Abstract
Adenomatoid tumors are benign neoplasms of mesothelial phenotype, commonly found in the genital tracts. Hepatic adenomatoid tumors are extremely uncommon and can present diagnostic challenges, particularly when mixed with other histologic elements. Here we report an adenomatoid tumor with prominent admixed cavernous hemangioma-like vessels in the liver of a 58-year-old woman. Grossly, the tumor appeared as a pedunculated lesion on the hepatic surface with cystic spaces filled with blood. Microscopic examination revealed numerous blood vessels of varying sizes intermingled with anastomosing tubular structures lined by cuboidal epithelioid cells that were immunohistochemically positive for mesothelial markers. Areas of typical cavernous hemangioma were also present within the same lesion. To our knowledge, this tumor represents the first documented instance of an adenomatoid tumor with prominent admixed cavernous hemangioma-like vessels in the liver. Our case report summarizes previously reported hepatic adenomatoid tumors and highlights their clinical, histopathologic, and immunohistochemical features. Given the various morphologic presentations, adenomatoid tumors can mimic other neoplasms. Their benign nature and excellent prognosis with complete surgical resection make accurate diagnosis crucial. Increased understanding and awareness of this uncommon tumor, combined with morphologic analysis and immunohistochemical studies, can help overcome diagnostic challenges to ensure accurate diagnosis and appropriate clinical management.
Introduction
Adenomatoid tumors are benign neoplasms of mesothelial phenotype that are most commonly found in the genital tracts.1–4 Extragenital adenomatoid tumors, including those occurring in the liver,5–11 are rare and can pose significant diagnostic challenges. Hepatic adenomatoid tumors generally present with an excellent prognosis following complete surgical resection. However, these tumors can exhibit a variety of morphologic features, mimicking other types of neoplasms such as vascular lesions and carcinomas, leading to potential diagnostic confusion. The tumor presented here is particularly unique as it represents the first known instance of an adenomatoid tumor with prominent admixed cavernous hemangioma-like vessels in the liver. We report this tumor to highlight the importance of considering adenomatoid tumors in the differential diagnosis when encountering well-circumscribed, hypervascular hepatic lesions and to emphasize the utility of combining careful morphologic analysis with immunohistochemical work-up to prevent misdiagnosis and improper clinical management.
Case Report
A 58-year-old woman with a notable medical history of Lynch syndrome and ductal carcinoma in-situ of the breast underwent regular surveillance following bilateral mastectomy, total hysterectomy and salpingo-oophorectomy. On abdominal computed tomography (CT) scan, she was found to have a heterogeneously enhancing mass arising from the superior pole of the right kidney. A follow-up CT with contrast showed a mild interval increase in the size of the mass, concerning for a slow-growing renal cell carcinoma. The mass was found to be inseparable from the medial aspect of the posterior right hepatic lobe, measuring 2.1 × 1.9 cm.
The patient was scheduled for right partial nephrectomy to address the suspected renal lesion. However, during the procedure, the surgeons discovered that the mass was actually of hepatic origin. Upon elevating the liver edge, it was observed that the mass dangled from the inferior surface of the right hepatic lobe as an exophytic lesion anchored by a 5 mm vascular stalk. Consequently, the surgical approach was modified, and the patient underwent wedge resection of the liver mass. The mass showed no association with the right kidney. The Gerota fascia and the perinephric fat were not entered during the resection.
Pathological Findings
The specimen was received as a well-circumscribed pedunculated mass lesion measuring 2.1 × 1.8 × 1.2 cm. It was attached to the surface of a 0.5 × 0.4 × 0.2 cm portion of nonneoplastic liver tissue via a short narrow (2-3 mm) vascular stalk and appeared encapsulated with continuation from the Glisson capsule (Figure 1A). The cut surfaces of the lesion were pink-to-tan with areas of pale-to-white discoloration and displayed multiple small thin-walled cystic spaces filled with clotted blood. No signs of necrosis were identified.
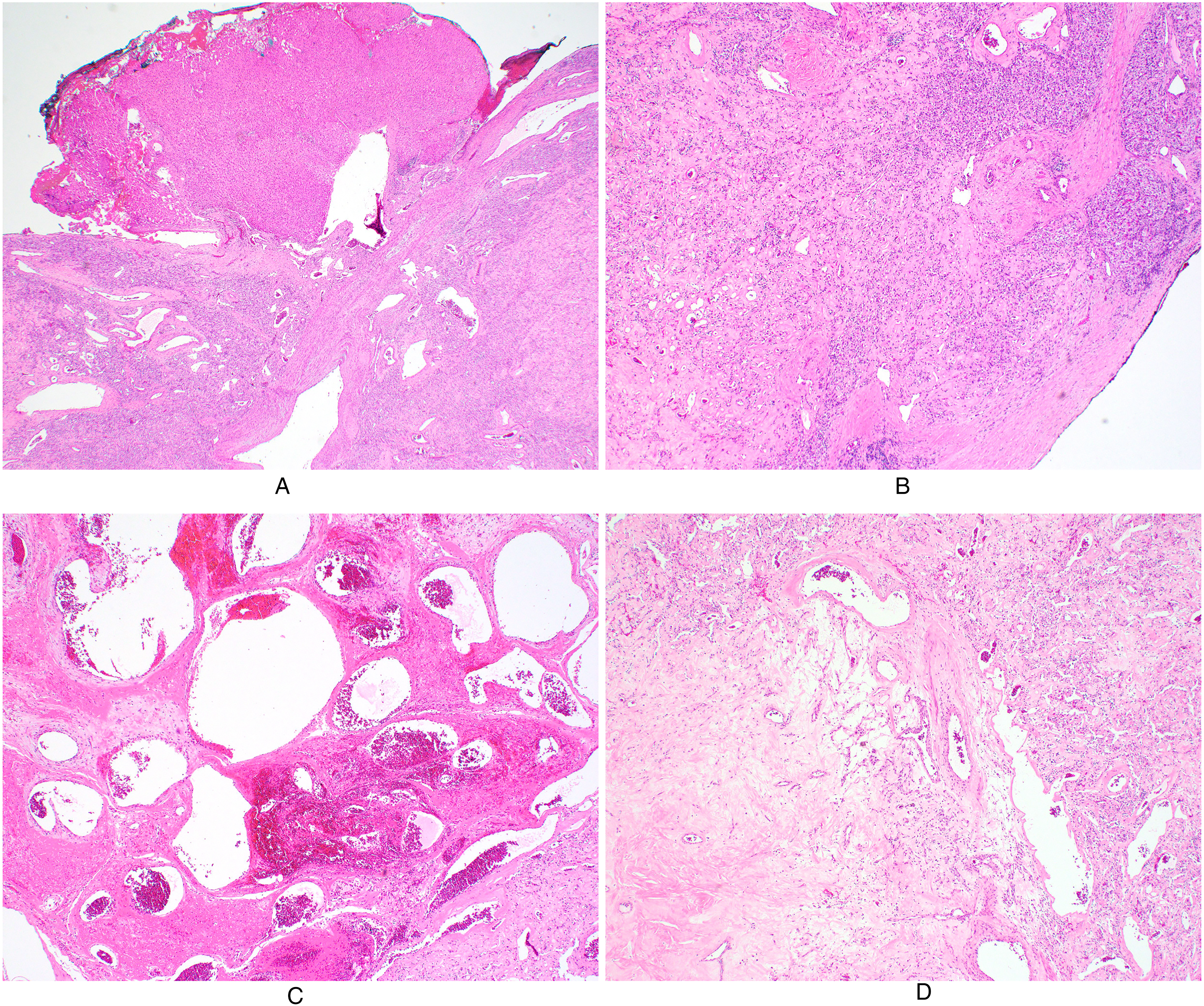
Histologic features on low-power view. (a) The tumor is attached to the surface of the liver via a short narrow vascular stalk. It appears encapsulated with the capsule continuous from the Glisson capsule of the liver (H&E, original magnification ×20). (b) The lesion consists of intermingled blood vessels of varying sizes and anastomosing tubular structures that appear compressed in areas (H&E, original magnification ×40). (c) Focal areas of typical cavernous hemangioma are present within the lesion (H&E, original magnification ×40). (d) Large paucicellular areas with hyalinized or edematous stroma are noted (H&E, original magnification ×40).
Microscopically, the lesion consisted of numerous blood vessels of varying sizes and anastomosing tubular structures. These two components closely intermingled in most areas (Figure 1B), but focal areas of typical cavernous hemangioma were present (Figure 1C). Large paucicellular areas with hyalinized or edematous stroma were also noted (Figure 1D). No hepatocytes or bile ducts were present within the lesion. The vascular channels were lined by flat endothelial cells (Figure 2A), which were immunohistochemically positive for CD31, CD34 (Figure 2B), and ERG. The tubular structures were lined by a single layer of cuboidal epithelioid cells that exhibited clear and vacuolated cytoplasm, relatively uniform and round nuclei, small nucleoli, and indistinct cell borders (Figure 3A). No significant nuclear atypia or mitosis was observed in epithelioid cells. There were hypercellular areas where the tubular architecture became indistinctive (Figure 3B) and hypocellular areas where epithelioid cells formed short cords or small clusters (Figure 3C). Some tubular structures appeared to have appreciable lumens whereas others did not with a compressed appearance.
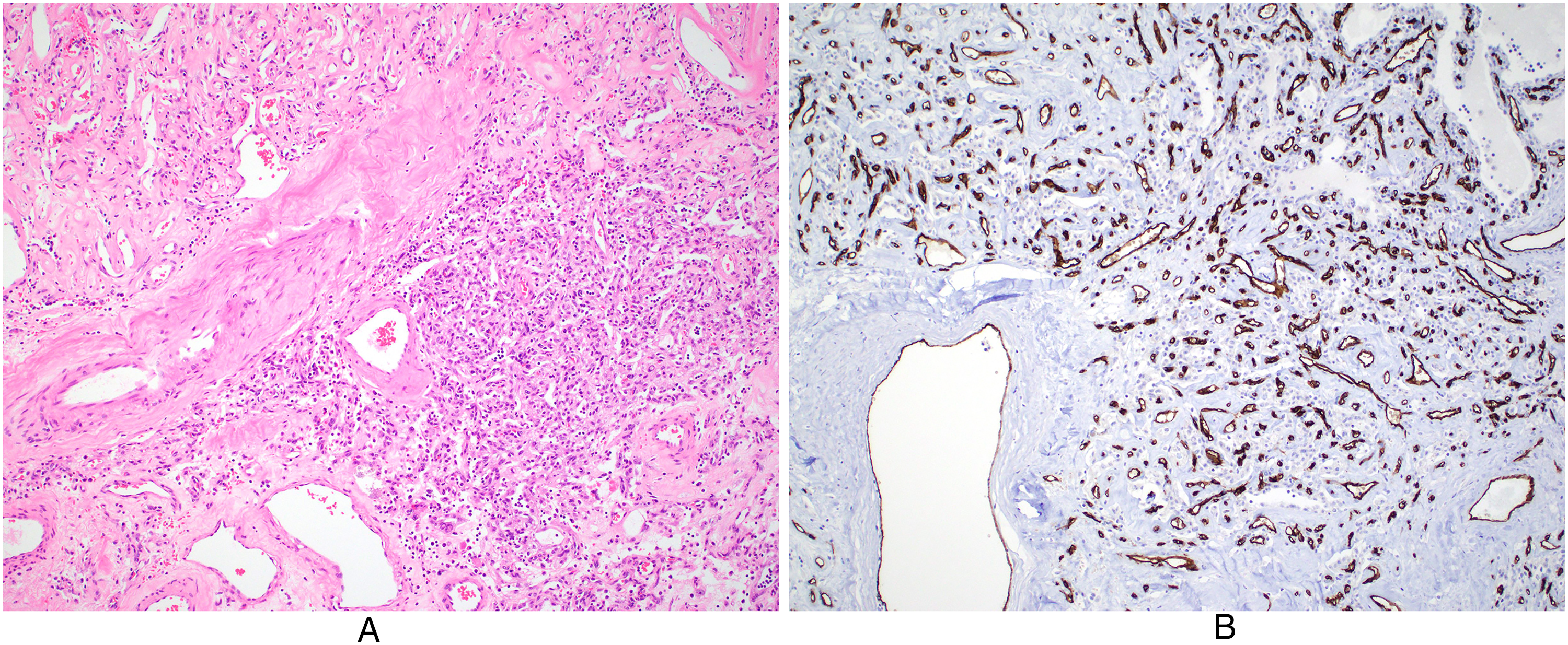
Hypervascularity. (a) There are numerous blood vessels of varying size ranging from capillaries to veins and arteries (H&E, original magnification ×100). (b) The vascular channels are highlighted by CD34 immunostaining (original magnification ×100).
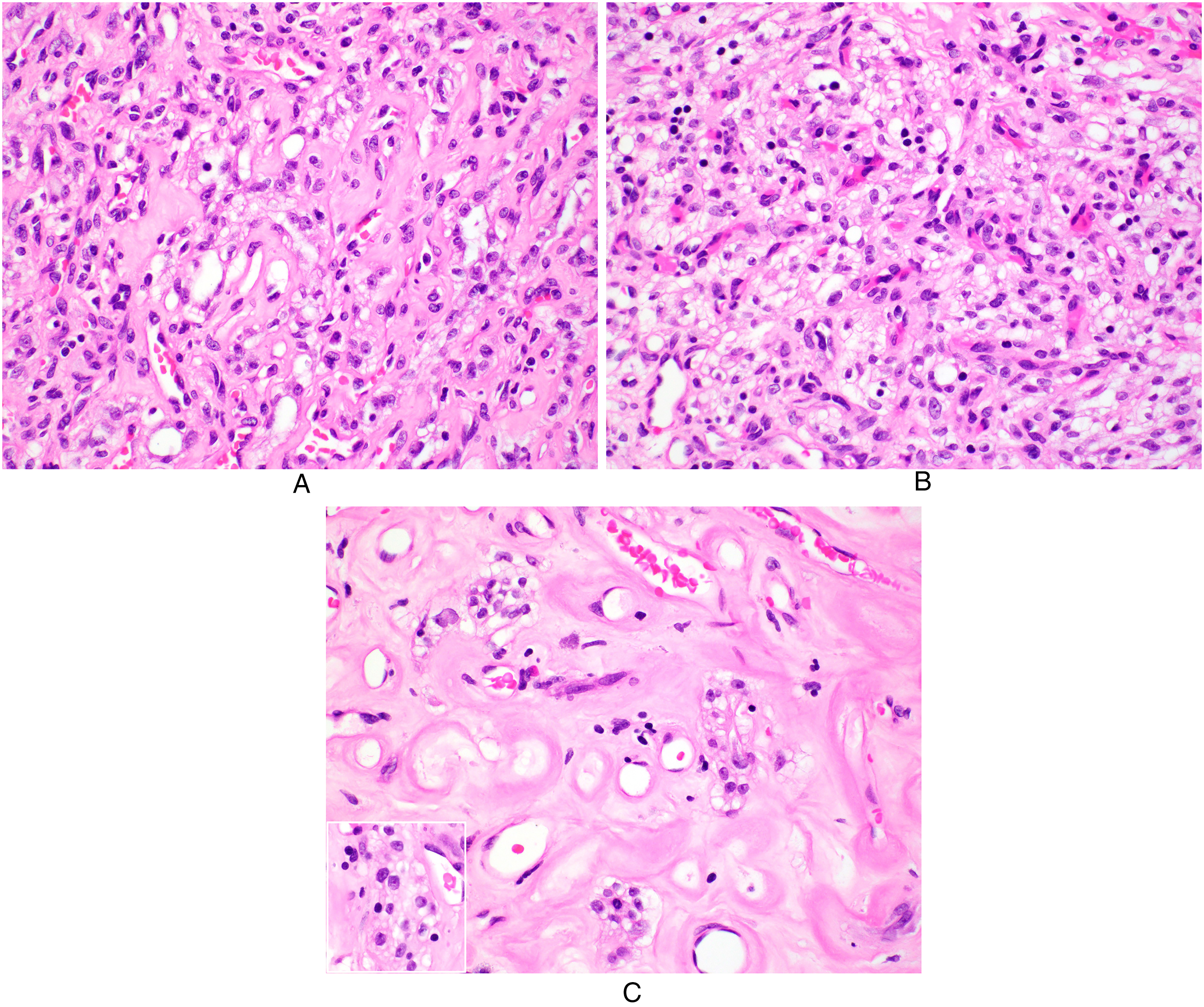
Histologic features on high-power view. (a) The tubular structures are lined by a single layer of cuboidal epithelioid cells that exhibit clear and vacuolated cytoplasm, relatively uniform and round nuclei, small nucleoli, and indistinct cell borders. No significant nuclear atypia or mitosis is observed (H&E, original magnification ×400). (b) In hypercellular areas, the tubular architecture becomes indistinctive (H&E, original magnification ×400). (c) In hypocellular areas, the epithelioid cells form short cords or small clusters in a hyalinized stroma (H&E, original magnification ×400; inset, higher magnification).
Immunohistochemically, the epithelioid cells were positive for pankeratin (AE1/AE3; Figure 4A), keratin 7 (Figure 4B), keratin 19, GLUT1, and mesothelial markers including WT1 (Figure 4C), calretinin (Figure 4D), keratin 5/6, and podoplanin (D2-40). These cells were negative for CEA, Ber-EP4, claudin-4, PAX8, RCC, arginase-1, hepar-1, and TTF1. Immunostaining for BAP1 showed retained nuclear expression. The proliferation index, as determined by Ki67 immunostaining, was estimated to be <5%. The nuclear expression of DNA mismatch repair proteins (MLH1, MSH2, MSH6, PMS2) was intact in epithelioid and endothelial cells.
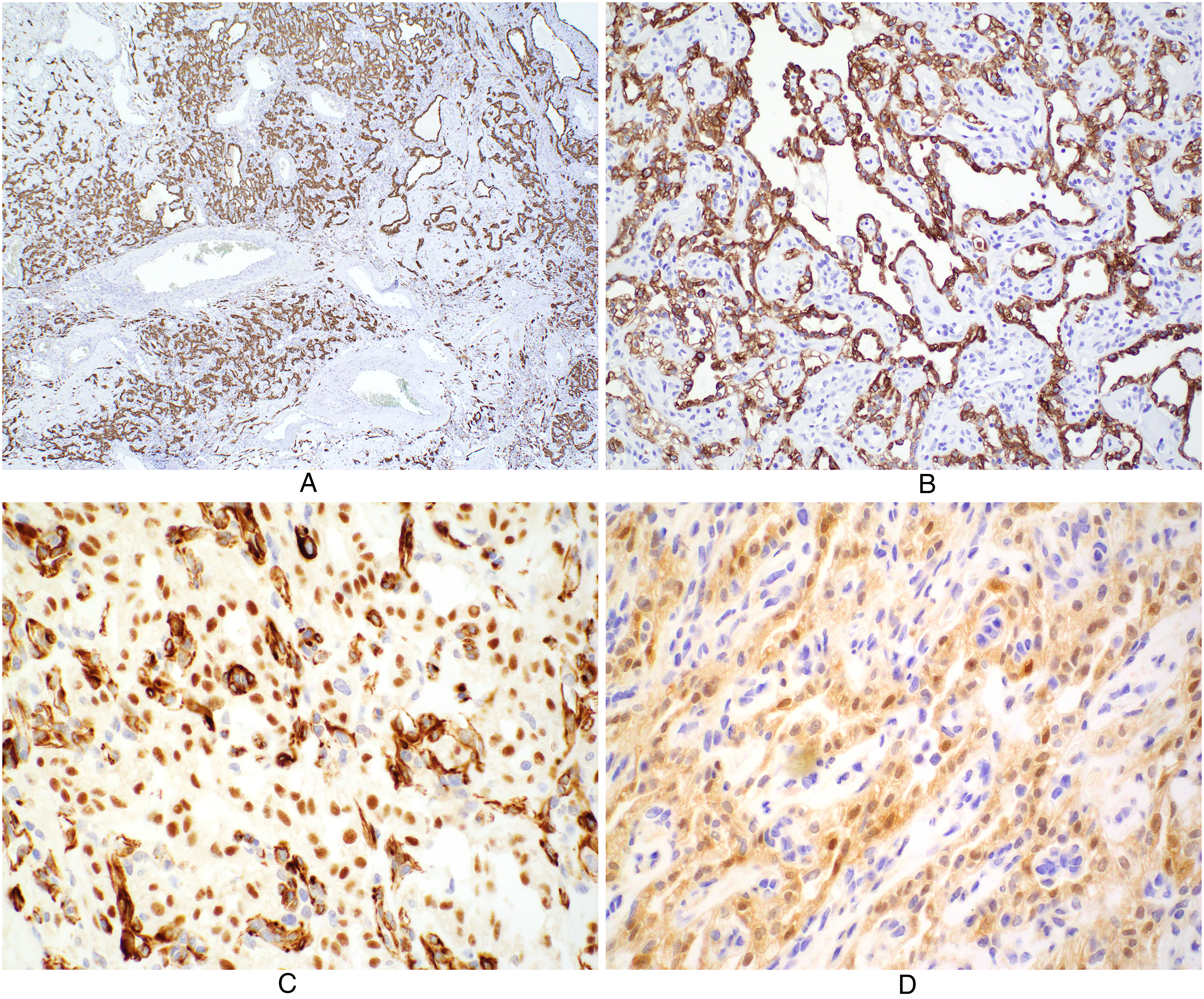
Immunohistochemical findings. The epithelioid cells are positive for (a) pankeratin (AE1/AE3; original magnification ×40), (b) keratin 7 (original magnification ×200), (c) WT1 (original magnification ×400), and (d) calretinin (original magnification ×400).
Discussion
We report an exceedingly rare case of hepatic adenomatoid tumor with prominent admixed cavernous hemangioma-like vessels, which was initially thought to be a right renal mass on CT scan. Intraoperatively, the tumor was found to be a pedunculated lesion emanating from the inferior surface of the liver. The diagnosis was challenging based on the findings of gross and histologic examination with differentials that might include hepatocellular neoplasm, intrahepatic cholangiocarcinoma, clear cell renal cell carcinoma, adrenocortical neoplasm, perivascular epithelioid cell tumor (PEComa), hemangioblastoma, epithelioid hemangioendothelioma, among others. Immunohistochemical studies were extremely helpful in establishing the diagnosis.
Adenomatoid tumors are benign neoplasms of mesothelial phenotype. 1 They are predominantly found in the genital tracts.2–4 Instances of extragenital manifestations have been documented, 12 including those in the adrenal glands,13–15 heart, 16 pleura, 17 and mediastinum. 18 Uncommon examples involving the digestive system, including the pancreas, 19 small intestine, 20 mesocolon and omentum, 21 appendix, 22 and liver,5–11 have also been reported. As shown in Table 1, 5 of 7 reported hepatic adenomatoid tumors were single lesions, while 2 reported tumors also involved the peritoneum and retroperitoneum. Six reported tumors consisted solely of adenomatoid components. One reported tumor also had a concurrent but separate hemangioma in the liver as an independent lesion. 9 The tumor we reported is unique because a component of typical cavernous hemangioma is present concurrently within the same lesion.
Data summary of reported adenomatoid tumors of the liver.
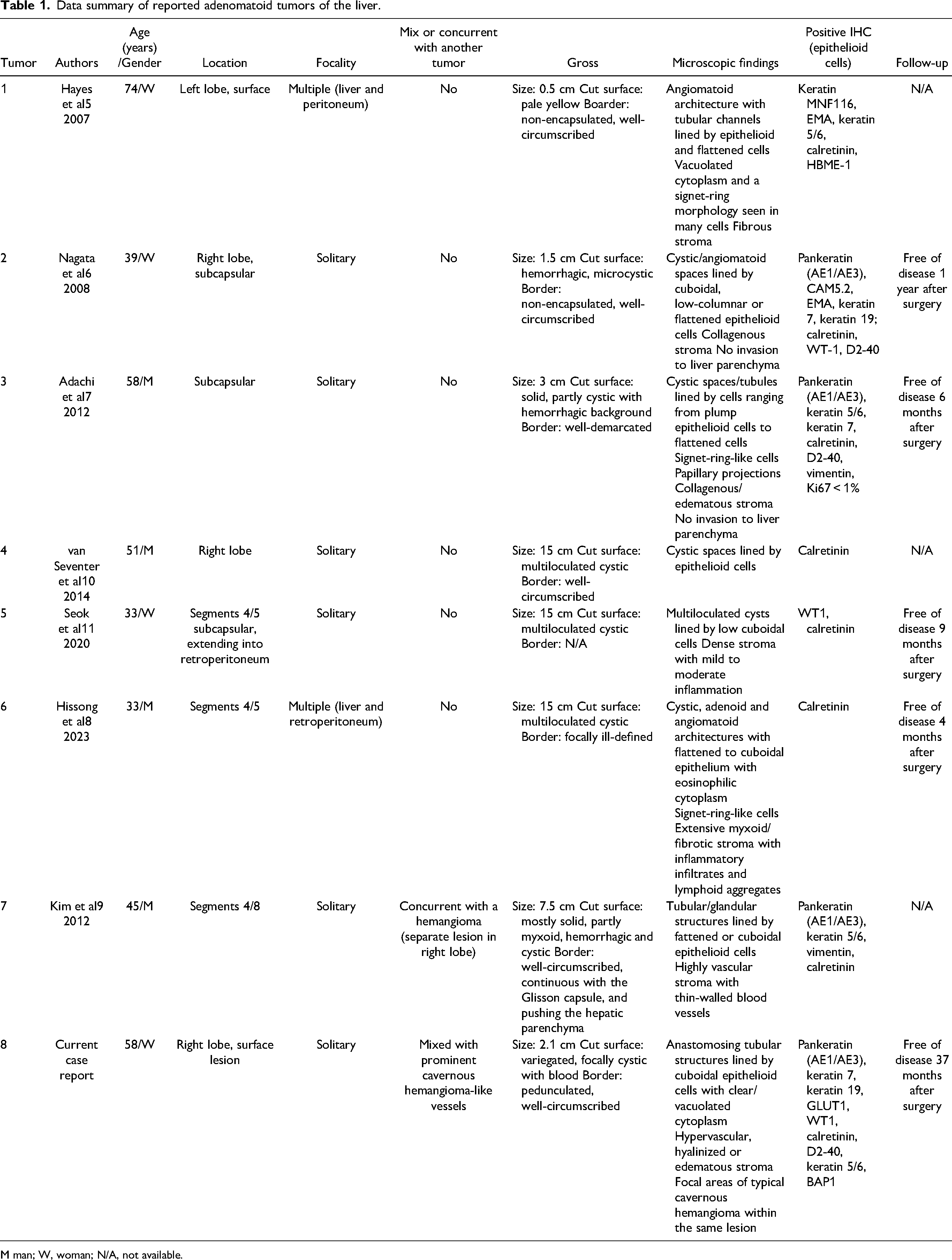
M man; W, woman; N/A, not available.
Adenomatoid tumors of the liver usually present as well-circumscribed lesions with a cystic and hemorrhagic appearance.5–11 They are almost always described as surface or subcapsular lesions (Table 1), supporting their mesothelial phenotype and possibly origin from the hepatic capsule. They often display architectural heterogeneity with a range of histologic patterns, including adenomatoid, angiomatoid, solid, cystic, and micropapillary forms. Tumor cells in previous case reports are often described as flattened, cuboidal to low-columnar epithelioid cells, forming tubular or cystic structures, with occasional signet-ring-like morphology.8,9 Nuclear atypia is not identified in our tumor or in 6 previously reported tumors;5,6,8–11 however, one tumor is described to have moderate nuclear pleomorphism.7 No mitosis, necrosis, or invasive growth is observed in reported tumors.
Interestingly, when reviewing the diagnostic process for reported hepatic adenomatoid tumors, we noted that vascular lesions were almost always at the top of the differential diagnosis.5–9 This is not surprising because most reported hepatic adenomatoid tumors are hypervascular or angiomatoid with a cystic and hemorrhagic gross appearance. The resemblance to vascular lesions is particularly distinctive for hepatic adenomatoid tumors because hypervascularity has rarely been described in adenomatoid tumors in other locations. Therefore, when a hypervascular hepatic lesion is encountered, adenomatoid tumor should be included in the differential diagnosis, particularly when it is subcapsular or on surface in location. Immunohistochemical stains for mesothelial and endothelial markers are very helpful in the distinction. It is interesting to note that our tumor is also hypervascular with numerous variably sized blood vessels intermixed with clusters of adenomatoid tumor cells. However, convincing areas of classical cavernous hemangioma are present within the same lesion, which makes this tumor unique.
The presence of epithelioid cells with tubular or glandular structures, occasional signet-ring-like morphology, and positive keratin immunohistochemistry may also cause confusion with carcinomas such as intrahepatic cholangiocarcinoma. Apparently, invasive growth patterns, nuclear atypia, mitosis, and necrosis are useful features for the diagnosis of carcinomas and are typically lacking in adenomatoid tumors. Immunohistochemical expression of mesothelial markers can further help the distinction. Another differential is malignant mesothelioma, which should also be distinguishable from adenomatoid tumor based on cytomorphologic features. The loss of BAP1 nuclear expression, the loss of MTAP (methylthioadenosine phosphorylase) cytoplasmic expression, the complete loss of merlin expression, and homozygous deletion of CDKN2A (by FISH) are useful diagnostic markers for mesothelioma. 23
The treatment choice for adenomatoid tumors is surgical resection, which is considered curative. In 7 reported hepatic adenomatoid tumors, follow-up data are available for 4 patients who were all free of disease spanning a period of 4-12 months post-surgery.6–8,11 Our patient has also been disease-free with no recurrence or metastasis for 37 months. She continues to undergo cancer surveillance given the history of Lynch syndrome.
In summary, we present the first hepatic adenomatoid tumor with prominent admixed cavernous hemangioma-like vessels. The diagnosis is challenging due to its rarity and mixed adenomatoid and angiomatous morphology. Adenomatoid tumor should be included in the differential diagnosis when a well-circumscribed subcapsular or surface lesion is encountered in the liver, particularly when it is hypervascular and exhibits anastomotic tubular structures with bland cytology. It is important to distinguish adenomatoid tumors from carcinomas, mesothelioma, or other neoplasms with malignant potential to avoid unnecessary treatment after complete surgical resection. Combined morphologic analysis and immunohistochemical work-up can help achieve the accurate diagnosis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Ethical Approval
IRB approval is not required for retrospective single-case report.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
