation.
A biopsy of the ampulla revealed adenomatous mucosal tissue fragments with low-grade dysplasia and the notable presence of goblet cells (Figure 1). Consequently, a cholecystectomy with Whipple operation was planned.
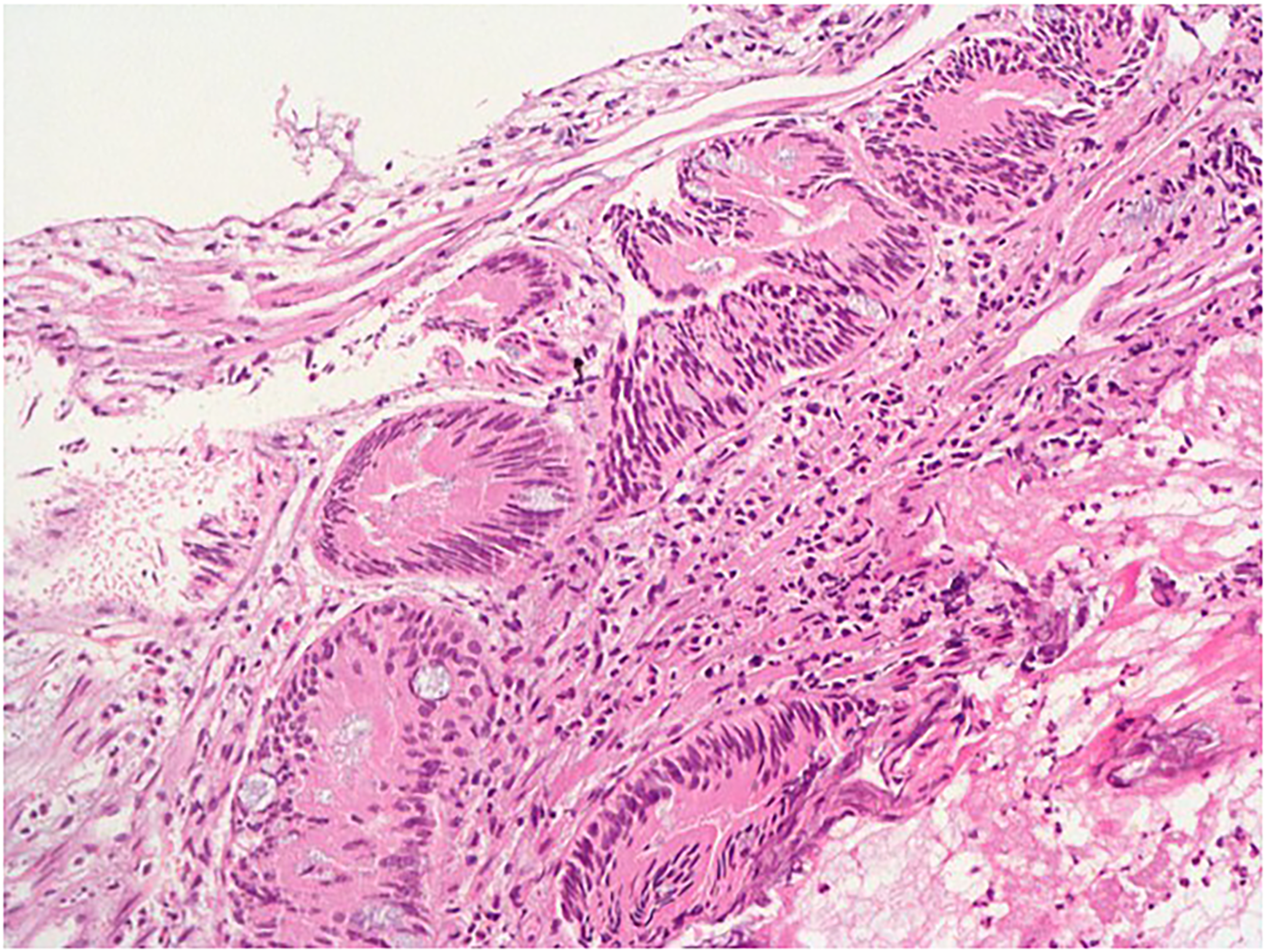
The first biopsy of the ampulla shows a low-grade adenomatous type of dysplasia (H&E, ×200).
During surgery, endomucosal ampullectomy was performed, preserving the muscular layer. The resected specimen, along with the 2 regional lymph nodes, was submitted to the pathology department for intraoperative consultation. Accordingly, a tumor of approximately 8 mm in diameter was observed in the ampullary region. During intraoperative consultation, histopathological evaluation revealed tumor areas with a trabecular architectural pattern and relatively monotonous cells showing salt-and-pepper chromatin, reported as a tumor suggestive of neuroendocrine differentiation (Figure 2a-b). It was also reported that the lymph nodes were reactive. Based on this result, Whipple surgery was not performed due to the small tumor size and negative lymph node involvement. Surgical management was limited to ampullectomy. The material detected by the pathology department was processed in its entirety by marking surgical margins.
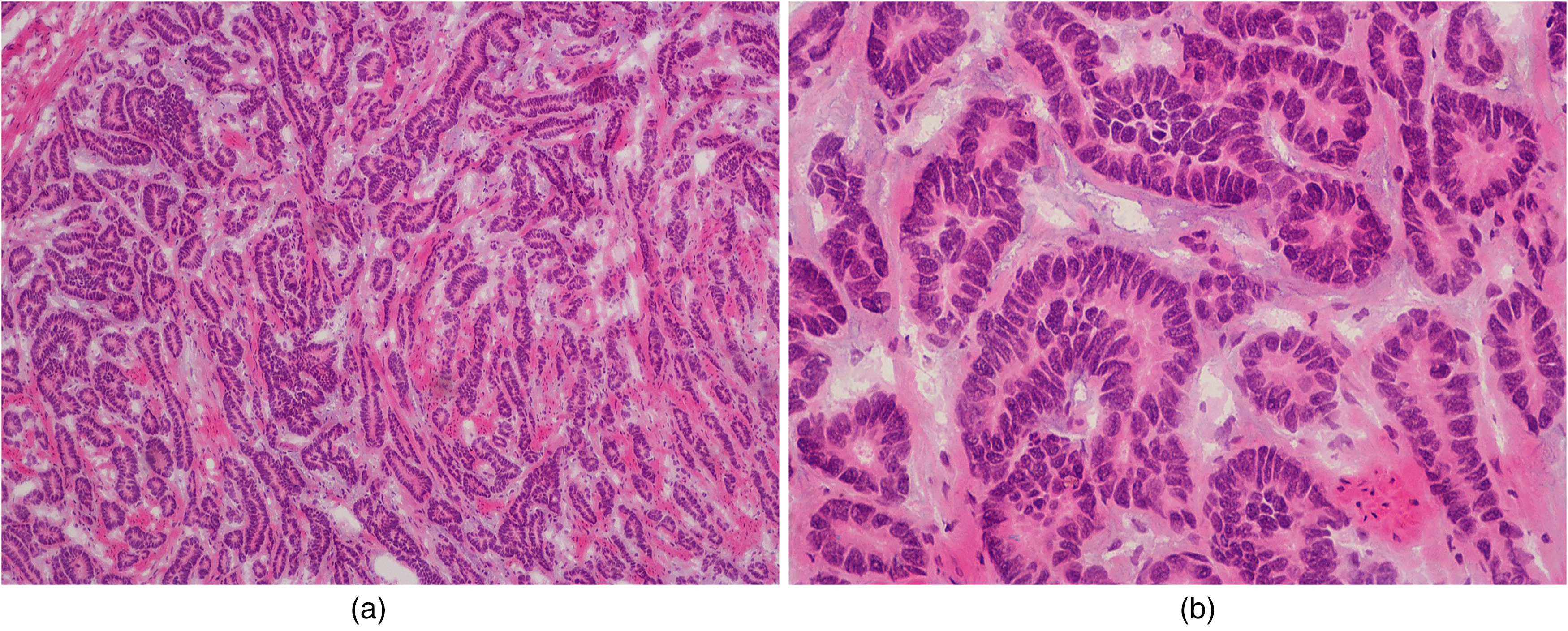
Frozen section analysis reveals a tumor with neuroendocrine features. (a) H&E ×100; (b) H&E ×400.
In the sections obtained after formalin fixation, a well-differentiated tumor with neuroendocrine differentiation was observed. Trabecular and glandular patterns, and nuclear salt-and-pepper chromatin were observed in the tumor cells. Invasion into the muscularis propria was noted, corresponding to a pathological T-stage of pT2. No lymphovascular invasion was observed, and neuroendocrine tumors and MiNEN-group tumors were considered in the differential diagnosis. However, careful examination revealed mucin in the cytoplasm of some tumor cells with neuroendocrine features. Immunohistochemically, the tumor cells were positive for synaptophysin and chromogranin. Alcian blue staining confirmed the presence of cytoplasmic mucin, and diffuse CDX2 expression was observed. Similar to the previous biopsy, the surface epithelium showed adenomatous dysplasia. The Ki-67 proliferation index of the tumor was approximately 80% (Figure 3a-f), and the surgical resection margins were tumor-free.
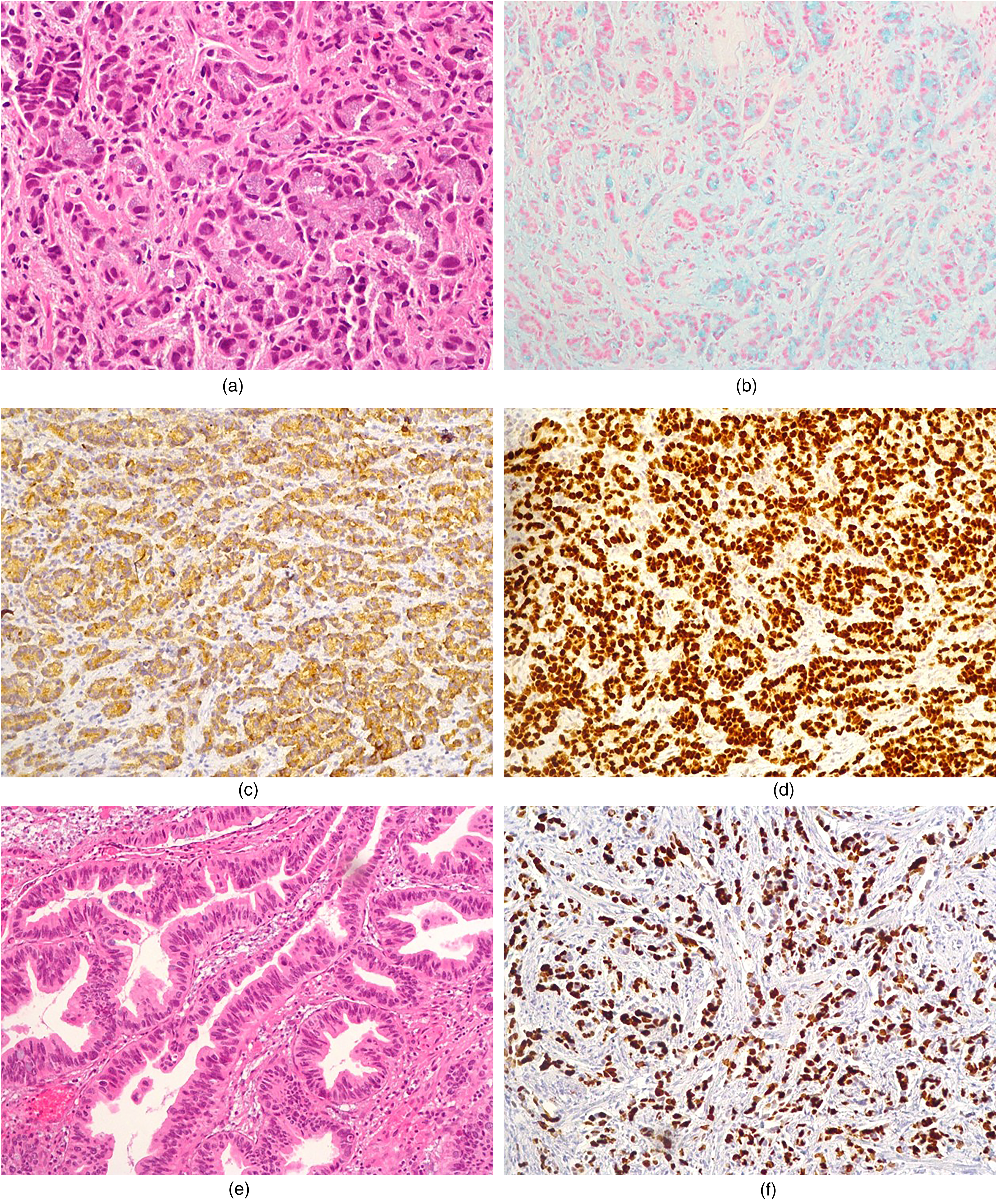
Morphological observation of the tumor showing neuroendocrine differentiation and mucin material in the cytoplasm of some cells (shown with an arrow). (a) H&E ×200. Cytoplasmic mucin staining is observed with Alcian blue histochemistry; (b) Alcian blue ×100. Tumor cells are positive with synaptophysin; (c) Synaptophysin ×100. CDX2 immunohistochemistry, an intestinal marker, is also widely positive in the same area; (d) CDX2 ×100. Adenomatous type dysplasia is observed in the surface epithelium; (e) H&E ×200. Ki67 proliferation index is quite high; (f) Ki-67 ×100.
DNA and RNA were extracted from formalin-fixed, paraffin-embedded tissue blocks. For DNA analysis, the genes listed in the targeted panel were sequenced using the BRG Hema Kit via next-generation sequencing (NGS) on the DNBSEQ-400 platform. During data evaluation, all coding exonic regions and 20 base pairs of intronic boundaries were analyzed, achieving 100% coverage at a minimum read depth of 20×. The obtained variants were analyzed using the Genomize Seq V8.13 database.
For RNA analysis, gene fusions included in the corresponding panel were investigated using NGS on the DNBSEQ-400 system. All coding regions of these genes (excluding intronic, 5′, and 3′ UTR regions) were examined, and exon–intron junctions (±10 bp) were included in the analysis. The detected variants were interpreted using the Nanodigmbio Bioinformatic Analysis Platform (v1.4) and the Personal Cancer Genome Report database (Tables 1 and 2).
NGS Solid Cancer Panel.
RNA Fusion Panel.
According to these analyses, only a duplication in the KMT2C gene, a stop codon mutation in the SOS1 gene, and an indel in the MED12 gene were detected. No fusion transcripts involving the genes included in the panel were detected in the fusion analysis.
As a result, the diagnosis was confirmed as “amphicrine carcinoma” After 6 months, multiple control biopsies were obtained from the anastomotic line, and no pathological evidence of recurrence was observed. The patient was planned for follow-up every 6 months with clinical, laboratory and imaging findings and no evidence of recurrence or metastasis has been detected at most recent follow-up.
Discussion
The term amphicrine tumor has been used for many years and was first described in 1965. 7 Amphicrine tumors can be observed anywhere in the gastrointestinal tract. A well-known amphicrine tumor is the appendiceal goblet cell carcinoid; however, owing to its clinical and molecular similarity to adenocarcinoma, it has since been reclassified as goblet cell adenocarcinoma in further studies. 8 Although amphicrine carcinomas are rare tumors, they are most frequently reported in the gastric region, 6 and their occurrence in the ampullary region is uncommon. A previous report involved a male patient who underwent pancreaticoduodenectomy for an ampullary amphicrine carcinoma staged as pT3N1, with a Ki-67 proliferation index of 15% to 20%. This patient showed no signs of recurrence after 1 year of follow-up. 9 Another report of duodenal amphicrine carcinoma involved a female patient who continued treatment and follow-up with platinum chemotherapy after distal gastrectomy surgery. The tumor stage was pT3, and the Ki-67 proliferation index was 60%. 10 A third report describes a tumor located in the right colon of a female patient. The tumor stage was pT2N0, and the Ki-67 proliferation index was approximately 90%. Right hemicolectomy was performed, and notably, a mismatch repair defect was detected in this patient. 11 No lymph node metastases were observed in any of these patients. In our study, similar findings were observed for pathological tumor stages, and no recurrence was reported during the follow-up, as with the other patients. The patient was treated conservatively and did not receive adjuvant chemotherapy. To date, no recurrence or metastases have been reported.
The precise classification of amphicrine tumors within the spectrum of neuroendocrine differentiation neoplasms remains uncertain. In the early years, these tumors were thought to be formed by metaplasia or synchronous differentiation of tumor cells. 12 However, their pathogenesis has been better understood through molecular studies in recent years.
In our report, we identified mutations in the SOS1, KMT2C, and MED12 genes. Although the clinical or therapeutic significance of the detected mutations has not yet been determined, SOS1 is known to be part of the RAS pathway, activating RAS through the conversion of GDP to GTP. 13 This finding suggests that the tumor may be independent of KRAS activation. Given that SOS1 is linked to KRAS activation, ongoing research is exploring therapeutic options targeting the SOS1 pathway in KRAS-mutant carcinomas.13,14
KMT2C, an epigenetic regulator of the KMT2 gene family, is overexpressed in various cancers such as acute myeloid leukemia, glioma and thymoma. High expression correlates with poor overall survival in lung adenocarcinoma, whereas mutations are linked to epithelial–mesenchymal transition and worse prognosis in gastric cancer. 15 Regarding gastric NECs, 1 study identified several mutations specific to pure NECs, including KMT2C mutations. In the present study, KMT2C missense mutations were identified in pure NECs; however, in our report, a duplication mutation was observed. Although the mutation types differ, they may have similar functional consequences. 16
MED12 is a key component of the mediator complex involved in regulating RNA polymerase II-dependent transcription and plays critical roles in gene expression control, cell differentiation, and development. Mutations in MED12 have been linked to various diseases, including cancer and intellectual disabilities. 17 Several studies have investigated the role of MED12 in different cancer types. For example, MED12 maintains basal cell identity by binding to the mediator complex for enhancer activation of ΔNp63 in pancreatic ductal adenocarcinoma. In prostate cancer, MED12 and CDK8/19 affect proliferation by regulating androgen receptor activity and enhancing the response to enzalutamide. Furthermore, in breast cancer, MED12 suppresses NEAT1 transcription through the control of histone acetylation in p53 wild-type cells; its loss leads to increased NEAT1 expression and chemotherapy resistance. These findings demonstrate the multifaceted role of MED12 in transcriptional regulation and resistance to cancer treatment.18–20
All 3 mutated genes identified in our study have been implicated, either directly or indirectly, in the progression and treatment of various cancer types. However, this is the first report of their detection in amphicrine carcinoma. Although the clinical significance and impact on this tumor subtype remain unclear, we believe that these findings provide valuable insights into the existing literature.
In one study, a pan-cancer transcriptome analysis was performed on gastric and intestinal amphicrine carcinomas. Accordingly, it was determined that amphicrine tumors had more homogeneous mRNA expression with adenocarcinomas but were more heterogeneous with neuroendocrine tumors. However, definitive conclusions could not be drawn due to the small number of patients. 21 Sun et al similarly evaluated a small number of gastric amphicrine carcinomas and MINEN tumors based on their molecular features. Although similar features were observed between the 2 groups, the C5 complement copy number was significantly higher in amphicrine carcinomas. 6
In conclusion, considering the latest information, we believe that this type of tumor should be included as a separate histological type from adenocarcinomas, neuroendocrine carcinomas, and MiNEN tumors. This will also support a more standardized approach in terms of treatment and follow-up protocols for reports of this rare tumor group.
Footnotes
Consent to Participate
All authors have verbally consented to participate.
Consent for Publication
All authors have verbally consented to the publication of the article.
Ethics Approval
Ethical approval for this study was waived, as Eskişehir Osmangazi University Faculty of Medicine does not require IRB approval for retrospective studies using anonymized archival pathology material.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was not sought, as the requirement was waived for retrospective studies using de-identified samples.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All authors have approved the availability of the data and consented to its sharing as stated in this article.
Use of AI
Artificial intelligence assistance was employed exclusively for linguistic refinement and grammar correction.
