Abstract
We report a patient in whom a primary high-grade serous carcinoma (HGSC) of the fallopian tube transformed into a carcinosarcoma at the site of peritoneal dissemination, and immunohistological analysis suggested the involvement of an epithelial–mesenchymal transition (EMT). The patient, a 70-year-old woman, had an abdominal mass palpated on admission, and a laparotomy was performed after a close examination. The resected right fallopian tube was cystically dilated, and a solid mass was observed in its lumen. The histological diagnosis was HGSC of the right fallopian tube with a papillary or complex tubular structure composed of tumor cells with marked nuclear irregularities. p53 was overexpressed, and no mesenchymal tumor component was observed. The resected left-sided abdominal mass of the omentum was a solid with a long diameter of 100 mm. Microscopically, the tumor exhibited a mixture of HGSC and high-grade sarcoma with nonspecific differentiation. Furthermore, a heterologous chondrosarcoma was subsequently observed from the high-grade sarcoma. The HGSC component was E-cadherin positive. The high-grade sarcoma component was positive for EMT-related proteins such as zinc finger E-box–binding homeobox 1 (ZEB1) and twist family bHLH transcription factor 1 (TWIST1). The chondrosarcoma component was ZEB1 positive and TWIST1 negative. p53 overexpression was found in all 3 components. The tumor of the omentum suggested that an EMT phenomenon was involved in the tumorigenesis. In this scenario, the primary HGSC of the fallopian tube with obvious invasion demonstrated that the conversion from carcinoma to sarcoma by EMT occurs only with peritoneal dissemination.
Keywords
Introduction
A carcinosarcoma of the fallopian tubes or ovaries is a very rare malignant neoplasm. Carcinosarcomas of the uterine corpus, which occur relatively frequently, are often composed of the carcinomatous components of a high-grade serous carcinoma (HGSC) and a homologous or heterologous sarcomatous component.1,2 The sarcomatous component in a carcinosarcoma of the uterine corpus is thought to be a transdifferentiate carcinomatous component that exhibits a sarcomatous morphology, and a carcinosarcoma of the uterine corpus has been proposed as a model for an epithelial–mesenchymal transition (EMT).1,3 The involvement of an EMT in the tumorigenesis of carcinosarcomas of the fallopian tubes or ovaries, as well as those of the uterine corpus, has been considered but not fully proven. In addition, the mechanisms of carcinosarcoma tumorigenesis remain unclear, including under what conditions carcinomatous components transdifferentiate into sarcomatous components and whether the mechanisms of transdifferentiation are the same in primary tumors and metastases or disseminated tumors. We report an example of a primary HGSC of the fallopian tube that transformed into heterologous carcinosarcoma at the site of peritoneal dissemination, with immunohistochemical evidence of EMT involvement.
Case Presentation
A 70-year-old woman developed a left-side abdominal swelling and recorded a weight loss of 3 kg in one month. She had past histories of hypertension, chronic hepatitis C, and breast fibroadenoma. There was no relevant family history. A soft mass with slight poor mobility was touching the left side of the abdomen, and no pain was noted on palpation. Computed tomography showed an irregularly shaped, heterogeneously contrasted 80 × 80 × 50 mm mass at the pelvic inlet. It was a hypervascularized mass with prominent surrounding vascular structures in the arterial phase. Tumor markers were within the normal range. Laparoscopy revealed a 110 × 80 × 50 mm omental mass on the left side of the abdomen, 2 nodules in the gastric wall and omentum, and a swelling of the right fallopian tube; therefore, the patient underwent a laparotomy. The right ovary, left adnexa, and uterus were grossly normal. A left-sided omental mass, 2 nodules in the gastric wall and omentum, and the right fallopian tube were resected.
The resected specimens were originally prepared from 10% buffered formalin-fixed, paraffin-embedded tissue according to our routine hospital procedure. A histopathological examination was performed using hematoxylin and eosin staining. Immunohistochemistry was performed using an autoimmunostainer (Leica BOND–III system: Leica Biosystems). The antigens used are listed in Table 1.
Antigens Used for Immunohistochemical Study.

Abbreviations: WT1, Wilms tumor-1; ZEB1, zinc finger E-box–binding homeobox 1; TWIST1, twist family bHLH transcription factor 1.
Pathological Findings
The right fallopian tube was cystically dilated to 60 × 50 × 10 mm (Figure 1A). Homogenous solid tumors were observed in the lumen of the fallopian tube, histologically showing an adenocarcinoma with papillary and complex ductal growth (Figure 1B). The tumor cells with marked nuclear irregularities infiltrated the stroma and formed labyrinthine architectures with slit-like spaces (Figure 1C). There were no morphological findings of a solid, endometrioid-like, and transitional pattern. Immunohistochemically, tumor cells exhibited diffuse positivity for Wilms tumor-1 (WT1) (Figure 1D) and an aberrant p53 pattern with strong diffuse staining (Figure 1E). E-cadherin was diffusely positive on the cytoplasmic membrane of the tumor cells, supporting the diagnosis of an epithelial tumor (Figure 1F). We diagnosed an HGSC of the fallopian tube. The morphological features of the epithelial tumor with cell-to-cell adhesion were clearly observed in all of the split specimens, and no sarcoma component was observed (Supplemental Figure).
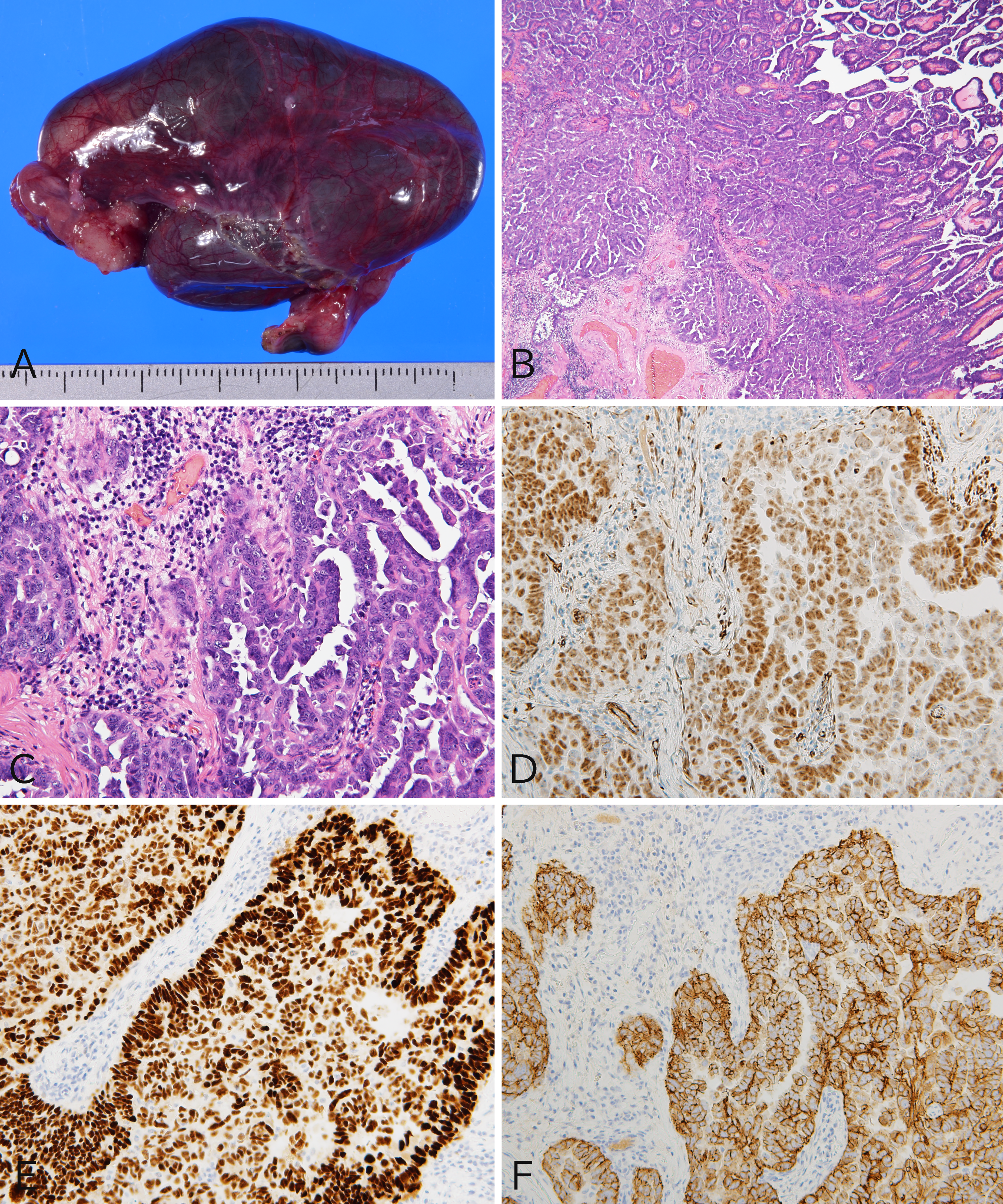
Pathological findings for high-grade serous carcinoma (HGSC) of the right fallopian tube. (A) The fallopian tube was cystically dilated. (B) Histologically, the tumor in the fallopian tube was an invasive adenocarcinoma with papillary and ductal growth. (C) The tumor glands showed a complex structure with slit-like lumens, and the proliferating tumor cells had a conspicuous nuclear irregularity. Tumor cells were positive for Wilms tumor-1 (D) and p53 overexpression (E), supporting the diagnosis of an HGSC. (F) E-cadherin was positive in all tumor cells. B and C, hematoxylin-eosin staining; B, ×40; C-F, ×200.
The left-sided abdominal tumor in the omentum was a solid, elastic-soft mass with a long diameter of 100 mm. The cut surface of the tumor was lobulated, opalescent, and hemorrhagic (Figure 2A). Histologically, the tumor was observed to have carcinomatous and sarcomatous components (Figure 2B). The carcinomatous component was an HGSC with complex ductal structures (Figure 2C). The sarcomatous component was observed to have a homologous element consisting of a high-grade sarcoma with nonspecific differentiation (Figure 2D) and a heterologous element of a chondrosarcoma. There was a continuous transition from the HGSC to a high-grade sarcoma (Figure 2E) and from the high-grade sarcoma to a chondrosarcoma (Figure 2F), although there was no continuity between the HGSC component and the chondrosarcoma. An immunohistochemical analysis was performed on the 3 components (Figure 3A). WT1 was positive only in the HGSC component (Figure 3B), and aberrant p53 expression with strong diffuse staining was observed in all components of the HGSC, high-grade sarcoma, and chondrosarcoma (Figure 3C). E-cadherin and the EMT-related transcription factors, twist family bHLH transcription factor 1 (TWIST1) and zinc finger E-box–binding homeobox 1 (ZEB1), were also examined. E-cadherin was positive only in the HGSC component (Figure 3D). Twist family bHLH transcription factor 1 was positive only in the high-grade sarcoma component (Figure 3E), and ZEB1 was positive in 2 components of high-grade sarcoma and chondrosarcoma (Figure 3F), as shown in Table 2. The tumor of the omentum was diagnosed as a carcinosarcoma with a heterologous component. No tumors were found histologically in the uterine cervix or endometrium. On the basis of the surgical and pathological findings, we concluded that the HGSC of the fallopian tube transformed into a carcinosarcoma with a heterologous chondrosarcoma at the site of peritoneal dissemination.
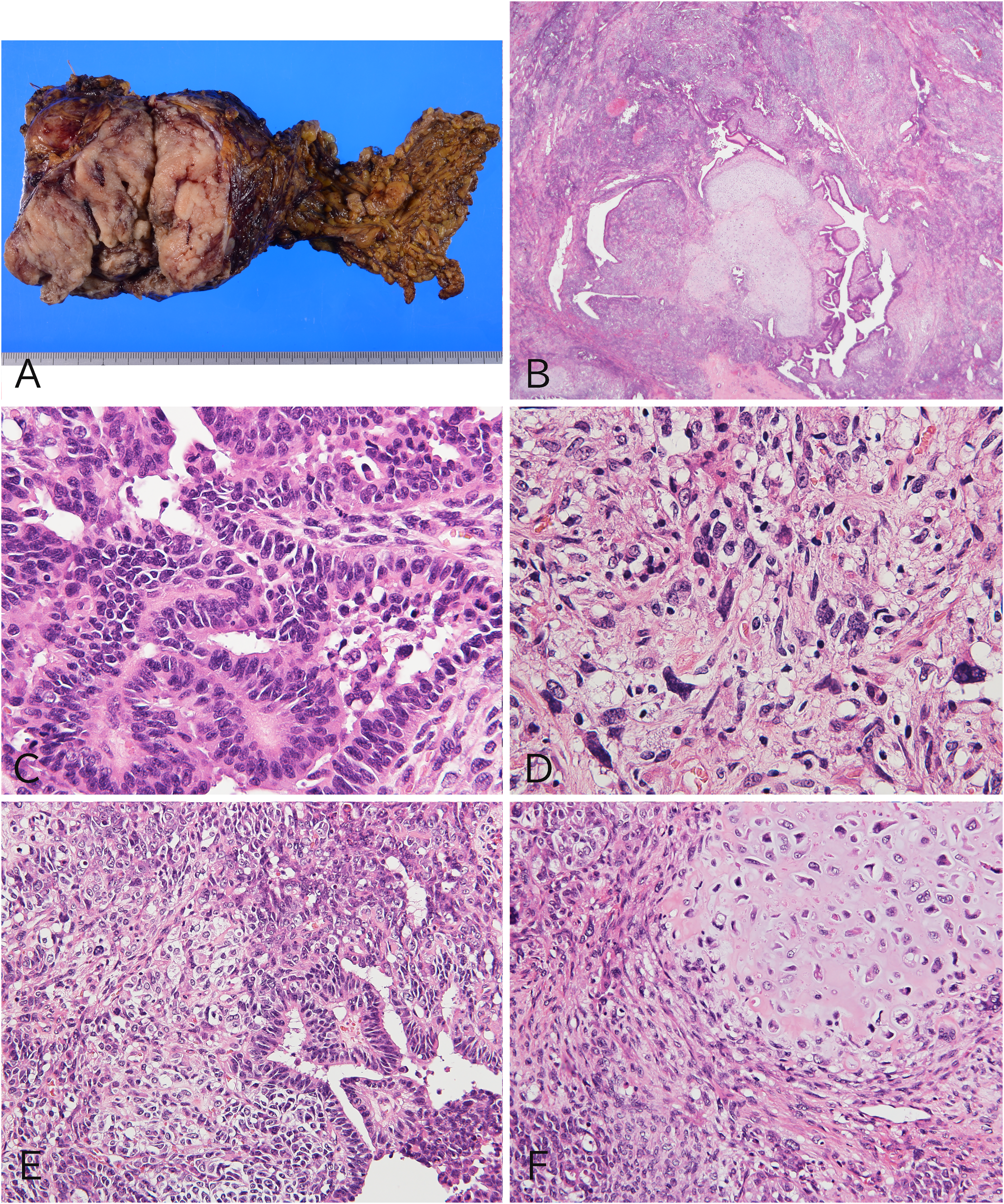
Pathological findings of carcinosarcoma of the omentum. (A) A grayish-white, solid, and elastic-soft mass was observed in the omentum. (B) An epithelial component forming a complex glandular architectural feature and a solid mesenchymal component that were proliferating. (C) The epithelial component was a high-grade serous carcinoma (HGSC). (D) The homologous mesenchymal component was a high-grade sarcoma. (E) The HGSC component had transitioned to a high-grade sarcoma. (F) The high-grade sarcoma component had transformed into a heterologous chondrosarcoma. B-F, hematoxylin-eosin staining; B, ×20; C and D, ×400; E and F, ×200.
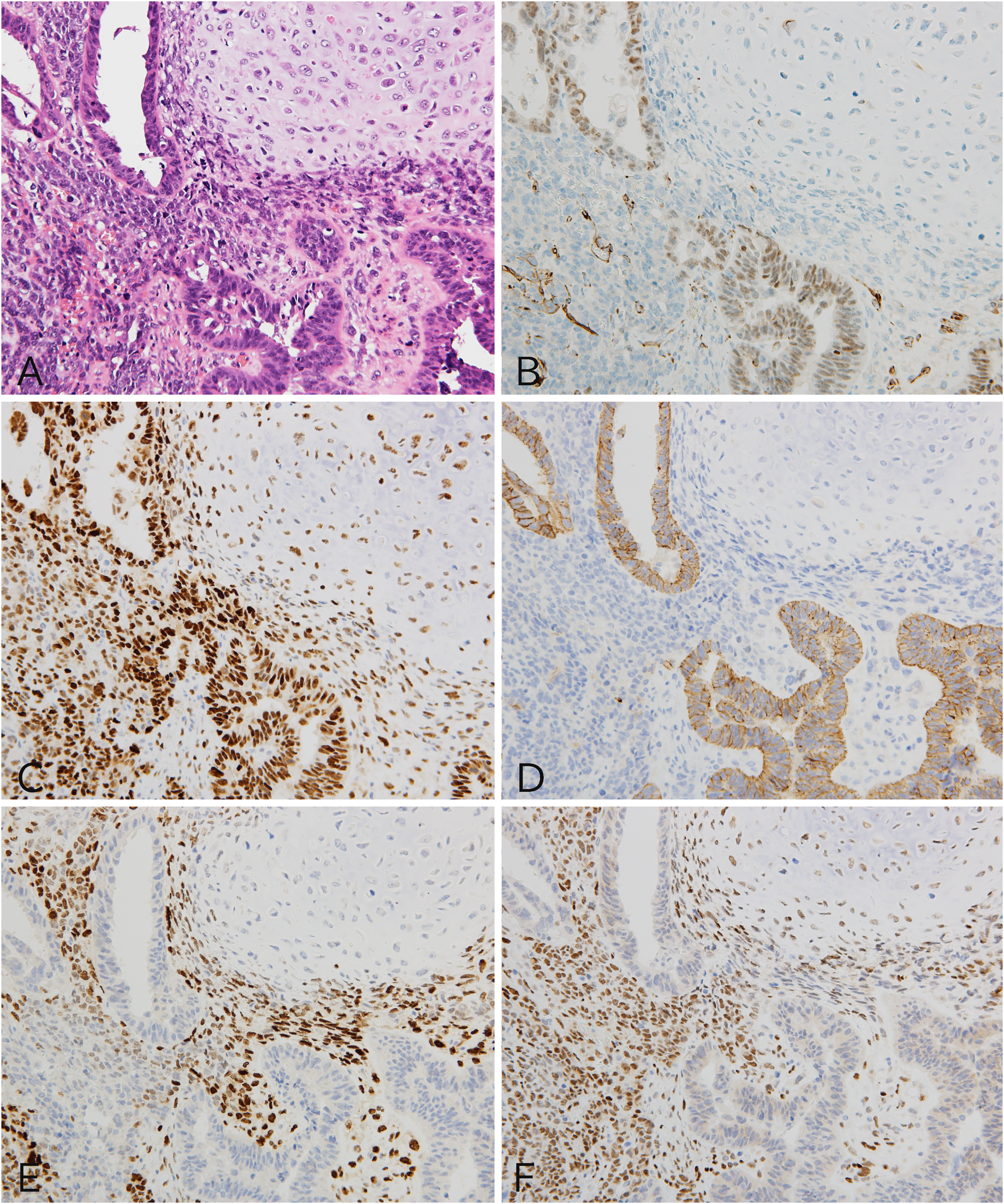
Immunohistochemical characteristics of the 3 components. (A) Transitional areas of a high-grade serous carcinoma (HGSC), a high-grade sarcoma, and a chondrosarcoma were observed (hematoxylin-eosin staining). (B) Wilms tumor-1 was positive in one HGSC component. (C) p53 overexpression was observed in all 3 components. (D) E-cadherin was positive for the HGSC component. (E) Twist family bHLH transcription factor 1 was immunostained for the high-grade sarcoma component. (F) Zinc finger E-box–binding homeobox 1 was positive for the high-grade sarcoma component and chondrosarcoma component. A-F, ×200.
Comparison of Immunohistochemical Findings in 3 Morphological Components.
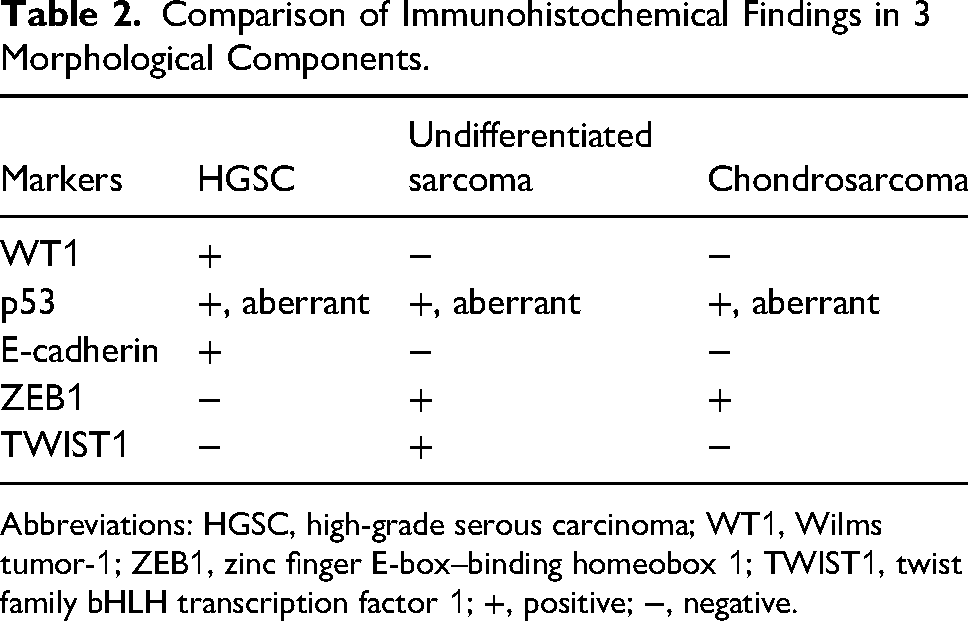
Abbreviations: HGSC, high-grade serous carcinoma; WT1, Wilms tumor-1; ZEB1, zinc finger E-box–binding homeobox 1; TWIST1, twist family bHLH transcription factor 1; +, positive; −, negative.
The patient was diagnosed with stage IIIC fallopian tube cancer and underwent chemotherapy with 6 courses of paclitaxel–carboplatin (TC), followed by interval debulking surgery, namely a total abdominal hysterectomy, a left salpingo-oophorectomy, lymph node dissection, and a pelvic peritonectomy. Not all specimens were sampled and retrieved, but histological findings for HGSCs were observed at several sampled sites. After adjuvant chemotherapy with 2 courses of TC, niraparib maintenance monotherapy was administered. The patient has remained alive for 2 years and 3 months without recurrence.
Discussion
Recently, ovarian HGSCs have been thought to be derived from a serous tubal intraepithelial carcinoma (STIC) of the fallopian tube epithelium. 4 Ardighieri et al reported that in 8 of 10 occurrences of the simultaneous presence of a pelvic extrauterine carcinosarcoma and a fallopian tube STIC, the same TP53 gene mutation was found in both. 5 Based on the similarity in morphology and p53 staining of the HGSC components, this tumor was interpreted as a dissemination of the fallopian tube HGSC. Interestingly, only the peritoneal dissemination was transformed into a carcinosarcoma in this patient, even though there was an obvious invasive carcinoma in the fallopian tube. There are no reports of such a phenomenon other than that by Nakashima et al in which the fallopian tube carcinoma was microinvasive. 6 Carcinosarcomas are known to exhibit different biological behaviors in different organs. 7 This report suggests that the tumor microenvironments, including organ differences, might influence a tumorigenesis in the transformation from a carcinoma to a sarcoma. Additional tumors must be accumulated to elucidate the diversity in tumorigenesis of carcinosarcomas.
In the present patient, a serous carcinoma disseminated in the peritoneum was considered to have transformed into a high-grade sarcoma, and the high-grade sarcoma into a chondrosarcoma, based on the overexpression of p53 in the 3 components and their morphological continuity. In addition, the expression status of E-cadherin and EMT-related transcription factors, ZEB1 and TWIST1, was clarified by immunohistochemistry. Epithelial–mesenchymal transition is induced by the activations of certain pathways such as transforming growth factor β, which promotes the EMT-related transcription factors, tyrosine kinase receptors, and Wnt. 8 Zinc finger E-box–binding homeobox 1 binds to E-box regulatory genes involved in the promoter region of E-cadherin and promotes the EMT process by repressing E-cadherin expression. 9 Twist family bHLH transcription factor 1 also cooperates with SNAI1 to induce ZEB1 expression. 10 The EMTs of the fallopian tube and peritoneal carcinosarcoma are little known, but it has been suggested that ZEB1 gene expression plays a critical role in the suppression of EMT in a study of uterine and ovarian carcinosarcomas.8,11 In this study, immunohistochemistry revealed that E-cadherin may be regulated by ZEB1 and TWIST1 during the process of transforming from an HGSC component to a high-grade sarcoma component.
Heterologous carcinosarcomas accounted for 41.2% of carcinosarcomas with high-grade carcinoma components in the uterine corpus and heterologous carcinosarcomas were associated with poorer progression-free survival and cause-specific overall survival than homologous carcinosarcomas. 12 However, the formation mechanism of the heterologous component is not clear, and moreover, it is not known how EMT-related transcription factors such as ZEB1 and TWIST1 are involved. At least, it suggested that TWIST1 was not involved in the chondrosarcoma formation mechanism in this tumor. On the other hand, it was reported that the expression of SIRT1, which enhances the expression of TWIST1, promotes the metastasis of a soft tissue chondrosarcoma by inducing EMT, and that was a poor prognostic factor for chondrosarcoma. 13 Epithelial–mesenchymal transition–related transcription factors were involved in promoting therapy resistance and tumor aggressiveness in both carcinoma and lymphoma, leukemia, and sarcoma. 14 Immunohistological TWIST1 expression is reportedly a poor prognostic factor in patients with osteosarcoma. 15 Although this has not been proven in chondrosarcoma, the TWIST1-negative chondrosarcoma component may indicate a good prognostic biological behavior among chondrosarcomas.
In summary, HGSC of the fallopian tube presented with a heterologous carcinosarcoma with a chondrosarcoma at the site of the peritoneal dissemination. Using morphologic and immunohistochemical findings as a basis, we hypothesized that a carcinosarcoma was a series of tumors that changed into an HGSC, a high-grade sarcoma, and a chondrosarcoma. The sarcoma component expressed TWIST1 and ZEB1, demonstrating the involvement of EMT in the carcinosarcoma tumorigenesis.
Supplemental Material
sj-docx-1-ijs-10.1177_10668969241271963 - Supplemental material for A Case Study of a High-Grade Serous Carcinoma of the Fallopian Tube Transformed into Carcinosarcoma at the Site of Peritoneal Dissemination With Immunohistological Evidence of an Epithelial–Mesenchymal Transition
Supplemental material, sj-docx-1-ijs-10.1177_10668969241271963 for A Case Study of a High-Grade Serous Carcinoma of the Fallopian Tube Transformed into Carcinosarcoma at the Site of Peritoneal Dissemination With Immunohistological Evidence of an Epithelial–Mesenchymal Transition by Naoko Taninaka, Kazuyuki Ishida, Atsuko Takada-Owada, Shuhei Noda, Masato Onozaki, Hadzki Matsuda, Yuko Kaneko, Akira Mitsuhashi and Akihiko Toyoda in International Journal of Surgical Pathology
Footnotes
Acknowledgments
The authors gratefully acknowledge the technical assistance of Chiaki Satoh and Ayako Shimizu of the Department of Diagnostic Pathology, Dokkyo Medical University. The authors are also grateful to Mihoko Ishikawa and other laboratory technicians of the Department of Pathology, Dokkyo Medical University Hospital for their support.
Author Contributions
Naoko Taninaka and Kazuyuki Ishida were responsible for the acquisition and interpretation of patient data and manuscript preparation. Atsuko Takada-Owada, Yuko Kaneko, and Akira Mitsuhashi critically revised the manuscript. Shuhei Noda, Masato Onozaki, Hadzki Matsuda, and Akihiko Toyoda performed histological diagnoses and immunohistochemical analyses. All the authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Informed consent was obtained from all participants and/or their legal guardians.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Trial Registration
This is case report of Pathology.
Supplemental Material
Supplemental material for this article is available online.
