Abstract
Introduction
The significance of radiation therapy in cancer treatment comes with associated complications, including fibrosis, osteonecrosis, and the development of secondary malignancies, such as malignant peripheral nerve sheath tumors (MPNSTs). We emphasize the importance of understanding these complications for an effective patient management.
Methods
We report a 47-year-old man with a history of squamous cell carcinoma of the tongue, treated with surgery, chemotherapy, and radiation therapy. The patient later presented with symptoms that led to the discovery of an intraosseous MPNST.
Results
Histopathological examination revealed characteristic features of MPNST, including spindle cells arranged is sweeping fascicles with contrasting hypercellular and hypocellular areas, producing a marble-like pattern, with atypical wavy, buckled, hyperchromatic nuclei, and brisk mitotic activity. Immunohistochemical analysis showed patchy positive staining for S100 and SOX10, and a complete loss of H3K27me3 expression. This report underscores the challenge of diagnosing secondary malignancies post-radiation therapy and the importance of careful histological examination to differentiate them from other conditions.
Conclusions
In conclusion, radiation-induced secondary malignancies are a significant late side effect of radiation therapy that can profoundly impact treatment decision-making and requires a high index of suspicion during post radiation surveillance. Malignant peripheral nerve sheath tumor serves as a pertinent example, highlighting the importance of considering long-term risks when developing optimal management plans for cancer patients.
Keywords
Introduction
Radiation therapy continues to stand as a fundamental pillar in cancer treatment, improving outcomes and increasing rates of remission. However, alongside these advancements, it also causes serious complications. Long-term complications associated with radiotherapy include the development of secondary malignancies.1,2 The risk of such secondary neoplasm is directly proportional to the administered radiation dose. 3 Radiation-induced secondary malignancies typically occur in 2 peaks: an initial peak within 3 years of radiation exposure, mainly involving hematological malignancies, such as acute leukemias, and a second peak emerging over 10 years post-therapy, primarily involving solid malignancies. 4
The most common solid malignancy associated with radiotherapy comprises malignant fibrous histiocytoma, leiomyosarcoma, osteosarcoma, angiosarcoma, and malignant peripheral nerve sheath tumor (MPNST).5–7 We report a post-radiation intraosseous MPNST in a middle-aged patient with no history of neurofibromatosis.
Case Report
We present a 47-year-old man with a history of squamous cell carcinoma of the tongue, surgically treated 10 years ago. The initial treatment involved surgical resection followed by adjuvant chemotherapy with cisplatin and radiation. The patient later experienced right-sided jaw pain, spontaneous loss of tooth #31, and a 10-pound weight loss. A CT scan revealed an enhancing mass, destroying the right retromolar trigone and extending along with the posterior mandibular body (Figures 1 and 2).
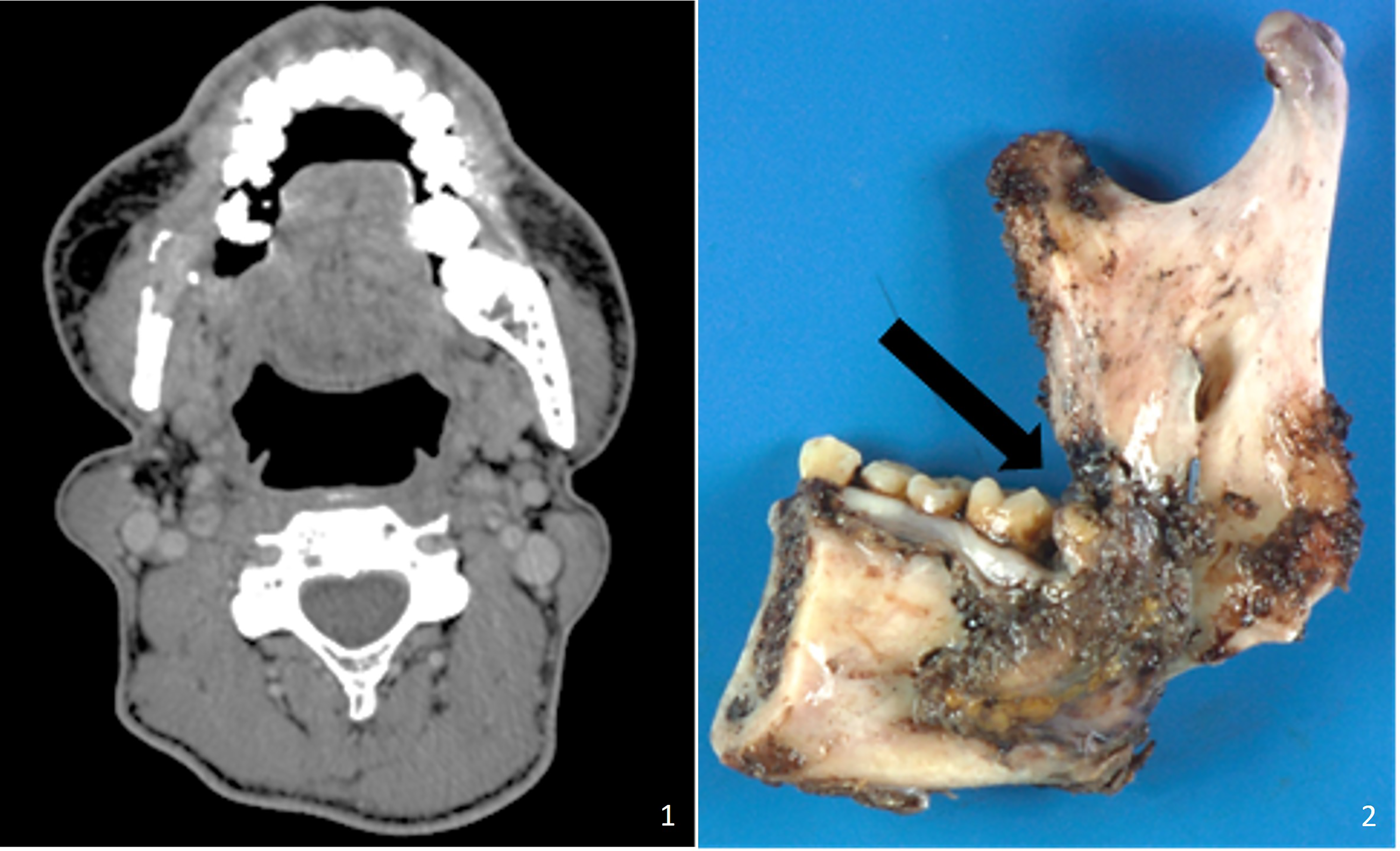
CT scan shows a mass, associated with a pathologic fracture, which destroys the right retromolar trigone, extending along with the posterior mandibular body.
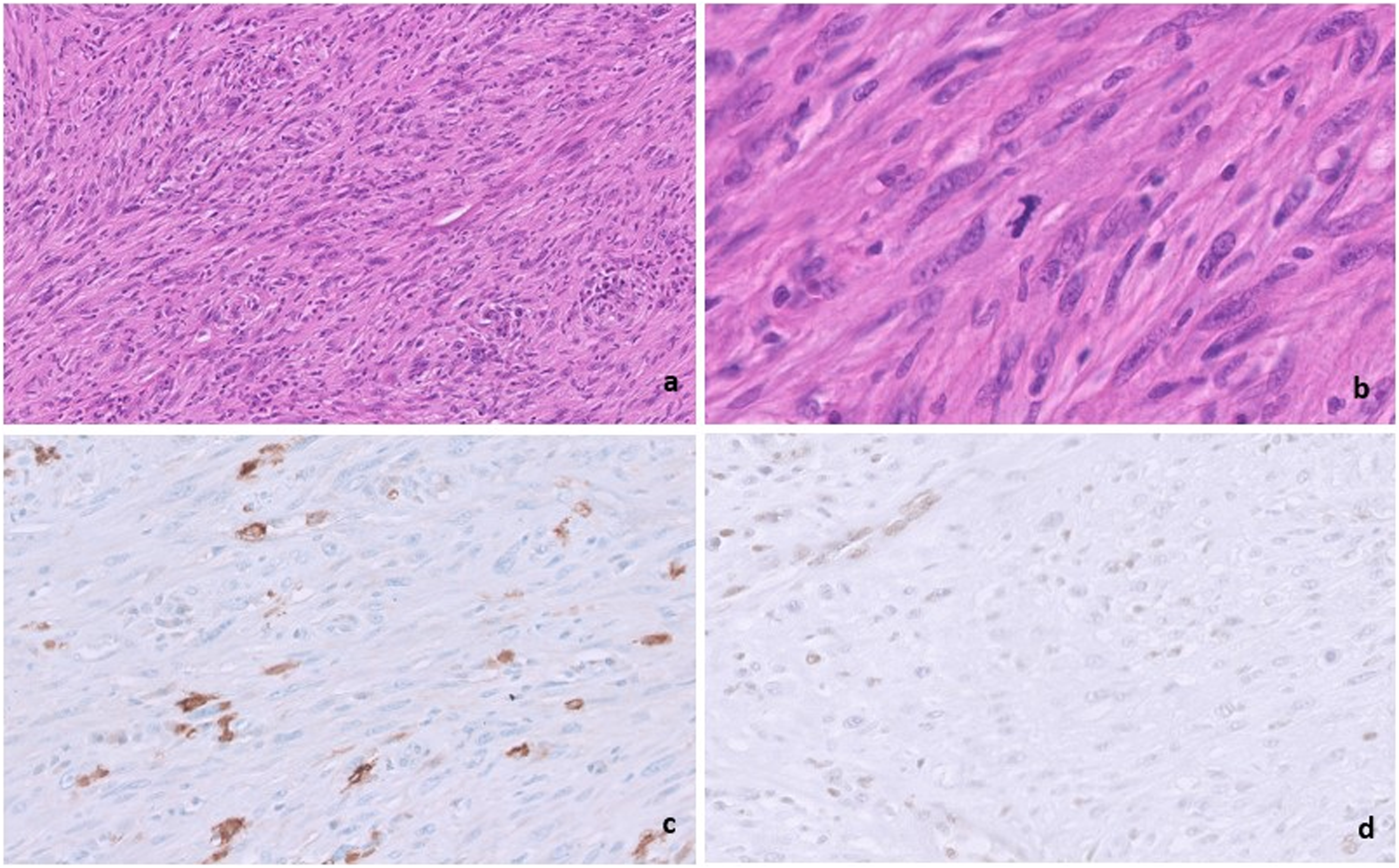
A 4 cm mandibular fracture. (A) Tumor composed of alternating hypercellular and hypocellular areas. (B) Fascicles of spindle cells with mildly pleomorphic hyperchromatic nuclei and pale wavy cytoplasm. Mitotic figures are conspicuous. (C) S100 staining is patchy positive. (D) Staining with H3K27me3 is lost in the tumor cells. Endothelial cells act as internal control.
Pathological Features
A retromolar biopsy exhibited benign squamous mucosa with lymphoplasmacytic infiltration. Subsequently, a right segmental mandibulectomy and reconstruction was performed. Gross examination of the specimen revealed a 4-cm fracture extending from the mandibular angle to the joint. Serial sectioning unveiled a 3.3-cm tan-white, firm mass extending into the fracture, abutting the medial and lateral soft tissues, and invading the roots of the overlying teeth. Histopathological examination revealed sweeping fascicules of spindle cells in a marble-like pattern produced by the alternating density of the hypercellular and hypocellular areas. The cells presented hyperchromatic, wavy, buckled nuclei, and conspicuous mitotic activity. Immunohistochemical analysis revealed patchy positive staining for S100 and SOX10, and a complete loss of H3K27me3 expression was observed (Figure 2A-D).
Discussion
Common solid malignancies associated with radiotherapy comprise malignant fibrous histiocytoma, leiomyosarcoma, osteosarcoma, angiosarcoma, and MPNST.5–7
Malignant peripheral nerve sheath tumor commonly arises in patients with neurofibromatosis and can rarely occur as a complication of radiotherapy, with approximately 10% of all MPNSTs attributed to radiation treatment.2–5,8–13 We report a rare post-radiation intraosseous MPNST in a middle-aged patient, with no history of neurofibromatosis.
At low power, MPNSTs exhibit a marbled appearance due to alternating hypocellular and hypercellular areas with perivascular accentuation. They typically feature uniform spindle cells with hyperchromatic, thin, wavy, or focally buckled nuclei. Some may display uniform cellularity with fibrosarcoma-like fascicular growth, which can lead to a differential diagnosis with synovial sarcoma. Immunohistochemistry often reveals patchy or focal expression of S100 and SOX10. Desmin, myogenin, and MyoD1 may show positivity in MPNST with rhabdomyosarcomatous heterologous components. Notably, the loss of nuclear H3K27me3 in high-grade sporadic and radiation-associated MPNSTs is crucial for arriving at this diagnosis.14,15
Malignant fibrous histiocytoma (more accurately known as undifferentiated pleomorphic sarcoma) usually shows highly pleomorphic tumor cells, including multinucleated tumor giant cells haphazardly scattered in fibrous stroma. There are no distinct areas of storiform growth pattern. 16
Leiomyosarcomas usually show diffuse hypercellularity with spindle cells with plump, blunt-ended nuclei and moderate to abundant, pale to brightly eosinophilic, fibrillary cytoplasm. These cells are set in long intersecting fascicles parallel and perpendicular to the plane of section, showing storiform or palisaded patterns. Moderate to severe nuclear pleomorphism is usually noted. Immunohistochemistry is positive for smooth muscle markers, such as SMA, MSA, desmin, and h-caldesmon. 17
Osteosarcomas are tumors composed of sarcomatous tumor cells that produce malignant bone or osteoid. The tumor cells may have densely eosinophilic cytoplasm resembling osteoblasts and vary in size with nuclear atypia. Immunohistochemistry analysis has limited value in the diagnosis of osteosarcoma. 18
Angiosarcoma has a wide morphologic appearance, ranging from lesions that are cytologically bland and vasoformative, to solid sheets of highly pleomorphic cells. It typically presents as numerous irregularly shaped anastomosing vascular channels lined by atypical endothelial cells with a highly infiltrative architecture. Tumor cells are typically plump, pleomorphic and mitotically active. They can be spindle, polygonal, epithelioid and primitive round, blue cells, forming papillae, or solid nests. Because of the heterogeneous histologic features, the histological identification of an angiosarcoma can be challenging. Angiosarcoma expresses endothelial cell markers, including CD31, CD34, ERG, FLI1, VEGF, and factor VIII. 19
Establishing the diagnosis of MPNST can be difficult due to significant morphological overlap with all these other entities. Besides S100 protein and SOX10, no other easily applicable markers are available that are specific for MPNST. Loss-of-function mutations in EED and SUZ12, which can occur in MPNST after radiotherapy, can serve as a diagnostic tool for identifying this entity.14,15,20 In our patient, the complete loss of H3K27me3 expression was crucial in arriving at the final diagnosis and ruling out other differentials.
Conclusion
In conclusion, while radiation therapy remains an indispensable tool in the fight against cancer, its efficacy comes with a spectrum of potential complications. The possibility of secondary malignancies, though rare, underscores the importance of carefully weighing the benefits against the risks associated with radiation treatment. Vigilance in monitoring for both immediate- and long-term effects is crucial in optimizing patient outcomes and ensuring the continued advancement of cancer care.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
