Abstract
Biliary adenofibroma (BAF) is a rare hepatic tumor with a high risk for malignant transformation. Since BAF shares many characteristics with other hepatic lesions, diagnosis usually requires meticulous evaluation. The literature review revealed 29 reported patients of BAF, with malignant transformation occurring in 45% of patients. High Ki-67 (>40%) and mutated/overexpressed p53 staining were associated with malignancy, but some patients also had malignant features with low Ki-67 (≤10%) and wild-type p53. We report an elderly female patient who was initially diagnosed with benign liver hemangioma based on an abdominal ultrasound. The mass enlarged by 4 cm raising concerns about potential complications, including hemorrhage, which led to hepatic segmental resection. Histopathological examination revealed predominantly BAF features with focal areas that showed malignant cholangiocarcinoma characteristics.
Background
Biliary adenofibromas (BAFs) are rare benign liver tumors composed of glandular and fibrous elements. These tumors often incidentally appear in imaging studies because of nonspecific symptoms. 1 While rare, malignant transformation into cholangiocarcinoma is a real risk, highlighting the need for early and accurate diagnosis.2,3 On histological examination, BAF manifests as a well-circumscribed mass with glands embedded in a densely fibrous stroma. Because this topic has been reported in only a few studies, no comprehensive data have focused on natural history, optimal management strategies, or long-term outcomes. The diagnosis is challenging because of the rarity and overlapping clinical, radiological, and histomorphologic features of these tumors with other liver lesions. 4 Therefore, increased awareness and documentation within the medical community are essential to develop guidelines for management and monitoring.
Case Report
The patient was a woman with a clinical history of diverticulitis and a liver mass that had been present for the past 5 years, initially presumed to be a hemangioma. Imaging of the liver revealed an exophytic mass in the left liver lobe, which had increased in size by 4 cm since it was first imaged 2 years prior. The mass was suspicious for a growing hemangioma, a benign and typically harmless lesion. The surgical team performed a left lateral liver resection because the continuous growth posed potential complications, including the risk of rupture and hemorrhage. The mass was observed to occupy segments 2 and 3 during the surgery and was excised without complications. The patient was discharged a few days later.
Pathological Findings
Histological examination (Figure 1) revealed well-differentiated cholangiocarcinoma within a preexisting BAF. The resection margin was positive, and the pathological stage was pT1b NX Mx according to the AJCC eighth edition. These findings did not correlate with the initial preoperative diagnosis of liver hemangioma. The mass measured 11 cm at its largest dimension and was found abutting the liver capsule. However, the invasive component measured approximately 2 cm. It displayed complex architectural features, including ductular structures, intracystic papillary proliferation, and cribriform patterns. These histological characteristics and a Ki-67 proliferative index of approximately 40% (Figure 2D) strongly indicated malignant transformation The benign component of the BAF is characterized by complex, nonmucinous tubuloacinar glandular structures surrounded by moderately cellular fibrous stroma containing myofibroblastic spindle cells. KRT7 and KRT19 demonstrated strong, diffuse staining of the epithelial components in BAF and cholangiocarcinoma, with both lesions exhibiting positive resection margins (Figure 2A-C). Additionally, it exhibited a low Ki-67 proliferative index of less than 10%, indicating minimal cellular proliferation within this component. Immunohistochemical analysis showed an absence of nuclear β-catenin staining (Figure 2E and F) and focal overexpression of p53 (Figure 2G), indicating molecular alterations within the lesion. The surgical resection margins were positive for malignancy, suggesting incomplete tumor excision. The non-neoplastic hepatic parenchyma also exhibited nodular regenerative hyperplasia, mild lymphocytic portal inflammation, and no portal fibrosis (stage 0/6), according to the Ishak fibrosis staging system. 5
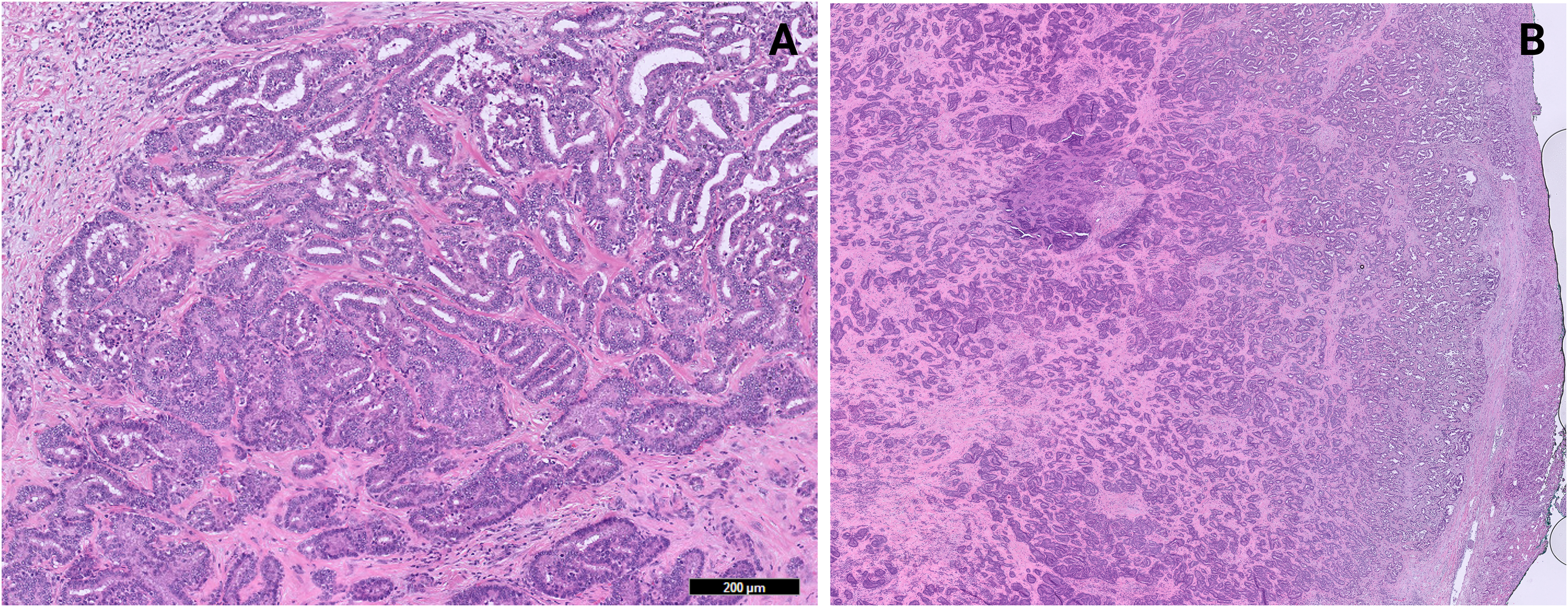
Histopathological features of biliary adenofibroma (BAF) and associated cholangiocarcinoma. (A) Biliary adenofibroma: Complex nonmucinous tubuloacinar glandular structures within a moderately cellular fibrous stroma containing myofibroblastic spindle cells (H&E, 10×). (B) Biliary adenofibroma with cholangiocarcinoma at the resection margins: The epithelial component comprises tubuloacinar and cystically dilated glands. In contrast, the stromal component consists of myofibroblastic cells in a background of moderate fibrosis (H&E, 2×).
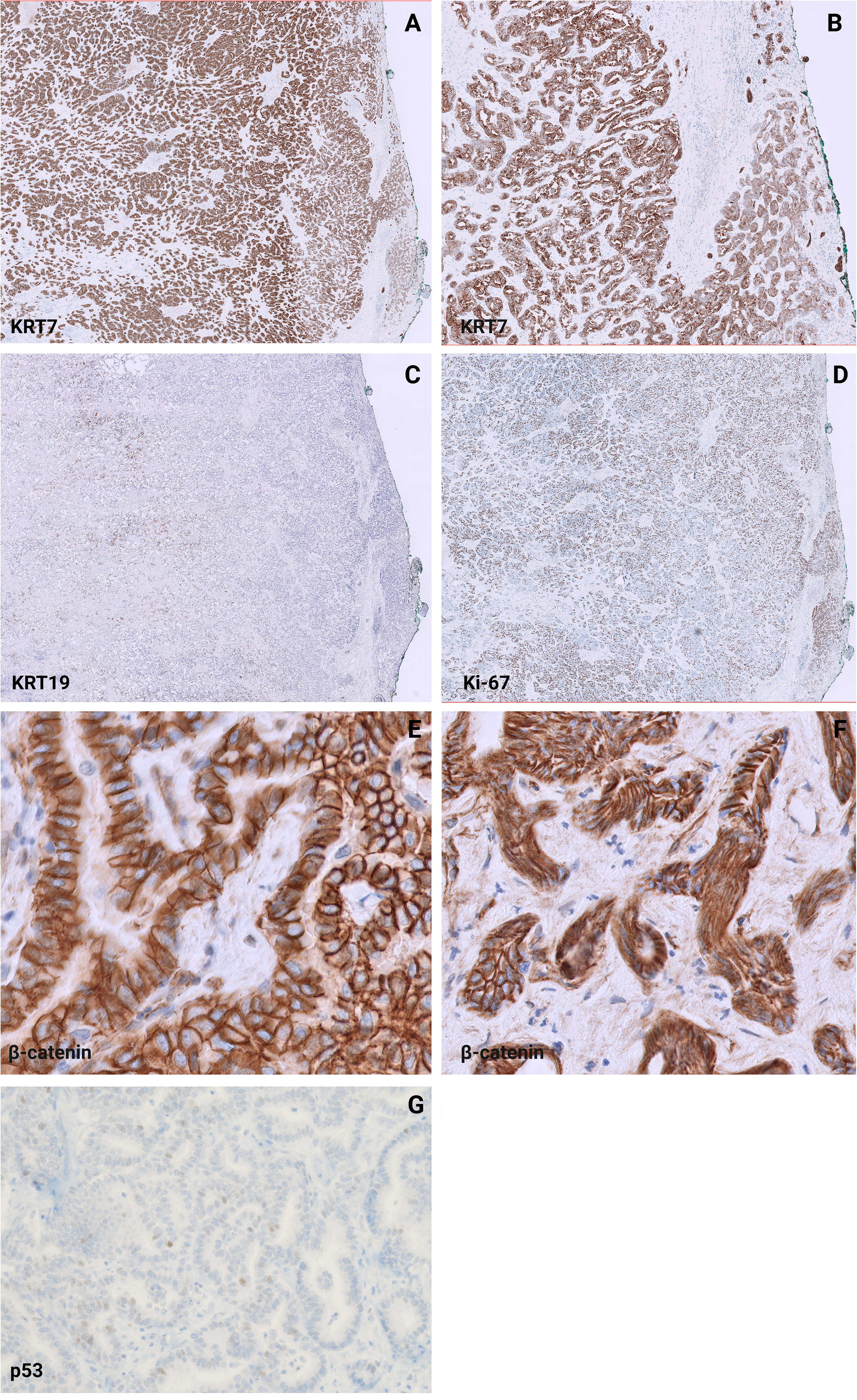
Immunohistochemical staining patterns in biliary adenofibroma and cholangiocarcinoma components. (A and B) Strong, diffuse KRT7 staining and (C) positive KRT19 staining both highlight the epithelial components of biliary adenofibroma and cholangiocarcinoma, with positive resection margins (KRT19, 2×). (A) Low magnification (2×); (B) Higher magnification (10×). (D) High Ki-67 proliferation index (≥40%) was observed in the cholangiocarcinoma lesion (Ki-67, 2×). (E and F) Absence of nuclear β-catenin stain (β-catenin, 40×). (G) Focal overexpression of p53 stain (p53, 20×).
Management and Follow-Up
This diagnosis significantly affected the patient's prognosis and necessitated a revised management plan. A postsurgical computed tomography (CT) (Figure 3) of the abdomen and pelvis revealed only postsurgical changes from left hepatic lobe resection. The patient was subsequently discussed at a multidisciplinary tumor board, and potential adjuvant chemotherapy was considered, depending on the final stage and the patient's response to the initial surgical intervention. The adjuvant chemotherapy plan included 8 cycles of capecitabine (Xeloda) while awaiting reresection. The patient reported overall well-being and denied symptoms including nausea, vomiting, fever, abdominal distension, wound dehiscence, or abdominal pain. Seven months post-resection, a posttreatment CT of the abdomen and pelvis showed no evidence of tumor recurrence, with only postsurgical changes and no ductal dilatation. The previously observed subcentimeter hepatic hypodensity remained stable. This transition from what was once a benign lesion to a malignant entity highlights the critical importance of thorough histopathological evaluation and comprehensive cancer care.
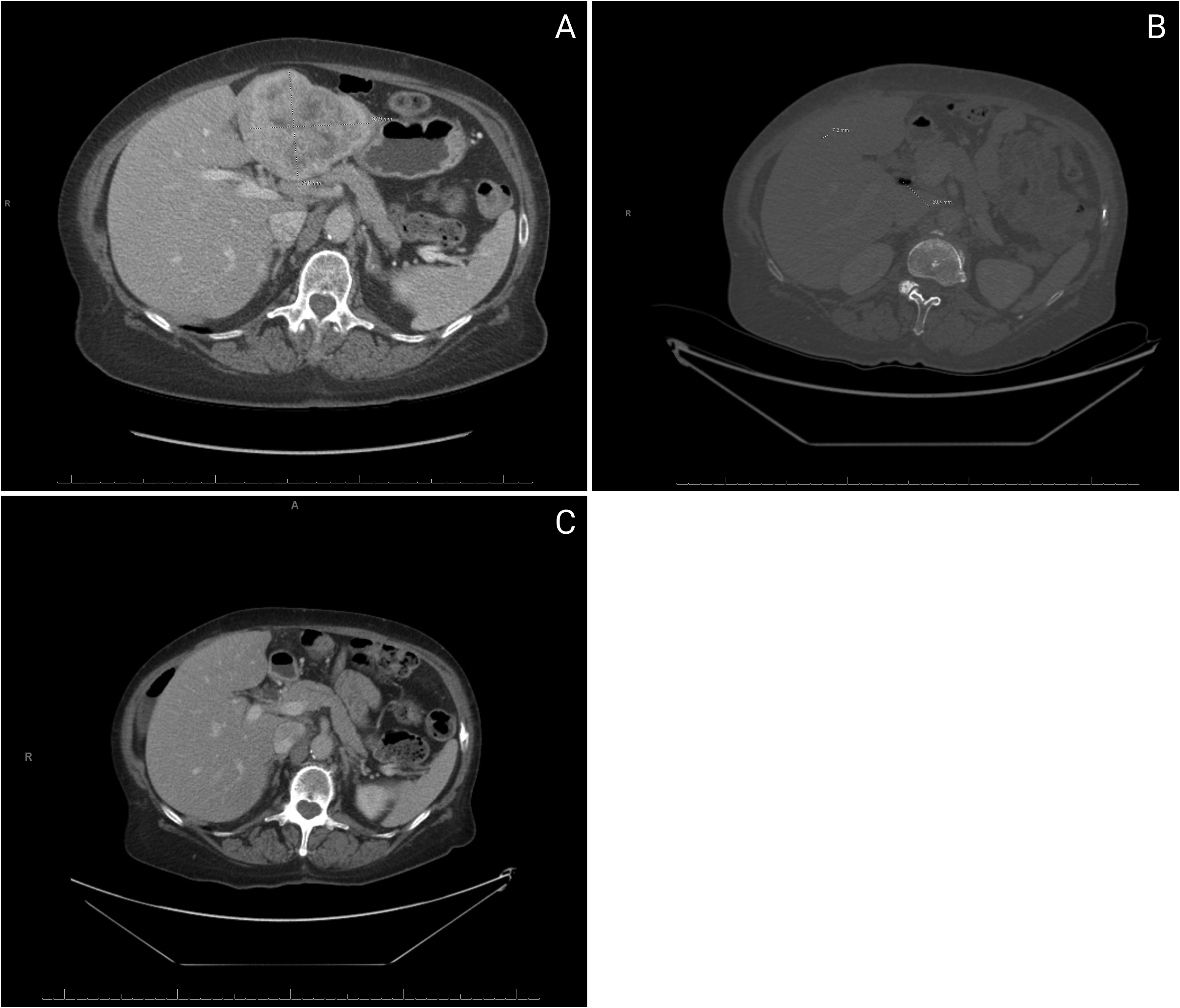
Computed tomography (CT) imaging of the lesion. (A) Preoperative CT demonstrates a normal-sized liver (19-cm oblique craniocaudal dimension) with preserved parenchymal architecture. A lobulated mass in the lateral segment (8.3 × 7.6 × 9 cm) exhibits peripheral arterial phase enhancement and centripetal progression matching blood pool density, consistent with atypical hemangioma. The portal vein is patent without thrombosis or flow obstruction, and there is no evidence of biliary ductal dilatation or hyperdense gallstones. (B) Postoperative CT revealed postoperative changes and no changes in the 0.6-cm hypodensity in the right hepatic lobe. (C) Posttreatment CT showed postsurgical changes and no ductal dilatation. The previously noted subcentimeter hypodensity in the liver is stable.
Discussion
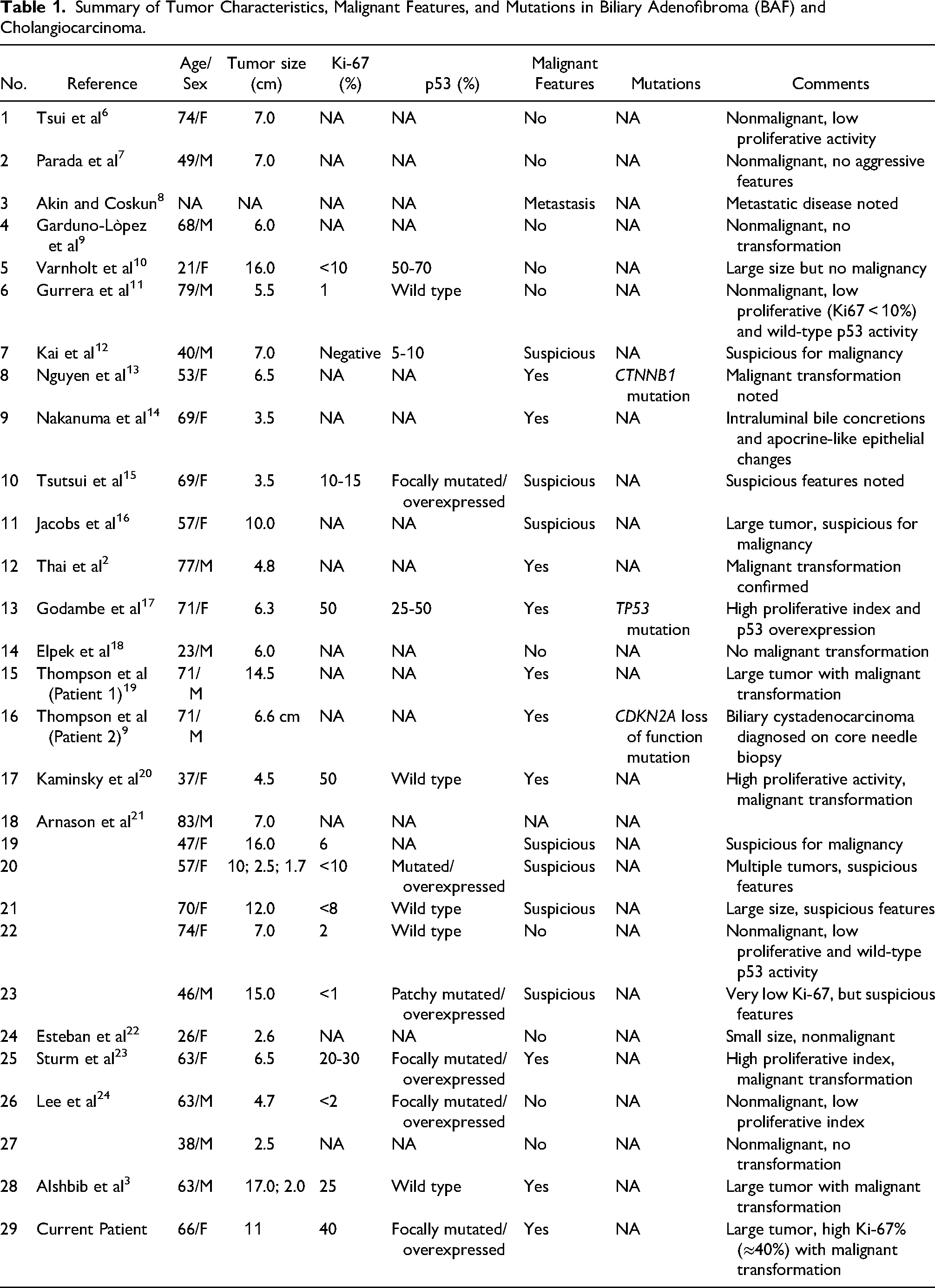
Biliary adenofibroma is a rare and complex benign hepatic tumor. After a review of the literature, we identified approximately 28 other reported patients. In Table 1, we summarize the pathological findings of these patients. After adding this patient, the conclusions are as follows: malignant transformation was observed in 13 out of 29 patients, indicating that approximately 45% of the reported patients in the literature developed malignancy. High Ki-67 (>10%) and mutated/overexpressed p53 staining frequently correlate with malignant features. However, some patients with low Ki-67 (≤10%) and wild-type p53 staining also had suspicious or confirmed malignancy; this finding infers that these markers are not absolute. Specific mutations, such as CTNNB1, CDKN2A, and TP53, have been identified in patients with malignant transformation, underscoring their potential role in the pathogenesis of biliary cancer.
Summary of Tumor Characteristics, Malignant Features, and Mutations in Biliary Adenofibroma (BAF) and Cholangiocarcinoma.
Although it is a benign tumor, there is a significant risk for malignant transformation; therefore, early and proper diagnosis is essential to prevent this progression. The clinical manifestations of BAF are primarily nonspecific and frequently include abdominal pain, jaundice, or a palpable mass. Even imaging features such as well-demarcated hepatic lesions with cystic components, as visualized by ultrasound, CT, and magnetic resonance imaging, are nonspecific. This lack of specificity makes the diagnosis challenging, as these lesions can easily be mistaken for other hepatic tumors.1,3 Biliary adenofibroma's etiology and risk factors remain poorly understood due to its rarity. Unlike intrahepatic cholangiocarcinoma, BAF is often reported in patients without a history of chronic liver disease, viral hepatitis, or other established risk factors for biliary neoplasms. Chau et al suggest a potential link between BAF and congenital ductal plate malformations, pointing to developmental abnormalities as a possible origin. 25 These findings highlight a distinct pathophysiology, separate from the inflammatory and fibrotic pathways commonly associated with intrahepatic cholangiocarcinoma.
Histologically, benign BAF comprises tubuloacinar glandular and microcystic structures lined by cuboidal epithelium in moderately cellular fibrous stroma, highly similar to low-grade renal tubulocystic carcinoma.15,23,26 In contrast, the histologic features of malignancy include ductular structures, intracystic papillary proliferation, and cribriform patterns, and the characteristics of malignant transformation include cellular atypia, hyperchromatic nuclei, increased mitotic activity, and capsular, lymphovascular, or perineural invasion.1,26 A hallmark of malignant transformation in BAF is the transition zone between the benign and malignant structures. It often shows cellular atypia, prominent nucleoli, and tissue infiltration. Interestingly, there may be evidence of perineural invasion in the benign-appearing areas, highlighting their malignant potential. 26
The differential diagnoses may include biliary adenoma, intrahepatic cholangiocarcinoma, hepatocellular carcinoma, and metastatic adenocarcinoma.3,27 They are distinct entities with unique characteristics. A biliary adenoma is a benign neoplasm of uniformly distributed small bile ducts in a fibrotic stroma, typically presenting as a solitary subcapsular nodule. It exhibits low Ki-67 proliferation (<10%) and positive p16INK4a immunoreactivity.28,29 In contrast, intrahepatic cholangiocarcinoma is a malignant epithelial neoplasm with biliary differentiation, often showing a mass-forming, periductal infiltrating, or intraductal growing pattern. 30 Intrahepatic cholangiocarcinoma typically displays strong p53 expression, negative p16 staining, and a higher Ki-67 index (>10%). 31 hepatocellular carcinoma, derived from hepatocytes, differs from intrahepatic cholangiocarcinoma in its enhancement pattern, showing arterial hyperenhancement and portal venous washout. 32 Metastatic adenocarcinoma can be distinguished from primary liver tumors using specific immunohistochemical markers: CDX2 for colorectal origin, TTF-1 for lung origin, and GATA3 for breast origin. 33 Additionally, most bile duct adenomas are positive for albumin in situ hybridization, which helps differentiate them from metastatic pancreatic adenocarcinoma. 34
Surgical complete excision remains the treatment of choice for BAF, especially in localized lesions. 35 When surgery is performed, adjuvant chemotherapy and radiotherapy are usually given to decrease the chance of recurrence.36,37 In unresectable or advanced BAF, systemic chemotherapy with gemcitabine and cisplatin remains a common standard. 38 Checkpoint inhibitors can also be considered in patients with high microsatellite instability. Close follow-up is essential for detecting disease recurrence early and managing potential treatment-related complications. 39
In a noteworthy study, Masetto et al conducted molecular analyses on biliary neoplasms, identifying recurring mutations in chromatin remodeling genes such as ARID1A, BAP1, and PBRM1. These mutations are commonly linked to intrahepatic cholangiocarcinoma. Additionally, the research uncovered clinically targetable mutations, including FGFR2-MCU fusions and IDH2 mutations, within biliary neoplasms. 26 Inhibitors such as pemigatinib (for FGFR2) and ivosidenib (for IDH) have demonstrated encouraging outcomes in cholangiocarcinoma and could potentially be applied to transformed BAF patients. 26 These therapeutic approaches underscore the critical role of genetic testing in managing patients with biliary tumors. Further studies are required in both the genetic and molecular levels of pathogenesis, the development of targeted therapies, and diagnostic accuracy. New case reports are crucial for understanding clinical features, histopathology, and genetics, and they are all helpful for proposing more appropriate diagnostic and therapeutic strategies for BAF disease.
Conclusion
Malignant transformation, particularly with gene mutations in CTNNB1, CDKN2A, and TP53, has been observed in BAFs. Given the significant risk of progression to cholangiocarcinoma, clinicians must maintain a high level of suspicion when diagnosing similar hepatic lesions. We also suggest establishing standardized guidelines for the management and follow-up of such rare tumors to improve the outcomes of such patients and direct appropriate indications for treatment. Further research into the molecular pathogenesis of BAF will help develop targeted therapies and improve diagnostic accuracy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
