Abstract
Mucinous cystadenocarcinoma of the breast is an exceedingly rare entity with less than 40 tumors reported to date. It is classified as a special subtype of breast carcinoma which exhibits low-grade indolent behavior despite having a triple-negative phenotype. Awareness of this rare tumor is essential to avoid misinterpreting it as a routine mucinous (colloid) carcinoma of the breast or metastasis from a nonmammary mucinous carcinoma. Though the clinical, histological, and immunohistochemical features of breast mucinous cystadenocarcinoma have been described thoroughly, data on molecular features remains scarce. Herein, we present 3 patients with mucinous cystadenocarcinoma, highlighting the molecular features in 2 patients and discussing the literature in this regard. Additionally, we report that these tumors have the potential for short-term recurrence despite complete surgical excision, and they therefore should probably be treated as any other triple-negative breast cancer (TNBC) with careful consideration of other factors such as patient's age, tumor size, nodal stage, and tumor grade. Review of the literature on this rare tumor together with findings from the current report suggests that MCAs at the molecular level are similar to TNBC, with frequent recurring variants in PI3K pathway genes, including PIK3CA, PTEN, and AKT1 genes. We report here for the first time the presence of GNAS variants in mucinous cystadenocarcinoma. This information could be useful to offer targeted treatment against PIK3CA and AKT1 in patients with this rare tumor.
Introduction
Mucinous cystadenocarcinoma of the breast is an exceedingly rare malignant breast neoplasm recognized only recently as a distinct entity in the WHO classification (fifth ed.) of breast tumors. 1 It was first described by Koenig and Tavassoli in 1998 as a primary breast carcinoma with histologic features similar to the mucinous carcinomas of the ovary and pancreas, 2 and since then less than 40 tumors have been described in the literature.3–15 Though many of these reports have thoroughly described the clinical, histological, and immunohistochemical features of mucinous cystadenocarcinoma, the data on molecular features remains sparse.6,9,11,12 Herein, we present 3 patients with mucinous cystadenocarcinoma of the breast, including a new patient (patient 1) with mucinous cystadenocarcinoma of the breast, another patient (patient 2) whose tumor was previously reported (patient 2 of Nayak et al, IJSP, 2018) and which recurred after 7 months of initial lumpectomy, and a third new patient (patient 3) with axillary lymph node metastases. 4 We present the mutational profile of 2 of these tumors, including the recurrence of 1, and discuss the literature on the mutational profile of this rare tumor. Lastly, we report the short-term recurrence of the tumor occurring 7 months after complete surgical excision in 1 patient (Patient #2), a deviation from the indolent behavior suggested in the literature.
Patient 1
The patient was a 50-year-old woman with no past medical history who identified a movable, nontender mass in the left breast. The patient's family history was significant for breast cancer in her mother and half-sister. She presented to an outside hospital where a diagnostic mammogram confirmed a 2.6 × 2.5 cm lobulated mass in the posterior inner left breast (Figure 1A). Subsequent ultrasound demonstrated that the mass was solid and heterogeneous with internal blood flow (Figure 1B). Core biopsy performed at the outside hospital was initially read as an invasive moderately differentiated mucinous carcinoma with negative staining for estrogen receptor (ER) protein, progesterone receptor (PR) protein, and HER2 oncoprotein expression. Further MRI imaging characterized the lesion as 4.1 × 3.3 × 2.1 cm in size, abutting the chest wall (Figure 1C). In addition, the MRI revealed left axillary adenopathy concerning metastatic disease; however, a core biopsy performed on the lymph node was negative for carcinoma.
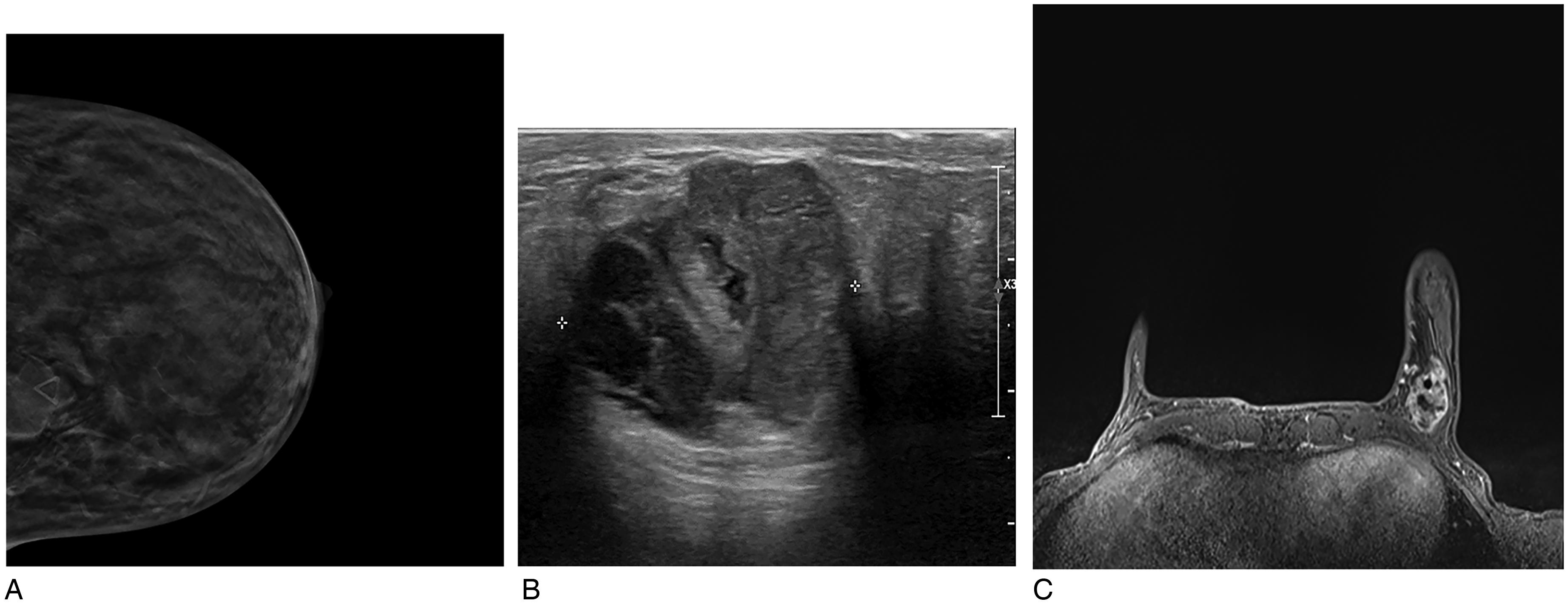
Imaging photographs from patient #1. (A) Original mammogram demonstrating lobular mass in left posterior inner breast; (B) Original ultrasound demonstrating heterogeneous, partially solid mass; (C) MRI imaging demonstrating heterogeneous lesion measuring 4.1 × 3.3 × 2.1 cm.
The patient transferred care to our hospital for treatment given the large tumor size and triple negative biomarker profile. At this time, the outside pathology slides were reviewed, and the diagnosis was modified to infiltrating poorly differentiated duct carcinoma with cystic and mucinous features, focal necrosis, and tumor giant cells (anaplastic features), suggestive of mucinous cystadenocarcinoma. Submitted immunohistochemical stains from the outside lab revealed that the tumor cells were positive for KRT7 and negative for KRT20, PAX8, CDX2, TTF1, NapsinA, GATA3, BRST2 (PIP), and mammaglobin. The outside block was retrieved and additional immunohistochemical stains demonstrated that the tumor cells were negative for WT1 and SOX10. Myoepithelial cell markers were negative around the cystic ducts supporting the invasive nature of the tumor. The previous lymph node biopsy's benign diagnosis was confirmed. Clinical and radiologic correlation was recommended to rule out a mucinous primary tumor in other visceral organs such as the pancreas, ovary, appendix, colon, or lung. Prior to determining a treatment plan, abdominal and chest CTs were performed, revealing an enlarged retroperitoneal lymph node measuring 4.4 × 2.7 cm. A biopsy of this lymph node yielded the diagnosis of a concurrent low-grade CD5-CD10- B-cell lymphoma. No other visceral organs showed imaging-detected abnormalities. The patient subsequently underwent a successful partial mastectomy of the left breast lesion with sentinel lymph node biopsies.
The partial mastectomy specimen revealed a white-tan, heterogeneous, partially encapsulated, and multi-lobulated mass with focal areas of hemorrhage, measuring 4.2 × 2.4 × 2.1 cm. Histologically, the tumor displayed cystic and papillary architecture with extracellular mucin, mimicking mucinous cystic neoplasms of the ovary and pancreas (Figure 2A). Tumor cells lining the cysts and papillae exhibited columnar features with abundant intracellular mucin, enlarged atypical grade 3 nuclei, and abundant mitotic activity (Figure 2B). Additionally, the invasive tumor was associated with foci of conventional ductal carcinoma in situ, high nuclear grade, and solid type. Surgical resection margins were negative for the tumor. Six lymph nodes submitted as sentinel nodes were all negative for tumor. Immunohistochemical stains were repeated on the partial mastectomy specimen, yielding similar results as on the biopsy (including negative staining for ER, PR, and HER2 expression). Given the clinical, histologic, and immunohistochemical findings, the diagnosis of mucinous cystadenocarcinoma of the breast was rendered. The patient was then treated with adjuvant chemotherapy with doxorubicin and cyclophosphamide followed by paclitaxel. The possibility of offering post-lumpectomy radiation was considered; however, the patient declined it and instead opted for bilateral mastectomy and breast reconstruction following the completion of chemotherapy. No residual tumor was identified in the mastectomy specimens. To date, the patient has been doing well (41 months follow-up) without any local or distant recurrence.
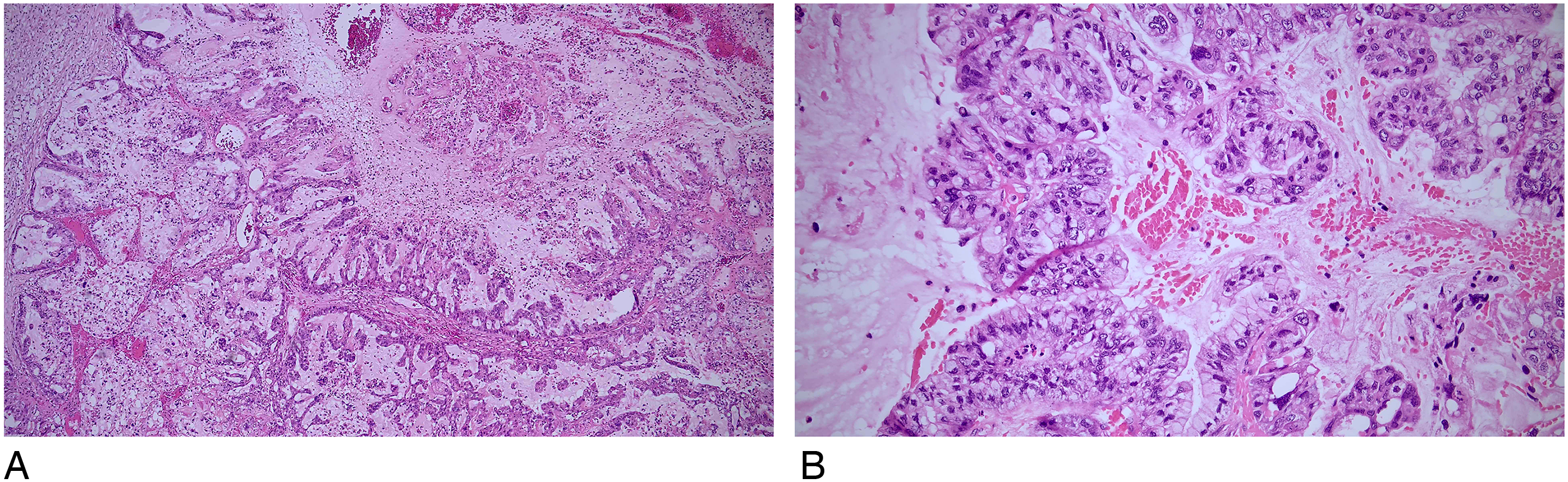
Pathology photomicrographs from patient #1. (A) Mastectomy specimen with mucinous and cystic tumor displaying complex papillary architecture and mimicking mucinous cystic neoplasms of ovary and pancreas (10x objective). (B) Tumor cells lining the cysts and papillae exhibited columnar stratified appearance, and contain abundant intracellular mucin, atypical grade 3 nuclei, and numerous mitotic figures (20x objective).
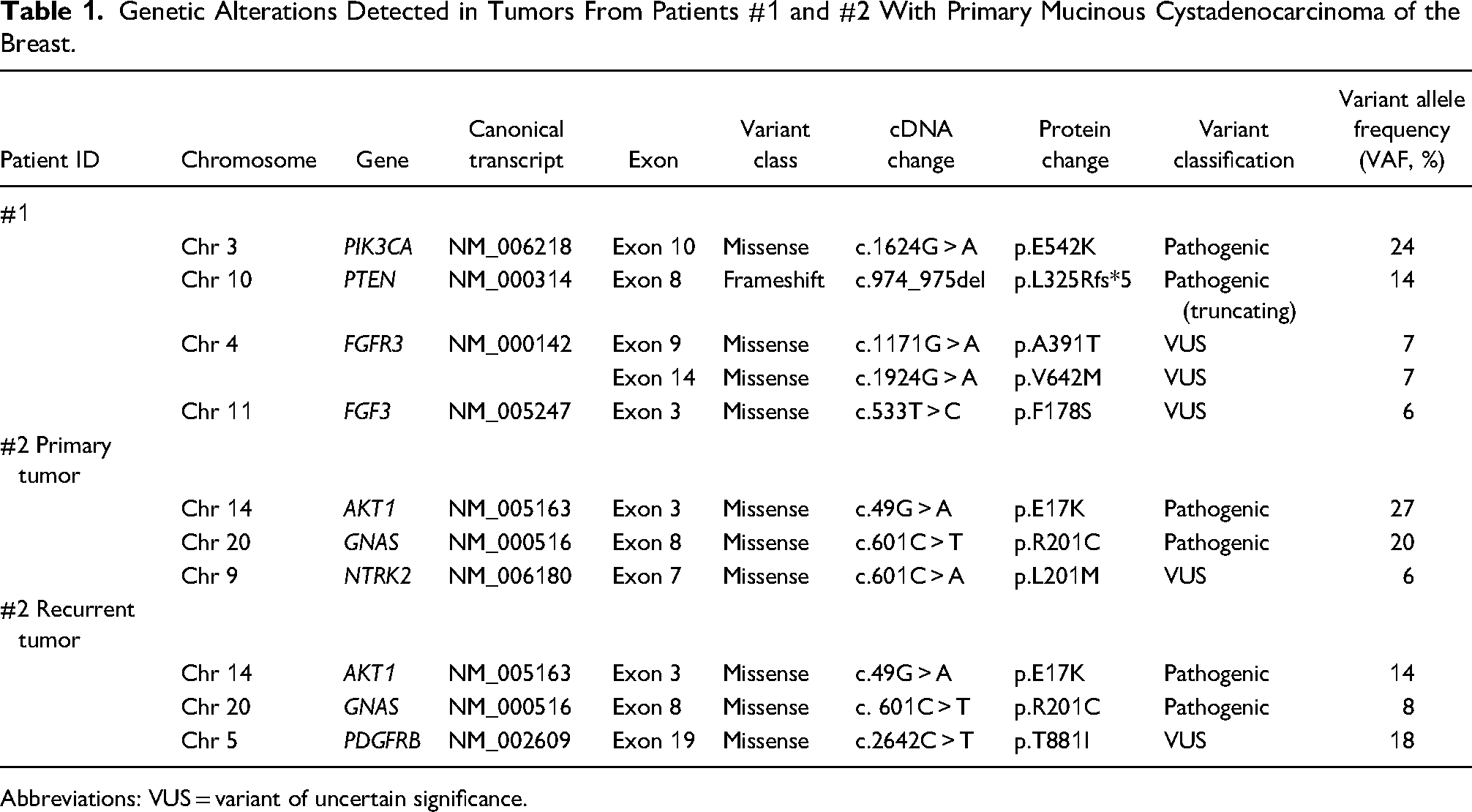
Gene sequencing studies were performed on the tumor for research purposes. DNA was isolated from formalin-fixed paraffin-embedded (FFPE) scrolls using the QIAGEN QIAmp DNA FFPE Advanced Kit. NGS libraries were prepared from 50 ng of tumor DNA using the DNA component of the AmpliSeq for Illumina Comprehensive Panel v3 following the protocol described in Document # 1000000039452 v06 (Illumina). This panel uses highly multiplex PCR to target 397 kb across 161 cancer-related genes. Libraries were sequenced on a NextSeq 2000 P1 flow cell using XLEAP chemistry and a loading concentration of 750pM. BCL to FASTQ conversion was performed in Illumina's cloud-based BaseSpace analysis platform using BCLConvert v2.5.0, followed by alignment and variant calling with the BaseSpace DRAGEN Amplicon App v4.3.6. Average depth of coverage ranged from 3600x to 6931x. Hard-filtered DRAGEN variant calls were further annotated with Annovar and filtered using BCFtools. In addition to the hard filter categories assigned to variants by DRAGEN, the minimum depth of coverage for a variant call was set at 100×, and the minimum allele frequency at 5. Variants falling outside of exons or splice sites, synonymous variants, and variants occurring in the Annovar gnomAD_genome_ All or gnomAD_exome_ All databases with a population frequency greater than 1% were also filtered. The results revealed pathogenic variants in PIK3CA and PTEN genes. Additionally, variants of uncertain significance (VUS) were noted in FGFR3 and FGF3 genes (see Table 1 for specific variants).
Genetic Alterations Detected in Tumors From Patients #1 and #2 With Primary Mucinous Cystadenocarcinoma of the Breast.
Abbreviations: VUS = variant of uncertain significance.
Patient 2: Previously Reported 4
The patient was a 68-year-old post-menopausal woman with no medical history, who palpated a tender breast mass in the left central breast. She presented to an outside hospital where mammography demonstrated a 4.5 cm, partially obscured mass with focal asymmetry. A subsequent ultrasound revealed an irregular 4.8 × 4.5 × 2.7 cm, heterogenous solid and cystic mass located in the 6:00 position at 6 cm from the nipple. The patient underwent a core biopsy at the outside institution which was diagnosed as well-differentiated mucinous carcinoma that was negative for ER, PR, and HER2 oncoprotein overexpression and HER2 gene amplification by FISH. Chest and abdominal CT scans were performed and revealed no other visceral tumors. The patient was referred to our institution for neoadjuvant treatment given the clinical T2 stage and triple-negative phenotype.
Similar to Patient 1, the core biopsy diagnosis was modified to poorly differentiated mucinous cystadenocarcinoma of the breast after a review of the outside biopsy slides and clinical correlation with body imaging. On histology, the core biopsy predominantly consisted of detached papillary fragments floating in extracellular mucin. The columnar tumor cells lining the papillae contained abundant intracytoplasmic mucin and highly pleomorphic nuclei with prominent nucleoli. Again, a small focus of conventional DCIS was noted at the edge of the core supporting the breast origin for this tumor. The morphology and immunophenotype (KRT7+, GATA3+, BRST2 (PIP)+, KRT20-, CDX2-, p63-, TTF1-, NapsinA-, PAX8-), together with triple-negative biomarker status, supported the diagnosis of mucinous cystadenocarcinoma of the breast. Given this new diagnosis of mucinous cystadenocarcinoma with presumably low-grade indolent behavior despite having a triple-negative biomarker phenotype, this patient underwent a partial mastectomy with sentinel node biopsy. The surgical specimen demonstrated a 6.2 × 4.4 × 4.0 cm cystic lesion with a lobulated, irregular border, and emulated the core biopsy histology except for showing focal areas of extravasated mucin with free-floating epithelial cell clusters as well. Two sentinel lymph nodes were negative for the tumor. The resection margins were negative for the tumor.
After surgical resection, the multidisciplinary tumor board determined that the patient did not require adjuvant chemotherapy or radiation therapy at this point given the rarity of the tumor and indolent clinical behavior reported in the literature. However, after 7 months of follow up the patient presented with ipsilateral local recurrence following which she underwent repeat partial mastectomy. The morphology of the 3.0 cm recurrent tumor was similar to the primary tumor with the exception of a large volume of tumor in the form of micropapillary clusters free floating in the extracellular mucin (Figure 3). Resection margins were free of tumor, and 2 additional sentinel lymph nodes were free from metastasis. The patient was offered chemotherapy which she declined; however, she did receive whole breast irradiation with a boost at the surgical bed. No further follow-up is available for this patient.
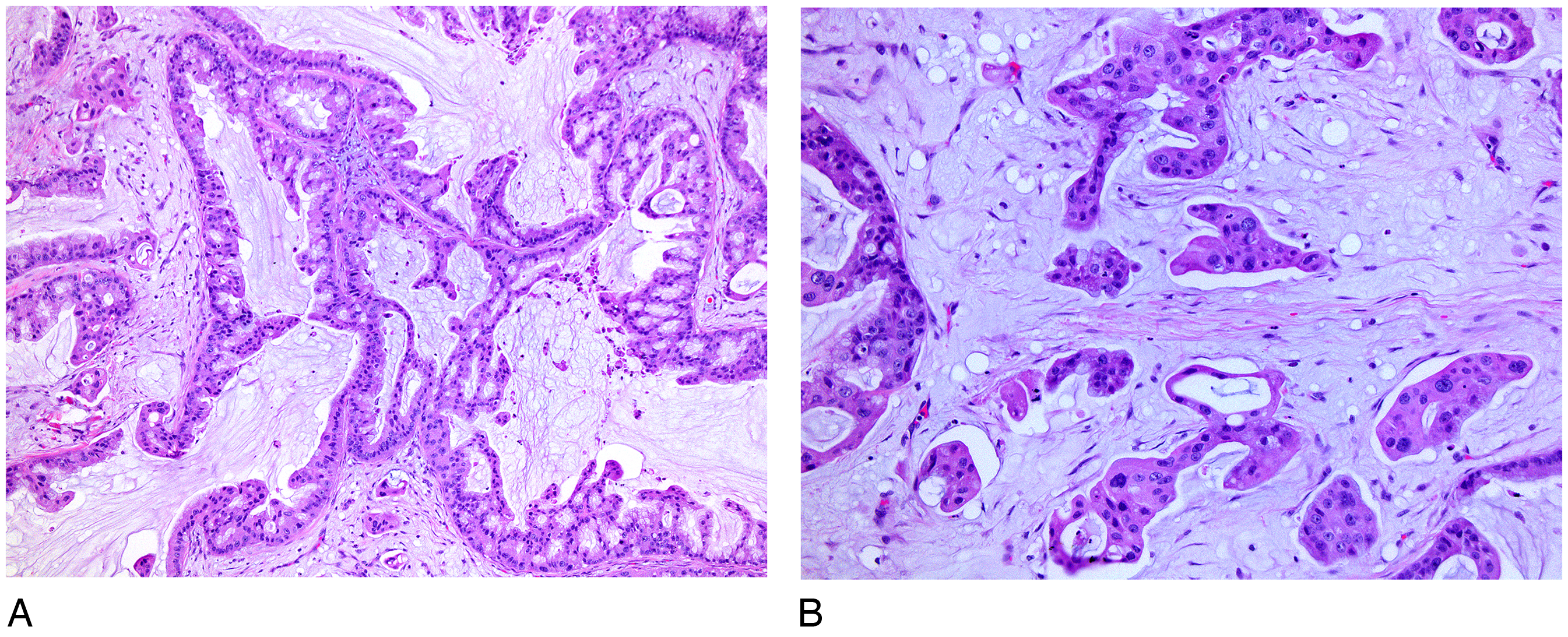
Pathology photomicrographs from patient #2 representing recurrent tumor after 7 months of surgical excision with clean margins. (A) Note the cystic, papillary, and mucinous architecture of the tumor with high-grade cytology. The recurrent tumor also contained clusters of tumor cells with micropapillary architecture floating within the extracellular mucin (6:00 location in the photomicrograph; 20x objective) (B) another field highlighting the tumor cells with micropapillary architecture floating within the extracellular mucin (40x objective).
The gene sequencing studies were performed later after we published the case report in 2018 4 as part of a research project. NGS testing was done on both the primary and the recurrent tumor using the same methodology and NGS platform as described above for patient #1. The results demonstrated pathogenic variants in AKT1 and GNAS genes in both primary and recurrent tumors supporting the oncogenic driver role of these variants. In addition, VUS in the NTRK2 gene and PDGFRB were noted in the primary and the recurrent tumor, respectively (see Table 1 for specific variants).
Patient 3
The patient was a 66-year-old post-menopausal woman with a medical history of left breast hormone receptor-positive invasive ducal carcinoma treated with breast conservative surgery and brachytherapy in 2012. The patient declined adjuvant endocrine therapy at the time. Eleven years later (2023), the patient presented to an outside hospital for a screening mammogram which demonstrated a new focal asymmetry corresponding to 2.3 × 1.8 × 2.9 cm irregular mass lesion on ultrasound at the prior lumpectomy site, along with a new prominent lymph node in the left axilla. Biopsy of the breast mass was reported as an invasive moderately differentiated mammary carcinoma with ductal and lobular features [ER positive (80%), PR positive (50%), and HER2 negative (score 1+)]. The concurrent biopsy of the axilla lymph node was however reported as an invasive mammary carcinoma with partial mucinous features [ER positive (50%); PR low positive (<5%); and HER2 negative (score 1+)]. Bilateral breast MRI performed prior to surgery identified another distinct 7 mm retroareolar mass, along with additional level 2 abnormal lymph nodes. Core biopsy of the retroareolar mass performed at the outside hospital was reported as well-differentiated invasive breast carcinoma with mucinous features, with features similar to that noted in the previously biopsied left axillary lymph node. Interestingly, ER, PR, and HER2 were negative in this sample [HER2 2+ by IHC and FISH negative (average HER2 copy number signals 2.5/nucleus, and HER2/CEP17 ratio of 1.5)]. Ki-67 was reported as 10%–20%. PET-CT of the whole body confirmed the disease in the left breast and axilla. There were no additional sites of suspicious FDG avid distant metastases. Following this, the patient underwent a total mastectomy with sentinel lymph nodes biopsy at the outside institution. The pathology report from the outside institution identified the index mass at the lumpectomy site as recurrent invasive breast carcinoma with ductal, lobular, and mucinous features. The tumor was graded as Nottingham histologic grade 2 of 3, measuring 30 mm grossly and associated with the prior lumpectomy scar. Additionally, a satellite focus of invasive breast carcinoma with entirely mucinous features, measuring 8 mm microscopically, was identified involving the subareolar tissue. This focus was morphologically identical to the mucinous component of the main tumor. Repeat immunohistochemical stains on the satellite retroareolar mass confirmed a triple-negative phenotype (negative for ER, PR, and HER2). Metastases were observed in 4 of 5 sentinel lymph nodes, which exhibited distinct areas of metastatic carcinoma with ductal and lobular features as well as regions of pure mucinous morphology. The largest involved node measured 75 mm and displayed multifocal extranodal extension. Repeat immunostaining for biomarkers on the metastatic carcinoma within the lymph nodes showed differing phenotypes between components. The metastatic carcinoma component with ductal and lobular features was positive for ER (90%–100%, moderate to strong) and PR (60%–70%, variably weak to strong) and negative for HER2 (score 0). In contrast, the metastatic carcinoma component with mucinous morphology was negative for ER (0%), PR (0%), and HER2 (score 0). Surgical margins of mastectomy were negative for tumor. The patient was placed on the KEYNOTE-522 regimen (chemotherapy plus pembrolizumab) and subsequently underwent left axillary lymph node dissection 7 months later to further determine the adjuvant treatment course. A residual focus of metastatic carcinoma in the axillary soft tissue as well as metastases in 3 new lymph nodes was noted.
At that time, the patient was referred to our institution for proton therapy and the slides were submitted for pathology review. Submitted slides from the original mastectomy specimen, the sentinel lymph nodes, and the subsequent axillary dissection demonstrated an invasive moderately differentiated mammary carcinoma with mixed ductal, lobular, and mucinous components, with focal areas of squamous metaplasia. The mucinous component of the tumor, both within the primary tumor, the satellite retroareolar tumor, and the lymph node metastases, distinctly demonstrated glands and cysts lined by cuboidal to polygonal tumor cells with abundant intracellular and extracellular mucin (Figure 4). A few cystically dilated ducts demonstrated distinct arborizing papillary projections (Figure 4D & 4E). In our opinion, this distinct morphology of the mucinous component, coupled with the squamous metaplasia and the triple negative immunophenotype, was akin to a mucinous cystadenocarcinoma of the breast than a conventional mucinous carcinoma. Therefore, we believe, this is a rare patient of invasive mammary carcinoma exhibiting unique morphology with a composite of ductal, lobular, and distinct mucinous cystadenocarcinoma components.
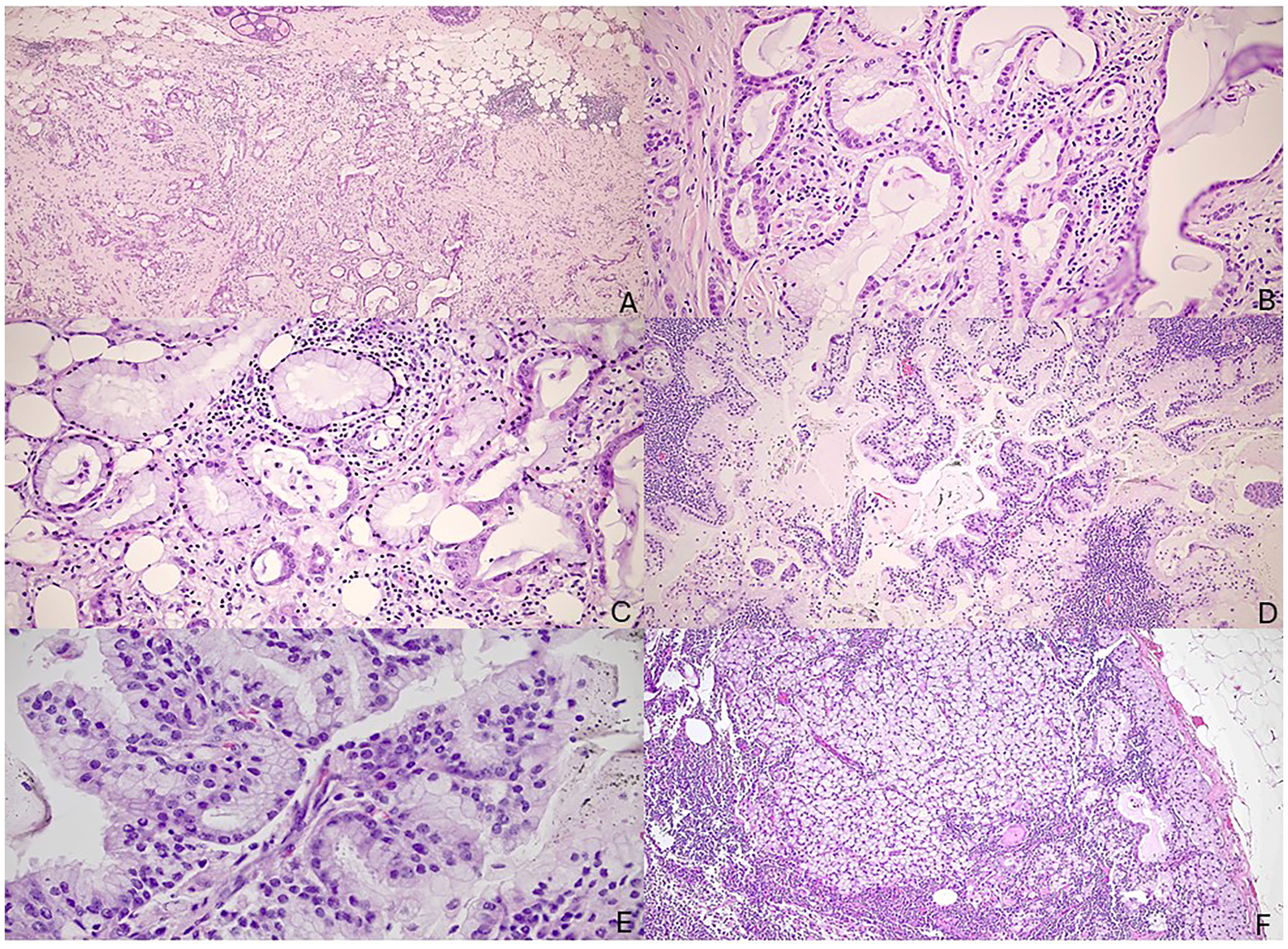
Pathology photomicrographs from patient #3. (A) Mastectomy specimen showing invasive mammary carcinoma with ductal, lobular and mucinous cystadenocarcinoma components with associated DCIS (5x objective); (B) Retroareolar satellite nodule demonstrating predominant mucinous cystadenocarcinoma component, negative for estrogen receptor, progesterone receptor, and HER2 protein (10x objective); (C) Higher magnification of the same (20x objective); (D) Sentinel lymph node demonstrating mucinous cystadenocarcinoma component with cystic and papillary architecture (5x objective); (E) Higher magnification view of the same highlighting moderate atypia (10x objective); (F) Residual mucinous cystadenocarcinoma component in the ipsilateral axillary node dissection 7 months after completion of adjuvant KEYNOTE chemotherapy (10x objective).
Gene sequencing studies were not performed for this patient's tumor, as the material was not available for additional testing. Since the initiation of proton therapy, the patient has undergone 3 months of treatment and continues to be followed with no evidence of recurrence.
Discussion
As alluded to earlier, the diagnosis of mucinous cystadenocarcinoma of the breast is exceedingly rare, with fewer than forty patients reported in the literature. 7 In our experience, this tumor is often erroneously diagnosed as a mucinous (colloid) carcinoma of the breast in general pathology settings because of the rarity of the tumor and the pathologist's unfamiliarity with the morphology. An accurate diagnosis is often made on a second opinion when the patient is referred to tertiary centers for consideration of neoadjuvant therapy given the triple-negative phenotype of the tumor. The 2 patients described here highlight that triple negative biomarker status in a tumor with distinctive mucinous, cystic, and papillary architecture serves as a useful diagnostic clue to the mucinous cystadenocarcinoma diagnosis and should prompt additional imaging workup to rule out primaries in other organ systems.
To date, only 4 studies have described the molecular findings of this rare tumor.6,9,11,12 Jain et al performed real-time polymerase chain reaction on their tumors and identified wild-type KRAS, NRAS, and BRAF genes. This contrasts with pancreatic, appendiceal, and colonic mucinous cystadenocarcinoma which often exhibit mutations in the above-mentioned genes, are morphologically similar on microscopy, and must be ruled out by clinical and radiologic investigations before the diagnosis of primary mucinous cystadenocarcinoma of the breast. 6 Lin et al performed a gene sequencing panel and identified variants in TP53, RB1, and BAP1. 11 Lei et al reported recurrent variants in PIK3CA, KRAS, MAP2K4, RB1, KDR, PKHD1, TERT, and TP53; with high tumor mutation burden of 9.27, and low microsatellite instability status in 1 patient of mucinous cystadenocarcinoma of the breast. 12 Further, immunohistochemical staining for P53 and RB1 proteins correlated well with the sequencing results, with overexpression of p53 protein and loss of RB1 protein expression in tumor cells. 12 Chen et al also documented a PIK3CA hot-spot variant in 1 of the 3 mucinous cystadenocarcinomas tested by whole genome sequencing, and akin to the study by Jain et al, reported no KRAS, NRAS, BRAF, or AKT variants in any of the 3 tumors. 9
Gene sequencing of the tumor in our patient (Patient 1) identified a pathogenic hot spot variant (NM_006218:c.1624G > A, p.E542K) in PIK3CA and a frameshift mutation in exon 9 of PTEN, whereas in the second patient (Patient 2) we identified pathogenic hot spot variants in AKT1 (NM_005163: c.49G > A, p.E17K) and GNAS (NM_000516: c.601C > T, p.R201C) in both the primary and recurrent tumors. Together with previous studies, our findings suggest that the variant profile of mucinous cystadenocarcinoma is closer to that of TNBC and is equally heterogeneous, with frequent PIK3CA, PTEN, and AKT1 variants. 1
Somatic activating variants of the PIK3CA gene are the common driver variants in breast cancer with a prevalence of up to 40% in primary breast cancer promoting uncontrolled cellular and tumor growth through dysregulation of PI3K/Akt/mTOR downstream signaling pathway. 16 The E542K gain-of-function mutation identified in the tumor from our Patient 1 is 1 of the 5 common hot spot variants identified in PIK3CA mutated breast cancers, with a frequency of 11% in all breast cancer subtypes, and 6% in TNBCs including those with indolent behavior, for example, low-grade acinic cell carcinomas, fibromatosis-like metaplastic carcinoma and low-grade adenosquamous carcinomas.17,18 This is particularly significant as this mutation is one of the few genomic aberrations predictive for drug response to PI3K-targeted therapy in advanced hormone receptor-positive HER2 negative breast cancers, 16 and the role of PIK3CA variants as predictive markers is currently under investigation in clinical trials focused on other breast cancer subtypes, including triple-negative breast cancers. 19 We therefore believe reporting the recurrent identification of PIK3CA hot spot variants in mucinous cystadenocarcinoma of the breast, as illustrated in both current and previous reports, is crucial and noteworthy. PIK3CA variants in breast cancer have been linked to prognosis as well, however, the results are conflicting. 16 Reinhardt K et al recently reported that the presence of 3 hot spot variants (E545 K, E542K, and H1047K) in PIK3CA did not have a significant association with recurrence-free interval in the overall cohort of 1123 tumors. Nonetheless, these variants had a favorable impact in early TNBC, indicated by a 2.3-times improved recurrence-free interval and a 3-times improved overall survival. 16 Given this data, it would be valuable to gather molecular information specifically related to PIK3CA variants in distinct subtypes of TNBCs exhibiting so-called indolent behavior, such as mucinous cystadenocarcinomas. This would allow for a thorough assessment of the prognostic implications.
The tumor from patient #1 in this report additionally exhibited a co-existent disease-associated loss of function variant in PTEN. The PTEN gene is a negative regulator in the PI3K/Akt/mTOR pathway and functions as a tumor suppressor in normal cells. Inactivating/loss of function variants in the PTEN gene in tumor cells results in inactivated PTEN protein leading to subsequent activation of downstream AKT pathway, promoting further cancer cell growth and survival. Somatic or germline variants in the PTEN gene have been documented in numerous breast cancers, with PTEN loss of function variants detected in 5%–10% of breast cancers, with frameshift representing the most frequent mechanism (as seen in our tumor). 20 The Cancer Genome Atlas (TCGA) study identified PTEN mutation/ loss of function across all subtypes of breast cancers though it was most common in Basal-like TNBCs. 21
Activating E17K variant is the most common somatic hot spot variant in AKT1 accounting for 1%–8% of breast cancers, particularly luminal type, and is frequently mutually exclusive with PIK3CA variants.21,22 Tumor from Patient #2 in our report exhibited the AKT1 variant without the PIK3CA variant in alignment with previous literature; and adds to the molecular literature on mucinous cystadenocarcinoma, as none of the other studies on mucinous cystadenocarcinoma to date have reported an AKT1 variant.6,9,11,12 Regarding GNAS (Patient 2), although GNAS variants have been described in breast cancers,21,23 they are frequently identified in mucinous neoplasms of the pancreas (especially intraductal papillary mucinous neoplasms (IPMNs) of the pancreas), mucinous appendiceal neoplasms, mucinous colorectal carcinomas, and mucinous lung carcinomas, highlighting a strong association with mucinous histology.24–26 Additionally, GNAS is implicated to promote mucin secretion through increased expression of the gel-forming mucins MUC2 and MUC5AC. 25 This particular association generates the question of a possible association between the GNAS variants and the mucinous histology of mucinous cystadenocarcinoma of the breast. More studies in this regard are however required. We, unfortunately, could not study the type of MUC expressed in these 2 tumors as our immunohistochemistry laboratory does not have MUC stains in its test menu.
In addition to adding molecular data to the existing literature, Patient 2 in this study highlights that the biological behavior of mucinous cystadenocarcinoma can be heterogeneous regarding its potential for local recurrence. Of note, we have reported another patient in the past who presented with local recurrence in the ipsilateral breast 8 years after the initial diagnosis. 4 To date, we have diagnosed a total of 4 patients with mucinous cystadenocarcinoma, of which 2 have recurred. Both patients with recurrence did not receive neoadjuvant chemotherapy due to the tumors’ presumed indolent behavior based on the literature, and the paucity of data supporting a role for neoadjuvant or adjuvant chemotherapy and radiation. Therefore, we would emphasize that mucinous cystadenocarcinomas of the breast may not always display indolent behavior and should be considered for treatment with adjuvant or neoadjuvant chemotherapy and/or adjuvant radiation therapy as in routine TNBCs with adequate considerations for a patient's age, tumor stage, and tumor grade. A review of the literature illustrates that in addition to local recurrence, these tumors do have the potential to metastasize to regional axillary lymph nodes or distant sites. Eight out of 40 (20%) reported patients presented with concurrent metastases to ipsilateral axillary lymph nodes,2–4,7,10,13–15 and one patient had concurrent ipsilateral axillary lymph node and bone metastases (see Table 2). 7 Patient #3 in this study demonstrated large volume metastases to ipsilateral axillary lymph nodes containing dominant mucinous cystadenocarcinoma component. None of the patients reported in the literature with metastases or recurrences have died of the disease, with follow-up periods limited to up to 96 months (Table 2).
Literature Review for Primary Mucinous Cystadenocarcinomas of the Breast Presenting With Metastases or Recurrence.
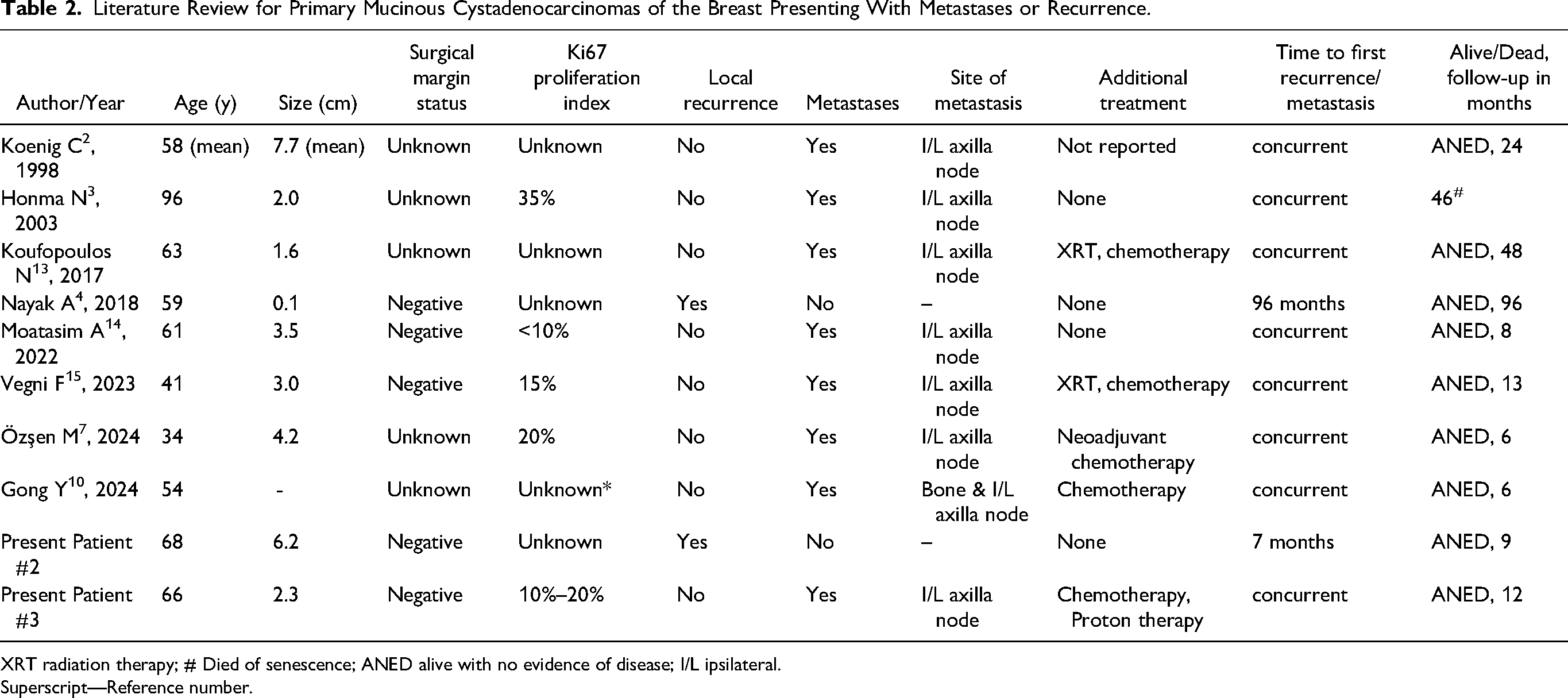
XRT radiation therapy; # Died of senescence; ANED alive with no evidence of disease; I/L ipsilateral.
Superscript—Reference number.
One might argue that the Ki-67 proliferation index could be useful for segregating patients with aggressive behavior to direct neoadjuvant therapy. However, given the lack of consensus over the utility of Ki-67 in triaging treatment and the appropriate cut-off percentage for Ki-67 in breast cancers, we do not routinely perform this test at our institution. A recent study by Srivastava et al further corroborates the ineffectiveness of the Ki-67 proliferation index in predicting the pathologic complete response to neoadjuvant therapy in triple-negative breast cancers with a low proliferation index. In thier study, 2 out of 18 patients (11%) treated with neoadjuvant therapy showed a pathologic complete response despite having a low Ki-67 index. 27
In conclusion, we report herein 3 patients with mucinous cystadenocarcinoma of the breast and contribute information on the somatic variant profile of this rare tumor. All tumors presented here highlight the common associated diagnostic error wherein mucinous cystadenocarcinoma is inadvertently diagnosed as a routine colloid carcinoma of the breast. To avoid this pitfall, we recommend careful attention to morphology and correlation with the biomarker profile in tumors with mucinous features. The second patient in our report further highlights that primary mucinous cystadenocarcinoma of the breast can defy the low-grade indolent behavior reported in the literature and may recur. A review of the literature on this rare tumor together with findings from the current report suggest that mucinous cystadenocarcinomas at the molecular level are similar to TNBC, with frequent recurring variants in PI3K pathway genes, including PIK3CA, PTEN, and AKT1 genes. We report here for the first time the presence of PTEN, AKT1, and GNAS variants in mucinous cystadenocarcinoma. A larger case series with molecular data on this rare subtype of breast cancer would be worthwhile to understand the exact biology and clinical behavior of this tumor. This information could be useful to offer targeted treatment against PIK3CA and AKT1 in patients with this rare tumor.
Footnotes
Acknowledgments
We acknowledge the Penn Genomics and Sequencing Core Facility (RRID:SCR_024999) and the Pathology Clinical Service Center of the University of Pennsylvania for providing the resources for molecular testing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require IRB approval for case reports with less than 3 patients.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
