Abstract
Malignancies arising from adrenal rests are extremely rare. They may occur in locations such as the retroperitoneum, testes/scrotum, ovaries, kidneys, anterior abdominal wall, spinal cord, and liver, in association with embryological development. In this article, we present a patient with ectopic adrenocortical cancer (ACC) located in the pancreas, along with a review of the relevant literature. A 57-year-old female patient was diagnosed with mixed-type breast cancer (lobular and ductal carcinoma). During further diagnostic evaluations, in addition to a malignant-appearing mass in the left breast, imaging revealed a second tumor in the left paraaortic region, located in the pancreas. This mass, measuring 6 cm, was well-circumscribed, partially necrotic, heterogeneous in appearance, and displayed no significant invasion of surrounding tissues. The patient exhibited no signs of hormonal hyperfunction, such as hypertension, electrolyte imbalances, cushingoid appearance, or hyperandrogenism. Following lumpectomy, the mass adjacent to the pancreas was surgically excised. Pathological examination revealed an oncocytic type of ACC, characterized by nuclear abnormalities, a high mitotic index, and high nuclear grade, with positivity for steroidogenic factor 1 (SF-1) (NR5A1) in immunohistochemical staining. Mitotane and glucocorticoid replacement therapy were added to the patient's treatment plan for ACC. At present, there are no signs of recurrence or metastasis. To our knowledge, this is the first reported example of ectopic ACC arising in the pancreas, diagnosed in a 57-year-old patient. ACCs should be considered in the differential diagnosis of tumors arising in nonadrenal regions.
Introduction
Adrenocortical cancer (ACC) is an uncommon malignant tumor with a low incidence rate of 0.7 to 2.0 patients per million people per year. It is more frequently diagnosed in women, comprising about 55% to 60% of all patients. Although it typically peaks in the first decade of life and in individuals aged 40 to 50, ACC can be diagnosed at any age. 1
The adrenal medulla originates in neural crest tissue, while the adrenal cortex originates from the coelomic mesoderm of the urogenital process. 2 Ectopic adrenal rests can sometimes be found in various locations, including the celiac plexus, ovaries, broad ligament, spermatic cord, testicles, bilateral lung, liver, spleen, pancreas, duodenum, and colon, due to the adrenal cortex's embryological development.3–7 While malignancies arising from adrenal rests are rare, when they do occur, the gender distribution is equal. These tumors have been reported in various locations such as the retroperitoneum, testis/scrotum, ovary, kidney, anterior abdominal wall, spinal cord, and liver.8–10 This article presents a unique tumor of ectopic ACC located in the pancreas, which, to our knowledge, has not been previously reported. Additionally, this patient is distinguished by the simultaneous presence of breast cancer, making it an intriguing clinical finding.
Patient
A 57-year-old female patient presented to the general surgery department with swelling, redness, and stiffness in her left breast. She did not have any prior medical conditions but had a family history of thyroid and lung cancer. No significant findings were observed during the physical examination except for the mass in her left breast. Mammography revealed multifocal masses in the lower half of the left breast, with a BI-RADS 6. The true-cut biopsy from the breast was compatible with a mixed type of infiltrative carcinoma comprising ductal and lobular components.
The thoracoabdominal computed tomography (CT) and FDG PET CT scans revealed a malignant mass in the left breast, as well as a second tumor in the left paraaortic area adjacent to the pancreas. The latter was 5.5 × 6 cm, well-circumscribed, locally necrotic, and had a heterogeneous appearance (Figure 1), with an SUVmax of 37.7 on the FDG PET CT scan. The adrenal glands appeared normal, except for a 1.5 to 2 cm nodule on the left side. A subsequent abdominal magnetic resonance imaging (MRI) revealed a mass with cystic and solid components originating from the pancreatic body and tail junction and growing toward the posterior and inferior regions. Notably, the patient did not exhibit any signs of hormonal hyperfunction, such as hypertension, electrolyte disturbance, cushingoid appearance, or hyperandrogenism.
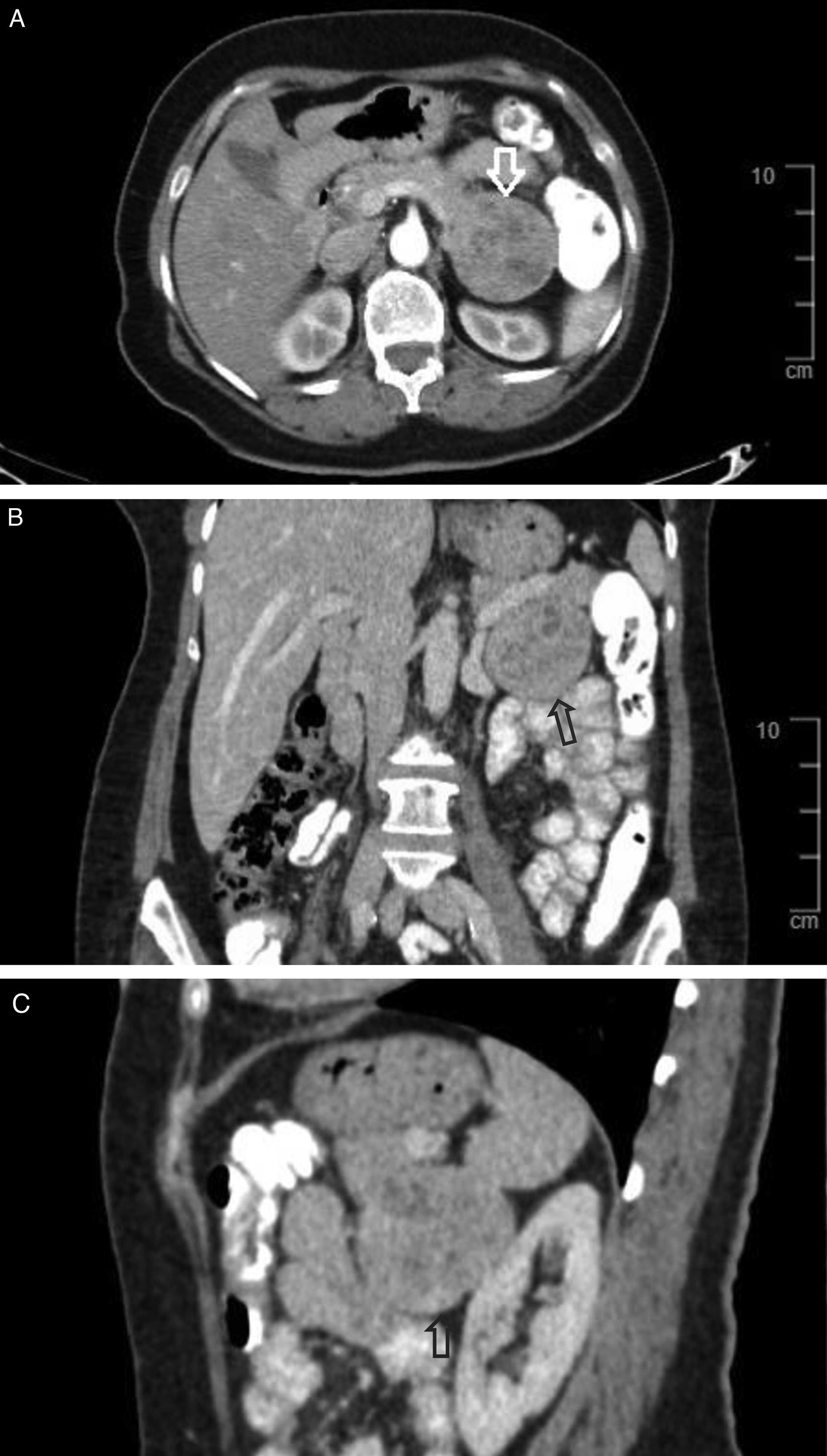
(A) Transverse computed tomography (CT) image demonstrating a well-circumscribed, heterogeneous, and partially necrotic mass located in the left paraaortic region adjacent to the pancreatic body-tail junction. (B) Coronal CT image of the left paraaortic mass. (C) Sagittal CT image showing the paraaortic mass, demonstrating that the lesion is independent of and not contiguous with the ipsilateral adrenal gland.
As an initial intervention, the patient underwent a lumpectomy and sentinel lymph node biopsy for a breast tumor. The final diagnosis revealed that the patient had a grade 2, ER-positive, PR- and HER2-negative mixed (ductal + lobular) infiltrative carcinoma with a tumor stage of T2N0M0. Subsequently, the patient underwent surgery to remove the mass adjacent to the pancreas and the left adrenal gland. A marginal excision of the mass was performed adjacent to the distal pancreas, yet the excision did not include pancreatic tissue. Radiologically and intraoperatively tumor had no macroscopic relation to the left adrenal gland. Ectopic adrenal tissue was incidentally found in the pathological examination and diagnosed through a clinical, radiological, and a pathological correlation. A histopathological examination of the mass revealed a cellular oncocytic neoplasm growing in diffuse sheets and a well-demarcated, expansile manner adjacent to a rim of adrenal tissue (Figure 2A). The tumor cells were large polygonal cells with significant nuclear atypia and lacked cytoplasmic inclusions (Figure 2B and C). Immunohistochemical analysis showed that the neoplasm was positive for melan-A (MLANA) (Figure 2D), SF-1 (NR5A1) (Figure 2E), calretinin (CALB2) (Figure 2F), and vimentin (VIM), which indicated an adrenocortical origin. However, it was negative for inhibin-α (INHA), synaptophysin (SYP), keratin AE1/AE3, EMA, chromogranin A (CHGA), PAX8, S100, HMB45, desmin (DES), Hep Par-1 (CPS1), and GATA3. The mitotic index was 6/50 in high-power fields (HPFs) (Figure 2C), while the Ki-67 proliferation index was 5%. No additional worrisome features were observed in the patient, such as venous, capsular, sinusoidal invasion, necrosis, or a tumor diameter >10 cm or weight >200 g. The surgical margins were found to be intact. A concomitant nodular neoplasm was also detected in the patient's left adrenal gland, but it exhibited benign features consistent with an adrenocortical adenoma. We employed the Lin-Weiss-Bisceglia (LWB) system, as recommended by the current World Health Organization (WHO) classification, for the evaluation of oncocytic ACCs. According to this system, the tumor was classified as malignant based on the presence of one major criterion: high mitotic activity (6 mitoses per 10 mm² or 50 HPFs at 0.2 mm² field size). Morphologically, the tumor exhibited features consistent with an oncocytic phenotype, including a diffuse growth pattern, nuclear atypia up to grade 3, and abundant eosinophilic granular cytoplasm in nearly all tumor cells. The Weiss score for the tumor was 4. The diagnosis of ectopic adrenocortical carcinoma was established through the correlation of radiological evidence showing no anatomical connection to the adrenal glands, intraoperative findings demonstrating no macroscopic relation to the left adrenal gland—indicating that the tumor was neither an extension nor a metastasis of an adrenal primary—and pathological confirmation of ectopic adrenal tissue displaying an adrenocortical immunophenotype.
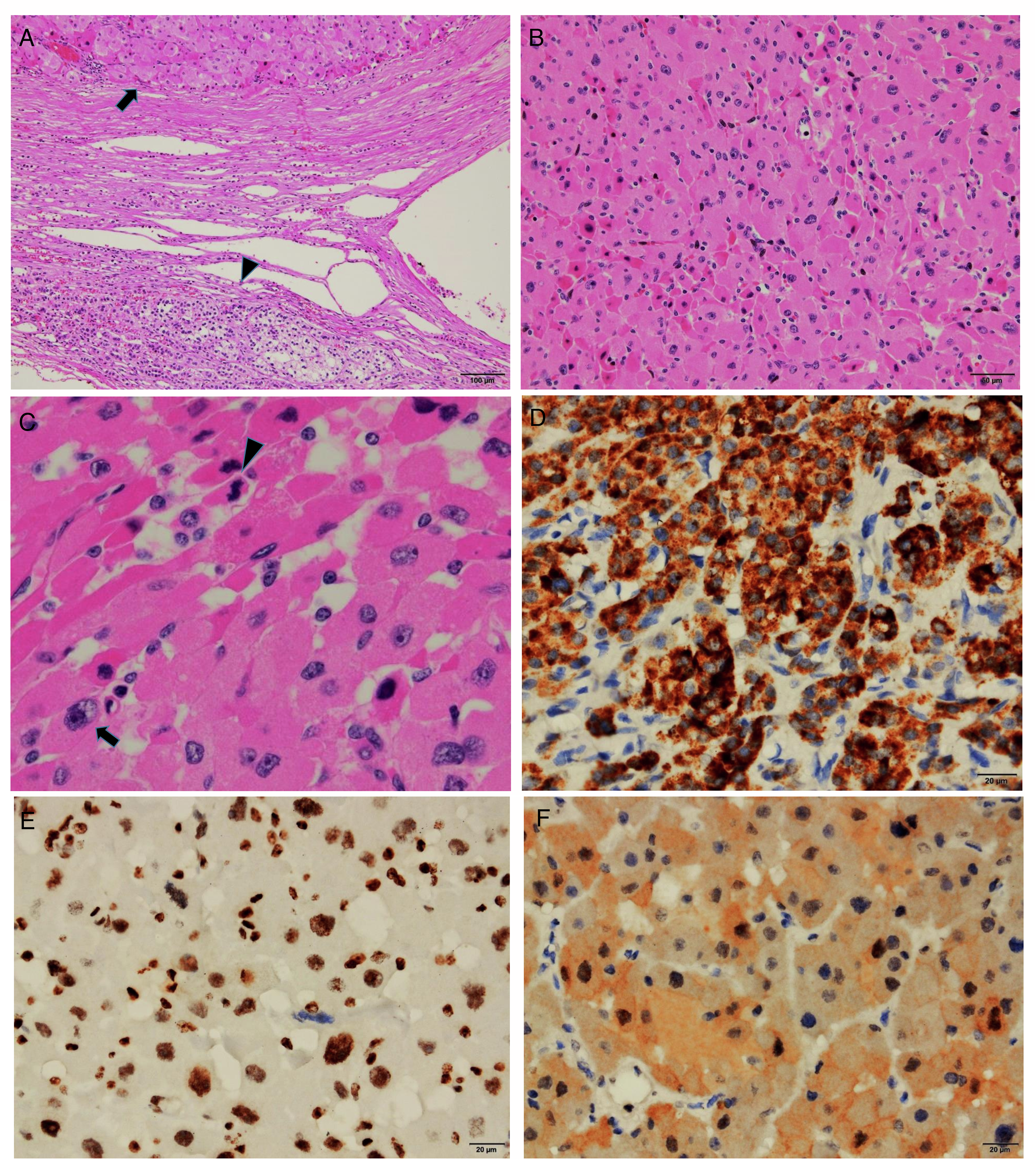
Pathological images of the removed mass. (A) Cellular neoplasm (arrow), well-demarcated from nonneoplastic adrenal tissue (arrowhead), Is seen (H&E, 100 ×). (B) Neoplasm consists of diffuse sheets of oncocytic cells with abundant granular eosinophilic cytoplasm (H&E, 200 ×). (C) Mitotically active malignant neoplasm (arrowhead) along with significant nuclear atypia (arrow) (H&E, 400 ×). (D) Immunohistochemically, neoplastic cells were strongly positive with melan-A (MLANA) (400 ×). (E) Immunohistochemically, neoplastic cells were strongly positive with SF-1 (NR5A1) (400 ×). (F) Calretinin (CALB2) (400 ×) was also focally positive in neoplastic cells.
The patient underwent radiotherapy as a treatment for breast cancer following 4 adjuvant cycles of docetaxel-cyclophosphamide. After that, she was treated with letrozole treatment. Mitotane and glucocorticoid replacement were included in her follow-up therapy for adrenocortical cancer. The subsequent follow-ups did not show any signs of recurrence or metastasis.
Discussion
This is the first case report of adrenocortical cancer localized in the pancreas, diagnosed at age 57. Another remarkable feature of the patient is the presence of concurrent breast cancer. Tumors of adrenal rest are typically not cancerous and are nonfunctional. They are often identified accidentally or after undergoing inguinal hernia surgery. They generally are detected incidentally or after inguinal hernia surgery.11,12 Occasionally, these tumors can be functional, producing glucocorticoids, mineralocorticoids, or androgens.13,14 It is important to note that a negative or low tumor marker result does not necessarily rule out malignancy, especially in cortisol-secreting tumors. This is because excess glucocorticoids can inhibit tumor markers. 15
ACCs that are associated with high levels of steroid hormones tend to grow at a faster rate and are more extensive. Typically, these tumors are larger, ranging from 10 to 13 cm. 16 Conversely, nonfunctional tumors are less common and typically smaller in size. 17 In our patient, faint clinical signs, a tumor diameter of 5.5 × 6 cm, and a Ki-67 proliferation index of 5% suggest a relatively less aggressive cancer.
Imaging, specifically MRI or CT, is necessary to evaluate ACCs. These tumors are generally heterogeneous with irregular borders and can contain areas of hemorrhage or necrosis. 18 Pathological diagnosis of our patient was ultimately challenging due to the tumor's unusual clinical presentation, variant histology, and subtle atypical features. Ectopic ACCs have been previously reported in the retroperitoneum, but a central relation to the pancreas is highly unexpected. Due to this abnormal localization, the oncocytic morphology, history of additional malignancy, and limited specificity of ancillary markers, an extensive workup was necessary to rule out other potential diagnoses, such as tumors of gonadal stromal lineage or nonsteroidogenic oncocytic malignancies. However, an accompanying adrenal rest was an important clue that aided in the diagnosis.
Determining the biological potential of oncocytic ACCs based on morphological criteria has proven challenging. However, the major criteria established by LWB indicate that a high mitotic index supports a carcinoma diagnosis. 19 In our patient, although only one major LWB criterion was met, the Weiss score for the tumor with its features listed above was 4, a threshold at which ectopic ACC has previously been reported. Additionally, the Helsinki score, proposed for oncocytic ACCs, was calculated as 8 (based on mitotic count: 3 and Ki-67 index: 5%). This value falls below the established malignancy threshold of 8.5. However, the applicability of this score in our patient is limited, as the Ki-67 index was not assessed using the recommended computer-assisted image analysis, which may affect reproducibility. 20 The sampling protocol followed for this specimen involved the standard approach of one tissue block per centimeter of tumor. Given that this diagnosis predated the widespread adoption of the Helsinki system, more extensive capsular sampling—as is sometimes employed in other endocrine tumors—might have provided additional diagnostic clarity, particularly in resolving the apparent discordance between the LWB scoring and Helsinki system. Reticulin staining, while considered by some as a supportive diagnostic tool, was not performed due to limited interobserver reliability in our experience and the absence of published data on its utility in ectopic ACCs. It is important to highlight that no standardized diagnostic criteria currently exist for ectopic ACCs. Several patients in the literature have also been diagnosed retrospectively, based on atypical clinical progression. Despite their location, ectopic ACCs have conventionally been assessed using the Weiss criteria, even when demonstrating oncocytic features. In some instances, infiltrative growth across natural anatomical boundaries has also been emphasized as an indicator of malignancy (see Table 1). Collectively, these observations point to the need for pathological standardization in the assessment of these rare presentations, encompassing aspects ranging from macroscopic sampling protocols to ancillary diagnostic techniques.
Summary of Published Case Reports of Ectopic Adrenal Cancer and Their Characteristics.
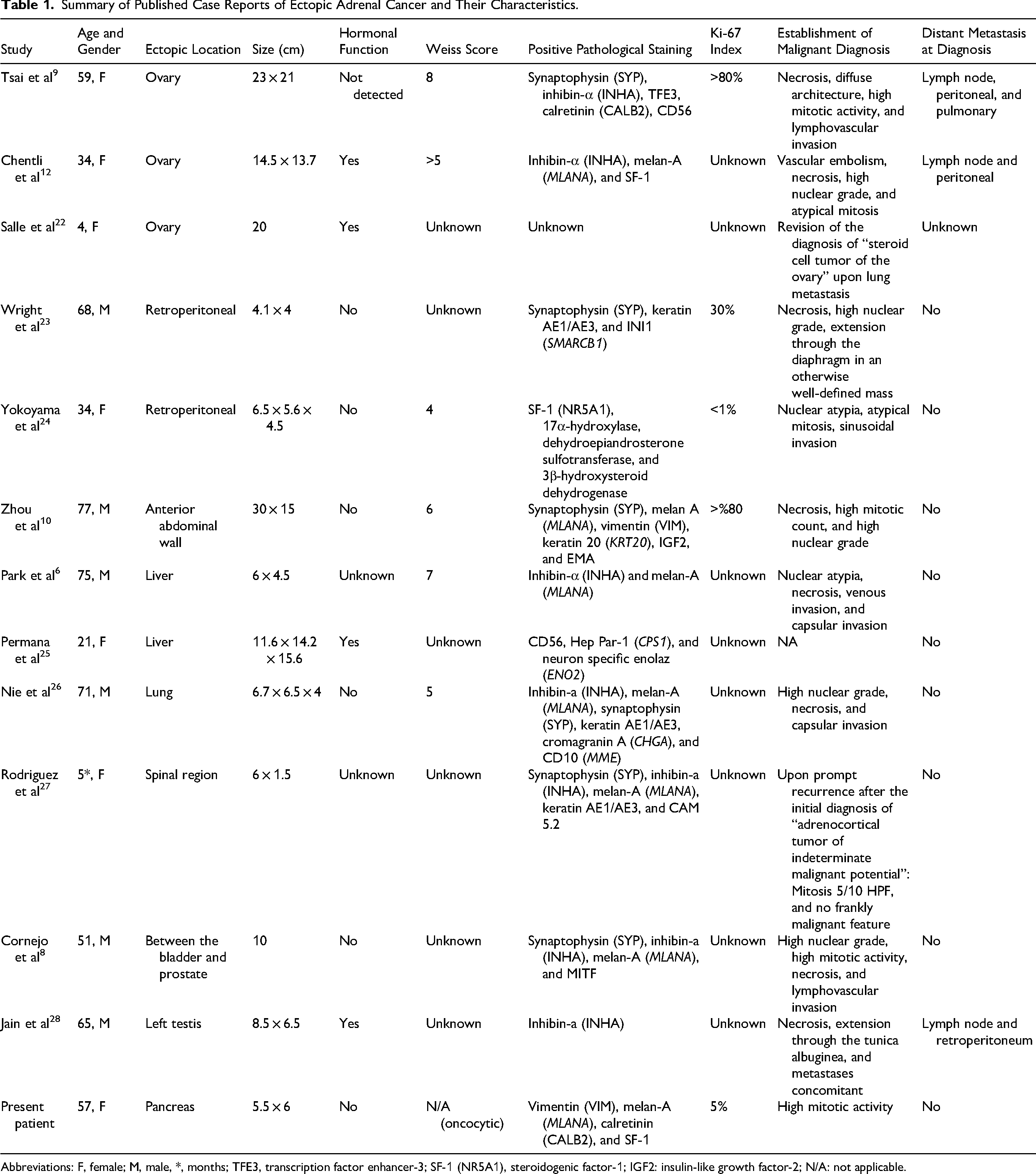
Abbreviations: F, female; M, male, *, months; TFE3, transcription factor enhancer-3; SF-1 (NR5A1), steroidogenic factor-1; IGF2: insulin-like growth factor-2; N/A: not applicable.
Recent updates to adrenocortical tumor classifications, published after the patient's diagnosis, have shown that ACCs with a lower Ki-67 index ≤20%, as observed in our patient with a Ki-67 index of 5%, are considered low-grade malignancies with a better prognosis. 21 Unfortunately, due to the rarity of these occurrences, there is limited data and experience with unusual presentations. Table 1 provides a review of reported patients of adrenocortical carcinoma arising from ectopic adrenal remnants, based on the published literature.
The primary treatment of adrenocortical cancers is surgery, 29 while mitotane can be used as medical therapy alone or in combination with chemotherapeutic drugs. 30 Although our patient involved a nonmetastatic, hormonally inactive, histologically low-grade ACC, adjuvant mitotane therapy was administered following multidisciplinary evaluation. This decision was made considering both the rarity of ectopic ACC and the limitations of existing prognostic tools.
Conclusion
This report describes an ectopic presentation of adrenocortical cancer in a 57-year-old female patient. The cancer was localized in the pancreas and was treated with surgical resection. Adrenocortical cancers should also be considered in nonadrenal tumors.
Footnotes
Ethical Approval
Hacettepe University does not require IRB approval for a case report.
Author Contributions
AOİ, AK, and UÜ: study design, data collection, drafting, and critical review of the manuscript. NA and BF: data collection and critical review of the manuscript. All authors approved the final version of the paper.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset regarding the patient's evaluation may be obtained from the corresponding author upon reasonable request.
