Abstract
The prognostic role of tumor cells in pancreatic ductal adenocarcinoma (PDAC) of the pancreatic head with direct microscopic infiltration (DMI) or in close proximity (≤1 mm) to the resection margin (RM) remains unclear. This single-center, retrospective study included specimens from 75 patients who underwent oncological resection of pancreatic head PDAC between February 2013 and July 2020. Two pathologists independently re-measured the distance between tumors and the multiple RMs. The impact of RM involvement for DMI, tumor cells within ≤1 mm, in general, and for individual RMs on overall survival (OS) and development of distant pulmonary (PM) and hepatic (HM) metastasis was analyzed. DMI of RMs was significantly associated with a shorter OS (median 5 vs 19 months, P = .02). The presence of tumor cells within ≤1 mm of RMs yielded a negative impact on OS with a trend toward significance (median 9 vs 21 months, P = .09). DMI and tumor cells within ≤1 mm of the pancreatic transection margin (PRM), individually, had a significant negative impact on OS (median 4 vs 19 months and 6 vs 19 months, P < .05), but not for any other individual RM. RM involvement of ≤1 mm of only the vascular circumferential resection margin (VCRM) resulted in a shorter time to HM development (P = 0.05). DMI of the posterior circumferential resection margin (PCRM) and VCRM, individually, showed shorter time to PM (P < .05). Potential clinical considerations include extended intraoperative evaluation of the PRM (1 mm) and intensified preoperative prediction of R1 resection as a basis for neoadjuvant therapy.
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the deadliest malignancies, with an overall 5-year survival rate of only 9%, resulting in approximately 466 000 related deaths worldwide each year.1,2 It is predicted that pancreatic cancer will become the second most frequent cause of cancer-related death in the United States by 2040. 3 Currently, oncologic resection is the only curative therapeutic approach. However, at the time of diagnosis, only about 20% of patients are eligible for oncologic resection. Even after tumor resection, overall 5-year survival rates remain as low as 20% to 27%.4–6 Therefore, precise pre-operative and postoperative therapeutic decision making is crucial under such unfavorable prognostic circumstances.
The histopathological examination of the surgical specimen from oncologic resection is substantial in determining postoperative prognosis. It is well established that residual disease in the form of direct microscopic infiltration (DMI) of a resection margin (RM) has poor prognostic influence. 7 However, is not clear what role tumor cells play when they are not directly infiltrating an RM but are in close proximity of it (≤1 mm) or further away (>1 mm). Furthermore, it is necessary to determine the impact of tumor cells infiltrating and being in proximity to different RMs. 7 During oncologic resection of the pancreatic head the surgical specimen has circumferential resection margins (CRMs): anterior (ACRM), posterior (PCRM), and vascular (VCRM), as well as a pancreatic transection margin (PRM). In resectable PDAC located in the pancreatic head, tumor involvement of an RM is almost exclusively located within the circumference of the PRM and the CRMs, whereas tumor involvement in the bile duct, gastric or duodenal, and jejunal transection margins is rare. 8
The purpose of this study is to incrementally analyze the impact of the distance and location of RMs on the survival and development of distant metastasis in PDAC of the pancreatic head.
Material and Methods
Patient Cohort
This retrospective single-center study included consecutive patients who underwent curative oncologic resection of the pancreatic head between February 2013 and July 2020 at the Department of Surgery, Campus Benjamin Franklin, Charité—Universitätsmedizin Berlin, and had histologically confirmed PDAC. Exclusion criteria were metastatic disease at the time of surgery (n = 4), retrospective nondeterminability of all RMs (n = 2), death within 30 days postoperatively (n = 6), and loss to follow up due to the patient residing outside of Germany (n = 1). After applying these exclusion criteria, a total of 75 patients (100%) were included in the study (Table 1). Overall survival (OS) was followed up for all patients until death or in censored cases for a median time of 63.5 months (range 39–109 months, n = 16, 21%). The development of distant hepatic and pulmonary metastasis was followed up for 54 patients (72%) either until metastasis or for a median time of 16.0 months (range 2–55 months).
Demographic and Pathological Characteristics of the Patient Cohort.

Abbreviations: G, grading; pL, lymphovascular invasion; pV: vascular invasion; Pn, perineural invasion.
This study was approved by the ethics committee of the Charité—Universitätsmedizin Berlin (No. EA4/020/19), and written informed consent was waived due to the retrospective study design.
Surgery
All patients included in this study underwent curative oncologic resection of the pancreatic head, either through pylorus-preserving pancreaticoduodenectomy (n = 48, 64%), Whipple procedure (n = 25, 33%), or total pancreatectomy (n = 2, 3%) as recommended by the interdisciplinary tumor board. Two patients (3%) had undergone neoadjuvant chemotherapy prior to tumor resection.
Sample Gross Examination and Macroscopic Examination
The gross examination of all specimens from oncologic resection was performed in a standardized fashion, as recently described and demonstrated in Figure 1A and B. 9 The specimens were fixed in 10% buffered formalin before gross examination. The anatomical elements (pylorus, duodenum, pancreas, bile duct) were measured, and the macroscopic appearance of the specimen was described (Figure 1C). The specimens were then marked using tissue dyes. Briefly, the ACRM of the pancreas was marked yellow (Figure 1D), the VCRM blue (Figure 1E), the PCRM black (Figure 1F), the PRM red, and the bile duct lumen until the major duodenal papilla was marked green.
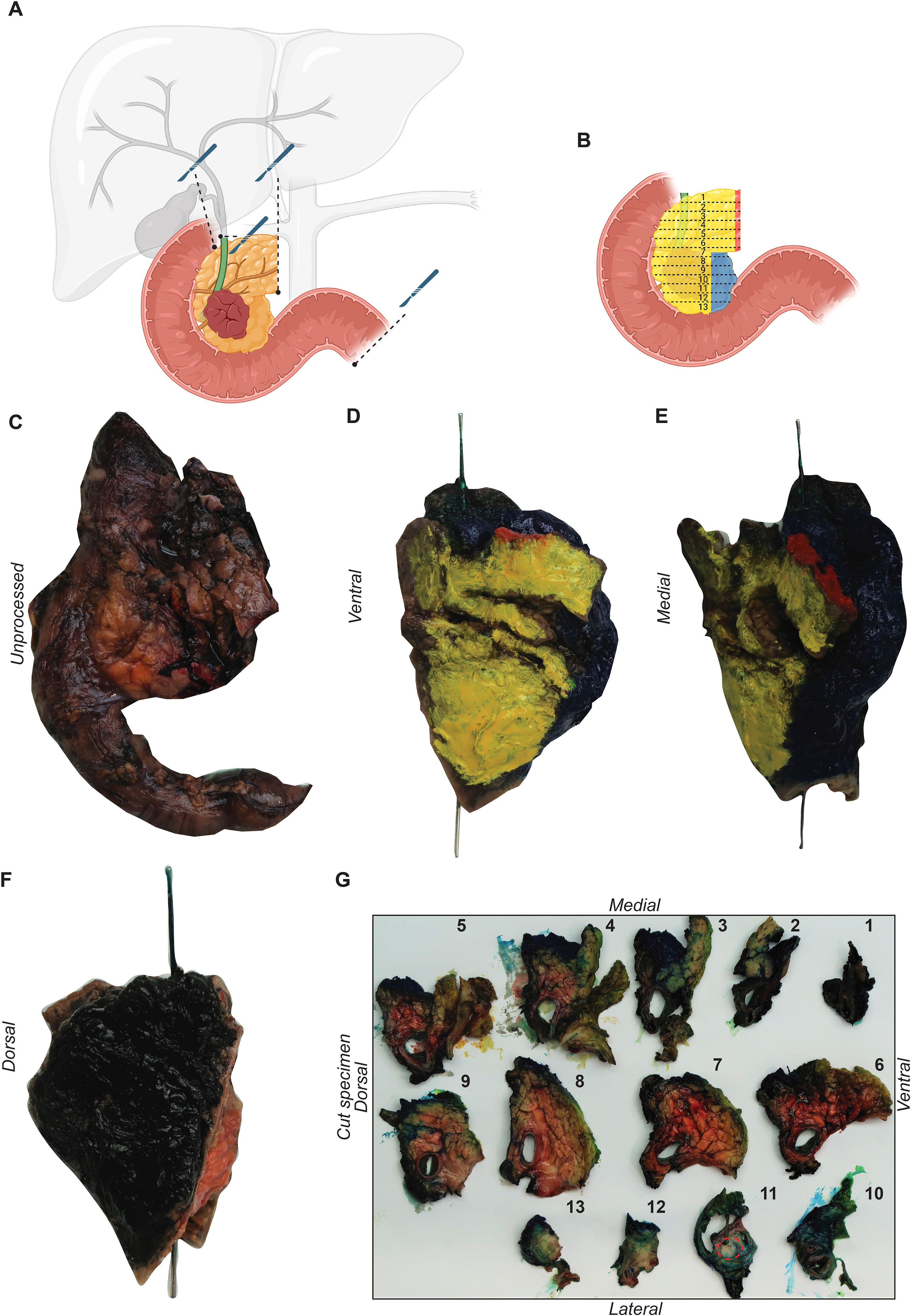
Tumor gross examination of a specimen obtained from oncological resection of the pancreatic head. (A) Depiction of the transection margins: pancreatic transection margin, distal bile duct, gastric/duodenal, and jejunal transection margin. (B) Graphical representation of the axial sectioning of the specimen from cranial to caudal. An unprocessed specimen of the pancreatic head, (C) and the same specimen stained according to our protocols: anterior—yellow (D) vascular margin—blue (E), posterior—black (F). (G) The specimen sectioned from cranial to caudal, with the tumor located in the uncinate process.
Next, the 4 transection margins (gastric/oral duodenal, aboral jejunal, pancreatic, and bile duct transection margin) were each embedded in 4 separate tissue-embedding cassettes. The remaining specimen was sliced in 5 mm sections from cranial to caudal perpendicular to the duodenal lumen (Figure 1G). Afterwards, a precise inspection of the slices was conducted to locate, describe, and measure the tumor. At this point, the macroscopic minimal distance of the tumor to the CRMs (ACRM, VCRM, and PCRM) and the PRM was determined and measured. The cranial and caudal ends of the specimen were further sliced and completely embedded. Next, the proximity of the tumors to the CRMs and the PRM were prepared and embedded. A minimum of 4 cassettes were prepared, 1 for each margin. Finally, all peripancreatic lymph nodes were detected and further embedded. An average of 20 tissue-embedding cassettes was prepared for each specimen. The embedded tissue blocks were cut into 2μm thick sections, and stained with hematoxylin and eosin (H&E), and one tissue block containing tumor tissue was stained with periodic acid-Schiff (PAS). Standard histological examination was performed according to the eighth edition of the TNM classification (AJCC) and included: T-stage, N-stage, grading (G1–3), lymphovascular (pL) and vascular invasion (pV), perineural invasion (Pn) and resection status. During routine diagnosis at our institution we considered a margin positive only if tumor cells were present in the colored surface of the specimen. Macroscopic residual disease (R2) was not present in any of the patients included in this study.
Microscopic Examination and R-Status Evaluation
For the purpose of this study, all included specimens were re-evaluated. For the re-evaluation of the RM status and the minimal distance between tumors to CRMs and PRM, all slides were retrieved from the archive of the Institute of Pathology, Charité—Universitätsmedizin Berlin. Two pathologists (MPD and SS) independently remeasured the distance between the tumor and the respective margins in 1 mm increments. In case of discrepancies these were further discussed with a senior pathologist from the Institute of Pathology, Charité—Universitätsmedizin Berlin. In cases of re-resections of the PRM, the specimens from the re-resection were used. None of the specimens included in this study showed tumor cells in the pyloric/duodenal, jejunal, or bile duct transection margins. In 2 specimens (2.7%), the ACRM, VCRM, and PCRM could not be re-evaluated and information regarding the DMI of these margins was retrieved from the original histopathology reports. Beyond DMI, these margins were excluded from further analysis within this study. When reported or referred to as results of this study, in a summarizing manner the PCRM, VCRM, ACRM, and PRM will be referred to as RMs for convenience.
Statistics
We compared the impact of different criteria of RM involvement on OS, as well as the development of distant hepatic and pulmonary metastases using Kaplan-Meier curves. The curves were compared using the log-rank test, and 95% confidence intervals (CI) were calculated for medians of OS. If there was a significant difference in survival analysis, we analyzed the association of the respective criterion of RM involvement with demographic and histopathological characteristics using the χ2 test. A P value of ≤.05 was considered statistically significant, while P values between .05 and .10 were considered a trend toward significance. Statistical analyses were performed using SPSS version 29.0 (IBM Corp. Released 2022. IBM SPSS Statistics for Windows, Version 29.0. Armonk, NY, USA: IBM Corp.).
Results
Demographic and Pathological Characteristics of Patient Cohort
The demographic and pathological characteristics of the patient cohort included in this study are presented in Table 1. The median OS of the entire patient cohort was 18 months (range 1–109, CI: 12.1–23.9). One-year, 3-year, and 5-year survival rates for the entire patient cohort were 61%, 27%, and 21%, respectively.
Distribution of Involved Resection Margins
Table 2 summarizes the specimens with DMI and presence of tumor cells within ≤1mm of an RM in terms of the total number of specimens as well as the involved RMs. Ten patients (13.3%) had tumors with DMI of at least 1 RM (Figure 2A-C). Of these 10 tumors, 8 (80.0%) showed DMI of only 1 RM (PCRM, n = 2, 20.0%; VCRM, n = 2, 20.0%; PRM, n = 4, 40.0%) and 2 tumors (20.0%) showed DMI of 2 RMs (PCRM & VCRM and PRM & VCRM).
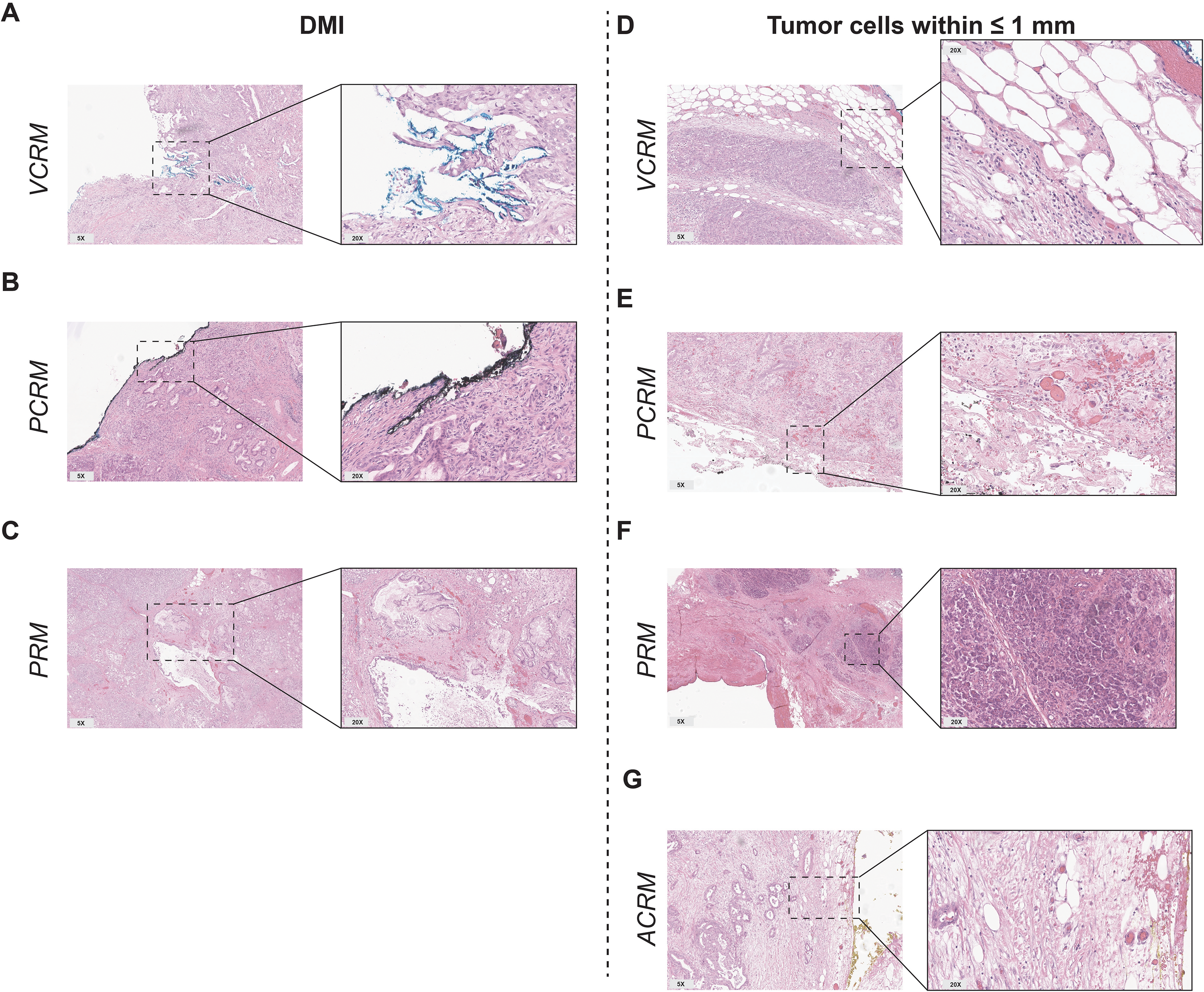
Microscopic examination of the resection margins. Representative H&E images showing tumor cells with DMI into the VCRM (A), PCRM (B), and PRM (C), and the presence of tumor cells within ≤1mm of the VCRM (D), PCRM (E), PRM (F), and ACRM (G). Abbreviations: ACRM, anterior circumferential resection margin; DMI, direct microscopic infiltration; PCRM, posterior circumferential resection margin; PRM, pancreatic transection margin; VCRM, vascular circumferential resection margin.
Distribution of Tumors With Direct Microscopic Infiltration and the Presence of Tumor Cells Within ≤ 1mm of the Specimen's Circumference.
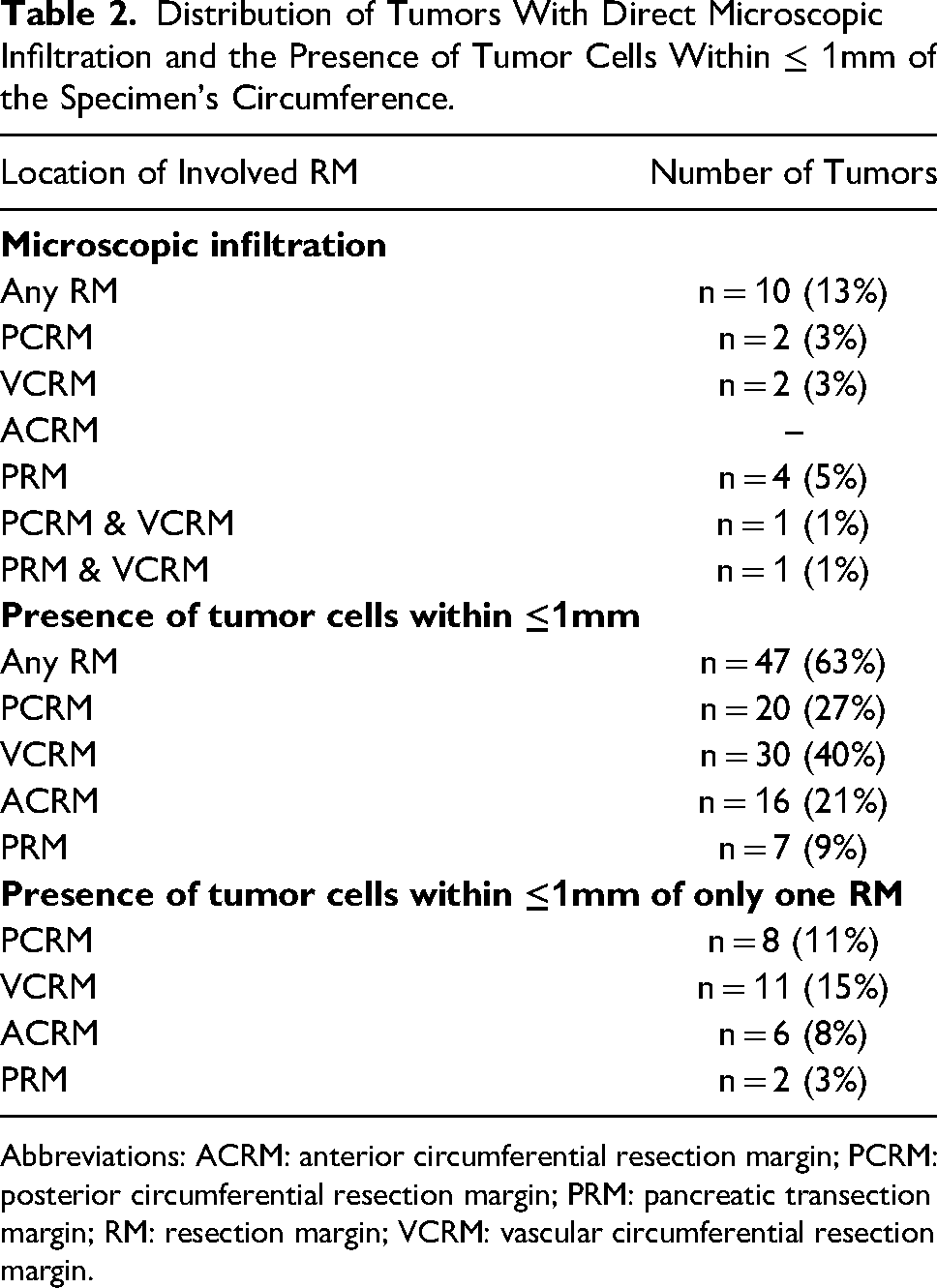
Abbreviations: ACRM: anterior circumferential resection margin; PCRM: posterior circumferential resection margin; PRM: pancreatic transection margin; RM: resection margin; VCRM: vascular circumferential resection margin.
Tumors in 47 patients (62.7%) showed the presence of tumor cells within ≤1mm of at least 1 RM (Figure 2D-G). Tumors in 27 patients (36.0%) showed the presence of tumor cells within ≤1mm of only 1 RM.
Table 3 summarizes the specimens that only showed the presence of tumor cells in >1 mm from any RM (n = 26, 34.7%). For the incremental analysis of the closest distance of tumor cells to any of the RMs (eg, specimens in which the distance of the tumor cells that showed the closest presence to any RM was at least >1mm but within 2 mm of the respective RM) subgroups were created in steps of 1 mm.
Subgroups in Steps of 1 mm for Incremental Analysis of Closest Distance to an RM.

Abbreviation: RM, resection margin.
Influence of Margin Involvement on Overall Survival
Influence of Direct Microscopic Infiltration of Resection Margins on Overall Survival
Survival analysis showed that DMI of an RM was significantly associated with a shorter OS (P = .02). Median OS in the group of patients with DMI of an RM was 5 months (CI: 1.9–8.1) versus 19 months (CI: 14.1–23.9) in the group of patients without DMI of an RM (Figure 3). DMI of an RM was significantly associated with male gender (P = .02). No significant association was found between DMI of a, RM and age (>65 years), advanced T stage (pT3/4), nodal metastasis (pN+), lymphovascular invasion (pL1), perineural invasion (Pn1), vascular invasion (pV1), or higher grade (G3) (P > .05, for all associations).
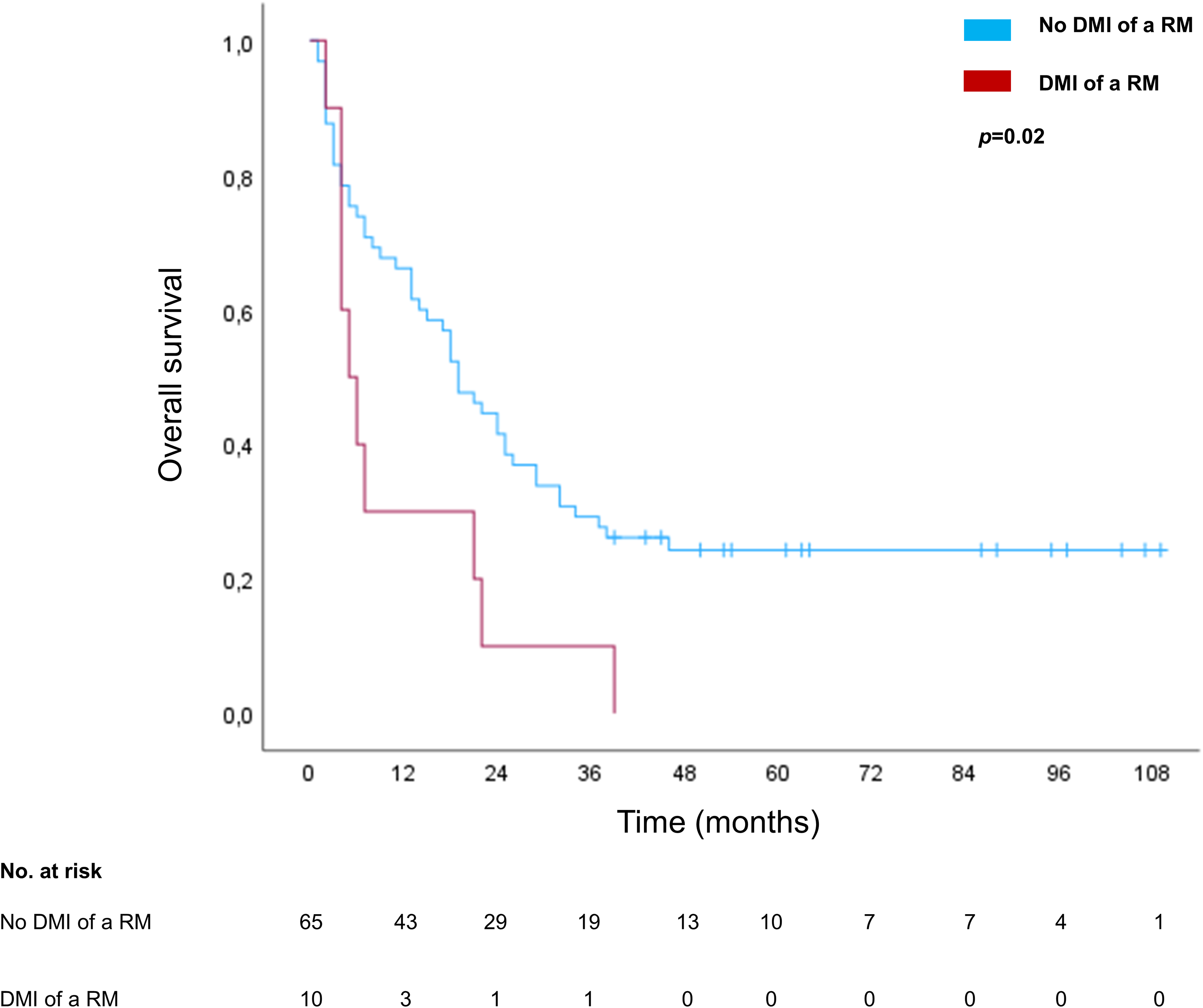
Survival analysis for DMI of an RM. The blue line represents the survival curve in the absence of DMI and the red line represents the survival curve in the presence of DMI of an RM. Tick marks indicate censoring. Abbreviations: DMI, direct microscopic infiltration; RM, resection margin.
To further distinguish the effect of DMI on OS, subgroup survival analysis was performed with respect to the location of DMI for each RM, individually. In this individual subgroup analysis, DMI of the PRM yielded a significantly shorter OS (median 4 months [CI: 1.9–6.1] versus 19 months [CI: 14.5–23.5], P = .02). For the individual DMI of other RMs (PCRM and VCRM), this subgroup survival analysis did not yield significance (P = .86 and .08, respectively). No significant association of PRM DMI with other demographic or histopathological characteristics was observed (gender, age >65 years, pT3/4, pN+, pL1, Pn1, pV1, G3; p > 0.05).
Influence of Presence of Tumors Cells Within ≤1 mm of Resection Margins on Overall Survival
Moreover, survival analyses were performed to investigate the influence of the presence of tumor cells within ≤1 mm of any RM on OS, as well as the presence of tumor cells within ≤1 mm of a specific RM, individually (Figure 4). The analysis regarding tumor cells within ≤1 mm of any RM showed a negative impact on OS with a trend toward significance (median OS 9 months [CI: 0.0–19.1] versus 21 months [CI: 13.5–28.5], P = .09).
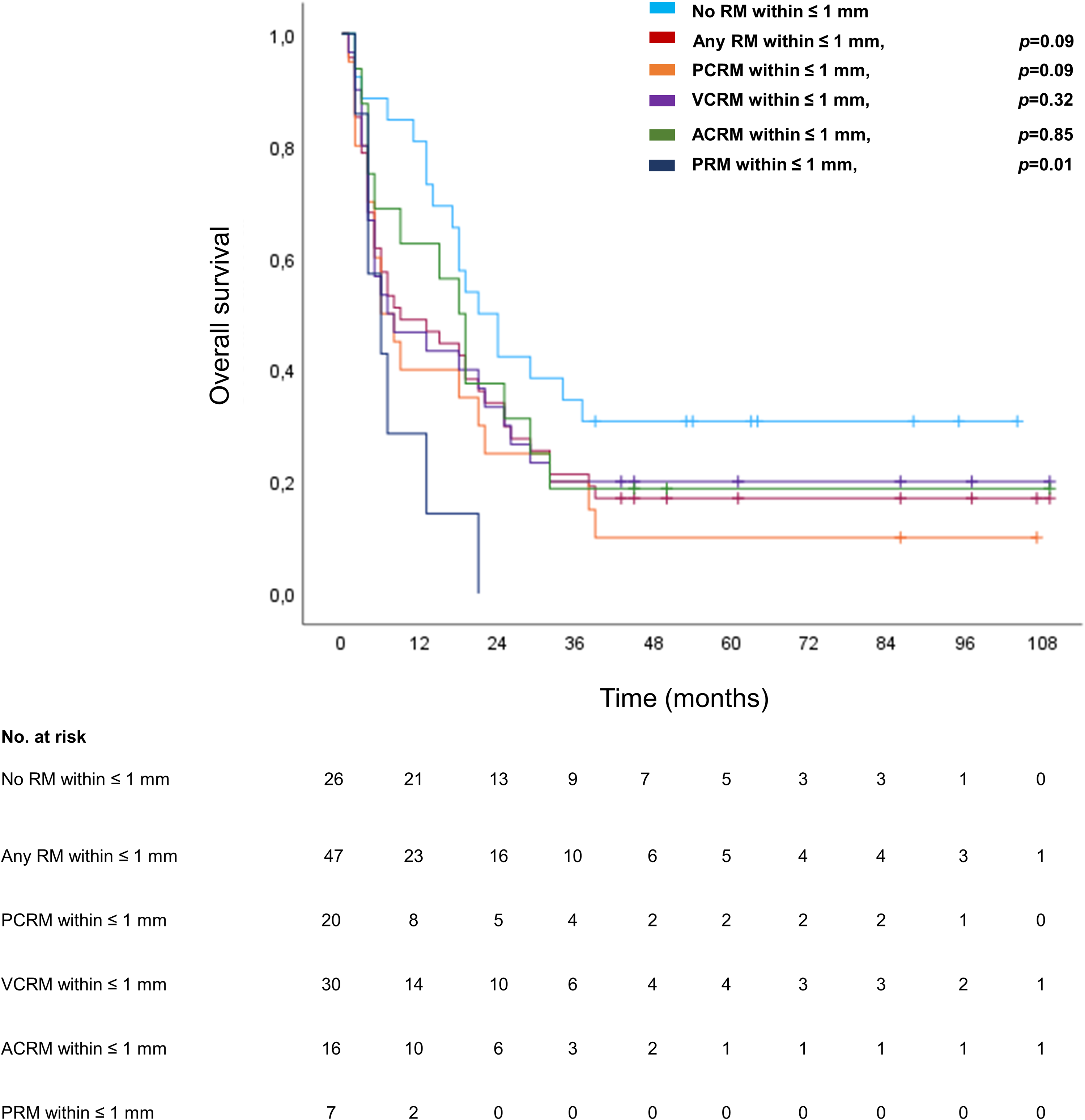
Survival analyses for the presence of tumor cells within ≤1 mm of RMs. The light blue line represents the survival curve in the absence of tumor cells within ≤1 mm of any RM; the red line represents specimens with tumor cells within ≤1 mm present at any RM; the orange line represents specimens with tumor cells within ≤1 mm of the PCRM; the purple line represents specimens with tumor cells within ≤1 mm of the VCRM; the green line represents specimens with tumor cells within ≤1 mm of the ACRM; and the dark blue line represents specimens with tumor cells within ≤1 mm of the PRM. Tick marks indicate censoring. P values refer to comparison of group of patients with presence versus absence of tumor cells within ≤1 mm of the specific RM analyzed. Abbreviations: ACRM, anterior circumferential resection margin; DMI, direct microscopic infiltration; PCRM, posterior circumferential resection margin; PRM, pancreatic transection margin; RM, resection margin; VCRM, vascular circumferential resection margin.
When analyzing the presence of tumor cells within ≤1 mm of a specific RM, individually, a significant impact on OS was seen for the PRM (median OS 6 months [CI: 0.9–11.1] versus 19 months [14.5–23.5], P = .01, Figure 4, dark blue line) and a trend toward significance for the PCRM (6 months [CI: 1.6–10.4] versus 19 months [12.8–25.2], P = .09, Figure 4, orange line). No difference in OS was seen for tumor cells within ≤1 mm of the VCRM and ACRM (P = .32 and .85, respectively). For the presence of tumor cells within ≤1 mm of the PRM a significant association was seen with male gender (P = .01) but not with other demographic or histopathological characteristics (age >65 years, pT3/4, pN+, pL1, Pn1, pV1, G3; P > .05).
To further endorse the impact of tumor cells within ≤1 mm of a specific RM, subgroup survival analysis was performed for the subset of specimens with tumor cells within ≤1 mm of only one specific RM (n = 27, 36%) for each RM, individually, compared to specimens with no tumor cells within ≤1 mm of any RM (n = 26, 35%). Also in this analysis, a significant impact on OS was seen for the PRM (median OS 2 vs 21 months (CI: 13.5–28.5), P < .01) but not for any other specific RM (P > .05). No significant association with demographic or other histopathological characteristics (gender, age >65 years, pT3/4, pN+, pL1, Pn1, pV1, G3; P > .05) was observed for PRM in this analysis.
Determination of a Prognostically Safe Distance to Resection Margins
To examine the impact of tumor distance of to the circumference beyond the presence of tumor within ≤1 mm of an RM or DMI, and to potentially determine a prognostically “safe distance” to the circumference, subgroups were formed for the incremental analysis of the closest distance of tumor cells to any RM in steps of 1 mm (see Table 3).
Survival analysis of these subgroups was performed and is presented in Figure 5. The shortest OS was seen in the group of tumors with DMI of one RM (Figure 5A, thick red line, 5-year survival rate 0%) and most patients survived over time in the subgroup of tumors with >5 mm distance to all RMs (Figure 5A, thick blue line, 5-year survival rate 60%). These 2 groups are intermediate to all other subgroups in the incremental analysis of the closest tumor distance to an RM (thin lines). However ,these intermediate subgroups, show only small differences, yielding no significant difference in OS between all groups (closest distance to an RM ≤ 1 mm but no DMI, > 1 mm & ≤2 mm, > 2 mm & ≤3 mm, >3 mm & ≤4 mm and >4 mm & ≤5 mm, 5-year survival rate ≤33% in all subgroups, P = .17).
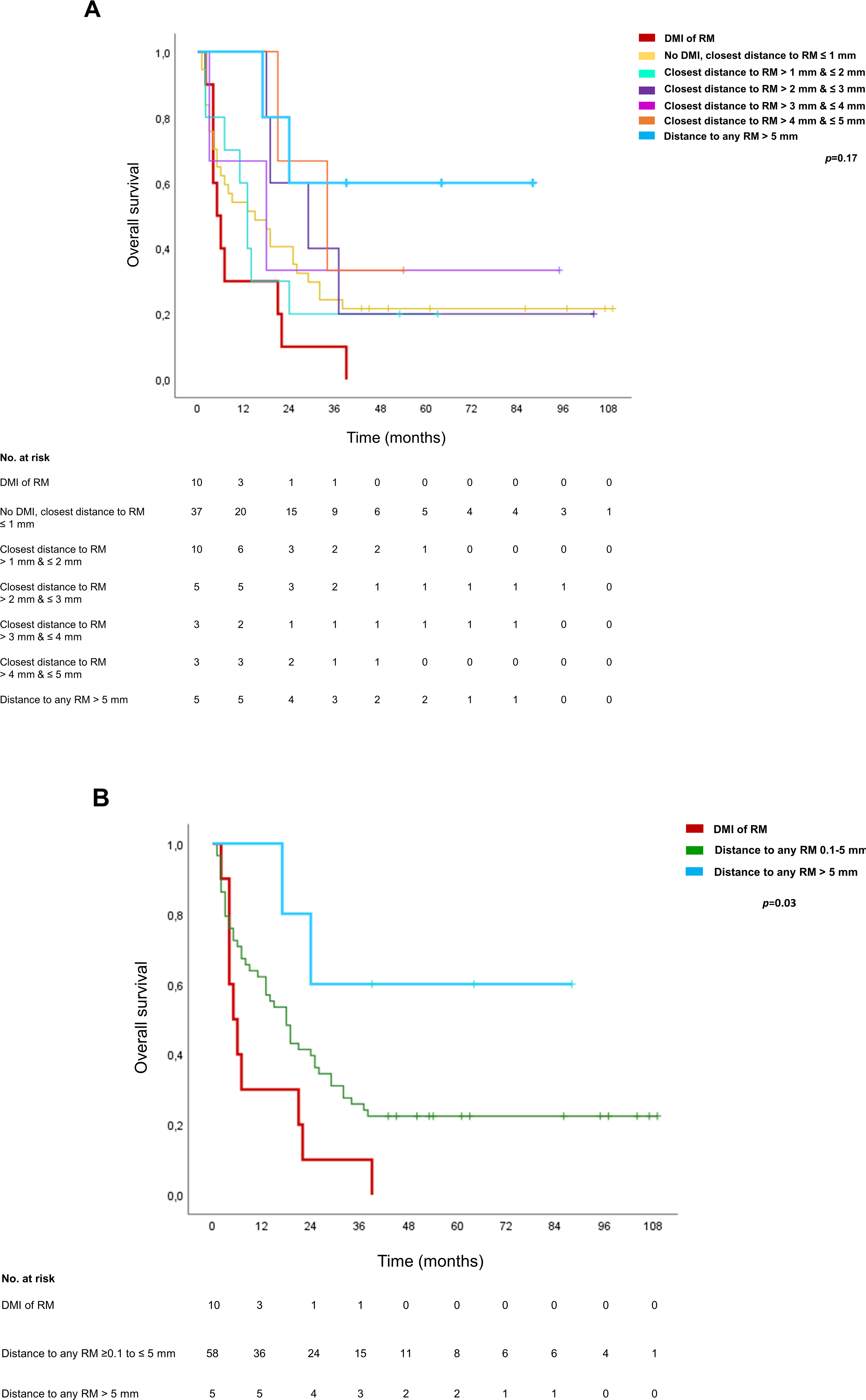
Survival analysis of subgroups formed by incremental analysis of the distance of tumor cells to the closest RM. (A) Survival analysis of all subgroups formed: DMI (thick red line), no DMI & closest RM ≤ 1 mm (yellow line), closest RM > 1 mm & ≤2 mm (turquoise line), >2 mm & ≤3 mm (purple line), >3 mm & ≤4 mm (pink line), >4 mm & ≤5 mm (orange) and >5 mm (thick light blue line). (B) Subgroup survival analysis : DMI (thick red line), distance to all RMs > 5 mm (thick blue line) and summary of all intermediate subgroups (≥0.1 to ≤5 mm, green line). Tick marks indicate censoring. Abbreviations: DMI, direct microscopic infiltration; RM, resection margin.
After combining the intermediate subgroups of closest distance to an RM from ≤1 mm (no DMI) to ≤5 mm (≥0.1 mm–≤5mm), survival analysis yields overall significance in the difference in OS of the 3 subgroups (Figure 5B, P = .03).
Resection Margins and Development of Metastatic Disease
Regarding distant hepatic and pulmonary metastases, 41% (n = 22) developed distant metastases. Hepatic metastases developed in 28% of the patients (n = 15) and pulmonary metastasis in 22% (n = 12). Five of these patients (9%) developed both hepatic and pulmonary metastases. The median time to liver metastasis was 3 (range 1–40) and 19 months (range 2–35) to pulmonary metastasis. Patients with liver metastases showed a significantly shorter OS compared to patients without metastases (median OS 18 months [CI: 4.1–31.9] vs 25 months [CI: 13.8–36.2], P = .02). No difference in OS was seen in patients with pulmonary metastases compared to patients without metastases (P > .05).
As previously presented for OS, we subsequently analyzed the impact of the different degrees of tumor distance to RMs in general and for the involvement of specific RMs individually on the development of hepatic and pulmonary metastasis. Among tumors that showed tumor cells within ≤1 mm of only one RM, the presence of tumor cells within ≤1 mm of only the VCRM led to a significantly shorter time to development of liver metastasis (50% of patients vs 5% of patients at 4 months, P = .05), whereas no difference was seen for any other RM (P > .05). Specifically, DMI of the PCRM and VCRM, showed significantly shorter time to pulmonary metastasis compared to no DMI of the respective RM (50% vs 2% and 100% vs 2% at 7 months, P < .05).
Discussion
The prognostic significance of residual disease after resection of malignant tumors is well established, and R-classification was added to histopathological TNM classification decades ago. 10 Since then, histopathological R-classification has been divided into microscopic residual disease (R1) and macroscopic residual disease (R2). 11 Traditionally, microscopic residual disease was defined as the presence of tumor cells directly within a resection margin on microscopic examination (DMI). Furthermore, in other malignancies, a negative prognostic role of tumor cells in the close proximity to resection margins (≤1 mm) has been demonstrated in addition to tumor cells directly infiltrating an RM. 12 These findings suggest that limiting histopathological examination and reporting to conventional R1 status of DMI may lead to occult residual disease. In the context of PDAC, this aspect is of paramount interest given its poor prognosis and thus, the imperative need to detect residual disease alongside reactive glands of associated chronic pancreatitis in a highly desmoplastic stromal reaction with diffuse infiltration. Additionally, given the complex anatomy in which the pancreatic head is embedded, the prognostic impact of margin involvement by PDAC may differ depending on the location of the involved margin.
In the present study, survival analysis of DMI of an RM was associated with a shorter OS. This represents the basis of the traditional R1 classification and is consistent with the existing literature.13–15 The OS analysis of the presence of tumor cells within ≤1 mm of an RM showed a negative impact with a trend toward significance. The significance of the presence of tumor cells within ≤1 mm of an RM in PDAC specimens remains equivocal in the existing literature. In a large study by Ghaneh et al, a negative impact on OS was seen for patients with conventional R1 resection status (DMI) but not for <1 mm positive RMs compared to patients with RM negative tumors (>1mm, median OS 18.7 and 25.4 months vs 24.9 months, P < .01). 7 Sugiura et al also did not observe a negative impact of the presence of tumor cells within ≤1 mm of an RM compared to RMs with >1 mm tumor clearance (median OS 30.4 vs 26.0 months, P > .05). In contrast, Jamieson et al showed a significant negative impact on OS for microscopic evidence of tumor ≤1 mm of an RM compared to tumor clearance >1 mm of RMs (median OS 15.4 vs 26.5 months, P = .01). 8 A negative impact on OS was also seen by Strobel et al between R0, R1 (<1 mm), and R1 (DMI) status (41.6, 27.5, and 23.4 months, P < .01). 16 Although final clarification appears to be pending, based on the existing data the Royal College of Pathologists as well as the American Joint Committee on Cancer (AJCC) consider tumor cells within 1 mm of a margin in PDAC as microscopic residual disease (R1).17,18
We also performed survival analyses of the individual RMs of the pancreatic head. For DMI as well as of tumor cells within ≤1 mm of an individual margin a significant impact on OS was seen for the PRM but not for any other margin. These results are consistent with the subgroup survival analysis of specimens in which tumor cells were present within ≤1 mm of only one specific RM. In this context, Tummer et al found no statistically significant association with OS for the location of an R1 margin using a 1 mm definition for R1. 19 Ghaneh et al found a negative impact on OS for PCRM DMI. 7 Similarly, Demir et al found a worse median OS for PCRM margin involvement compared to VCRM (median OS: 14.8 vs 31.0 months, P = .02). Thus, the findings of this study regarding the impact of VCRM and ACRM involvement are consistent with these data. For PCRM involvement of ≤1 mm, our data show a trend toward significance (P = .09). Additionally, our findings emphasize the prognostic impact of the PRM.
Subsequent to survival analyses of margin involvement (DMI and ≤1 mm), the impact of tumor distance >1 mm was examined in order to potentially identify a subset of patients with a higher survival rate and thereby determine a prognostically “safe distance.” Only small differences between distances were seen and OS analysis of all incremental subgroups of 1 mm from DMI to distance >5 mm did not reach significance. However, the survival rate in the subgroup of patients with margin clearance >5 mm, stood out (5-year survival rate 60%), and combining the intermediate subgroups as one (≥0.1 to ≤5 mm) yields overall significance in the difference of OS of the 3 subgroups formed, whereas no difference in OS was seen for clearance from 1 to 3 mm and clearance of 3 to 5 mm (P = 0.73). Similarly, Liu et al found a better OS for clearance of >5 mm than of >1 to ≤5 mm (P = .02) whereas no difference in OS was seen for clearance from 1 to 3 mm and clearance of 3 to 5 mm (P = .73). 20 However, Liu et al only examined the distance to the superior mesenteric artery (SMA) and all patients included in their study had received neoadjuvant therapy.
When analyzing the impact of margin involvement on the development of distant hepatic and pulmonary metastases, tumors with malignant cells within ≤1 mm of only the VCRM resulted in a significantly shorter time to liver metastasis. Individual DMI of the PCRM and VCRM showed significantly shorter time to pulmonary metastasis compared to no DMI of the respective RM. Similarly, Liu et al observed a higher rate of distant metastasis in patients with tumor involvement of ≤1 mm of the superior mesenteric artery compared to >1 mm, and Tummer et al noticed a shorter time from diagnosis to distant metastasis after R1 resection without specifying the site of margin involvement (≤1 mm).19,20 Portal vein invasion has also been seen to be associated with a higher incidence of distant metastasis. 21
Although the results of our study are encouraging, our present study has limitations. First, we did not subdivide the VCRM into SMA margin and superior mesenteric vein margin, which limits the interpretation of the individual impact of the superior mesenteric vessels involvement. Also, the size of our study cohort and, consequently the size of the subgroups formed is limited. For instance, only 2 of the patients included in this study (3%) received neoadjuvant therapy, not allowing a sufficient subgroup analysis of this subgroup. However, strict inclusion criteria were applied and specimens from each patient were re-examined for the purpose of this study by 2 pathologists independently with respect to the distance between the tumor and the respective margins. Future larger multi-institutional studies with further subdivision of margins would improve the statistical power of these results and allow a more precise differentiation of the margin impact on prognosis. The results of the survival analysis of the larger subgroup of patients after primary surgery (97%) are summarized in Supplemental Table S1.
Nevertheless, the results presented provide suggestions for clinical implications. Intraoperative histopathological examination of frozen sections of the PRM during oncologic resection of the pancreatic head is considered standard in specialized centers. Our results suggest that intraoperative evaluation of the PRM not only for DMI but also for tumor cells within ≤1 mm as a basis for margin revision may hold the potential to improve resections with margin clearance >1 mm of the PRM and thereby improve prognosis. 22 Currently, we perform frozen sections using OCT (optimal cutting temperature) as embedding media. Next, the tissue is frozen a −20°C to −15°C using a cryostat. The quality of the analysis is best for small tissue samples ideally up to 5 mm in diameter. The tissue is cut into 4 to 7 microns thick slides. Finally, the slides are stained with H&E. This procedure leads to multiple possible artifacts, such as ice crystals, overfreezing, and underfreezing, often making analysis difficult. In order to perform a more precise analysis of PRM the use of molecular biology techniques holds potential in the near future. Indeed, molecular based analysis of the intraoperative RM using methylation data would be of great use, as shown in recent studies on formalin fixed paraffin embedded (FFPE) samples using machine learning classifiers that can automatically detect PDAC. 23 In order to transfer this technique into the intraoperative setting nanopore sequencing of targeted DNA methylation regions needs to be employed. Moreover, preoperative imaging criteria as surrogate parameters for predicting margin positive resection (R1) in resectable PDAC should receive more attention. 24 In this way, preoperative prediction of R1 resection may represent a sufficient basis to guide neoadjuvant therapy to improve prognosis and R0 resection rates.25–27
In conclusion, the present study underlines the utmost importance of accurate histopathological preparation and examination of PDAC specimens including a differentiated analysis of each margin individually and its exact distance from the tumor not only as a basis for future studies but also for considerations in contemporary clinical decision making.
Supplemental Material
sj-docx-1-ijs-10.1177_10668969241229342 - Supplemental material for Stepwise Analysis of Resection Margin Impact on Survival and Distant Metastasis in Pancreatic Head Ductal Adenocarcinoma
Supplemental material, sj-docx-1-ijs-10.1177_10668969241229342 for Stepwise Analysis of Resection Margin Impact on Survival and Distant Metastasis in Pancreatic Head Ductal Adenocarcinoma by Florian N. Loch, Carsten Kamphues, Freschta Rieger, Katharina Beyer, Wael Rayya, Christian Schineis, Frederick Klauschen, David Horst, Simon Schallenberg and Mihnea P. Dragomir in International Journal of Surgical Pathology
Footnotes
Author Contributions
FNL, CK, SS, and MPD were involved in conceptualization, methodology, and project administration; FNL, FR, SS, and MPD in software and visualization; FNL, CK, SS, and MPD in validation; FNL, CK, FR, SS, and MPD in formal analysis, investigation, and writing—original draft preparation; FNL, CK, KB, WR, CS, FK, DH, SS, and MPD in resources; FNL and FR in data curation; and CK, KB, WR, CS, FK, and DH in supervision.
Data Availability Statement
The datasets used and/or analyzed during the current study are available upon reasonable request pending approval by the local data security authorities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The work of MPD was supported by Berlin Institute of Health (Junior Clinician Scientist Program) and DKTK Berlin (Young Investigator Grant 2022). The work of SS was supported by the Federal Ministry of Education and Research (BMBF).
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki. It was approved by the local ethics committee (No. EA4/020/19).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bundesministerium für Bildung und Forschung, (grant number 16LW0239K).
Informed Consent
The written informed consent was waived due to the retrospective study design.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
