Abstract
SMARCA4-deficient undifferentiated tumors (SMARCA4-UT) are a newly described entity and are typically seen in the thoracic cavity. However, these tumors have been described in other body sites, including the esophagus. These tumors are rare, aggressive neoplasms, characterized by the loss of protein product of SMARCA4 (Brahma-related gene-1) and the preservation of INI1 (SMARCB1) expression. Here, we present two tumors of SMARCA4-UT of the esophagus with its microscopic appearance and immunohistochemical profile. We also include a literature review of SMARCA4-deficient tumors of the tubular gastrointestinal tract with their immunohistochemical and mismatch repair profiles for each specimen. Due to its non-specific histologic appearance and variable staining in expanded immunohistochemical panels, this tumor frequently overlaps with other tumor types, making the diagnosis of SMARCA4-UT challenging. These tumors are often associated with intestinal metaplasia of the esophagus and are thought to represent a high-grade undifferentiated transformation of a conventional esophageal adenocarcinoma. These tumors are typically associated with poor clinical outcomes and have poor response to conventional therapies. Currently, there are no standard guidelines for treatment of these tumors; however, palliative radiotherapy and systemic chemotherapy may provide benefit. More recently, immunotherapy and novel therapeutic targets have shown some promise for these patients.
Introduction
Switch/sucrose non-fermentable (SWI/SNF) related, matrix associated, actin-dependent regulator of chromatin, subfamily A, member 4 (SMARCA4)-deficient undifferentiated tumors (SMARCA4-UT) are a group of recently characterized entities which includes several tumors arising in the brain, prostate, breast, pancreas, ovary, endometrium, lung, GI, and sinonasal tract. 1 The vast majority of SMARCA4-UT arise in the thoracic cavity. 2 These tumors tend to be smoking-related and display strong expression of SOX2, CD34, and SALL4. 3 Morphologically, these tumors share a number of characteristics including sheet-like proliferation of discohesive cells with focal rhabdoid appearance, large nuclei with prominent nucleoli, and variable necrosis and mitotic activity. While all these lesions share a common underlying abnormality, the pathogenesis of these tumors varies depending on the site. Here, we present two specimens of SMARCA4-deficient undifferentiated carcinomas of the esophagus and their diagnostic challenges.
Tumor #1
Biopsy of a hemorrhagic, fungating esophageal lesion revealed fragments of a highly necrotic tumor (Figure 1a) in a background of intestinal metaplasia with goblet cells (Figure 1b). The tumor was composed of sheets of discohesive cells with variable nuclear pleomorphism, prominent nucleoli, frequent apoptosis, and focal rhabdoid appearance. Immunohistochemical staining (IHC) was performed to determine lineage, however, tumor cells were negative for pan-keratin (Figure 1d), p40, CDX2, HER2, S100, CD45, SOX10, CD34, synaptophysin, chromogranin A, myogenin, DOG1, KIT (CD117), CD30, and ALK. MLH1, PMS2, MSH2, and MSH6 staining was intact, as was INI1 (SMARCB1) (Figure 1f). No definite mucin staining was identified on a PAS histochemical stain. The tumor cells were positive for CD138 (Figure 1e) and Vimentin. Staining with Brahma-related gene-1 (BRG1), the SMARCA4 protein product, demonstrated loss of expression within tumor cells with intact staining in adjacent stromal cells (Figure 1b). BRG1 staining was also preserved within foci of intestinal metaplasia. The specimen was reported as a SMARCA4-deficient undifferentiated carcinoma of the esophagus.
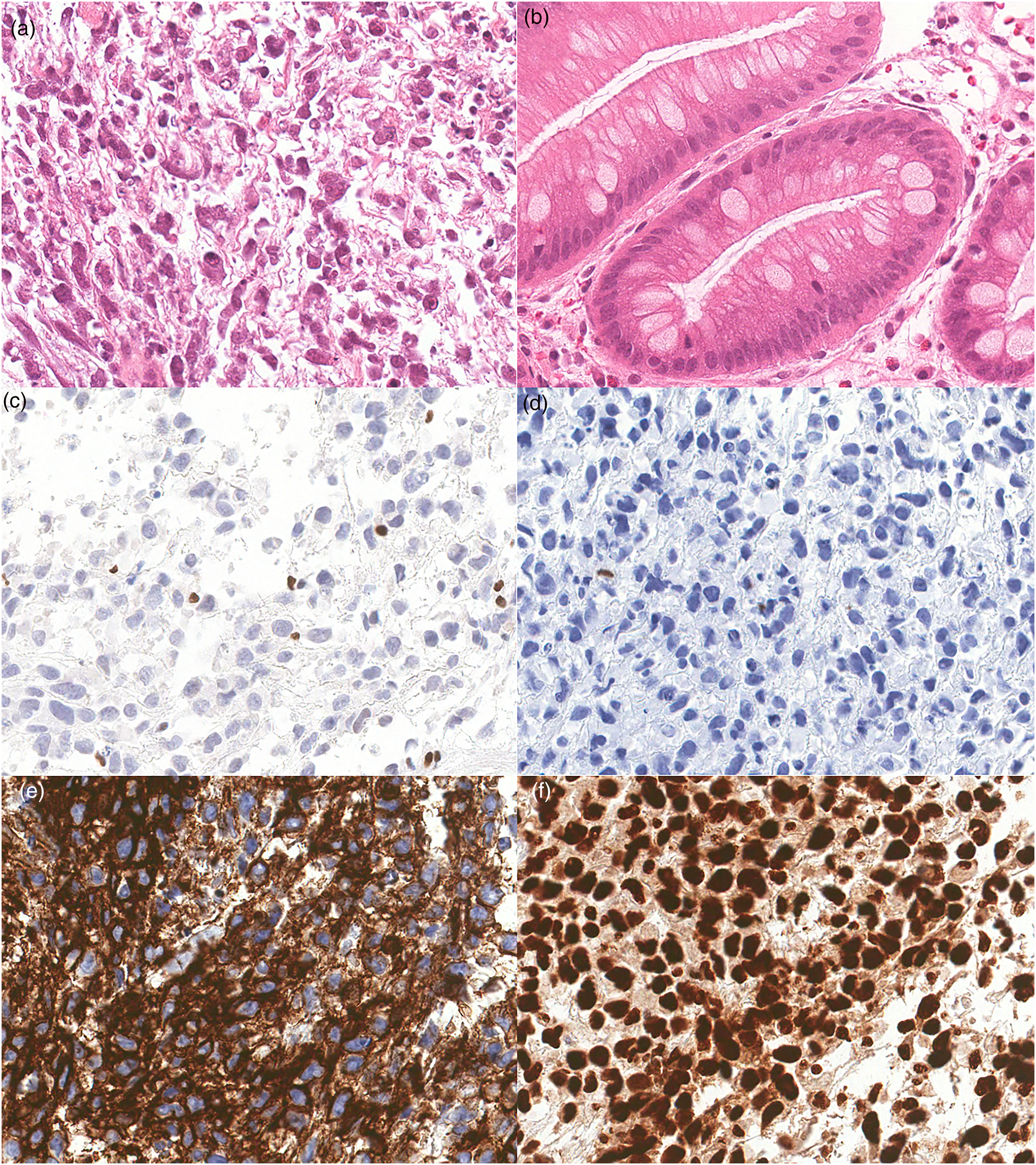
Representative H&E and immunohistochemical staining of SMARCA4-UT. (a) H&E staining of tumor showing rhabdoid morphology and necrosis (40x). (b) H&E staining of background Barrett Esophagus (20x). (c) Tumor cells exhibit loss of protein expression for BRG1 (SMARCA4) (20x). (d) Negative staining in tumor cells for Pan- KRT (20x). (e) Tumor cells stain positively for CD138. (f) Tumor cells show intact expression of INI1 (40x). SMARCA4-UT, SMARCA4-deficient undifferentiated tumors; BRG1, Brahma-related gene-1.
Tumor #2
Endoscopic biopsy of an esophageal mass lesion revealed a necrotic tumor (Figure 2a) arising in a background of intestinal metaplasia (Figure 2b). The tumor cells were arranged in sheets with nuclear pleomorphism, prominent nucleoli, frequent mitoses, and apoptosis with an epithelioid appearance (Figure 2). An extensive IHC panel was performed, and tumor cells were negative for keratin 7, MOC31, CD3, CD20, p40 (Figure 2e), Arginase, S100 (Figure 2f), CD117, DOG1, ERG, CD31, SMA, and CD30. Tumor cells were positive for CD34 (Figure 2c). Subsequent staining for BRG1 was performed which demonstrated loss of protein expression in the tumor cells (Figure 2d). This specimen was also reported as a SMARCA4-deficient undifferentiated esophageal tumor.
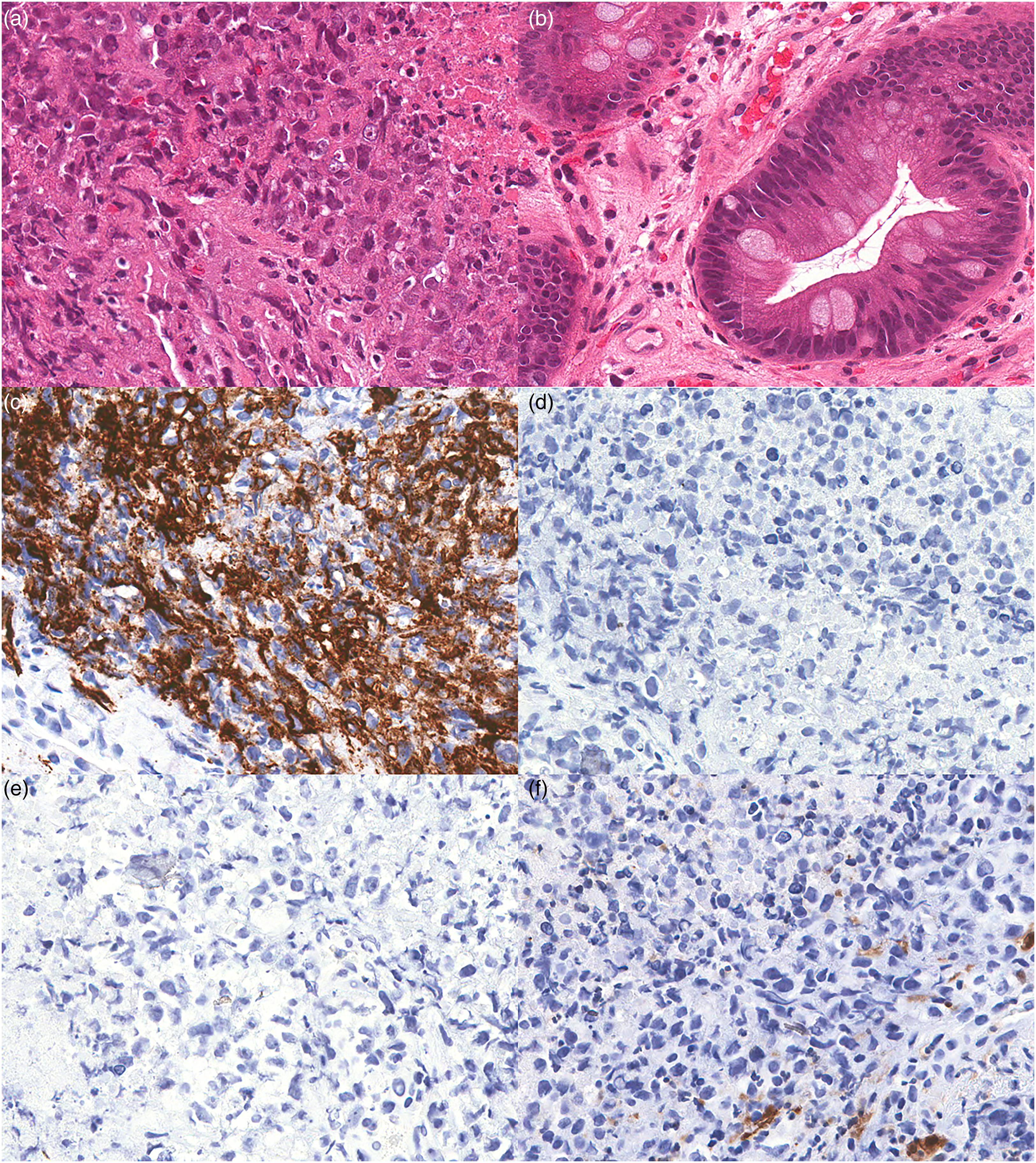
Representative H&E and immunohistochemical staining of SMARCA4-UT. (a) H&E staining of tumor showing rhabdoid morphology and necrosis (40x). (b) H&E staining of background Barrett Esophagus (20x). (c) Tumor cells stain positively for CD34 (20x). (d) Tumor cells exhibit loss of protein expression for BRG1 (20x). (e) p40 (20x), and (f) S100 (20x). SMARCA4-UT, SMARCA4-deficient undifferentiated tumors; BRG1, Brahma-related gene-1.
Discussion
SMARCA4 is a critical component of the SWI/SNF complex along with other members including ARID1A, SMARCB1, and SMARCA2. The main role of this complex is to facilitate transcriptional regulation and lineage specification through nucleosome mobilization and chromatin remodeling.4,5 In the esophagus, a minority of adenocarcinomas demonstrate loss in one or more components of the SWI/SNF complex. Most frequently, there is the loss of ARID1A (10%), however, the loss of SMARCA2 (9.9%), SMARCA4 (3.4%), and SMARCB1 (2%) is also seen. 5 Concordant ARID1A and MLH-1 loss has also been reported in 1-2% of cases. 6
The loss of SMARCA4 alone is insufficient for the classification as a SMARCA4-UT. The diagnosis of SMARCA4-UT requires characteristic morphology and lack of clear differentiation by morphology or immunohistochemistry as described. If such a tumor is suspected, IHC for all the components of the SWI/SNF complex may aid in diagnosis; however at initial diagnosis, a more comprehensive approach should be taken. A literature review of SMARCA4-UT of the tubular gastrointestinal tract demonstrates variable immunohistochemistry and mismatch repair (MMR) staining profiles that makes the diagnosis of this entity challenging (Table 1) and represent potential pitfalls.
Immunohistochemical Profile of SMARCA4-deficient Tumors of the Tubular Gastrointestinal Tract in the Literature.

IHC, immunohistochemical staining; MMR, mismatch repair.
These tumors typically have a relatively characteristic appearance with discohesive cells that have a rhabdoid/plasmacytoid appearance. However, this is not completely specific and other tumors can have an overlapping morphology. Based on morphology, the differential is typically broad and includes poorly differentiated carcinomas, melanoma, undifferentiated sarcomas, and hematolymphoid neoplasms. While there are a number of immunohistochemical stains that are more frequently positive in this lesion (Supplemental Table 2), as with any tumor with such a broad differential diagnosis, it is typically best to start with a small immunopanel that covers major lineages such as Pan- KRT, CD45, and S100.
SMARCA4-UT tend to display little differentiation by immunohistochemistry and routine screening panels highlighted above are often negative (Table 1). This prompts the need for larger immunopanels in an effort to demonstrate lines of differentiation. Focal and sometimes diffuse Keratin AE1/AE3 and CAM5.2 staining may be present and this can be very helpful to support this diagnosis and support the fact that this a poorly differentiated carcinoma.2,15 A subset of these tumors can also have focal staining of synaptophysin, chromogranin A, and CD56, which is a diagnostic pitfall that may result in the misclassification of the tumor as a high-grade neuroendocrine carcinoma. 15 CD138 and PAX5 staining has been reported in a SMARCA4-deficient tumor at the gastroesophageal junction (GEJ) 14 and is also a pitfall that could result in a misclassification as hematolymphoid neoplasm. CD34 staining may be variable, however, diffuse positive staining of an undifferentiated GEJ tumor can be seen and can also result in misclassification. 2 Notably, SALL4 staining is also often seen, which can result in the erroneous classification as a germ cell tumor.13,16 Given the undifferentiated nature of the tumor and the variable expression of some lineage markers, it is likely in the past that these tumors have been misclassified as hematolymphoid tumors, sarcomas, poorly differentiated carcinomas, vascular tumors, and germ cell tumors.
Similar to our specimen, MMR abnormalities and microsatellite instability-high (MSI-H) are not commonly seen in the esophagus. MMR loss by IHC has been reported in 3-6.6% of esophageal adenocarcinomas 18 and in a separate study determined that the MSI-H rate in esophageal squamous cell carcinoma and adenocarcinomas is 0.6%. 19 The rate of MMR loss in SMARCA4 undifferentiated carcinomas of the esophagus has not been characterized; however, elsewhere in the GI system, case reports of MMR loss in SMARCA4-deficient carcinomas have been demonstrated. Tessier-Cloutier et al reported an undifferentiated colonic carcinoma with SMARCA4, MLH1, and PMS2 loss. 17 There is great variability in MMR status among SMARCA4-deficient carcinomas in the GI tract with reports of tumors in the small intestine and stomach. 9 An analysis of the TCGA Pan-Cancer Atlas showed 14.7% of any gastric cancer with any SWI/SNF alteration had MSI. 20 Therefore, it appears that MMR deficiency is not mutually exclusive with SMARCA4 loss and should not preclude the classification as a SMARCA4-UT.
Barrett esophagus are present in both specimens. Though adenocarcinomas of esophagus and GE junction are frequently associated with Barret esophagus in the western world, the relationship of Barrett esophagus and the development of SMARCA4-UT is still unknown. The location of the tumors and the presence of intestinal metaplasia with goblet cells suggest the carcinoma lineage of the tumor which is in line with prior results demonstrating concurrence of Barrett esophagus with this lesion. 12 Moreover, further testing of BRG1 loss supports the diagnosis of SMARCA4-UT.
There is no standard treatment protocol in place for these patients, and in general, the prognosis for this entity is very poor. SMARCA4-UT carcinomas at extra-GI have showed inadequate response to systemic chemotherapy (albeit with limited short-term benefit with adriamycin and ifosfamide 21 ) and surgery 22 and variable response to palliative radiation therapy. 23 Patients with chemotherapy-resistant SMARCA4-UT have also shown some benefit using immune checkpoint inhibitors (ICIs) such as pembrolizumab and nivolumab and platinum-based chemotherapy.22,24 Due to growing body of literature suggesting the role of tumor mutation burden in the pathogenesis of SMARCA4-UT, the use of ICIs as first line-therapy in these patients is currently being evaluated. 24 More recently, clinical trials are underway and have shown some promise using novel therapeutic agents including BETi (bromodomain and extra-terminal motif protein inhibitors), enhancer of zeste homolog 2 (EZH2) (NCT03213665; NCT02875548; NCT02601950), cyclin-dependent kinase (CDK)4/6 inhibitors, and histone deacetylase inhibition.5,21,22,24–26 Therefore, it is important for pathologists to recognize SMARCA4 loss in poorly differentiated carcinomas both to direct patients towards the most effective existing therapies as well as allowing for patients the opportunity to be enrolled in existing clinical trials.
Conclusion
SMARCA4-UT present a unique diagnostic challenge for pathologists. While there are common morphologic features, the variability in histology, and IHC staining can lead to improper or delayed diagnoses. Recognizing this diagnostic entity earlier in the diagnostic work up can allow for more targeted and efficient IHC panels and limit the risk of misclassification. SMARCA4-UT are aggressive tumors with a poor clinical outcome, however, there are some emerging therapies which may hold promise in treating these tumors.
Supplemental Material
sj-docx-1-ijs-10.1177_10668969241228290 - Supplemental material for SMARCA4-Deficient Undifferentiated Tumor of the Esophagus: Diagnostic Pitfalls in Immunohistochemical Profiles
Supplemental material, sj-docx-1-ijs-10.1177_10668969241228290 for SMARCA4-Deficient Undifferentiated Tumor of the Esophagus: Diagnostic Pitfalls in Immunohistochemical Profiles by Rana Chakrabarti, Sherman Lin, Hui Wang and Matthew Cecchini in International Journal of Surgical Pathology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The patients had very aggressive clinical disease and unfortunately died shortly after the diagnosis was made. Therefore, it was not possible to obtain consent. Therefore, we cannot include patient details for this case. We have formatted this as a pitfall in pathology focusing on a review of the IHC profiles of these cases in the context of other cases published in the literature. The demographics of these cases is similar to other cases that have been presented in the literature.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
